
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Wenge Zheng | -- | 1854 | 2022-11-22 07:31:48 | | | |
| 2 | Wenge Zheng | + 83 word(s) | 3840 | 2022-11-22 07:50:14 | | | | |
| 3 | Jessie Wu | + 15 word(s) | 3855 | 2022-11-24 01:40:52 | | | | |
| 4 | Jessie Wu | + 4 word(s) | 3859 | 2022-11-24 01:42:53 | | | | |
| 5 | Jessie Wu | Meta information modification | 3859 | 2022-11-24 01:44:14 | | | | |
| 6 | Jessie Wu | Meta information modification | 3859 | 2022-11-24 01:44:50 | | | | |
| 7 | Jessie Wu | Meta information modification | 3859 | 2022-11-24 07:09:22 | | |
Video Upload Options
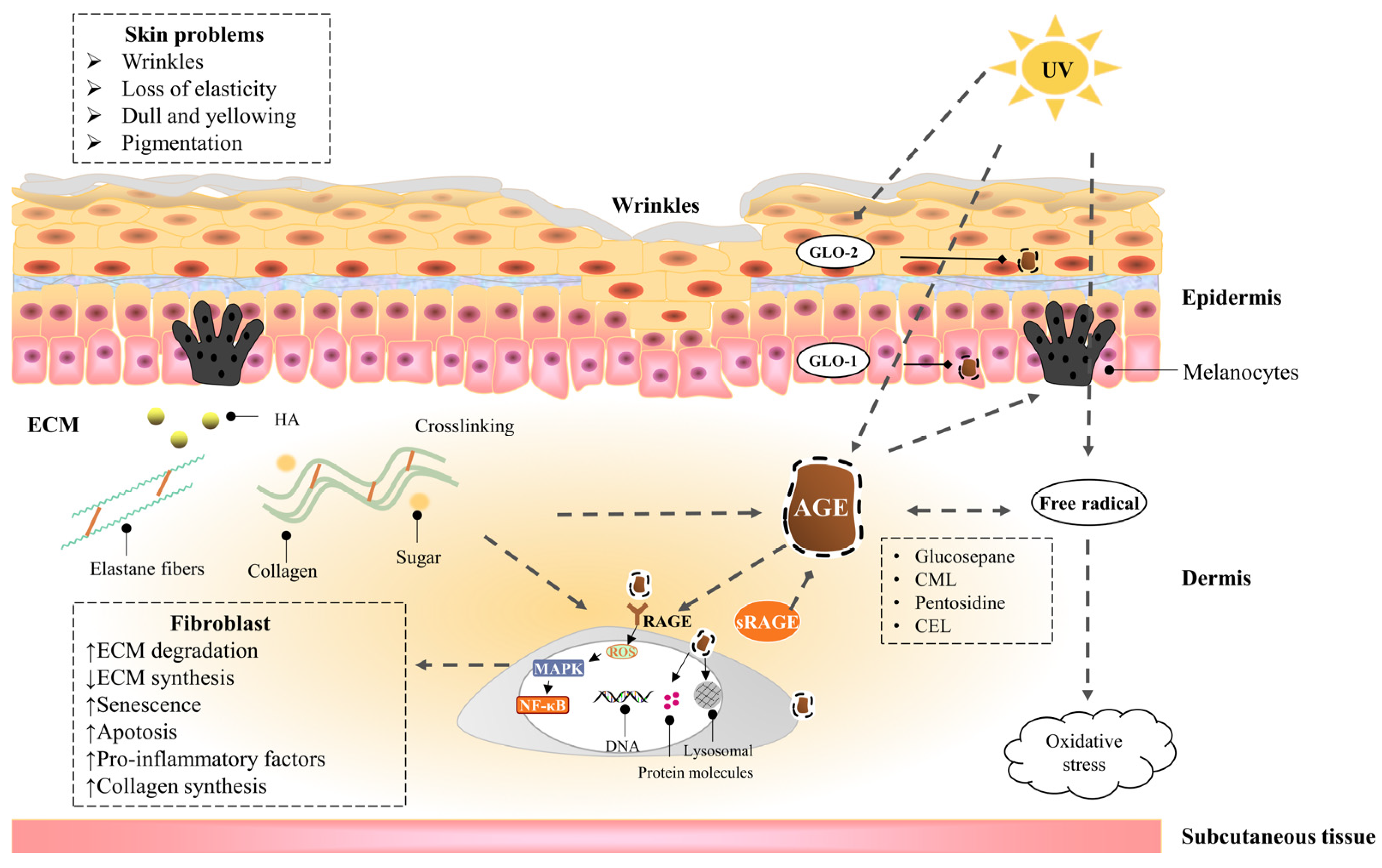
Skin saccharification, a non-enzymatic reaction between proteins, e.g., dermal collagen and naturally occurring reducing sugars, is one of the basic root causes of endogenous skin aging. During the reaction, a series of complicated glycation products produced at different reaction stages and pathways are usually collectively referred to as advanced glycation end products (AGEs). AGEs cause cellular dysfunction through the modification of intracellular molecules and accumulate in tissues with aging. AGEs are also associated with a variety of age-related diseases, such as diabetes, cardiovascular disease, renal failure (uremia), and Alzheimer’s disease. AGEs accumulate in the skin with age and are amplified through exogenous factors, e.g., ultraviolet radiation, resulting in wrinkles, loss of elasticity, dull yellowing, and other skin problems.
1. The Hazards of Skin Glycation
1.1. The Harm of High Glucose to the Skin
1.2. Advanced Glycaion End Products Induce Skin Aging
1.2.1. Epidermis
1.2.2. Dermis—Fibroblast
1.2.3. Dermis—Extracellular matrix (ECM)
1.3. UVA Induces Advanced Glycation End Products of the Skin
2. Inhibitors of Advanced Glycation End Products
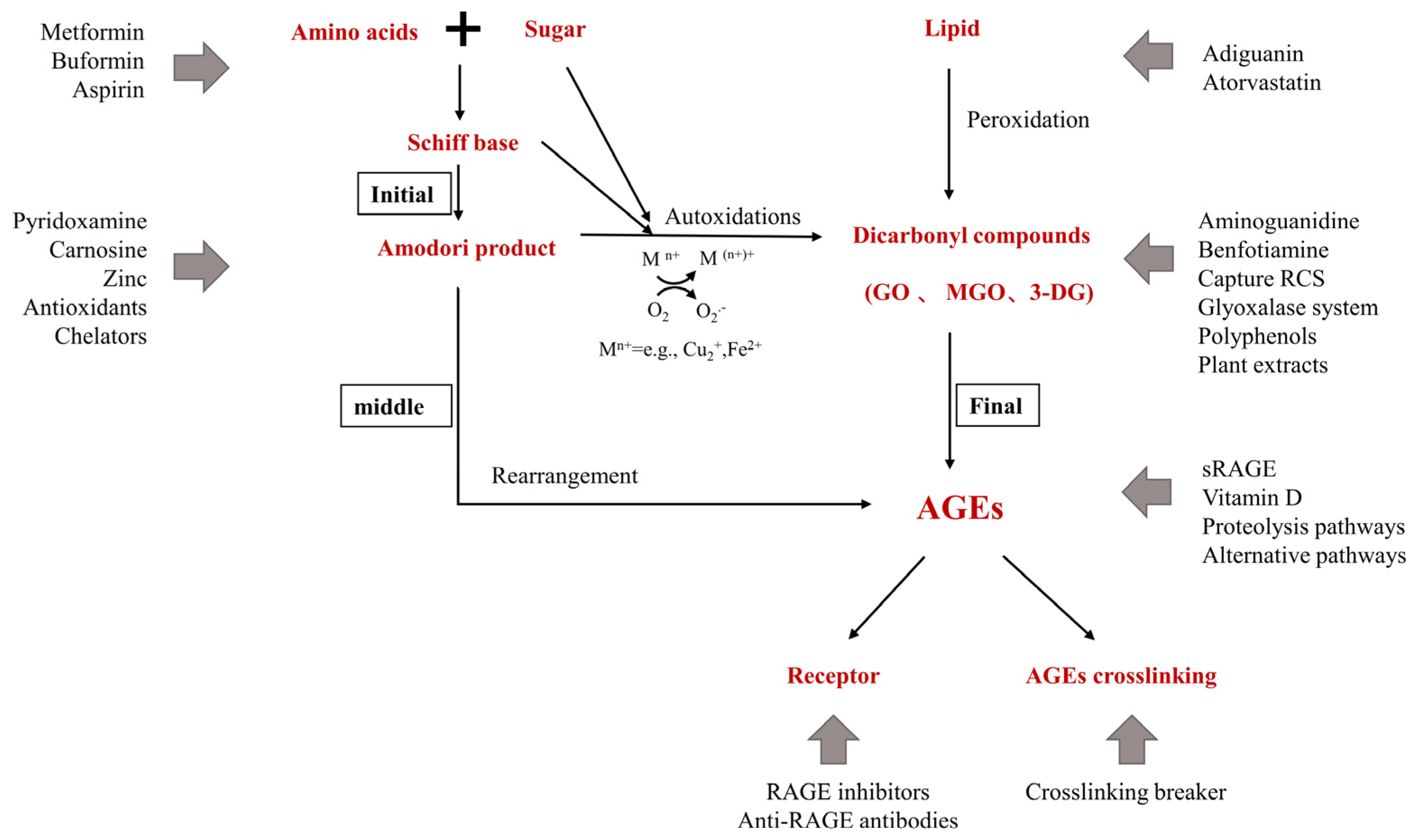
2.1. Pre-Amadori Inhibitors
2.2. Post-Amadori Inhibitors
2.3. Crosslinking Breaker
2.4. Indirect Advanced Glycation End Products Inhibitors
2.5. Natural Advanced Glycation End Products Inhibitors
2.6. Polyphenolic Compounds
2.7. Other Advanced Glycation End Products Inhibitors
References
- Cao, C.; Xiao, Z.; Wu, Y.; Ge, C. Diet and Skin Aging-From the Perspective of Food Nutrition. Nutrients 2020, 12, 870.
- Liu, X.; Yang, C.; Deng, Y.; Liu, P.; Yang, H.; Du, X.; Du, Y. Polygoni Multiflori Radix Preparat Delays Skin Aging by Inducing Mitophagy. BioMed Res. Int. 2021, 2021, 1–12.
- Gasser, P.; Arnold, F.; Peno-Mazzarino, L.; Bouzoud, D.; Luu, M.T.; Lati, E.; Mercier, M. Glycation Induction and Antiglycation Activity of Skin Care Ingredients On Living Human Skin Explants. Int. J. Cosmet. Sci. 2011, 33, 366–370.
- Danby, F.W. Nutrition and Aging Skin: Sugar and Glycation. Clin. Dermatol. 2010, 28, 409–411.
- Cibrian, D.; de la Fuente, H.; Sánchez-Madrid, F. Metabolic Pathways that Control Skin Homeostasis and Inflammation. Trends Mol. Med. 2020, 26, 975–986.
- Devarakonda, K.; Mobbs, C.V. Mechanisms and Significance of Brain Glucose Signaling in Energy Balance, Glucose Homeostasis, and Food-Induced Reward. Mol. Cell. Endocrinol. 2016, 438, 61–69.
- Quondamatteo, F. Skin and Diabetes Mellitus: What Do we Know? Cell Tissue Res. 2014, 355, 1–21.
- Argyropoulos, A.J.; Robichaud, P.; Balimunkwe, R.M.; Fisher, G.J.; Hammerberg, C.; Yan, Y.; Quan, T. Alterations of Dermal Connective Tissue Collagen in Diabetes: Molecular Basis of Aged-Appearing Skin. PLoS ONE 2016, 11, e153806.
- Mentink, C.J.; Hendriks, M.; Levels, A.A.; Wolffenbuttel, B.H. Glucose-Mediated Cross-Linking of Collagen in Rat Tendon and Skin. Clin. Chim. Acta 2002, 321, 69–76.
- Van Putte, L.; De Schrijver, S.; Moortgat, P. The Effects of Advanced Glycation End Products (Ages) On Dermal Wound Healing and Scar Formation: A Systematic Review. Scars Burn. Heal. 2016, 2, 1011166500.
- Shu, F.; Gao, H.; Wu, W.; Yu, S.; Zhang, L.; Liu, H.; Xiao, S.; Xia, Z.; Zheng, Y. Amniotic Epithelial Cells Accelerate Diabetic Wound Healing by Protecting Keratinocytes and Fibroblasts From High-Glucose-Induced Senescence. Cell Biol. Int. 2022, 46, 755–770.
- Sruthi, C.R.; Raghu, K.G. Advanced Glycation End Products and their Adverse Effects: The Role of Autophagy. J. Biochem. Mol. Toxicol. 2021, 35, e22710.
- Khalifah, R.G.; Baynes, J.W.; Hudson, B.G. Amadorins: Novel Post-Amadori Inhibitors of Advanced Glycation Reactions. Biochem. Biophys. Res. Commun. 1999, 257, 251–258.
- Osawa, T.; Kato, Y. Protective Role of Antioxidative Food Factors in Oxidative Stress Caused by Hyperglycemia. Ann. N. Y. Acad. Sci. 2005, 1043, 440–451.
- Yevdokimova, N.Y. High Glucose-Induced Alterations of Extracellular Matrix of Human Skin Fibroblasts are Not Dependent On Tsp-1-Tgfbeta1 Pathway. J. Diabetes Complicat. 2003, 17, 355–364.
- Bian, X.; Li, B.; Yang, J.; Ma, K.; Sun, M.; Zhang, C.; Fu, X. Regenerative and Protective Effects of Dmsc-Sevs On High-Glucose-Induced Senescent Fibroblasts by Suppressing Rage Pathway and Activating Smad Pathway. Stem Cell Res. Ther. 2020, 11, 166.
- Li, B.; Bian, X.; Hu, W.; Wang, X.; Li, Q.; Wang, F.; Sun, M.; Ma, K.; Zhang, C.; Chang, J.; et al. Regenerative and Protective Effects of Calcium Silicate On Senescent Fibroblasts Induced by High Glucose. Wound Repair Regen. 2020, 28, 315–325.
- Buranasin, P.; Mizutani, K.; Iwasaki, K.; Pawaputanon, N.M.C.; Kido, D.; Takeda, K.; Izumi, Y. High Glucose-Induced Oxidative Stress Impairs Proliferation and Migration of Human Gingival Fibroblasts. PLoS ONE 2018, 13, e201855.
- Liu, J.; Wu, Y.; Wang, B.; Yuan, X.; Fang, B. High Levels of Glucose Induced the Caspase-3/Parp Signaling Pathway, Leading to Apoptosis in Human Periodontal Ligament Fibroblasts. Cell Biophys. 2013, 66, 229–237.
- Soydas, T.; Sayitoglu, M.; Sarac, E.Y.; Cinar, S.; Solakoglu, S.; Tiryaki, T.; Sultuybek, G.K. Metformin Reverses the Effects of High Glucose On Human Dermal Fibroblasts of Aged Skin via Downregulating Rela/P65 Expression. J. Physiol. Biochem. 2021, 77, 443–450.
- Wang, X.; Jin, H.; Jiang, S.; Xu, Y. Microrna-495 Inhibits the High Glucose-Induced Inflammation, Differentiation and Extracellular Matrix Accumulation of Cardiac Fibroblasts through Downregulation of Nod1. Cell. Mol. Biol. Lett. 2018, 23, 23.
- Yue, E.; Yu, Y.; Wang, X.; Liu, B.; Bai, Y.; Yang, B. Anthocyanin Protects Cardiac Function and Cardiac Fibroblasts From High-Glucose Induced Inflammation and Myocardial Fibrosis by Inhibiting Il-17. Front. Pharmacol. 2020, 11, 593633.
- Chiu, H.C.; Fu, M.M.; Yang, T.S.; Fu, E.; Chiang, C.Y.; Tu, H.P.; Chin, Y.T.; Lin, F.G.; Shih, K.C. Effect of High Glucose, Porphyromonas Gingivalis Lipopolysaccharide and Advanced Glycation End-Products On Production of Interleukin-6/-8 by Gingival Fibroblasts. J. Periodont. Res. 2017, 52, 268–276.
- Lee, J.; Jeong, E.T.; Lim, J.M.; Park, S.G. Development of the Facial Glycation Imaging System for in Situ Human Face Skin Glycation Index Measurement. J. Cosmet. Dermatol. 2021, 20, 2963–2968.
- Nguyen, H.P.; Katta, R. Sugar Sag: Glycation and the Role of Diet in Aging Skin. Ski. Ther. Lett. 2015, 20, 1–5.
- Da, M.S.C.; Webb, M.; Waller, H.; Khunti, K.; Davies, M. Skin Autofluorescence, a Non-Invasive Marker of Advanced Glycation End Products: Clinical Relevance and Limitations. Postgrad. Med. J. 2017, 93, 289–294.
- Smit, A.J.; van de Zande, S.C.; Mulder, D.J. Skin Autofluorescence as Tool for Cardiovascular and Diabetes Risk Prediction. Curr. Opin. Nephrol. Hypertens. 2022, 31, 522–526.
- André, A.; Touré, A.K.; Stien, D.; Eparvier, V. 2,5-Diketopiperazines Mitigate the Amount of Advanced Glycation End Products Accumulated with Age in Human Dermal Fibroblasts. Int. J. Cosmet. Sci. 2020, 42, 596–604.
- Low, E.; Alimohammadiha, G.; Smith, L.A.; Costello, L.F.; Przyborski, S.A.; von Zglinicki, T.; Miwa, S. How Good is the Evidence that Cellular Senescence Causes Skin Ageing? Ageing Res. Rev. 2021, 71, 101456.
- D’Errico, M.; Lemma, T.; Calcagnile, A.; Proietti, D.S.L.; Dogliotti, E. Cell Type and Dna Damage Specific Response of Human Skin Cells to Environmental Agents. Mutat. Res. Mol. Mech. Mutagen. 2007, 614, 37–47.
- Iwamura, M.; Yamamoto, Y.; Kitayama, Y.; Higuchi, K.; Fujimura, T.; Hase, T.; Yamamoto, H. Epidermal Expression of Receptor for Advanced Glycation End Products (Rage) is Related to Inflammation and Apoptosis in Human Skin. Exp. Dermatol. 2016, 25, 235–237.
- Zhu, P.; Ren, M.; Yang, C.; Hu, Y.X.; Ran, J.M.; Yan, L. Involvement of Rage, Mapk and Nf-Kappab Pathways in Ages-Induced Mmp-9 Activation in Hacat Keratinocytes. Exp. Dermatol. 2012, 21, 123–129.
- Tian, M. Effects of Advanced Glycation End-Products (Ages) On Skin Keratinocytes by Nuclear Factor-Kappa B (Nf-Κb) Activation. Afr. J. Biotechnol. 2012, 11, 11132–11142.
- Farrar, M.D. Advanced Glycation End Products in Skin Ageing and Photoageing: What are the Implications for Epidermal Function? Exp. Dermatol. 2016, 25, 947–948.
- Yumnam, S.; Subedi, L.; Kim, S.Y. Glyoxalase System in the Progression of Skin Aging and Skin Malignancies. Int. J. Mol. Sci. 2020, 22, 310.
- Reichert, O.; Fleming, T.; Neufang, G.; Schmelz, M.; Genth, H.; Kaever, V.; Wenck, H.; Stab, F.; Terstegen, L.; Kolbe, L.; et al. Impaired Glyoxalase Activity is Associated with Reduced Expression of Neurotrophic Factors and Pro-Inflammatory Processes in Diabetic Skin Cells. Exp. Dermatol. 2017, 26, 44–50.
- Pageon, H.; Bakala, H.; Monnier, V.M.; Asselineau, D. Collagen Glycation Triggers the Formation of Aged Skin in Vitro. Eur. J. Dermatol. 2007, 17, 12–20.
- Okano, Y.; Masaki, H.; Sakurai, H. Dysfunction of Dermal Fibroblasts Induced by Advanced Glycation End-Products (Ages) and the Contribution of a Nonspecific Interaction with Cell Membrane and Ages. J. Dermatol. Sci. 2002, 29, 171–180.
- Guarneri, F.; Custurone, P.; Papaianni, V.; Gangemi, S. Involvement of Rage and Oxidative Stress in Inflammatory and Infectious Skin Diseases. Antioxidants 2021, 10, 82.
- Xu, X.; Zheng, Y.; Huang, Y.; Chen, J.; Gong, Z.; Li, Y.; Lu, C.; Lai, W.; Xu, Q. Cathepsin D Contributes to the Accumulation of Advanced Glycation End Products During Photoaging. J. Dermatol. Sci. 2018, 90, 263–275.
- Arseni, L.; Lombardi, A.; Orioli, D. From Structure to Phenotype: Impact of Collagen Alterations On Human Health. Int. J. Mol. Sci. 2018, 19, 1407.
- Fournet, M.; Bonté, F.; Desmoulière, A. Glycation Damage: A Possible Hub for Major Pathophysiological Disorders and Aging. Aging Dis. 2018, 9, 880.
- Pageon, H. Reaction of Glycation and Human Skin: The Effects On the Skin and its Components, Reconstructed Skin as a Model. Pathol. Biol. 2010, 58, 226–231.
- Jaisson, S.; Gillery, P. Methods to Assess Advanced Glycation End-Products. Curr. Opin. Clin. Nutr. Metab. Care 2021, 24, 411–415.
- Sjoberg, J.S.; Bulterijs, S. Characteristics, Formation, and Pathophysiology of Glucosepane: A Major Protein Cross-Link. Rejuvenation Res. 2009, 12, 137–148.
- Pennacchi, P.C.; de Almeida, M.E.; Gomes, O.L.; Faiao-Flores, F.; de Araujo, C.M.; Dos, S.M.; de Moraes, B.S.; Maria-Engler, S.S. Glycated Reconstructed Human Skin as a Platform to Study the Pathogenesis of Skin Aging. Tissue Eng. Part A 2015, 21, 2417–2425.
- Han, A.R.; Nam, M.H.; Lee, K.W. Plantamajoside Inhibits Uvb and Advanced Glycation End Products-Induced Mmp-1 Expression by Suppressing the Mapk and Nf-Kappab Pathways in Hacat Cells. Photochem. Photobiol. 2016, 92, 708–719.
- Crisan, M.; Taulescu, M.; Crisan, D.; Cosgarea, R.; Parvu, A.; Catoi, C.; Drugan, T. Expression of Advanced Glycation End-Products On Sun-Exposed and Non-Exposed Cutaneous Sites During the Ageing Process in Humans. PLoS ONE 2013, 8, e75003.
- Okano, Y.; Masaki, H.; Sakurai, H. Pentosidine in Advanced Glycation End-Products (Ages) During Uva Irradiation Generates Active Oxygen Species and Impairs Human Dermal Fibroblasts. J. Dermatol. Sci. 2001, 27 (Suppl. 1), S11–S18.
- Zhang, S.; Duan, E. Fighting Against Skin Aging: The Way From Bench to Bedside. Cell Transplant. 2018, 27, 729–738.
- Pageon, H.; Zucchi, H.; Ricois, S.; Bastien, P.; Asselineau, D. Uva Exposure Combined with Glycation of the Dermis are Two Catalysts for Skin Aging and Promotes a Favorable Environment to the Appearance of Elastosis. J. Aging Res. 2021, 2021, 1–13.
- Lee, E.J.; Kim, J.Y.; Oh, S.H. Advanced Glycation End Products (Ages) Promote Melanogenesis through Receptor for Ages. Sci. Rep. 2016, 6, 27848.
- Sanajou, D.; Ghorbani, H.A.; Argani, H.; Aslani, S. Age-Rage Axis Blockade in Diabetic Nephropathy: Current Status and Future Directions. Eur. J. Pharmacol. 2018, 833, 158–164.
- Deane, R.; Singh, I.; Sagare, A.P.; Bell, R.D.; Ross, N.T.; LaRue, B.; Love, R.; Perry, S.; Paquette, N.; Deane, R.J.; et al. A Multimodal Rage-Specific Inhibitor Reduces Amyloid Beta-Mediated Brain Disorder in a Mouse Model of Alzheimer Disease. J. Clin. Investig. 2012, 122, 1377–1392.
- Burstein, A.H.; Sabbagh, M.; Andrews, R.; Valcarce, C.; Dunn, I.; Altstiel, L. Development of Azeliragon, an Oral Small Molecule Antagonist of the Receptor for Advanced Glycation Endproducts, for the Potential Slowing of Loss of Cognition in Mild Alzheimer’s Disease. J. Prev. Alzheimer′s Dis. 2018, 5, 149–154.
- Manigrasso, M.B.; Rabbani, P.; Egana-Gorrono, L.; Quadri, N.; Frye, L.; Zhou, B.; Reverdatto, S.; Ramirez, L.S.; Dansereau, S.; Pan, J.; et al. Small-Molecule Antagonism of the Interaction of the Rage Cytoplasmic Domain with Diaph1 Reduces Diabetic Complications in Mice. Sci. Transl. Med. 2021, 13, f7084.
- Ahmad, S.; Khan, M.S.; Alouffi, S.; Khan, S.; Khan, M.; Akashah, R.; Faisal, M.; Shahab, U. Gold Nanoparticle-Bioconjugated Aminoguanidine Inhibits Glycation Reaction: An in Vivo Study in a Diabetic Animal Model. BioMed Res. Int. 2021, 2021, 5591851.
- Degenhardt, T.P.; Fu, M.X.; Voss, E.; Reiff, K.; Neidlein, R.; Strein, K.; Thorpe, S.R.; Baynes, J.W.; Reiter, R. Aminoguanidine Inhibits Albuminuria, but Not the Formation of Advanced Glycation End-Products in Skin Collagen of Diabetic Rats. Diabetes Res. Clin. Pr. 1999, 43, 81–89.
- Gibson, G.E.; Luchsinger, J.A.; Cirio, R.; Chen, H.; Franchino-Elder, J.; Hirsch, J.A.; Bettendorff, L.; Chen, Z.; Flowers, S.A.; Gerber, L.M.; et al. Benfotiamine and Cognitive Decline in Alzheimer’s Disease: Results of a Randomized Placebo-Controlled Phase Iia Clinical Trial. J. Alzheimer′s Dis. 2020, 78, 989–1010.
- Khalifah, R.G.; Chen, Y.; Wassenberg, J.J. Post-Amadori Age Inhibition as a Therapeutic Target for Diabetic Complications: A Rational Approach to Second-Generation Amadorin Design. Ann. N. Y. Acad. Sci. 2005, 1043, 793–806.
- Adrover, M.; Vilanova, B.; Frau, J.; Munoz, F.; Donoso, J. The Pyridoxamine Action On Amadori Compounds: A Reexamination of its Scavenging Capacity and Chelating Effect. Bioorganic Med. Chem. 2008, 16, 5557–5569.
- Ramis, R.; Ortega-Castro, J.; Caballero, C.; Casasnovas, R.; Cerrillo, A.; Vilanova, B.; Adrover, M.; Frau, J. How Does Pyridoxamine Inhibit the Formation of Advanced Glycation End Products? The Role of its Primary Antioxidant Activity. Antioxidants 2019, 8, 344.
- Voziyan, P.A.; Hudson, B.G. Pyridoxamine: The Many Virtues of a Maillard Reaction Inhibitor. Ann. New York Acad. Sci. 2005, 1043, 807–816.
- Sourris, K.C.; Watson, A.; Jandeleit-Dahm, K. Inhibitors of Advanced Glycation End Product (Age) Formation and Accumulation. Handb. Exp. Pharmacol. 2021, 264, 395–423.
- Lunceford, N.; Gugliucci, A. Ilex Paraguariensis Extracts Inhibit Age Formation More Efficiently than Green Tea. Fitoterapia 2005, 76, 419–427.
- Reddy, V.P.; Beyaz, A. Inhibitors of the Maillard Reaction and Age Breakers as Therapeutics for Multiple Diseases. Drug Discov. Today 2006, 11, 646–654.
- Yagi, M.; Inoue, K.; Sato, Y.; Ishizaki, K.; Sakiyama, C.; Yonei, Y. Antiglycative Effect of Black Galangal, Kaempferia Parviflora Wall. Ex. Baker (Zingiberaceae). Glycative Stress Res. 2021, 8, 1–7.
- Peng, X.; Ma, J.; Chen, F.; Wang, M. Naturally Occurring Inhibitors Against the Formation of Advanced Glycation End-Products. Food Funct. 2011, 2, 289–301.
- Yamagishi, S.; Nakamura, K.; Matsui, T.; Inoue, H. A Novel Pleiotropic Effect of Atorvastatin On Advanced Glycation End Product (Age)-Related Disorders. Med. Hypotheses 2007, 69, 338–340.
- Ishibashi, Y.; Matsui, T.; Takeuchi, M.; Yamagishi, S. Metformin Inhibits Advanced Glycation End Products (Ages)-Induced Renal Tubular Cell Injury by Suppressing Reactive Oxygen Species Generation Via Reducing Receptor for Ages (Rage) Expression. Horm. Metab. Res. 2012, 44, 891–895.
- Kiho, T.; Kato, M.; Usui, S.; Hirano, K. Effect of Buformin and Metformin On Formation of Advanced Glycation End Products by Methylglyoxal. Clin. Chim. Acta 2005, 358, 139–145.
- Mesias, M.; Navarro, M.; Gokmen, V.; Morales, F.J. Antiglycative Effect of Fruit and Vegetable Seed Extracts: Inhibition of Age Formation and Carbonyl-Trapping Abilities. J. Sci. Food Agric. 2013, 93, 2037–2044.
- Yousof, A.M.; Jannat, S.; Mizanur, R.M. Ginsenoside Derivatives Inhibit Advanced Glycation End-Product Formation and Glucose-Fructose Mediated Protein Glycation in Vitro Via a Specific Structure-Activity Relationship. Bioorganic Chem. 2021, 111, 104844.
- Shin, S.; Lee, J.A.; Kim, M.; Kum, H.; Jung, E.; Park, D. Anti-Glycation Activities of Phenolic Constituents From Silybum Marianum (Milk Thistle) Flower in Vitro and On Human Explants. Molecules 2015, 20, 3549–3564.
- Chayaratanasin, P.; Adisakwattana, S.; Thilavech, T. Protective Role of Clitoria Ternatea L. Flower Extract On Methylglyoxal-Induced Protein Glycation and Oxidative Damage to Dna. Bmc Complement. Med. Ther. 2021, 21, 80.
- Fernandes, A.; Vieira, N.C.; Santana, A.L.; Gandra, R.; Rubia, C.; Castro-Gamboa, I.; Macedo, J.A.; Macedo, G.A. Peanut Skin Polyphenols Inhibit Toxicity Induced by Advanced Glycation End-Products in Raw264.7 Macrophages. Food Chem. Toxicol. 2020, 145, 111619.
- Ramful, D.; Tarnus, E.; Rondeau, P.; Da, S.C.; Bahorun, T.; Bourdon, E. Citrus Fruit Extracts Reduce Advanced Glycation End Products (Ages)- And H2O2-Induced Oxidative Stress in Human Adipocytes. J. Agric. Food Chem. 2010, 58, 11119–11129.
- Shin, S.; Son, D.; Kim, M.; Lee, S.; Roh, K.B.; Ryu, D.; Lee, J.; Jung, E.; Park, D. Ameliorating Effect of Akebia Quinata Fruit Extracts On Skin Aging Induced by Advanced Glycation End Products. Nutrients 2015, 7, 9337–9352.
- Suantawee, T.; Wesarachanon, K.; Anantsuphasak, K.; Daenphetploy, T.; Thien-Ngern, S.; Thilavech, T.; Pasukamonset, P.; Ngamukote, S.; Adisakwattana, S. Protein Glycation Inhibitory Activity and Antioxidant Capacity of Clove Extract. J. Food Sci. Technol. 2015, 52, 3843–3850.
- Spagnuolo, L.; Della, P.S.; Fanali, C.; Dugo, L.; De Gara, L. Antioxidant and Antiglycation Effects of Polyphenol Compounds Extracted From Hazelnut Skin On Advanced Glycation End-Products (Ages) Formation. Antioxidants 2021, 10, 424.
- Khanam, A.; Ahmad, S.; Husain, A.; Rehman, S.; Farooqui, A.; Yusuf, M.A. Glycation and Antioxidants: Hand in the Glove of Antiglycation and Natural Antioxidants. Curr. Protein Pept. Sci. 2020, 21, 899–915.
- Peng, X.; Cheng, K.W.; Ma, J.; Chen, B.; Ho, C.T.; Lo, C.; Chen, F.; Wang, M. Cinnamon Bark Proanthocyanidins as Reactive Carbonyl Scavengers to Prevent the Formation of Advanced Glycation Endproducts. J. Agric. Food Chem. 2008, 56, 1907–1911.
- Khedher, M.R.B.; Hafsa, J.; Haddad, M.; Hammami, M. Inhibition of Protein Glycation by Combined Antioxidant and Antiglycation Constituents from a Phenolic Fraction of Sage (Salvia officinalis L.). Plant Foods Hum. Nutr. 2020, 75, 505–511.
- Verzelloni, E.; Pellacani, C.; Tagliazucchi, D.; Tagliaferri, S.; Calani, L.; Costa, L.G.; Brighenti, F.; Borges, G.; Crozier, A.; Conte, A.; et al. Antiglycative and Neuroprotective Activity of Colon-Derived Polyphenol Catabolites. Mol. Nutr. Food Res. 2011, 55 (Suppl. 1), S35–S43.
- Liu, W.; Ma, H.; Frost, L.; Yuan, T.; Dain, J.A.; Seeram, N.P. Pomegranate Phenolics Inhibit Formation of Advanced Glycation Endproducts by Scavenging Reactive Carbonyl Species. Food Funct. 2014, 5, 2996–3004.
- Khan, M.; Liu, H.; Wang, J.; Sun, B. Inhibitory Effect of Phenolic Compounds and Plant Extracts On the Formation of Advance Glycation End Products: A Comprehensive Review. Food Res. Int. 2020, 130, 108933.
- Edeas, M.; Attaf, D.; Mailfert, A.S.; Nasu, M.; Joubet, R. Maillard Reaction, Mitochondria and Oxidative Stress: Potential Role of Antioxidants. Pathol. Biol. 2010, 58, 220–225.
- Hajizadeh-Sharafabad, F.; Sahebkar, A.; Zabetian-Targhi, F.; Maleki, V. The Impact of Resveratrol On Toxicity and Related Complications of Advanced Glycation End Products: A Systematic Review. BioFactors 2019, 45, 651–665.
- Khangholi, S.; Majid, F.A.; Berwary, N.J.; Ahmad, F.; Aziz, R.B. The Mechanisms of Inhibition of Advanced Glycation End Products Formation through Polyphenols in Hyperglycemic Condition. Planta Med. 2016, 82, 32–45.
- Wang, Z.H. Anti-Glycative Effects of Asiatic Acid in Human Keratinocyte Cells. BioMedicine 2014, 4, 19.
- Ahmad, S.; Alouffi, S.; Khan, S.; Khan, M.; Akasha, R.; Ashraf, J.M.; Farhan, M.; Shahab, U.; Khan, M.Y. Physicochemical Characterization of in Vitro Ldl Glycation and its Inhibition by Ellagic Acid (Ea): An in Vivo Approach to Inhibit Diabetes in Experimental Animals. BioMed Res. Int. 2022, 2022, 5583298.
- Abbas, G.; Al-Harrasi, A.S.; Hussain, H.; Hussain, J.; Rashid, R.; Choudhary, M.I. Antiglycation Therapy: Discovery of Promising Antiglycation Agents for the Management of Diabetic Complications. Pharm. Biol. 2016, 54, 198–206.
- Khan, M.; Otaibi, A.A.; Alsukaibi, A.; Alshammari, E.M.; Al-Zahrani, S.A.; Sherwani, S.; Khan, W.A.; Saha, R.; Verma, S.R.; Ahmed, N. Biophysical, Biochemical, and Molecular Docking Investigations of Anti-Glycating, Antioxidant, and Protein Structural Stability Potential of Garlic. Molecules 2022, 27, 1868.
- Ashraf, J.M.; Shahab, U.; Tabrez, S.; Lee, E.J.; Choi, I.; Ahmad, S. Quercetin as a Finer Substitute to Aminoguanidine in the Inhibition of Glycation Products. Int. J. Biol. Macromol. 2015, 77, 188–192.
- Alam, M.M.; Ahmad, I.; Naseem, I. Inhibitory Effect of Quercetin in the Formation of Advance Glycation End Products of Human Serum Albumin: An in Vitro and Molecular Interaction Study. Int. J. Biol. Macromol. 2015, 79, 336–343.
- Chen, X.Y.; Huang, I.M.; Hwang, L.S.; Ho, C.T.; Lo, C.Y. Anthocyanins in Blackcurrant Effectively Prevent the Formation of Advanced Glycation End Products by Trapping Methylglyoxal. J. Funct. Foods 2014, 8, 259–268.
- Meeprom, A.; Sompong, W.; Chan, C.B.; Adisakwattana, S. Isoferulic Acid, a New Anti-Glycation Agent, Inhibits Fructose- and Glucose-Mediated Protein Glycation in Vitro. Molecules 2013, 18, 6439–6454.
- Arfin, S.; Siddiqui, G.A.; Naeem, A.; Moin, S. Inhibition of Advanced Glycation End Products by Isoferulic Acid and its Free Radical Scavenging Capacity: An in Vitro and Molecular Docking Study. Int. J. Biol. Macromol. 2018, 118, 1479–1487.
- Adisakwattana, S.; Sompong, W.; Meeprom, A.; Ngamukote, S.; Yibchok-Anun, S. Cinnamic Acid and its Derivatives Inhibit Fructose-Mediated Protein Glycation. Int. J. Mol. Sci. 2012, 13, 1778–1789.
- Ghodsi, R.; Kheirouri, S. Carnosine and Advanced Glycation End Products: A Systematic Review. Amino Acids 2018, 50, 1177–1186.
- Narda, M.; Peno-Mazzarino, L.; Krutmann, J.; Trullas, C.; Granger, C. Novel Facial Cream Containing Carnosine Inhibits Formation of Advanced Glycation End-Products in Human Skin. Ski. Pharmacol. Physiol. 2018, 31, 324–331.
- Kheirouri, S.; Alizadeh, M. Vitamin D and Advanced Glycation End Products and their Receptors. Pharmacol. Res. 2020, 158, 104879.
- Kheirouri, S.; Alizadeh, M.; Maleki, V. Zinc Against Advanced Glycation End Products. Clin. Exp. Pharmacol. Physiol. 2018, 45, 491–498.





