
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Diego Mariano | + 291 word(s) | 291 | 2022-02-21 04:26:14 | | | |
| 2 | Diego Mariano | Meta information modification | 291 | 2022-02-21 14:16:28 | | | | |
| 3 | Catherine Yang | -30 word(s) | 261 | 2022-02-23 06:43:57 | | | | |
| 4 | Diego Mariano | + 247 word(s) | 508 | 2022-08-30 21:32:22 | | | | |
| 5 | Diego Mariano | + 507 word(s) | 1015 | 2024-01-29 14:42:07 | | |
Video Upload Options
β-glucosidases (EC. 3.2.1.21) are enzymes that hydrolyze glucosidic bonds of oligosaccharides, in special disaccharides, such as cellobiose, realizing glucose at the end of the process. They are highly used in second-generation biofuel production.
Introduction
β-glucosidase represents a category of heterologous enzymes that break down glycosidic bonds in disaccharides, oligosaccharides, and so on. The applications of β-glucosidases extend to diverse biotechnological industries, such as enhancing aroma in juices and wine, hydrolyzing soybean isoflavone glycosides, reducing toxicity in animal feed, and facilitating cellulose degradation for biofuel production [5]. Cellulose, comprising approximately 40 to 50% of plant biomass weight, stands as the predominant biomass source on Earth. It consists of glucose monomers linked by β-1,4 glucosidic bonds. The glucose derived from cellulose breakdown holds the potential for fermentation to yield bioethanol, offering a sustainable and eco-friendly alternative for fuel production [5].
In second-generation biofuel production, they act in synergy with other classes of enzymes, such as endo-glucanases and exo-glucanases [2][3], to obtain fermentable sugars from biomass. Endoglucanases function within the cellulose chain, generating oligosaccharides of varying lengths, while exoglucanases operate on these oligosaccharides, primarily yielding cellobiose [5]. β-glucosidases play a crucial role in this enzymatic system by breaking β-1,4 glucosidic bonds with the assistance of a water molecule, resulting in the production of two glucose molecules. It is widely recognized that β-glucosidases are pivotal in removing cellobiose, a potent inhibitor of both endoglucanases and exoglucanases within this enzymatic process. However, the literature has described that most β-glucosidase enzymes are inhibited by glucose, which has been considered one of the most impacting bottlenecks for highly efficient industrial production [4].
Recently, a more efficient class of β-glucosidases has been described in the literature. They are called glucose-tolerant due to their high resistance to inhibition, even in high glucose concentrations [5]. This class of enzymes has excellent potential for use in industrial applications such as biofuel production. Hence, glucose-tolerant β-glucosidase enzymes have been an essential target of several studies to detect mutations that transform non-tolerant enzymes into glucose-tolerant enzymes [6].
β-glucosidases have been organized into glycoside hydrolase families 1, 3, 5, 9, and 30, with many of them falling into families 1 and 3 [5]. Most glucose-tolerant β-glucosidases are obtained from glycoside-hydrolase family 1 (GH1). GH1 family enzymes belong to the GH-A clan, characterized by proteins with a TIM barrel fold and conserved catalytic amino acids on β-strands 4 and 7 of the barrel [5]. GH1 β-glucosidases employ a retention mechanism on the anomeric carbon, utilizing two glutamates as a catalytic nucleophile and catalytic acid/base. These enzymes have garnered attention for their remarkable resistance to product inhibition, making them highly valuable for cellulose degradation.
In the next section, you will learn a little more about the sequences and structures of beta-glucosidases available in public databases.
Sequence and structure
The primary structure of a protein corresponds to its sequence. We can generally obtain protein sequences from our own databases, such as UniProt (http://www.uniprot.com.br). However, due to their biotechnological importance, beta-glucosidases have a series of databases of both sequences and three-dimensional structures. Two examples are the BETAGDB and Glutantbase databases.
Below, you can see the sequence of the beta-glucosidase from a metagenome sample collected in the South China Sea. This sequence was obtained in the BETAGDB database (available at http://bioinfo.dcc.ufmg.br/betagdb/). GH1 beta-glucosidases present approximately 400 residues (each letter represents an amino acid residue).
>Yang_Y et al. (2015) tr|D5KX75|D5KX75_9BACT Beta-glucosidase OS=uncultured bacterium PE=3 SV=1 MTKISLPTCSPLLTKEFIYGVATASFQIEGGSAHRLPCIWDTFCDTPGKIADNSNGHVAC DHYNNWKQDIDLIESLGVDAYRLSISWPRVITKSGELNPEGVKFYTDILDELKKRNIKAF VTLYHWDLPQHLEDEGGWLNRETAYAFAHYVDLITLAFGDRVHSYATLNEPFCSAFLGYE IGIHAPGKVGKQYGRKAAHHLLLAHGLAMTVLKQNSPTTLNGIVLNFTPCYSISEDADDI AATAFADDYLNQWYMKPIMDGTYPAIIEQLPSAHLPDIHDGDMAIISQSIDYLGINFYTR QFYKAHPTEIYEPIEPTGPLTDMGWEIYPKSFTELLVTLNNTYTLPPIFITENGAAMPDS YNNGEINDVDRLDYYNSHLNAVHNATEQGVRIDGYFAWSLMDNFEWAEGYLKRFGIVYVD YSTQQRTIKNSGLAYKALISNR |
Another example is the β-glucosidase structures obtained from fungi, such as Humicola grisea and Humicola insolens. The sequence below illustrates the primary structure of a fungi β-glucosidase structure.
>de Giuseppe et al. (2014); Benoliel et al. (2010) tr|O93784|O93784_HUMGT Beta-glucosidase OS=Humicola grisea var. thermoidea GN=bgl4 PE=1 SV=1 PDB=4mdo MSLPPDFKWGFATAAYQIEGSVNEDGRGPSIWDTFCAIPGKIADGSSGAVACDSYKRTKE DIALLKELGANSYRFSISWSRIIPLGGRNDPINQKGIDHYVKFVDDLIEAGITPFITLFH WDLPDALDKRYGGFLNKEEFAADFENYARIMFKAIPKCKHWITFNEPWCSAILGYNTGYF APGHTSDRSKSPVGDSAREPWIVGHNILIAHARAVKAYREDFKPTQGGEIGITLNGDATL PWDPEDPADIEACDRKIEFAISWFADPIYFGKYPDSMRKQLGDRLPEFTPEEVALVKGSN DFYGMNHYTANYIKHKTGVPPEDDFLGNLETLFYNKYGDCIGPETQSFWLRPHAQGFRDL LNWLSKRYGYPKIYVTENGTSLKGENDMPLEQVLEDDFRVKYFNDYVRAMAAAVAEDGCN VRGYLAWSLLDNFEWAEGYETRFGVTYVDYANDQKRYPKKSAKSLKPLFDSLIRKE |
Below, you can see a comparison between both sequences generated using the Clustal Omega web tool (available at https://www.ebi.ac.uk/Tools/msa/clustalo/). Humicola insolens β-glucosidase is shown above, and Uncultured bacteria's β-glucosidase sequence from the South China Sea is shown below. The asterisks (*) represent conserved amino acid residues, while the punctuation mark (:) represents a modification for an amino acid residue with similar characteristics. Take note that β-glucosidase sequences from the same family present similar sequences.
Humicola ----------MSLPPDFKWGFATAAYQIEGSVNEDGRGPSIWDTFCAIPGKIADGSSGAV 50 uncultured MTKISLPTCSPLLTKEFIYGVATASFQIEGGS--AHRLPCIWDTFCDTPGKIADNSNGHV 58 * :* :*.***::****. * *.****** ******.*.* * Humicola ACDSYKRTKEDIALLKELGANSYRFSISWSRIIPLGGRNDPINQKGIDHYVKFVDDLIEA 110 uncultured ACDHYNNWKQDIDLIESLGVDAYRLSISWPRVITKSGE---LNPEGVKFYTDILDELKKR 115 *** *:. *:** *::.**.::**:**** *:* .*. :* :*:..*..::*:* : Humicola GITPFITLFHWDLPDALDKRYGGFLNKEEFAADFENYARIM-FKAIPKCKHWITFNEPWC 169 uncultured NIKAFVTLYHWDLPQHLEDE-GGWLNRETA-YAFAHYVDLITLAFGDRVHSYATLNEPFC 173 .*. *:**:*****: *:.. **:**:* * :*. :: : : : : *:***:* Humicola SAILGYNTGYFAPGHTSDRSKSPVGDSAREPWIVGHNILIAHARAVKAYREDFKPTQGGE 229 uncultured SAFLGYEIGIHAPGKVGKQYGR----K------AAHHLLLAHGLAMTVLKQNSPTTLN-- 221 **:***: * .***:...: . ..*::*:**. *:.. ::: * . Humicola IGITLNGDATLPWDPEDPADIEACDRKIEFAISWFADPIYFGKYPDSMRKQLGDRLPEFT 289 uncultured -GIVLNFTPCY-SISEDADDIAATAFADDYLNQWYMKPIMDGTYPAIIEQLPSAHLPDIH 279 **.** ** ** * :: .*: .** *.** :.: . :**:: Humicola PEEVALVKGSNDFYGMNHYTANYIKHKTGVPPEDDFLGNLETLFYNKYGDCIGPETQSFW 349 uncultured DGDMAIISQSIDYLGINFYTRQFYKAHPTEI-------------YEP-IEPTGPLTDMGW 325 ::*::. * *: *:*.** :: * : *: : ** *: * Humicola LRPHAQGFRDLLNWLSKRYGYPKIYVTENGTSLKGENDMPLEQVLEDDFRVKYFNDYVRA 409 uncultured E-IYPKSFTELLVTLNNTYTLPPIFITENGAAMPDSYNN---GEINDVDRLDYYNSHLNA 381 : :.* :** *.: * * *::****::: .. : ::* *:.*:*.::.* Humicola MAAAVAEDGCNVRGYLAWSLLDNFEWAEGYETRFGVTYVDYANDQKRYPKKSAKSLKPLF 469 uncultured VH-NATEQGVRIDGYFAWSLMDNFEWAEGYLKRFGIVYVDYSTQQRTIKNSG-LAYKALI 439 : .:*:* .: **:****:********* .***:.****:.:*: :.. : * *: Humicola DSLIRKE 476 uncultured ----SNR 442 :. |
Also, proteins from the GH1 family have a conserved folding structure called TIM-barrel [7]. Figure 1 illustrates the three-dimensional structure of GH1 β-glucosidase from the fungus Humicola insolens complexed with a glucose molecule [8].
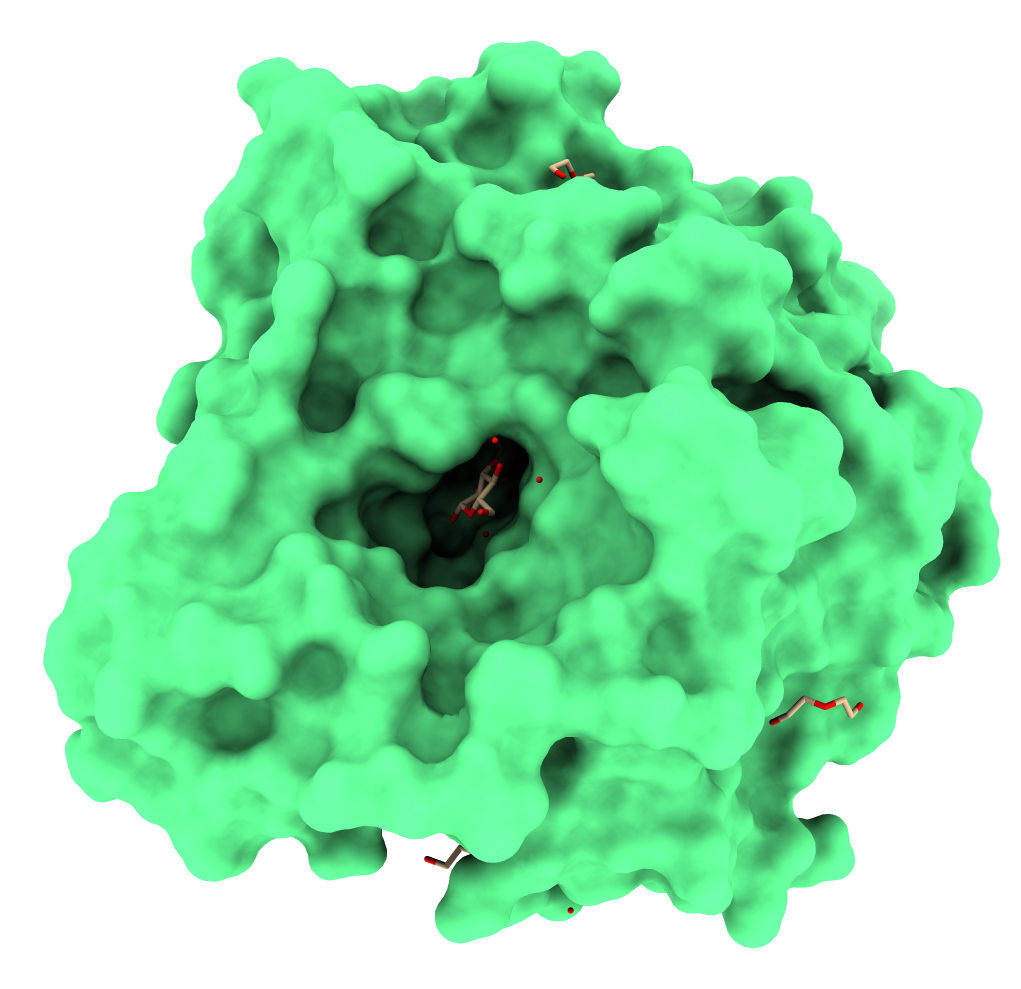 Figure 1. GH1 β-glucosidase from the fungus Humicola insolens. Obtained from PDB ID: 4MDP. Figure generated using ChimeraX [9].
Figure 1. GH1 β-glucosidase from the fungus Humicola insolens. Obtained from PDB ID: 4MDP. Figure generated using ChimeraX [9].
Glucose tolerance mechanisms
Some beta-glucosidases have been described in the literature as resistant to inhibition by glucose. They are called glucose-tolerant. Several recent studies have addressed the reasons for this resistance to inhibition. Recently, a study [2] demonstrated that glucose-tolerant beta-glucosidases have a mechanism for removing glucose from the catalytic pocket region. This was termed the "slingshot mechanism". This mechanism involves the participation of an amino acid residue, usually an aspartate, which performs hydrogen bond interactions with the glucose structure, acting as a type of catapult that pulls the glucose out of the site. Studies have shown that replacing this aspartate with histidine increases the inhibition effect, possibly by removing this ability to remove glucose from the site. It is worth mentioning that the catalytic pocket entry of glucose-tolerant beta-glucosidases is narrower, being formed by a series of hydrophobic residues. These residues are responsible for positioning glucose so the slingshot mechanism can release it. At the same time, the interactions performed by the catalytic residues with glucose are replaced by bonds between them [2].
Conclusion
In conclusion, β-glucosidase enzymes have excellent potential for second-generation biofuel production. However, genetic engineering applications are still necessary to improve the activity of non-tolerant enzymes. Additionally, bioinformatics applications have been successfully used to bring new insights to detect sites for mutations that could improve their activity.
References
- James R. Ketudat Cairns; Asim Esen; β-Glucosidases. Cellular and Molecular Life Sciences 2010, 67, 3389-3405, 10.1007/s00018-010-0399-2.
- Leon Sulfierry Corrêa Costa; Diego César Batista Mariano; Rafael Eduardo Oliveira Rocha; Johannes Kraml; Carlos Henrique Da Silveira; Klaus Roman Liedl; Raquel Cardoso De Melo-Minardi; Leonardo Henrique Franca De Lima; Molecular Dynamics Gives New Insights into the Glucose Tolerance and Inhibition Mechanisms on β-Glucosidases.. Molecules 2019, 24, 3215, 10.3390/molecules24183215.
- Diego Mariano; Naiara Pantuza; Lucianna H. Santos; Rafael E. O. Rocha; Leonardo Lima; Lucas Bleicher; Raquel Cardoso De Melo-Minardi; Glutantβase: a database for improving the rational design of glucose-tolerant β-glucosidases. BMC Molecular and Cell Biology 2020, 21, 1-15, 10.1186/s12860-020-00293-y.
- Andreza P. Garbin; Nayara F.L. Garcia; Gabriela F. Cavalheiro; Maria Alice Silvestre; André Rodrigues; Marcelo F. DA Paz; Gustavo G. Fonseca; Rodrigo S.R. Leite; β-glucosidase from thermophilic fungus Thermoascus crustaceus: production and industrial potential. Anais da Academia Brasileira de Ciências 2020, 93, e20191349, 10.1590/0001-3765202120191349.
- Diego Mariano; C. Leite; L.H.S. Santos; L.F. Marins; K.S. Machado; Adriano Velasque Werhli; L.H.F. Lima; R.C. De Melo-Minardi; Characterization of glucose-tolerant β-glucosidases used in biofuel production under the bioinformatics perspective: a systematic review. Genetics and Molecular Research 2013, 16, 1, 10.4238/gmr16039740.
- Diego César Batista Mariano; Lucianna Helene Santos; Karina Dos Santos Machado; Adriano Velasque Werhli; Leonardo Henrique França De Lima; Raquel Cardoso De Melo-Minardi; A Computational Method to Propose Mutations in Enzymes Based on Structural Signature Variation (SSV). International Journal of Molecular Sciences 2019, 20, 333, 10.3390/ijms20020333.
- Nozomi Nagano; Christine A Orengo; Janet Thornton; One Fold with Many Functions: The Evolutionary Relationships between TIM Barrel Families Based on their Sequences, Structures and Functions. Journal of Molecular Biology 2002, 321, 741-765, 10.1016/s0022-2836(02)00649-6.
- Priscila Oliveira de Giuseppe; Tatiana De Arruda Campos Brasil Souza; Flavio Henrique Moreira Souza; Leticia Maria Zanphorlin; Carla Botelho Machado; Richard Ward; Joao Atilio Jorge; Rosa Dos Prazeres Melo Furriel; Mario Tyago Murakami; Structural basis for glucose tolerance in GH1 β-glucosidases. Acta Crystallographica Section D Biological Crystallography 2014, 70, 1631-1639, 10.1107/s1399004714006920.
- Eric F. Pettersen; Thomas D. Goddard; Conrad C. Huang; Elaine C. Meng; Gregory S. Couch; Tristan I. Croll; John H. Morris; Thomas E. Ferrin; UCSF ChimeraX : Structure visualization for researchers, educators, and developers. Protein Science 2020, 30, 70-82, 10.1002/pro.3943.





