Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Faheem Ahmad | + 2764 word(s) | 2764 | 2022-02-21 04:21:21 | | | |
| 2 | Beatrix Zheng | + 527 word(s) | 3291 | 2022-02-28 03:43:47 | | | | |
| 3 | Beatrix Zheng | + 2 word(s) | 3293 | 2022-02-28 04:20:42 | | | | |
| 4 | Beatrix Zheng | Meta information modification | 3293 | 2022-03-02 07:20:03 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Ahmad, F. Plant-Mediated Nanoparticle Synthesis. Encyclopedia. Available online: https://encyclopedia.pub/entry/19958 (accessed on 05 June 2026).
Ahmad F. Plant-Mediated Nanoparticle Synthesis. Encyclopedia. Available at: https://encyclopedia.pub/entry/19958. Accessed June 05, 2026.
Ahmad, Faheem. "Plant-Mediated Nanoparticle Synthesis" Encyclopedia, https://encyclopedia.pub/entry/19958 (accessed June 05, 2026).
Ahmad, F. (2022, February 28). Plant-Mediated Nanoparticle Synthesis. In Encyclopedia. https://encyclopedia.pub/entry/19958
Ahmad, Faheem. "Plant-Mediated Nanoparticle Synthesis." Encyclopedia. Web. 28 February, 2022.
Copy Citation
The green synthesis of nanoparticles(NPs) is gaining attention owing to its facilitation of the development of alternative, sustainable, safer, less toxic and environment-friendly approaches. Thus, green nanotechnology using plant extract opens up new possibilities for the synthesis of novel nanoparticles with the desirable characteristics required for developing biosensors, biomedicine, cosmetics and nano-biotechnology, and in electrochemical, catalytic, antibacterial, electronics, sensing and other applications.
biosynthesis
eco-friendly
green chemistry
nanoparticle
plant extract
sustainable application
1. Introduction
The nanotechnology sector has proven to be one of the most active research fields [1]. Owing to their broad uses in catalysis, sensing, electronics, photonics and medicines, the synthesis of nanoparticles has gained significant attention in recent decades [2]. Scientists have understood the potential of biological organisms to reduce metal precursors since the nineteenth century, but the mechanisms are still not known. Researchers have drawn attention towards biological methods due to the success of nanoparticle synthesis using natural reduction, capping and stabilizing agents, and avoiding harmful chemicals and high energy consumption [3][4][5]. A wide variety of products (e.g., Quantum dots (Q-dots) of cadmium sulphide, titanium oxide hybrid-based electrochemical biosensors and oxorubicin-loaded heparinized nanoparticles) can be developed through nanotechnology, and applicable to a broad array of scientific fields, including optoelectronics, biosensors, nano-biotechnology, biomedicine and others [6][7][8][9]. Creation, exploitation and synthesis are nanotechnology concepts that typically consider materials smaller than 1 mm in dimension [10]. Many different methods, such as physical, chemical and green (biological) techniques, have been used to synthesize nanoparticles [11][12][13]. The stabilized nanoparticles are formed by reducing ions through reduction (palladium nanoparticles(NPs)), nucleation (silver NPs) and growth system (silver NPs) [14][15][16]. Green chemistry, which uses chemical principles to reduce or eliminate the use of hazardous substances, has led to considerable reductions in toxic residues, which are harmful to man and the environment.
Green chemistry may be defined as chemical-assisted pollution-prevention strategies employed in specific domains such as green analytical chemistry, ecologically friendly analytical chemistry and clean analytical methodologies [17]. Thus, green synthesis is regarded as a viable approach for nanoparticle synthesis since it is biocompatible, inert and environmentally safe [18].
2. Biosynthesis of Novel Metal Nanoparticles Using Plant Extracts
Nanoparticles with sizes ranging from 1 to 100 nm bind larger particles to atomic or molecular structures [19]. They are synthesized via different approaches, mainly divided into physical and chemical processes (Figure 1). The physical process involves laser ablation, condensation, evaporation, etc., whereas the chemical process involves hydrazine, sodium borohydride, green synthesis, etc. Using plant species to produce nanoparticles has been termed a green technique (Figure 1 and Figure 2) and the most reliable environmentally sustainable approach [20][21]. Nowadays, researchers are attracted towards biological synthesis, including the use of natural reducing, capping and stabilizing agents and without using hazardous, high-cost chemicals and high power consumption [22] (Figure 1 and Figure 2). NPs are extensively utilized in human contact areas (medicine, [23][24] and agriculture, [25][26]), and synthesis methods that do not use harmful compounds are increasingly required.
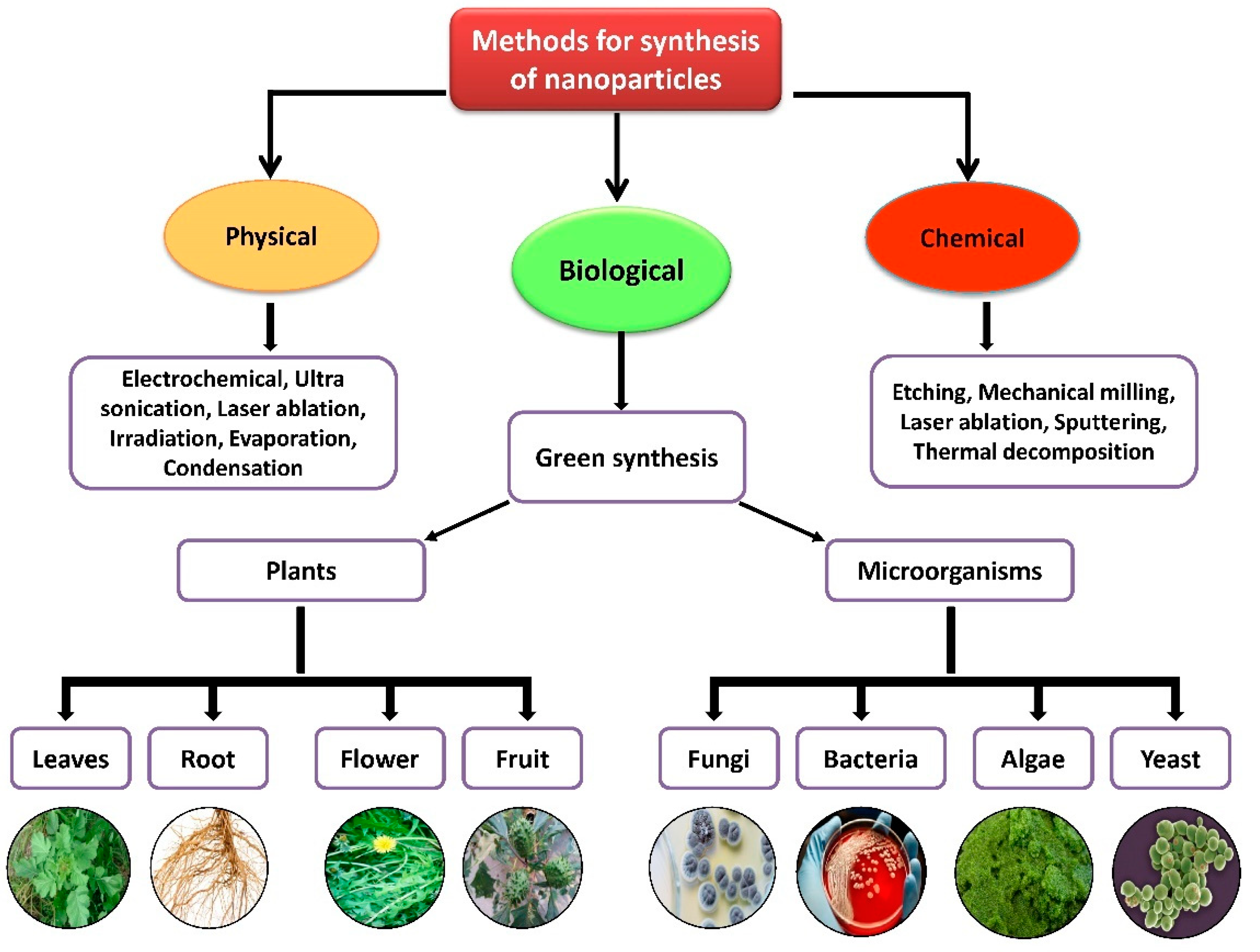
Figure 1. Different methods of nanoparticle synthesis.
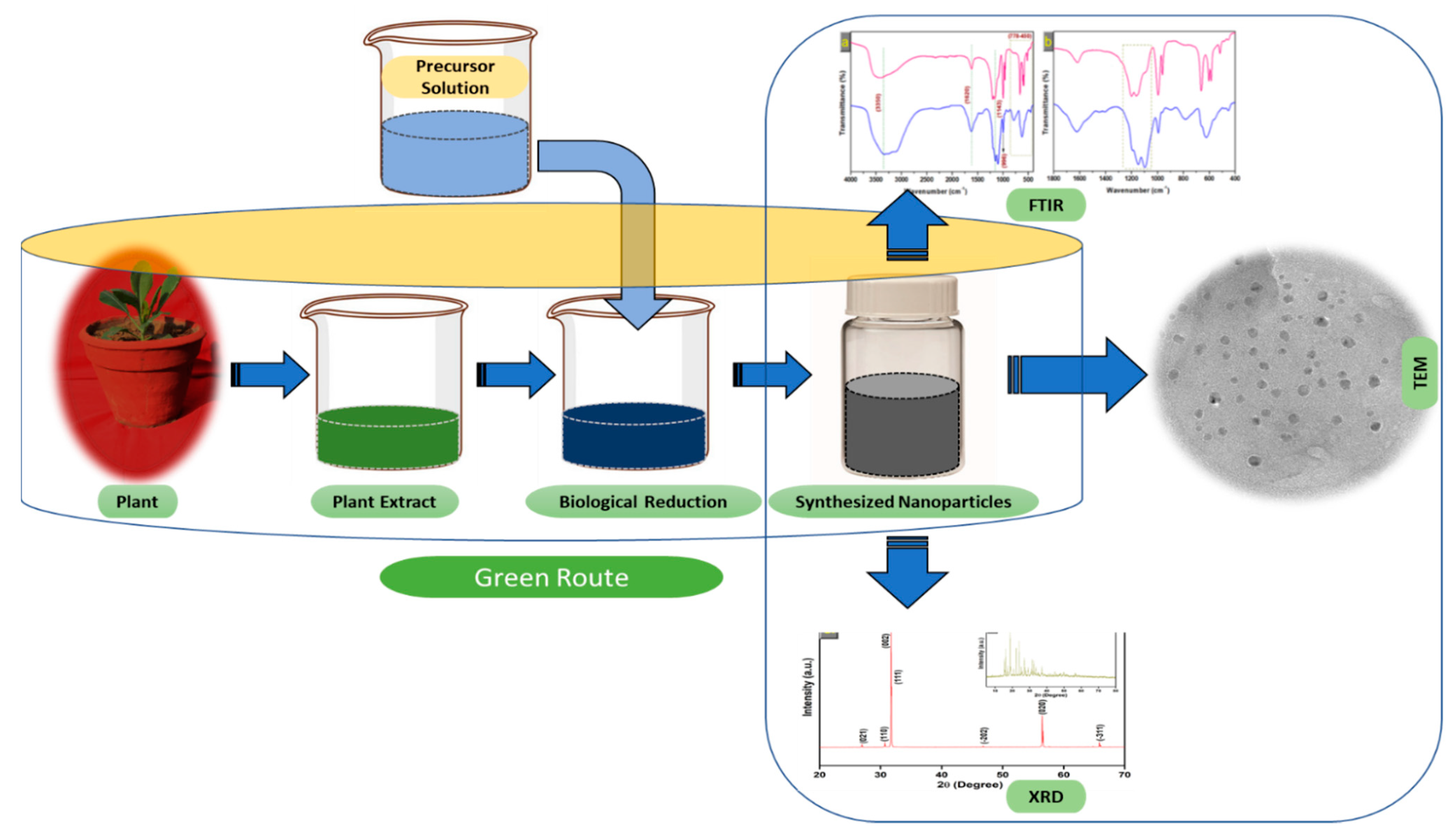
Figure 2. The schematic diagram for the biosynthesis of nanoparticles (NPs) via a green route using plant extract.
2.1. Mechanism of Nanoparticle Synthesis
Extensive research has been published on the testing and assessing of plants to prepare metallic nanoparticles (Figure 2), but the underlying principle for synthesizing nanomaterials has received comparatively less scientific attention [27][28]. The general tools, steps and materials involved in nanoparticle synthesis include reducing agents, capping agents, solvents, metal salts, nucleation, growth, aggregation, stabilization and characterization (Figure 3). Chemical reduction is commonly used in nanoparticle synthesis. Most methods utilize highly reactive reducing agents such as amino acids, citric acid, aldehydes, flavonoids, NADP reductase, tartaric acids, secondary metabolites, etc. Two researchers reported that the reduction potential of each metal is different and greatly affect the reduction of metals or metal precursors during synthesis. If the positive reduction potential is more, the metal precursor can be reduced at a faster rate. The nucleation and growth phases will be close to equilibrium when the reducing rate is slow [29][30]. In one-step synthesis, the slow reduction rate is also a key factor in the production of Au−Pd core–shell NPs. The finding reported the reduction potentials of PdCl42−/Pd and AuCl4−/Au are 0.59 and 0.99 eV, respectively. As confirmed from the TEM analysis, during reaction the Au particles were synthesized earlier then Pd at different time intervals. This is highly consistent with PdCl42−/Pd and AuCl4−/Au’s redox potential difference, and it is believed that this difference is very important for the development of the core–shell NPs [30]. In the water-soluble components of geranium leaves, Shankar et al. [31] recognized proteins and secondary metabolites. They suggested that terpenoids aid in reducing silver ions, which are then oxidized to carbonyl groups. In a study with tamarind leaf broth, the probability of an acid (tartaric acid) functional group operating as a capping medium and being essential for forming bio-reduced gold nanoparticles was studied by Ankamwar et al. [32]. This entry investigated the way that alfalfa roots can absorb silver from agar media in the form of Ag(0) and transmit it to the shooting segment in the identical oxidation number [33]. The synthesized nanoparticles’ general characterization was carried out through scanning electron microscopy (SEM), transmission electron microscopy (TEM), energy-dispersive X-ray spectroscopy (EDX), ultraviolet–visible spectroscopy (UV–Vis), Fourier-transform infrared spectroscopy (FTIR) and X-ray diffraction (XRD). Microscopy (SEM and TEM) is used to determine the shape, size and particle aggregation of the desired nanoparticles without any comparison with standard materials [34]. Spectrometric techniques are the most widely used tactic for nanoparticle characterization. EDX is used to confirm the composition and distribution of the nanoparticles through spectrum and element mapping. The UV–Vis spectrometry investigates nanoparticles on the basis of particle aggregation and average particle size [35]. The basic principle of this method is absorption of plasmas by free electrons attached on the surface of nanoparticles. They interact with the electromagnetic field and shift towards higher wavelength values because the size of nanoparticles is directly proportional to higher values of wavelength. Furthermore, FTIR and XRD are applied for the determination of structural characteristics and crystallinity of formed particles.
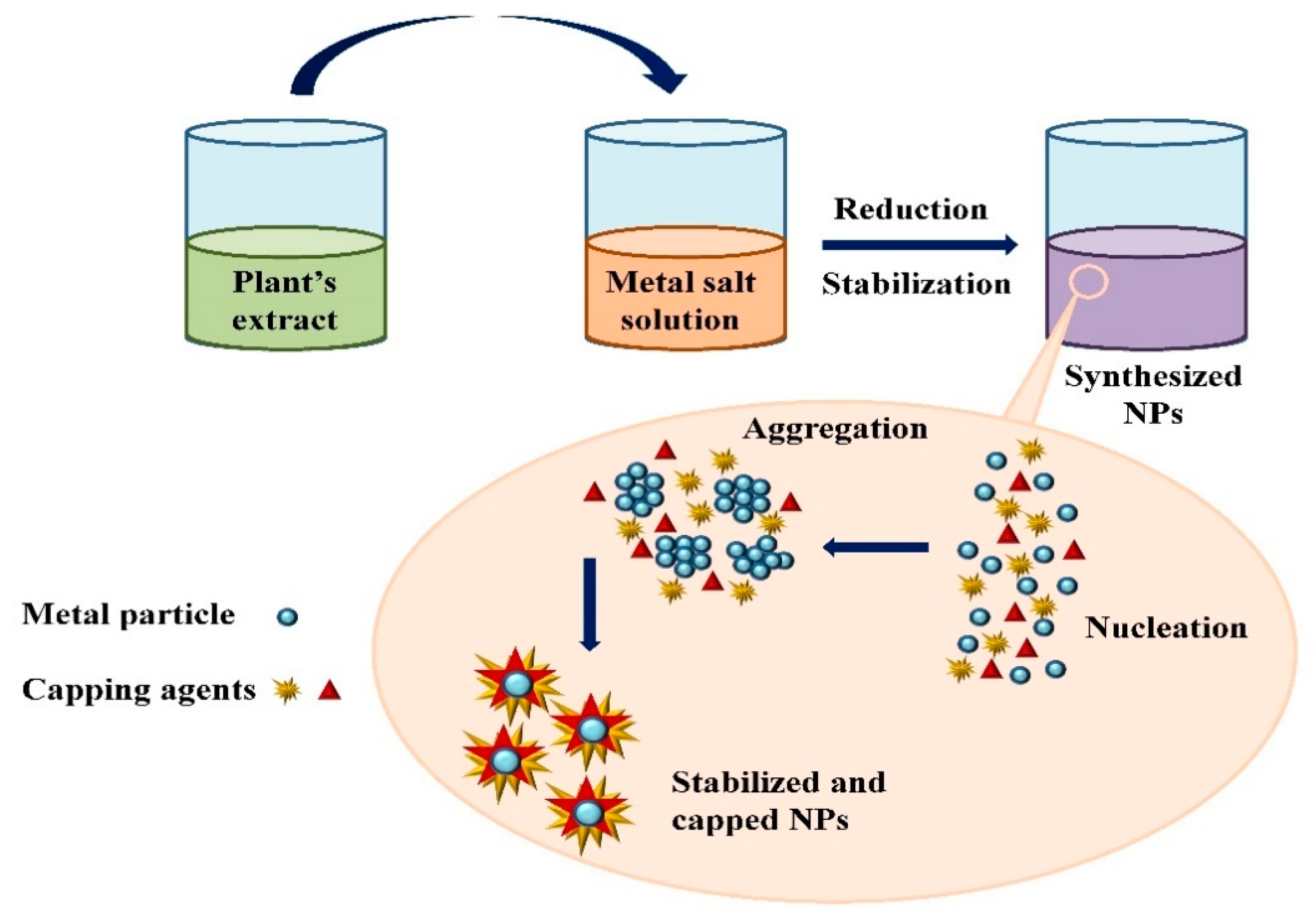
Figure 3. Mechanism of nanoparticle synthesis using phytoextracts.
The information on the production of various metallic NPs such as silver, gold, zinc, palladium and titanium using various plant extracts is summarized here.
2.2. Silver Nanoparticles
Silver nanoparticles (AgNPs) are commonly utilized nanoparticles and have attracted much study interest due to their distinctive properties. They are widely used in emerging biomedical and industrial applications [36]. AgNPs exhibit completely different characteristics from bulk materials derived from the same material due to their elevated surface/volume ratio [37]. In recent times, the synthesis of silver NPs by bio-organisms containing phytochemical agents has become an important goal for workers. Various unique secondary metabolites derived from plant extracts such as sugars, alkaloids, phenolic acids, flavonoids and terpenoids are responsible for bio-reducing ionic silver metal into nanoparticles [21][38][39].
Biosynthesis of AgNPs by Tribulus terrestris [40] and Astragalus tribuloides Delile [41] has already been reported. Spherical silver nanoparticles of size 2–6 nm were obtained from Cycas leaf [42]. For the synthesis of AgNPs, the affinity of Curcuma longa bark and powder extracts was determined. It was found that bark extract could produce more AgNPs than powder extract [43]. Kumar and Yadav [44] investigated Lonicera japonica plant leaf extract to develop silver and gold nanostructures. The particles obtained were different in size and shape; AgNPs were spherical to plate-like poly-shaped, and their size was 36–72 nm. Banerjee and Narendhirakannan [45] utilized seed extract of Syzygium cumuni to form crystalline silver nanoparticles. There is considerable data available on how to make silver nanoparticles from the latex of the Plumeria rubra plant [46]. Ponarulselvam et al. [47] evaluated Catharanthus roseus to produce silver nanoparticles because of the presence of vincristine and vinblastin. Sathishkumar et al. [48] prepared silver nanoparticles using Cinnamomum zeylanicum bark extract and powdered bark extract and studied the variations in the biogenic nanoparticles.
AgNPs were synthesized with a 58–458 nm range in size from the leaf extract of Mukia maderaspatana [49]. Pedalium murex was also reported to synthesize AgNPs by Anandalakshmi et al. [50]. The TEM micrographs revealed that the produced AgNPs were circular with a mean value of 50 nm. Raju et al. [51] utilized living peanut plants to synthesize AgNPs. The TEM examination showed that the biosynthesized AgNPs were of different shapes (spherical, hexagonal, triangular, square and rod-shaped) and sizes. Most of the formed AgNPs were spherical and 56 nm in average size. The EDX technique confirmed that the formed NPs were of silver. Some reports on plant-assisted synthesis of silver nanoparticles are enlisted below in Table 1.
Table 1. Plant-assisted synthesis of silver nanoparticles.
| Plant Name | Parts Used | Size (nm) | Shapes | Reference |
|---|---|---|---|---|
| Morinda citrifolia L. | Leaves, fruit pulp, seeds | 3–11 | Spherical | [52] |
| Nymphae odorata | Leaves | 15 ± 5 | Spherical | [53] |
| Capparis zeylanica | Leaves | 23 | Spherical | [54] |
| Caesalpinia pulcherrima | Leaves | 9 | Spherical | [55] |
| Carya illinoinensis | Leaves | 12–30 | Spherical | [56] |
| Mentha piperita | Leaves extract | 35 | Spherical | [57] |
| Jatropha curcas | Latex | 10–20 | Face-centered cubic | [58] |
| Acalypha indica | Leaves extract | 20–30 | Spherical | [59] |
| Hibiscus rosa sinensis | Leaves | 14 | Spherical/prism | [60] |
| Cycas | Leaves | 2–6 | Spherical | [42] |
| Ceratonia siliqua | Leaves extract | 5–40 | Spherical | [61] |
| Suaeda monoica | Leaves | 31 | Spherical | [62] |
| Catharanthtus roseus | Leaves | 35–55 | Cubical | [47] |
| Ocimum sanctum | Leaves extract | 10–20 | Spherical | [63] |
| Ocimum tenuiflorum | Leaves | 25–40 | Spherical | [64] |
| Ginkgo biloba | Leaves | 15–500 | Cubic | [65] |
| Tanacetum vulgare | Fruit | 16 | Spherical | [66] |
| Argemone mexicana | Leaves extract | 30 | Spherical, hexagonal | [67] |
| Sesuvium portulacastrum | Callus extract | 5–20 | Spherical | [68] |
| Syzygium cumini | Leaves and seed | 29–92 | Spherical | [45][69] |
| Cinnamomum camphora | Sun dried leaves | 3.2–20 | Cubic hexagonal crystalline | [70] |
| Melia azedarach | Leaves | 78 | Spherical | [71] |
| Rhododedendron dauricam | Flower extract | 25–40 | Spherical | [72] |
| Lippia citriodora | Leaves extract | 15–30 | Crystalline | [73] |
| Tribulus terrestris | Fruit | 16–28 | Spherical | [40] |
| Citrullusm colocynthis | Leaves | 31 | Spherical | [74] |
2.3. Gold Nanoparticles
Gold nanoparticles (AuNPs) are the most appealing new metal NPs due to their remarkable uses in catalysis, gene expression, nonlinear optics, nanoelectronics and disease diagnostics fields [75]. Gold nanoparticles made using either phytochemicals or other extract constituents are stable for a limited period [76]. According to Sharma et al. [77], tea leaf extract can be employed in gold NP preparation. Suman et al. [78] synthesize gold NPs of size range 8–17 nm from the root extracts of Morinda citrifolia at ambient temperature. The biogenic production of gold nanoparticles exploiting Nyctanthes arbortristis alcoholic extract led to the creation of spherical-shaped nanostructures of size 19.8 ± 5.0 nm [79]. The synthesis of AuNPs was reported with Bael (Aegle marmelos) leaves and the particles obtained were round and 4–10 nm in size [80].
Lee et al. [34] performed the synthesis of AuNPs from the peel aqueous extract of Garcinia mangostana. The aqueous solution of gold in contact with G. mangostana extract was reduced to gold metal ions and synthesized AuNPs. The FTIR results suggested that the reducing agent found in the aqueous solution of G. mangostana is strongly associated with anthocyanins, benzophenones, flavonoids and phenols. The synthesized AuNPs were spherical with a size range of 32.96 ± 5.25 nm that was analyzed by TEM. Rodríguez-León et al. [81] synthesized AuNPs from the bark extract of Mimosa tenuiflora at different metallic (acting as precursor) concentrations.
AuNPs were made from the aqueous suspension of Azadirachta indica [82]. When the A. indica extract was mixed with Au(III) solution, the nanoparticle formation commenced. Kasthuri et al. [83] constructed gold nanoparticles with triangular and hexagonal shapes from HAuCl4 solution and a diluted extract possessing phyllanthin (derived from Phyllanthus amarus). Aromal and Philip [84] synthesized AuNPs using Benincasa hispida seed extract as either a reducing or capping agent. Carboxylic groups (COOH) found in the plant extract change to COO- during the reduction process. The protein’s COOH group works as a surfactant, adhering to the surface of the AuNPs and then stabilizing AuNPs via electrostatic stabilization. The synthesized AuNPs were observed to have a crystalline nature and were 10–30 nm in size. Some reports on the plant-assisted synthesis of gold nanoparticles are listed below in Table 2.
Table 2. Plant-assisted synthesis of gold nanoparticles.
| Plant Name | Parts Used | Size (nm) | Shapes | Reference |
|---|---|---|---|---|
| Parkia biglobosa | Leaves | 1–35 | Truncated, pentagonal, spherical, triangular | [35] |
| Curcuma pseudomontana | Rhizome | 20 | Spherical | [85] |
| Lawsonia inermis | Leaves | 20 | Spherical | [86] |
| Cinnamon | Bark | 35 | Spherical | [87] |
| Croton Caudatus Geisel | Leaves | 20 | Spherical | [13] |
| Tamarind | Leaves | 20–40 | Triangle | [32] |
| Aloe vera | Plant extract | 50/350 | Crystalline | [88] |
| Mentha, Ocimum, Eucalyptus | Leaves | 3–16 | Spherical | [89] |
| Canna indica, Quisqualis indica | Leaves and flower | 30–130 | Polymorphic/stable | [90] |
| Murraya koenigii | Leaves | 20 | Spherical | [91] |
| Aegle marmelos | Leaves | 4–10 | Spherical | [80] |
| Rosa hybrid | Rose petals | 10 | Cubic | [92] |
| Terminalia chebula | Plant extract | 6–60 | Anisotropic | [93] |
| Momordica charantia | Fruit | 30–40 | Cubical | [94] |
| Phyllanthus amarus | Leaves | 65–99 | Cubic | [95] |
| Mangifera indica | Leaves | 17–20 | Spherical | [96] |
| Stevia rebaudiana | Leaves | 8–20 | Octahedral | [97] |
| Nyctanthes arbortristis | Flower extract | 19.8 | Spherical, hexagonal | [79] |
| Trigonella foneum-graecum | Leaves | 15–25 | Spherical | [75] |
| Tanacetum vulgare | Fruit | 11 | Triangular | [66] |
| Cuminum cyminum | Seeds | 1–10 | Spherical | [98] |
| Sorbus aucuparia | Leaf extract | 16–18 | Spherical, triangular, hexagonal | [99] |
2.4. Zinc Nanoparticles
Zinc oxide (ZnO) is an inorganic metal oxide with a vast range of nanostructures. Zinc nanoparticles (ZnNPs) have gained considerable attention due to their low cost, large surface area, white appearance, UV-filtering, antifungal, antibacterial and photochemical properties, and high catalytic activity [100][101]. There are several reports of ZnO nanoparticle synthesis using various plant extracts [102][103][104][105]. Plant extracts contain some phytochemicals (i.e., polyphenols, saponins, terpenoids) that act as reducing and stabilizing agents in the reaction system. Phytochemicals are synthesized in the plant parts, including root, stem, leaf, fruit and seed. These phytochemicals lower the metal’s valence to zero, then calcinate it to add oxide. Additionally, zinc ions interact with the polyphenols in the plant extract to form a complex. After that, zinc hydroxide (Zn(OH)2) is formed via hydrolysis, and then ZnO nanoparticles are synthesized after complex calculations [106].
During the literature survey, it was observed that members of the Fabaceae, Rutaceae, Apocynaceae, Solanaceae and Lamiaceae families are most commonly employed for the production of ZnNPs (Table 3). Plants from the family Lamiaceae, such as Anisochilus carnosus, Plectranthus amboinicus and Vitex negundo were used to produce ZnO nanoparticles of different sizes and shapes, including hexagonal, spherical, quasi-spherical and rod-shaped particles. The findings indicated that the particle sizes decrease when plant extract concentration increases [107][108]. All experiments displayed nanoparticles in the same size range with spherical and hexagonal disc shapes, which XRD and TEM analysis characterized. Singh et al. [109] synthesized ZnO NPs using Calotropis procera latex that were spherical and 5 nm to 40 nm in size. Ramesh et al. [110] used the floral extract of Cassia auriculata to react with Zn(NO3)2 solution resulting in the development of ZnNPs with a particle size ranging from 110 nm to 280 nm. Some reports on the plant-assisted synthesis of zinc nanoparticles are listed below in Table 3.
Table 3. Plant-assisted synthesis of zinc nanoparticles.
| Plant Name | Parts Used | Size (nm) | Shapes | Reference |
|---|---|---|---|---|
| Artemisia pallens | Leaves along with stem | 50–100 | Hexagonal | [105] |
| Cayratia pedata | Leaves | 52.24 | Spherical | [111] |
| Euphorbia hirta | Leaves | 20–50 | Spherical | [112] |
| Eucalyptus globules | Leaves | 52–70 | Spherical, elongated | [104] |
| Tecoma castanifolia | Leaves | 70–75 | Spherical | [113] |
| Zingiber officinale | Root | 30–50 | Spherical | [114] |
| Azadirachta indica | Leaves | 50 | Spindle shaped | [115] |
| Catharanthus roseus | Leaves | 23–57 | Spherical | [116] |
| Solanum nigrum | Leaves | 20–30 | Hexagonal | [117] |
| Olea europea | Leaves | 18–30 | Crystalline | [118] |
| Azadirachta indica | Leaves | 25 | Crystalline | [119] |
| Nyctanthes arbor-tristis | Flowers | 12–32 | Crystalline | [120] |
| Hibiscus rosa-sinensis | Leaves | 30–35 | Crystal, spongy | [121] |
| Ruta graveolens | Stem | 28 | Spherical | [102] |
| Aloe vera | Leaves | 22.18 | Hexagonal | [122] |
| Ocimum tenuiflorum | Leaves | 11–25 | Hexagonal | [123] |
| Sargassum muticum | Leaves | 30–57 | Hexagonal | [124] |
| Calotropis gigantea | Leaves | 1.5–8.5 | Spherical | [103] |
| Beta vulgaris | Root | 52–76 | Hexagonal | [125] |
| Curcuma longa | Root | 20–80 | Hexagonal | [126] |
| Nephelium lappaceum | Peel | 20 | Spherical | [127] |
| Artocarpus gomezianus | Fruit | 50 | Spherical | [128] |
| Senna auriculata | Leaves | 2 | Spherical | [129] |
| Brassica oleraceae | Leaves | 1–100 | Spherical and sheet shaped | [130] |
| Acalypha Indica | Leaves | 100–200 | Cube | [131] |
| Plectranthus amboinicus | Leaves | 20–50 | Crystalline | [132] |
| Coptidis rhizome | Rhizome | 2.9–25.2 | Spherical and rod shaped | [133] |
| Ginger | Rhizome | 23–26 | Crystalline | [134] |
2.5. Titanium Nanoparticles
Titanium dioxide nanoparticles (TiNPs) have drawn great attention because of their appropriate electrical band structure, high specific surface area and quantum efficacy, stability, and chemical innerness [135]. TiNPs have a wide applicability in lowering the toxicity of synthetic dyes [136] and pharmaceutical medicines [137], wastewater treatment [138], etc. The synthesis of TiO2 nanoparticles on a wide scale using biological methods has stimulated the interest of researchers due to its low cost, environmental friendliness and reproducibility. Nowadays, there are many reports on the biosynthesis of TiO2 nanoparticles by using microbes (such as bacteria and fungi), algae, plant parts and enzymes. The aqueous extract of Eclipta prostrata produce nanoparticles with a spherical shape and sizes ranging from 36 nm to 68 nm, confirmed by XRD and TEM analysis [139]. Subhashini and Nachiyar [140] used the leaf extract of Albizia saman for the production of titanium NPs via a green route. The aqueous TiO2 solution was added dropwise into the leaf extract with stirring at 50 °C resulting in the formation of anatase crystals of TiO2 nanoparticles. The synthesized TiO2 nanoparticles were found to be 41 nm in size and confirmed by XRD analysis. Jalill et al. [141] synthesized the anatase form of TiO2 nanoparticles by using the plant extract of Curcuma longa (because of its terpenoid and flavonoid contents). The nanoparticles that were developed were identified by the techniques of XRD, FTIR, SEM and EDX that revealed the aggregated, circular structure and a particle size of 160–220 nm. TiNPs were synthesized by the utilization of herbal extract (as a bio-reductant) of Echinacea purpurea [142]. The particle size of the synthesized TiO2 nanoparticles was found to be in the 120 nm range. The leaf extract of Psidium guajava includes alcohol and primary and aromatic amines, which aid in producing TiO2 nanoparticles. Some reports on the plant-assisted synthesis of titanium nanoparticles are listed below in Table 4.
Table 4. Plant-assisted synthesis of titanium nanoparticles.
| Plant Name | Parts Used | Size (nm) | Shapes | Reference |
|---|---|---|---|---|
| Ledebouria revoluta | Bulb | 47 | Tetragonal | [143] |
| Pouteria campechiana | Leaves | 73–140 | Spherical | [144] |
| Syzygium cumini | Leaves | 22 | Spherical round | [145] |
| Mentha arvensis | Leaves | 20–70 | Spherical | [146] |
| Azadirachta indica | Leaves | 15–50 | Spherical | [147] |
| Psidium guajava | Leaves | 32.58 | Spherical | [148] |
| Nyctanthes arbor-tristis | Leaves | 100–150, 100–200 | Cubic, crystalline, Spherical | [149] |
| Calotropis gigantea | Flower | 10–52 | Crystalline, Spherical oval | [150] |
| Salvia officinalis | Leaves | 15–20 | Spherical | [136] |
| Solanum trilobatum | Leaves | 70 | Spherical, oval | [151] |
| Azadirachta indica | Leaves | 124 | Spherical | [152] |
| Annona squamosal | Leaves | 40–60 | Spherical | [153] |
| Jatropha curcas, citrus aurantium | Leaves | 25–50 | Spherical | [154] |
| Jatropha curcas | Latex | 25–50 | Spherical, uneven | [155] |
| Euphorbia prostrata | Leaves | 81–84 | Spherical | [156] |
| Citrus sinensis | Fruit peel | 19 | Tetragonal | [157] |
| Cassia auriculata | Leaves | 38 | Spherical | [158] |
| Ocimum basilicum | Leaves | 50 | Hexagonal | [159] |
| Hibiscus-rosa-sinensis | Petals | 7–24 | Spherical | [12] |
| Erythrina variegates | Leaves | 39 | Crystalline, spherical | [160] |
2.6. Palladium Nanoparticles
The major studies of most researchers were focused on the biological synthesis of palladium nanoparticles (PdNPs) via plant materials because it is cost-effective, sustainable, and human- and eco-friendly. Plant extracts contain a number of primary and secondary metabolites that transform metal (Pd) salts to PdNPs. Siddiqi and Husen [161] reported that the shape, size and stability of PdNPs depends on concentrations of plant extract, pH, temperature and incubation time. Plant sources including the extracts of leaves, flowers, seeds, fruits, peels and roots were extensively utilized to synthesize Pd nanoparticles.
Gurunathan et al. [162] synthesized Pd nanoparticles from a plant extract of Evolvulus alsinoides. This plant extract has various natural antioxidants, including alkaloids, flavonoids, saponins, tannin, steroids and phenol, which work as reducing and capping tools to synthesize Pd nanoparticles. Nasrollahzadeh et al. [163] used the leaf extract of Hippophae rhamnoides to synthesize PdNPs because the leaf extract has polyphenols that play an important role as reducing and capping agents for nanostructure development. The formed NPs were found to be spherically shaped and ranging from 2.5 nm to 14 nm, which was confirmed by TEM. Pd nanoparticles have been synthesized from the root extract of Salvadora persica, which contains polyphenols that act as reductant and stabilizing agents [164]. The average particle size of synthesized NPs was 10 nm at 90°C, which was revealed from the UV spectrum of the colloidal solution. Palladium NPs were generated with the bark extract of Cinnamomum zeylanicum and PdCl2 solution at 30 °C [165]. Khan et al. [166] carried out the plant-assisted synthesis of PdNPs from the extract of Pulicaria glutinosa and PdCl2. After stirring the mixture of PdCl2 + extract at 90 °C for 2 h, the colour changed from pale yellow to dark brown, indicating the production of PdNPs, validated by UV–visible spectroscopy. A TEM monograph revealed the particle size of the obtained Pd nanoparticles ranged between 20 nm and 25 nm. The particle size of the synthesized NPs was found to be between 10 nm and 50 nm. The biosynthesis of Pd nanoparticles from the leafy solution of Glycine max has been reported [167]. The shape of the particles was found to be uniformly spherical with a 15 nm diameter, which was confirmed by TEM micrograph. Jia et al. [168] performed the synthesis of Pd nanoparticles utilizing Gardenia jasminoides extract containing various antioxidants such as geniposide, crocins, crocetin and chlorogenic acid, which reduce and stabilize the nanoparticles. There are some reports on plant-assisted synthesis of palladium nanoparticles listed below in Table 5.
Table 5. Plant-assisted synthesis of palladium nanoparticles.
| Plant Name | Parts Used | Size (nm) | Shapes | Reference |
|---|---|---|---|---|
| Peganum harmala | Seed | 22.5 ± 5.7 | Spherical | [169] |
| Coleus amboinicus | Leaves | 40–50 | Spherical | [170] |
| Anogeissus latifolia | Gum ghatti | 4.8 ± 1.6 | Spherical | [171] |
| Filicium decipiens | Leaves | 2–22 | Spherical | [172] |
| Cinnamomum camphora | Leaves | 3.2–6 | Multiple | [173] |
| Pulicariaglutinosa | Leaves | 3–5 | Spherical | [166] |
| Musa paradisica | Peeled banana | 50 | Crystalline | [174] |
| Cinnamom zeylanicum | Bark | 15–20 | Crystalline | [165] |
| Catharanthus roseus | Leaves | 38 | Spherical | [175] |
| Curcuma longa | Tuber | 10–15 | Spherical | [176] |
| Glycine max | Leaves | 15 | Spherical | [167] |
References
- Sergeev, G.B.; Shabatina, T.I. Cryochemistry of nanometals. Colloids Surf. A Physicochem. Eng. Asp. 2008, 313–314, 18–22.
- Duan, H.; Wang, D.; Li, Y. Green chemistry for nanoparticle synthesis. Chem. Soc. Rev. 2015, 44, 5778–5792.
- Korbekandi, H.; Iravani, S.; Abbasi, S. Production of nanoparticles using organisms Production of nanoparticles using organisms. Crit. Rev. Biotechnol. 2009, 29, 279–306.
- Luangpipat, T.; Beattie, I.R.; Chisti, Y.; Haverkamp, R.G. Gold nanoparticles produced in a microalga. J. Nanopart. Res. 2011, 13, 6439–6445.
- Arumugam, A.; Karthikeyan, C.; Haja Hameed, A.S.; Gopinath, K.; Gowri, S.; Karthika, V. Synthesis of cerium oxide nanoparticles using Gloriosa superba L. leaf extract and their structural, optical and antibacterial properties. Mater. Sci. Eng. C 2015, 49, 408–415.
- Mehata, M.S. Enhancement of Charge Transfer and Quenching of Photoluminescence of Capped CdS Quantum Dots. Sci. Rep. 2015, 5, 12056.
- Shetti, N.P.; Bukkitgar, S.D.; Reddy, K.R.; Reddy, C.V.; Aminabhavi, T.M. Nanostructured titanium oxide hybrids-based electrochemical biosensors for healthcare applications. Colloids Surf. B Biointerfaces 2019, 178, 385–394.
- Zhang, M.; Yuan, P.; Zhou, N.; Su, Y.; Shao, M.; Chi, C. pH-Sensitive N-doped carbon dots–heparin and doxorubicin drug delivery system: Preparation and anticancer research. RSC Adv. 2017, 7, 9347–9356.
- Niculescu, A.G.; Chircov, C.; Bîrcă, A.C.; Grumezescu, A.M. Nanomaterials Synthesis through Microfluidic Methods: An Updated Overview. Nanomaterials 2021, 11, 864.
- Chung, I.M.; Park, I.; Seung-Hyun, K.; Thiruvengadam, M.; Rajakumar, G. Plant-Mediated Synthesis of Silver Nanoparticles: Their Characteristic Properties and Therapeutic Applications. Nanoscale Res. Lett. 2016, 11, 1–14.
- El-Khatib, A.M.; Badawi, M.S.; Ghatass, Z.F.; Mohamed, M.M.; Elkhatib, M. Synthesize of Silver Nanoparticles by Arc Discharge Method Using Two Different Rotational Electrode Shapes. J. Clust. Sci. 2018, 29, 1169–1175.
- Kumar, P.S.M.; Francis, A.P.; Devasena, T. Biosynthesized and Chemically Synthesized Titania Nanoparticles: Comparative Analysis of Antibacterial Activity. J. Environ. Nanotechnol. 2014, 3, 73–81.
- Kumar, P.V.; Kala, S.M.J.; Prakash, K.S. Green synthesis of gold nanoparticles using Croton Caudatus Geisel leaf extract and their biological studies. Mater. Lett. 2019, 236, 19–22.
- Chen, Y.H.; Yeh, C.S. Laser ablation method: Use of surfactants to form the dispersed Ag nanoparticles. Colloids Surf. A Physicochem. Eng. Asp. 2002, 197, 133–139.
- Sen, P.; Ghosh, J.; Abdullah, A.; Kumar, P. Preparation of Cu, Ag, Fe and Al nanoparticles by the exploding wire technique. J. Chem. Sci. 2003, 115, 499–508.
- Sangar, S.; Sharma, S.; Vats, V.K.; Mehta, S.K.; Singh, K. Biosynthesis of silver nanocrystals, their kinetic profile from nucleation to growth and optical sensing of mercuric ions. J. Clean. Prod. 2019, 228, 294–302.
- Melchert, W.R.; Reis, B.F.; Rocha, F.R.P. Green chemistry and the evolution of flow analysis. A review. Anal. Chim. Acta 2012, 714, 8–19.
- Malik, P.; Shankar, R.; Malik, V.; Sharma, N.; Mukherjee, T.K. Green Chemistry Based Benign Routes for Nanoparticle Synthesis. J. Nanopart. 2014, 2014, 1–14.
- Thakkar, K.N.; Mhatre, S.S.; Parikh, R.Y. Biological synthesis of metallic nanoparticles. Nanomed. Nanotech. Biol. Med. 2010, 6, 257–262.
- Gowramma, B.; Keerthi, U.; Rafi, M.; Muralidhara Rao, D. Biogenic silver nanoparticles production and characterization from native stain of Corynebacterium species and its antimicrobial activity. 3 Biotech 2015, 5, 195–201.
- Makarov, V.V.; Love, A.J.; Sinitsyna, O.V.; Makarova, S.S.; Yaminsky, I.V.; Taliansky, M.E.; Kalinina, N.O. “Green” nanotechnologies: Synthesis of metal nanoparticles using plants. Acta Nat. 2014, 6, 35–44.
- Mukherjee, P.; Ahmad, A.; Mandal, D.; Senapati, S.; Sainkar, S.R.; Khan, M.I.; Parishcha, R.; Ajaykumar, P.V.; Alam, M.; Kumar, R.; et al. Fungus-Mediated Synthesis of Silver Nanoparticles and Their Immobilization in the Mycelial Matrix: A Novel Biological Approach to Nanoparticle Synthesis. Nano Lett. 2001, 1, 515–519.
- Wang, L.; Yao, J.; Zhang, X.; Zhang, Y.; Xu, C.; Lee, R.; Yu, G.; Yu, B.; Teng, L. Delivery of paclitaxel using nanoparticles composed of poly (ethylene oxide)-b-poly (butylene oxide) (PEO-PBO). Colloids Surf. B Biointerfaces 2018, 161, 464–470.
- Niculescu, A.G.; Grumezescu, A.M. Polymer-Based Nanosystems—A Versatile Delivery Approach. Materials 2021, 14, 6812.
- Ahirwar, D.; Bano, M.; Khan, I.; Gound, S.S.; Ud Din Sheik, M.; Mondal, R.; Khan, F. Facile synthesis of macroporous Ag and CuO monoliths as an efficient nonenzymatic electrochemical sensor and antimicrobial agent. J. Solid State Chem. 2019, 273, 233–242.
- Zare, M.; Namratha, K.; Alghamdi, S.; Mohammad, Y.H.E.; Hezam, A.; Zare, M.; Drmosh, Q.A.; Byrappa, K.; Nanjegowda, B.; Chandrashekar, S.R.; et al. Novel green biomimetic approach for synthesis of ZnO-Ag nanocomposite; antimicrobial activity against food-borne pathogen, biocompatibility and solar. Sci. Rep. 2019, 9, 8303.
- Nadagouda, M.N.; Varma, R.S. Green synthesis of silver and palladium nanoparticles at room temperature using coffee and tea extract. Green Chem. 2008, 10, 859–886.
- Elavazhagan, T.; Arunachalam, K.D. Memecylon edule leaf extract mediated green synthesis of silver and gold nanoparticles. Int. J. Nanomed. 2011, 6, 1265–1278.
- Lee, Y.W.; Kim, M.; Kim, Z.H.; Han, S.W. One-step synthesis of core-shell nanooctahedron. J. Am. Chem. Soc. 2009, 131, 17036–17037.
- Zhang, G.; Liu, Z.; Xiao, Z.; Huang, J.; Li, Q.; Wang, Y.; Sun, D. Ni2P-graphite nanoplatelets supported Au-Pd core-shell nanoparticles with superior electrochemical properties. J. Phys. Chem. C 2015, 119, 10469–10477.
- Shankar, S.S.; Ahmad, A.; Sastry, M. Geranium Leaf Assisted Biosynthesis of Silver Nanoparticles. Biotechnol. Prog. 2003, 19, 1627–1631.
- Ankamwar, B.; Chaudhary, M.; Sastry, M. Gold Nanotriangles Biologically Synthesized using Tamarind Leaf Extract and Potential Application in Vapor Sensing. Synth. React. Inorg. Met. Nano-Metal Chem. 2005, 35, 19–26.
- Gardea-Torresdey, J.L.; Gomez, E.; Peralta-Videa, J.R.; Parsons, J.G.; Troiani, H.; Jose-Yacaman, M. Alfalfa sprouts: A natural source for the synthesis of silver nanoparticles. Langmuir 2003, 19, 1357–1361.
- Xin Lee, K.; Shameli, K.; Miyake, M.; Kuwano, N.; Bt Ahmad Khairudin, N.B.; Bt Mohamad, S.E.; Yew, Y.P. Green Synthesis of Gold Nanoparticles Using Aqueous Extract of Garcinia mangostana Fruit Peels. J. Nanomater. 2016, 2016, 7.
- Davids, J.S.; Ackah, M.; Okoampah, E.; Fometu, S.S.; Guohua, W.; Jianping, Z. Biocontrol of Bacteria Associated with Pine Wilt Nematode, Bursaphelenchus xylophilus by using Plant mediated Gold Nanoparticles. Int. J. Agric. Biol. 2021, 26, 517–526.
- Jiang, H.; Manolache, S.; Wong, A.C.L.; Denes, F.S. Plasma-enhanced deposition of silver nanoparticles onto polymer and metal surfaces for the generation of antimicrobial characteristics. J. Appl. Polym. Sci. 2004, 93, 1411–1422.
- Thirunavoukkarasu, M.; Balaji, U.; Behera, S.; Panda, P.K.; Mishra, B.K. Biosynthesis of silver nanoparticle from leaf extract of Desmodium gangeticum (L.) DC. and its biomedical potential. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2013, 116, 424–427.
- Parlinska-Wojtan, M.; Kus-Liskiewicz, M.; Depciuch, J.; Sadik, O. Green synthesis and antibacterial effects of aqueous colloidal solutions of silver nanoparticles using camomile terpenoids as a combined reducing and capping agent. Bioprocess Biosyst. Eng. 2016, 39, 1213–1223.
- Rodríguez-León, E.; Iñiguez-Palomares, R.; Navarro, R.E.; Herrera-Urbina, R.; Tánori, J.; Iñiguez-Palomares, C.; Maldonado, A. Synthesis of silver nanoparticles using reducing agents obtained from natural sources (Rumex hymenosepalus extracts). Nanoscale Res. Lett. 2013, 8, 1–9.
- Gopinath, V.; MubarakAli, D.; Priyadarshini, S.; Priyadharsshini, N.M.; Thajuddin, N.; Velusamy, P. Biosynthesis of silver nanoparticles from Tribulus terrestris and its antimicrobial activity: A novel biological approach. Colloids Surf. B Biointerfaces 2012, 96, 69–74.
- Sharifi-Rad, M.; Pohl, P.; Epifano, F.; Álvarez-Suarez, J.M. Green Synthesis of Silver Nanoparticles Using Astragalus tribuloides Delile. Root Extract: Characterization, Antioxidant, Antibacterial, and Anti-Inflammatory Activities. Nanomaterials 2020, 10, 2383.
- Jha, A.K.; Prasad, K. Green Synthesis of Silver Nanoparticles Using Cycas Leaf. Int. J. Green Nanotechnol. Phys. Chem. 2010, 1, 110–117.
- Sathishkumar, M.; Sneha, K.; Yun, Y.S. Immobilization of silver nanoparticles synthesized using Curcuma longa tuber powder and extract on cotton cloth for bactericidal activity. Bioresour. Technol. 2010, 101, 7958–7965.
- Kumar, V.; Yadav, S.K. Synthesis of stable, polyshaped silver, and gold nanoparticles using leaf extract of Lonicera japonica L. Int. J. Green Nanotechnol. Biomed. 2011, 3, 281–291.
- Banerjee, J.; Narendhirakannan, R.T. Biosynthesis of silver nanoparticles from Syzygium cumini (L.) seed extract and evaluation of their in vitro antioxidant activities. Dig. J. Nanomater. Biostruct. 2011, 6, 961–968.
- Patil, C.D.; Patil, S.V.; Borase, H.P.; Salunke, B.K.; Salunkhe, R.B. Larvicidal activity of silver nanoparticles synthesized using Plumeria rubra plant latex against Aedes aegypti and Anopheles stephensi. Parasitol. Res. 2012, 110, 1815–1822.
- Ponarulselvam, S.; Panneerselvam, C.; Murugan, K.; Aarthi, N.; Kalimuthu, K.; Thangamani, S. Synthesis of silver nanoparticles using leaves of Catharanthus roseus Linn. G. Don and their antiplasmodial activities. Asian Pac. J. Trop. Biomed. 2012, 2, 574–580.
- Sathishkumar, M.; Sneha, K.; Won, S.W.; Cho, C.W.; Kim, S.; Yun, Y.S. Cinnamon zeylanicum bark extract and powder mediated green synthesis of nano-crystalline silver particles and its bactericidal activity. Colloids Surf. B Biointerfaces 2009, 73, 332–338.
- Harshiny, M.; Matheswaran, M.; Arthanareeswaran, G.; Kumaran, S.; Rajasree, S. Enhancement of antibacterial properties of silver nanoparticles-ceftriaxone conjugate through Mukia maderaspatana leaf extract mediated synthesis. Ecotoxicol. Environ. Saf. 2015, 121, 135–141.
- Anandalakshmi, K.; Venugobal, J.; Ramasamy, V. Characterization of silver nanoparticles by green synthesis method using Pedalium murex leaf extract and their antibacterial activity. Appl. Nanosci. 2016, 6, 399–408.
- Raju, D.; Paneliya, N.; Mehta, U.J. Extracellular synthesis of silver nanoparticles using living peanut seedling. Appl. Nanosci. 2014, 4, 875–879.
- Morales-Lozoya, V.; Espinoza-Gómez, H.; Flores-López, L.Z.; Sotelo-Barrera, E.L.; Núñez-Rivera, A.; Cadena-Nava, R.D.; Alonso-Nuñez, G.; Rivero, I.A. Study of the effect of the different parts of Morinda citrifolia L. (noni) on the green synthesis of silver nanoparticles and their antibacterial activity. Appl. Surf. Sci. 2021, 537, 147855.
- Gudimalla, A.; Jose, J.; Varghese, R.J.; Thomas, S. Green Synthesis of Silver Nanoparticles Using Nymphae odorata Extract Incorporated Films and Antimicrobial Activity. J. Polym. Environ. 2021, 29, 1412–1423.
- Nilavukkarasi, M.; Vijayakumar, S.; Prathip Kumar, S. Biological synthesis and characterization of silver nanoparticles with Capparis zeylanica L. leaf extract for potent antimicrobial and anti proliferation efficiency. Mater. Sci. Energy Technol. 2020, 3, 371–376.
- Moteriya, P.; Chanda, S. Green Synthesis of Silver Nanoparticles from Caesalpinia pulcherrima Leaf Extract and Evaluation of Their Antimicrobial, Cytotoxic and Genotoxic Potential (3-in-1 System). J. Inorg. Organomet. Polym. Mater. 2020, 30, 3920–3932.
- Dalir, S.J.B.; Djahaniani, H.; Nabati, F.; Hekmati, M. Characterization and the evaluation of antimicrobial activities of silver nanoparticles biosynthesized from Carya illinoinensis leaf extract. Heliyon 2020, 6, e03624.
- Khatoon, A.; Khan, F.; Ahmad, N.; Shaikh, S.; Rizvi, S.M.D.; Shakil, S.; Al-Qahtani, M.H.; Abuzenadah, A.M.; Tabrez, S.; Ahmed, A.B.F.; et al. Silver nanoparticles from leaf extract of Mentha piperita: Eco-friendly synthesis and effect on acetylcholinesterase activity. Life Sci. 2018, 209, 430–434.
- Bar, H.; Bhui, D.K.; Sahoo, G.P.; Sarkar, P.; De, S.P.; Misra, A. Green synthesis of silver nanoparticles using latex of Jatropha curcas. Colloids Surf. A Physicochem. Eng. Asp. 2009, 339, 134–139.
- Krishnaraj, C.; Jagan, E.G.; Rajasekar, S.; Selvakumar, P.; Kalaichelvan, P.T.; Mohan, N. Synthesis of silver nanoparticles using Acalypha indica leaf extracts and its antibacterial activity against water borne pathogens. Colloids Surf. B Biointerfaces 2010, 76, 50–56.
- Philip, D. Green synthesis of gold and silver nanoparticles using Hibiscus rosa sinensis. Phys. E Low-Dimens. Syst. Nanostruct. 2010, 42, 1417–1424.
- Awwad, A.M.; Salem, N.M.; Abdeen, A.O. Green synthesis of silver nanoparticles using carob leaf extract and its antibacterial activity. Int. J. Ind. Chem. 2013, 4, 29.
- Satyavani, K.; Gurudeeban, S.; Ramanathan, T.; Balasubramanian, T. Toxicity study of silver nanoparticles synthesized from Suaeda monoica on Hep-2 cell line. Avicenna J. Med. Biotechnol. 2012, 4, 35–39.
- Philip, D.; Unni, C. Extracellular biosynthesis of gold and silver nanoparticles using Krishna tulsi (Ocimum sanctum) leaf. Phys. E Low-Dimens. Syst. Nanostruct. 2011, 43, 1318–1322.
- Patil, R.S.; Kokate, M.R.; Kolekar, S.S. Bioinspired synthesis of highly stabilized silver nanoparticles using Ocimum tenuiflorum leaf extract and their antibacterial activity. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2012, 91, 234–238.
- Song, J.Y.; Kim, B.S. Rapid biological synthesis of silver nanoparticles using plant leaf extracts. Bioprocess Biosyst. Eng. 2009, 32, 79–84.
- Dubey, S.P.; Lahtinen, M.; Sillanpää, M. Tansy fruit mediated greener synthesis of silver and gold nanoparticles. Process Biochem. 2010, 45, 1065–1071.
- Singh, A.; Jain, D.; Upadhyay, M.K.; Khandelwal, N.; Verma, H.N. Green synthesis of silver nanoparticles using Argemone mexicana leaf extract and evaluation of their antimicrobial activities. Dig. J. Nanomater. Biostruct. 2010, 5, 483–489.
- Nabikhan, A.; Kandasamy, K.; Raj, A.; Alikunhi, N.M. Synthesis of antimicrobial silver nanoparticles by callus and leaf extracts from saltmarsh plant, Sesuvium portulacastrum L. Colloids Surf. B Biointerfaces 2010, 79, 488–493.
- Kumar, V.; Yadav, S.C.; Yadav, S.K. Syzygium cumini leaf and seed extract mediated biosynthesis of silver nanoparticles and their characterization. J. Chem. Technol. Biotechnol. 2010, 85, 1301–1309.
- Huang, J.; Li, Q.; Sun, D.; Lu, Y.; Su, Y.; Yang, X.; Wang, H.; Wang, Y.; Shao, W.; He, N.; et al. Biosynthesis of silver and gold nanoparticles by novel sundried Cinnamomum camphora leaf. Nanotechnology 2007, 18, 105104.
- Sukirtha, R.; Priyanka, K.M.; Antony, J.J.; Kamalakkannan, S.; Thangam, R.; Gunasekaran, P.; Krishnan, M.; Achiraman, S. Cytotoxic effect of Green synthesized silver nanoparticles using Melia azedarach against in vitro HeLa cell lines and lymphoma mice model. Process Biochem. 2012, 47, 273–279.
- Mittal, A.K.; Kaler, A.; Banerjee, U.C. Free radical scavenging and antioxidant activity of silver nanoparticles synthesized from flower extract of Rhododendron dauricum. Nano Biomed. Eng. 2012, 4, 118–124.
- Cruz, D.; Falé, P.L.; Mourato, A.; Vaz, P.D.; Luisa Serralheiro, M.; Lino, A.R.L. Preparation and physicochemical characterization of Ag nanoparticles biosynthesized by Lippia citriodora (Lemon Verbena). Colloids Surf. B Biointerfaces 2010, 81, 67–73.
- Satyavani, K.; Gurudeeban, S.; Ramanathan, T.; Balasubramanian, T. Biomedical potential of silver nanoparticles synthesized from calli cells of Citrullus colocynthis (L.) Schrad. J. Nanobiotechnol. 2011, 9, 1–8.
- Aromal, S.A.; Philip, D. Green synthesis of gold nanoparticles using Trigonella foenum-graecum and its size-dependent catalytic activity. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2012, 97, 1–5.
- Singh, A.K.; Talat, M.; Singh, D.P.; Srivastava, O.N. Biosynthesis of gold and silver nanoparticles by natural precursor clove and their functionalization with amine group. J. Nanopart. Res. 2010, 12, 1667–1675.
- Sharma, R.K.; Gulati, S.; Mehta, S. Preparation of gold nanoparticles using tea: A green chemistry experiment. J. Chem. Educ. 2012, 89, 1316–1318.
- Suman, T.Y.; Radhika Rajasree, S.R.; Ramkumar, R.; Rajthilak, C.; Perumal, P. The Green synthesis of gold nanoparticles using an aqueous root extract of Morinda citrifolia L. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2014, 118, 11–16.
- Das, R.K.; Gogoi, N.; Bora, U. Green synthesis of gold nanoparticles using Nyctanthes arbortristis flower extract. Bioprocess Biosyst. Eng. 2011, 34, 615–619.
- Jha, A.K.; Prasad, K. Biosynthesis of gold nanoparticles using bael (Aegle marmelos) leaf: Mythology meets technology. Int. J. Green Nanotechnol. Biomed. 2011, 3, 92–97.
- Rodríguez-León, E.; Rodríguez-Vázquez, B.E.; Martínez-Higuera, A.; Rodríguez-Beas, C.; Larios-Rodríguez, E.; Navarro, R.E.; López-Esparza, R.; Iñiguez-Palomares, R.A. Synthesis of Gold Nanoparticles Using Mimosa tenuiflora Extract, Assessments of Cytotoxicity, Cellular Uptake, and Catalysis. Nanoscale Res. Lett. 2019, 14, 334.
- Anuradha, J.; Tasneem, A.; Abbasi, S.A. “Green” Synthesis of Gold Nanoparticles with Aqueous Extracts of Neem (Azadirachta indica). Res. J. Biotechnol. 2010, 5, 75–79.
- Kasthuri, J.; Kathiravan, K.; Rajendiran, N. Phyllanthin-assisted biosynthesis of silver and gold nanoparticles: A novel biological approach. J. Nanopart. Res. 2009, 11, 1075–1085.
- Aromal, S.A.; Philip, D. Benincasa hispida seed mediated green synthesis of gold nanoparticles and its optical nonlinearity. Phys. E Low-Dimens. Syst. Nanostruct. 2012, 44, 1329–1334.
- Muniyappan, N.; Pandeeswaran, M.; Amalraj, A. Green synthesis of gold nanoparticles using Curcuma pseudomontana isolated curcumin: Its characterization, antimicrobial, antioxidant and anti-inflammatory activities. Environ. Chem. Ecotoxicol. 2021, 3, 117–124.
- Kumari, P.; Meena, A. Green synthesis of gold nanoparticles from Lawsoniainermis and its catalytic activities following the Langmuir-Hinshelwood mechanism. Colloids Surf. A Physicochem. Eng. Asp. 2020, 606, 125447.
- ElMitwalli, O.S.; Barakat, O.A.; Daoud, R.M.; Akhtar, S.; Henari, F.Z. Green synthesis of gold nanoparticles using cinnamon bark extract, characterization, and fluorescence activity in Au/eosin Y assemblies. J. Nanopart. Res. 2020, 22, 1–9.
- Chandran, S.P.; Chaudhary, M.; Pasricha, R.; Ahmad, A.; Sastry, M. Synthesis of gold nanotriangles and silver nanoparticles using Aloe vera plant extract. Biotechnol. Prog. 2006, 22, 577–583.
- Jha, A.K.; Prasad, K. Biosynthesis of gold nanoparticles using common aromatic plants. Int. J. Green Nanotechnol. Biomed. 2012, 4, 219–224.
- Shukla, D.; Vankar, P.S. Synthesis of plant parts mediated gold nanoparticles. Int. J. Green Nanotechnol. Biomed. 2012, 4, 277–288.
- Philip, D.; Unni, C.; Aromal, S.A.; Vidhu, V.K. Murraya Koenigii leaf-assisted rapid green synthesis of silver and gold nanoparticles. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2011, 78, 899–904.
- Noruzi, M.; Zare, D.; Khoshnevisan, K.; Davoodi, D. Rapid green synthesis of gold nanoparticles using Rosa hybrida petal extract at room temperature. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2011, 79, 1461–1465.
- Kumar, K.M.; Mandal, B.K.; Sinha, M.; Krishnakumar, V. Terminalia chebula mediated green and rapid synthesis of gold nanoparticles. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2012, 86, 490–494.
- Pandey, S.; Oza, G.; Mewada, A.; Sharon, M. Green Synthesis of Highly Stable Gold Nanoparticles using Momordica charantia as Nano fabricator. Arch. Appl. Sci. Res. 2012, 4, 1135–1141.
- Annamalai, A.; Babu, S.T.; Jose, N.A.; Sudha, D.; Lyza, C.V. Biosynthesis and characterization of silver and gold nanoparticles using aqueous leaf extraction of Phyllanthus amarus Schum. & Thonn. World Appl. Sci. J. 2011, 13, 1833–1840.
- Philip, D. Rapid green synthesis of spherical gold nanoparticles using Mangifera indica leaf. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2010, 77, 807–810.
- Mishra, A.N.; Bhadauria, S.; Gaur, M.S.; Pasricha, R.; Kushwah, B.S. Synthesis of Gold Nanoparticles by Leaves of Zero-Calorie Sweetener Herb ( Stevia rebaudiana ) and Their Nanoscopic Characterization by Spectroscopy and Microscopy. Int. J. Green Nanotechnol. Phys. Chem. 2010, 1, 118–124.
- Sneha, K.; Sathishkumar, M.; Lee, S.Y.; Bae, M.A.; Yun, Y.S. Biosynthesis of Au nanoparticles using cumin seed powder extract. J. Nanosci. Nanotechnol. 2011, 11, 1811–1814.
- Dubey, S.P.; Lahtinen, M.; Särkkä, H.; Sillanpää, M. Bioprospective of Sorbus aucuparia leaf extract in development of silver and gold nanocolloids. Colloids Surf. B Biointerfaces 2010, 80, 26–33.
- Kajbafvala, A.; Ghorbani, H.; Paravar, A.; Samberg, J.P.; Kajbafvala, E.; Sadrnezhaad, S.K. Effects of morphology on photocatalytic performance of Zinc oxide nanostructures synthesized by rapid microwave irradiation methods. Superlattices Microstruct. 2012, 51, 512–522.
- Kumar, S.S.; Venkateswarlu, P.; Rao, V.R.; Rao, G.N. Synthesis, characterization and optical properties of zinc oxide nanoparticles. Int. Nano Lett. 2013, 3, 1–6.
- Lingaraju, K.; Raja Naika, H.; Manjunath, K.; Basavaraj, R.B.; Nagabhushana, H.; Nagaraju, G.; Suresh, D. Biogenic synthesis of zinc oxide nanoparticles using Ruta graveolens (L.) and their antibacterial and antioxidant activities. Appl. Nanosci. 2016, 6, 703–710.
- Chaudhuri, S.K.; Malodia, L. Biosynthesis of zinc oxide nanoparticles using leaf extract of calotropis gigantea: Characterization and its evaluation on tree seedling growth in nursery stage. Appl. Nanosci. 2017, 7, 501–512.
- Ahmad, H.; Venugopal, K.; Rajagopal, K.; De Britto, S.; Nandini, B.; Pushpalatha, H.G.; Konappa, N.; Udayashankar, A.C.; Geetha, N.; Jogaiah, S. Green synthesis and characterization of zinc oxide nanoparticles using eucalyptus globules and their fungicidal ability against pathogenic fungi of apple orchards. Biomolecules 2020, 10, 425.
- Gomathi, R.; Suhana, H. Green synthesis, characterization and antimicrobial activity of zinc oxide nanoparticles using Artemisia pallens plant extract. Inorg. Nano-Metal Chem. 2021, 51, 1663–1672.
- Bala, N.; Saha, S.; Chakraborty, M.; Maiti, M.; Das, S.; Basu, R.; Nandy, P. Green synthesis of zinc oxide nanoparticles using Hibiscus subdariffa leaf extract: Effect of temperature on synthesis, anti-bacterial activity and anti-diabetic activity. RSC Adv. 2015, 5, 4993–5003.
- Ambika, S.; Sundrarajan, M. Antibacterial behaviour of Vitex negundo extract assisted ZnO nanoparticles against pathogenic bacteria. J. Photochem. Photobiol. B Biol. 2015, 146, 52–57.
- Anbuvannan, M.; Ramesh, M.; Viruthagiri, G.; Shanmugam, N.; Kannadasan, N. Anisochilus carnosus leaf extract mediated synthesis of zinc oxide nanoparticles for antibacterial and photocatalytic activities. Mater. Sci. Semicond. Process. 2015, 39, 621–628.
- Singh, R.P.; Shukla, V.K.; Yadav, R.S.; Sharma, P.K.; Singh, P.K.; Pandey, A.C. Biological approach of zinc oxide nanoparticles formation and its characterization. Adv. Mater. Lett. 2011, 2, 313–317.
- Ramesh, P.; Rajendran, A.; Sundaram, M. Green Synthesis of Zinc Oxide Nanoparticles Using Flower Extract Cassia Auriculata. J. Nanosci. Nanotechnol. 2014, 2, 41–45.
- Jayachandran, A.; Aswathy, T.R.; Nair, A.S. Green synthesis and characterization of zinc oxide nanoparticles using Cayratia pedata leaf extract. Biochem. Biophys. Rep. 2021, 26, 100995.
- Ahmad, W.; Kalra, D. Green synthesis, characterization and anti-microbial activities of ZnO nanoparticles using Euphorbia hirta leaf extract. J. King Saud Univ.-Sci. 2020, 32, 2358–2364.
- Sharmila, G.; Thirumarimurugan, M.; Muthukumaran, C. Green synthesis of ZnO nanoparticles using Tecoma castanifolia leaf extract: Characterization and evaluation of its antioxidant, bactericidal and anticancer activities. Microchem. J. 2019, 145, 578–587.
- Raj, L.F.A.; Jayalakshmy, E. Biosynthesis and characterization of zinc oxide nanoparticles using root extract of Zingiber officinale. Orient. J. Chem. 2015, 31, 51–56.
- Noorjahan, C.M.; Shahina, S.K.J.; Deepika, T.; Rafiq, S. Green Synthesis and Characterization of Zinc Oxide Nanoparticles from Neem (Azadirachta indicia). Int. J. Sci. Eng. Technol. Res. 2015, 4, 5751–5753.
- Savithramma, N.; Bhumi, G. Biological Synthesis of Zinc oxide Nanoparticles from C atharanthus roseus (l.) G. Don. Leaf extract and validation for antibacterial activity. Int. J. Drug Dev. Res. 2014, 6, 208–214.
- Ramesh, M.; Anbuvannan, M.; Viruthagiri, G. Green synthesis of ZnO nanoparticles using Solanum nigrum leaf extract and their antibacterial activity. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2015, 136, 864–870.
- Awwad, A.M.; Albiss, B.; Ahmad, A.L. Green synthesis, characterization and optical properties of zinc oxide nanosheets using Olea europea leaf extract. Adv. Mater. Lett. 2014, 5, 520–524.
- Oudhia, A.; Kulkarni, P.; Sharma, S. Green Synthesisof ZnO nanotubes for Bioapplications. J. Adv. Eng. Res. Stud. 2015, 280–281.
- Jamdagni, P.; Khatri, P.; Rana, J.S. Green synthesis of zinc oxide nanoparticles using flower extract of Nyctanthes arbor-tristis and their antifungal activity. J. King Saud Univ.-Sci. 2018, 30, 168–175.
- Devi, R.; Gayathri, R. Green Synthesis of Zinc Oxide Nanoparticles by using Hibiscus rosa-sinensis. Int. J. Curr. Eng. Technol. 2014, 44, 2444–2446.
- Varghese, E.; George, M. Green synthesis of zinc oxide nanoparticles. Int. J. Adv. Res. Sci. Eng. 2015, 4, 307–314.
- Raut, S.; Thorat, P.V.; Thakre, R. Green Synthesis of Zinc Oxide (ZnO) Nanoparticles Using Ocimum Tenuiflorum Leaves. Int. J. Sci. Res. 2013, 4, 2319–7064.
- Azizi, S.; Ahmad, M.B.; Namvar, F.; Mohamad, R. Green biosynthesis and characterization of zinc oxide nanoparticles using brown marine macroalga Sargassum muticum aqueous extract. Mater. Lett. 2014, 116, 275–277.
- Kumar, M.A.P.; Suresh, D.; Nagabhushana, H.; Sharma, S.C. Beta vulgaris aided green synthesis of ZnO nanoparticles and their luminescence, photocatalytic and antioxidant properties. Eur. Phys. J. Plus 2015, 130, 1–7.
- Fatimah, I.; Yudha, S.P.; Mutiara, N.A.L. Green synthesis of ZnO nanoparticles via complex formation by using Curcuma longa extract. Proceedings of 6th Nanoscience and Nanotechnology Symposium (NNS2015), Sukarta, Indonesia, 4–5 November 2015; Purwano, A., Nur, A., Rahmawati, F., Dyartanti, E.R., Jumari, A., Eds.; AIP Publishing LLC: Melville, NY, USA, 2015.
- Yuvakkumar, R.; Suresh, J.; Hong, S.I. Green synthesis of zinc oxide nanoparticles. Adv. Mater. Res. 2014, 952, 137–140.
- Suresh, D.; Shobharani, R.M.; Nethravathi, P.C.; Pavan Kumar, M.A.; Nagabhushana, H.; Sharma, S.C. Artocarpus gomezianus aided green synthesis of ZnO nanoparticles: Luminescence, photocatalytic and antioxidant properties. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2015, 141, 128–134.
- Sindhura, K.S.; Tnvkv, P.; Selvam, P.P.; Hussain, O.M. Green Synthesis of Zinc Nanoparticles from Senna Auriculata and Influence on Peanut Pot-Culture. Int. J. Res. Agric. Sci. 2015, 2, 61–69.
- Raj, A.; Lawrence, R.S.; Jalees, M.; Lawrence, K. Anti-Bacterial Activity of Zinc Oxide Nanoparticles Prepared from Brassica Oleraceae Leaves Extract. Int. J. Adv. Res. 2015, 3, 322–328.
- Gnanasangeetha, D.; Thambavani, D. Biological and Physical Sciences Biogenic Production of Zinc Oxide Nanoparticles Using Acalypha Indica. J. Chem. Biol. Phys. Sci. 2014, 4, 238–246.
- Vijayakumar, S.; Vinoj, G.; Malaikozhundan, B.; Shanthi, S.; Vaseeharan, B. Plectranthus amboinicus leaf extract mediated synthesis of zinc oxide nanoparticles and its control of methicillin resistant Staphylococcus aureus biofilm and blood sucking mosquito larvae. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2015, 137, 886–891.
- Nagajyothi, P.C.; Sreekanth, T.V.M.; Tettey, C.O.; Jun, Y.I.; Mook, S.H. Characterization, antibacterial, antioxidant, and cytotoxic activities of ZnO nanoparticles using Coptidis Rhizoma. Bioorganic Med. Chem. Lett. 2014, 24, 4298–4303.
- Janaki, A.C.; Sailatha, E.; Gunasekaran, S. Synthesis, characteristics and antimicrobial activity of ZnO nanoparticles. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2015, 144, 17–22.
- Lai, Y.; Wang, L.; Liu, D.; Chen, Z.; Lin, C. TiO2-Based Nanomaterials: Design, Synthesis, and Applications. J. Nanomater. 2015, 2015, 1–3.
- Altikatoglu Yapaoz, M.; Attar, A. Salvia officinalis-derived rutile TiO2NPs: Production, characterization, antibacterial evaluation and its effect on decolorization. Mater. Res. Express 2019, 6, 55039.
- Mahmoud, W.M.M.; Rastogi, T.; Kümmerer, K. Application of titanium dioxide nanoparticles as a photocatalyst for the removal of micropollutants such as pharmaceuticals from water. Curr. Opin. Green Sustain. Chem. 2017, 6, 1–10.
- Abel, S.; Jule, L.T.; Belay, F.; Shanmugam, R.; Dwarampudi, L.P.; Nagaprasad, N.; Krishnaraj, R. Application of Titanium Dioxide Nanoparticles Synthesized by Sol-Gel Methods in Wastewater Treatment. J. Nanomater. 2021, 2021, 3039761.
- Rajakumar, G.; Rahuman, A.A.; Priyamvada, B.; Khanna, V.G.; Kumar, D.K.; Sujin, P.J. Eclipta prostrata leaf aqueous extract mediated synthesis of titanium dioxide nanoparticles. Mater. Lett. 2012, 68, 115–117.
- Subhashini, D.; Nachiyar, C.V. Albizia saman: A Green Route for the Reduction of Bulk TiO2. Int. J. Chem Tech. Res. 2014, 6, 5137–5141.
- Jalill, R.D.A.; Nuaman, R.S.; Abd, A.N. Biological synthesis of Titanium Dioxide nanoparticles by Curcuma longa plant extract and study its biological properties. World Sci. News 2016, 49, 204–222.
- Dobrucka, R. Synthesis of titanium dioxide nanoparticles using Echinacea purpurea herba. Iran. J. Pharm. Res. 2017, 16, 753–759.
- Aswini, R.; Murugesan, S.; Kannan, K. Bio-engineered TiO2 nanoparticles using Ledebouria revoluta extract: Larvicidal, histopathological, antibacterial and anticancer activity. Int. J. Environ. Anal. Chem. 2021, 101, 2926–2936.
- Narayanan, M.; Devi, P.G.; Natarajan, D.; Kandasamy, S.; Devarayan, K.; Alsehli, M.; Elfasakhany, A.; Pugazhendhi, A. Green synthesis and characterization of titanium dioxide nanoparticles using leaf extract of Pouteria campechiana and larvicidal and pupicidal activity on Aedes aegypti. Environ. Res. 2021, 200, 111333.
- Sethy, N.K.; Arif, Z.; Mishra, P.K.; Kumar, P. Green synthesis of TiO2 nanoparticles from Syzygium cumini extract for photo-catalytic removal of lead (Pb) in explosive industrial wastewater. Green Process. Synth. 2020, 9, 171–181.
- Ahmad, W.; Jaiswal, K.K.; Soni, S. Green synthesis of titanium dioxide (TiO2) nanoparticles by using Mentha arvensis leaves extract and its antimicrobial properties. Inorg. Nano-Metal Chem. 2020, 50, 1032–1038.
- Thakur, B.K.; Kumar, A.; Kumar, D. Green synthesis of titanium dioxide nanoparticles using Azadirachta indica leaf extract and evaluation of their antibacterial activity. S. Afr. J. Bot. 2019, 124, 223–227.
- Santhoshkumar, T.; Rahuman, A.A.; Jayaseelan, C.; Rajakumar, G.; Marimuthu, S.; Kirthi, A.V.; Velayutham, K.; Thomas, J.; Venkatesan, J.; Kim, S.K. Green synthesis of titanium dioxide nanoparticles using Psidium guajava extract and its antibacterial and antioxidant properties. Asian Pac. J. Trop. Med. 2014, 7, 968–976.
- Sundrarajan, M.; Gowri, S. Green synthesis of titanium dioxide nanoparticles by nyctanthes arbor-tristis leaves extract. Chalcogenide Lett. 2011, 8, 447–451.
- Marimuthu, S.; Rahuman, A.A.; Jayaseelan, C.; Kirthi, A.V.; Santhoshkumar, T.; Velayutham, K.; Bagavan, A.; Kamaraj, C.; Elango, G.; Iyappan, M.; et al. Acaricidal activity of synthesized titanium dioxide nanoparticles using Calotropis gigantea against Rhipicephalus microplus and Haemaphysalis bispinosa. Asian Pac. J. Trop. Med. 2013, 6, 682–688.
- Rajakumar, G.; Rahuman, A.A.; Jayaseelan, C.; Santhoshkumar, T.; Marimuthu, S.; Kamaraj, C.; Bagavan, A.; Zahir, A.A.; Kirthi, A.V.; Elango, G.; et al. Solanum trilobatum extract-mediated synthesis of titanium dioxide nanoparticles to control Pediculus humanus capitis, Hyalomma anatolicum anatolicum and Anopheles subpictus. Parasitol. Res. 2014, 113, 469–479.
- Sankar, R.; Rizwana, K.; Shivashangari, K.S.; Ravikumar, V. Ultra-rapid photocatalytic activity of Azadirachta indica engineered colloidal titanium dioxide nanoparticles. Appl. Nanosci. 2015, 5, 731–736.
- Velayutham, K.; Rahuman, A.A.; Rajakumar, G.; Santhoshkumar, T.; Marimuthu, S.; Jayaseelan, C.; Bagavan, A.; Kirthi, A.V.; Kamaraj, C.; Zahir, A.A.; et al. Evaluation of Catharanthus roseus leaf extract-mediated biosynthesis of titanium dioxide nanoparticles against Hippobosca maculata and Bovicola ovis. Parasitol. Res. 2012, 111, 2329–2337.
- Nwanya, A.C.; Ugwuoke, P.E.; Ejikeme, P.M.; Oparaku, O.U.; Ezema, F.I. Jathropha curcas and citrus aurantium leaves dye extract for use in dye sensitized solar cell with TiO2 films. Int. J. Electrochem. Sci. 2012, 7, 11219–11235.
- Hudlikar, M.; Joglekar, S.; Dhaygude, M.; Kodam, K. Green synthesis of TiO2 nanoparticles by using aqueous extract of Jatropha curcas L. latex. Mater. Lett. 2012, 75, 196–199.
- Zahir, A.A.; Chauhan, I.S.; Bagavan, A.; Kamaraj, C.; Elango, G.; Shankar, J.; Arjaria, N.; Roopan, S.M.; Rahuman, A.A.; Singh, N. Green synthesis of silver and titanium dioxide nanoparticles using Euphorbia prostrata extract shows shift from apoptosis to G0/G1 arrest followed by necrotic cell death in Leishmania donovani. Antimicrob. Agents Chemother. 2015, 59, 4782–4799.
- Rao, K.G.; Ashok, C.; Rao, K.V.; Chakra, C.S.; Rajendar, V. Synthesis of TiO2 nanoparticles from orange fruit waste. Int. J. Multidiscip. Adv. Res. Trends 2015, 2, 82–90.
- Valli, D.G.; Geetha, S. A green method for the synthesis of titanium dioxide nanoparticles using Cassia auriculata leaves extract. Eur. J. Biomed. Pharm. Sci. 2015, 2, 490–497.
- Salam, H.A.; Sivaraj, R. Ocimum basilicum L. var. purpurascens Benth.-Lamiaceae Mediated Green Synthesis and Characterization of Titanium Dioxide Nanoparticles. Adv. Biores. 2014, 5, 10–16.
- Valli, G.; Jayalakshmi, A. Erythrina variegate leaves extract assisted synthesis of titanium dioxide nanoparticles in an eco-friendly approach. Eur. J. Biomed. Pharm. Sci. 2015, 2, 1228–1236.
- Siddiqi, K.S.; Husen, A. Green Synthesis, Characterization and Uses of Palladium/Platinum Nanoparticles. Nanoscale Res. Lett. 2016, 11, 482.
- Gurunathan, S.; Kim, E.S.; Han, J.W.; Park, J.H.; Kim, J.-H.H.; Grumezescu, A.M. Green chemistry approach for synthesis of effective anticancer palladium nanoparticles. Molecules 2015, 20, 22476–22498.
- Nasrollahzadeh, M.; Sajadi, S.M.; Maham, M. Green synthesis of palladium nanoparticles using Hippophae rhamnoides Linn leaf extract and their catalytic activity for the Suzuki-Miyaura coupling in water. J. Mol. Catal. A Chem. 2015, 396, 297–303.
- Khan, M.; Albalawi, G.H.; Shaik, M.R.; Khan, M.; Adil, S.F.; Kuniyil, M.; Alkhathlan, H.Z.; Al-Warthan, A.; Siddiqui, M.R.H. Miswak mediated green synthesized palladium nanoparticles as effective catalysts for the Suzuki coupling reactions in aqueous media. J. Saudi Chem. Soc. 2017, 21, 450–457.
- Sathishkumar, M.; Sneha, K.; Kwak, I.S.; Mao, J.; Tripathy, S.J.; Yun, Y.S. Phyto-crystallization of palladium through reduction process using Cinnamom zeylanicum bark extract. J. Hazard. Mater. 2009, 171, 400–404.
- Khan, M.M.; Khan, M.M.; Kuniyil, M.; Adil, S.F.; Al-Warthan, A.; Alkhathlan, H.Z.; Tremel, W.; Tahir, M.N.; Siddiqui, M.R.H. Biogenic synthesis of palladium nanoparticles using Pulicaria glutinosa extract and their catalytic activity towards the Suzuki coupling reaction. Dalt. Trans. 2014, 43, 9026–9031.
- Kumar Petla, R.; Vivekanandhan, S.; Misra, M.; Kumar Mohanty, A.; Satyanarayana, N. Soybean (Glycine Max) Leaf Extract Based Green Synthesis of Palladium Nanoparticles. J. Biomater. Nanobiotechnol. 2012, 3, 14–19.
- Jia, L.; Zhang, Q.; Li, Q.; Song, H. The biosynthesis of palladium nanoparticles by antioxidants in Gardenia jasminoides Ellis: Long lifetime nanocatalysts for p-nitrotoluene hydrogenation. Nanotechnology 2009, 20, 385601–385611.
- Fahmy, S.A.; Fawzy, I.M.; Saleh, B.M.; Issa, M.Y.; Bakowsky, U.; Azzazy, H.M.E.S. Green synthesis of platinum and palladium nanoparticles using Peganum harmala L. Seed alkaloids: Biological and computational studies. Nanomaterials 2021, 11, 965.
- Bathula, C.; Subalakshmi, K.; Kumar, A.; Yadav, H.; Ramesh, S.; Shinde, S.; Shrestha, N.K.; Mallikarjuna, K.; Kim, H. Ultrasonically driven green synthesis of palladium nanoparticles by Coleus amboinicus for catalytic reduction and Suzuki-Miyaura reaction. Colloids Surf. B Biointerfaces 2020, 192, 111026.
- Kora, A.J.; Rastogi, L. Green synthesis of palladium nanoparticles using gum ghatti (Anogeissus latifolia) and its application as an antioxidant and catalyst. Arab. J. Chem. 2018, 11, 1097–1106.
- Sharmila, G.; Farzana Fathima, M.; Haries, S.; Geetha, S.; Manoj Kumar, N.; Muthukumaran, C. Green synthesis, characterization and antibacterial efficacy of palladium nanoparticles synthesized using Filicium decipiens leaf extract. J. Mol. Struct. 2017, 1138, 35–40.
- Yang, X.; Li, Q.; Wang, H.; Huang, J.; Lin, L.; Wang, W.; Sun, D.; Su, Y.; Opiyo, J.B.; Hong, L.; et al. Green synthesis of palladium nanoparticles using broth of Cinnamomum camphora leaf. J. Nanopart. Res. 2010, 12, 1589–1598.
- Bankar, A.; Joshi, B.; Kumar, A.R.; Zinjarde, S. Banana peel extract mediated novel route for the synthesis of palladium nanoparticles. Mater. Lett. 2010, 64, 1951–1953.
- Kalaiselvi, A.; Roopan, S.M.; Madhumitha, G.; Ramalingam, C.; Elango, G. Synthesis and characterization of palladium nanoparticles using Catharanthus roseus leaf extract and its application in the photo-catalytic degradation. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2015, 135, 116–119.
- Sathishkumar, M.; Sneha, K.; Yun, Y. Palladium Nanocrystal Synthesis Using Curcuma longa Tuber Extract. Int. J. Mater. Sci. 2009, 4, 11–17.
More
Information
Subjects:
Plant Sciences; Nanoscience & Nanotechnology
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
6.5K
Revisions:
4 times
(View History)
Update Date:
02 Mar 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





