
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Lu Jianhong | -- | 1592 | 2024-03-14 10:30:52 | | | |
| 2 | Wendy Huang | + 816 word(s) | 2408 | 2024-03-15 08:27:39 | | | | |
| 3 | Lu Jianhong | + 625 word(s) | 3033 | 2024-03-16 14:02:49 | | | | |
| 4 | Wendy Huang | -4 word(s) | 3029 | 2024-03-19 08:24:46 | | |
Video Upload Options
α-linolenic acid (ALA) belongs to the family of n-3 polyunsaturated fatty acids (n-3 PUFAs) and contains a carbon–carbon double bond on the third carbon atom at the methyl end of the carbon chain. This family of essential fatty acids also includes eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA). ALA has gradually attracted increased attention due to its nutritional and medicinal advantages. Studies have shown that ALA exerts beneficial effects on a variety of diseases, including cancer.
1. Introduction
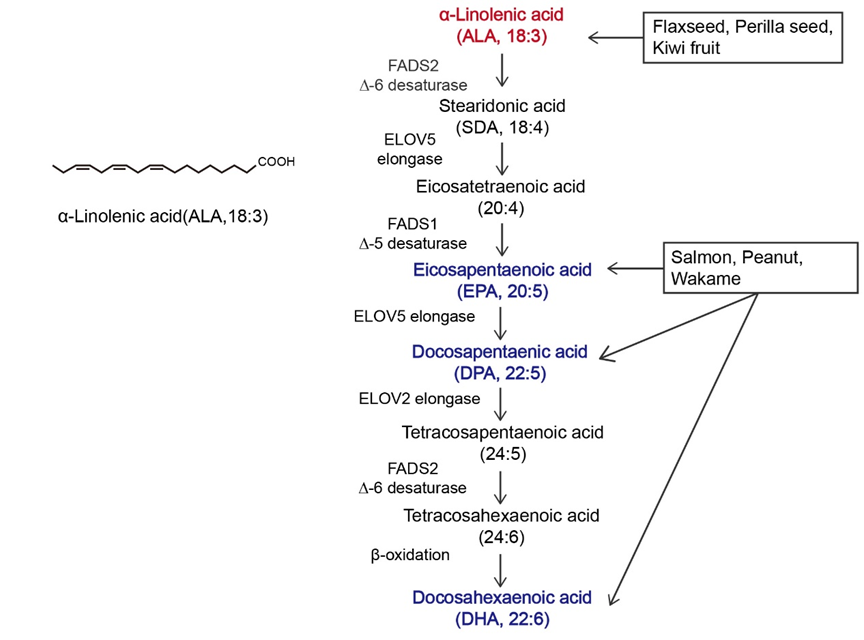
Figure 1.Overview of α-linolenic acid (ALA). Molecular structure of ALA and the in vivo metabolic pathway by which n-3 polyunsaturated fatty acids (n-3 PUFAs) are generated from ALA. The colors in the figure are intended to emphasize the members of n-3 PUFAs. The vertical arrow represents the metabolism of ALA to other n-3 PUFAs in vivo; Arrows in other directions represent different dietary sources of n-3 PUFAs.
According to the International Agency for Research on Cancer GLOBOCAN 2020 cancer incidence and mortality estimates, in 2020, there were 19.3 million new cancer cases and nearly 10 million deaths worldwide [30], except for melanoma cell cancer. As a populous country, China’s new cancer cases in 2020 accounted for 24% of the world’s new cancer cases [31]. Cancer has surpassed cardiovascular disease as the leading cause of death in China. A prominent feature of tumors is that their growth and proliferation are uncontrolled, and invasion and metastasis are the main problems facing current cancer treatment. Although the etiology of cancer is not yet fully understood, it can be roughly divided into two categories, endogenous and exogenous, and nutrients are the factors most closely related to daily life [32]. Nutrient intake can regulate the tumor microenvironment, thereby affecting cancer cell proliferation, apoptosis, and invasion. Current cancer treatment strategies, including surgery, radiotherapy, and chemotherapy, reduce the quality of life of patients, and diet has gradually become one of the most common treatment methods due to its high acceptance by patients and low toxicity and side effects [13][14]. Most of the initial dietary studies focused on limiting the proliferation of tumor cells by reducing the supply of major nutrients to tumors [33][34][35]. With further research, supplementation with specific nutrients, including histidine and mannose, has also become a strategy for the clinical treatment of cancer [36][37].The n-3 PUFA family has attracted considerable attention for its anticancer effects and use as a dietary supplement.
| Cancer | Effect | Effector Molecules | Change in Ex-Pression |
|---|---|---|---|
| PCa (prostate cancer) [52] |
anti-inflammatory effect | PG/LTs | downregulation |
| BC (breast cancer) [38][39] |
anti-inflammatory effect/inhibition of tumor metastasis | COX2/PGE2/Twist 1 | downregulation |
| HCC (hepatocellular carcinoma) [40][41] |
inhibition of proliferation | Farnesoid X receptor | upregulation |
| CRC (colorectal cancer) [42][43] |
induction of apoptosis | caspase 3 | downregulation |
| PCA (pancreatic cancer) [44] |
anti-inflammatory effect | IL-1β/IL-6 | downregulation |
2. Inhibition of Proliferation
3. Induction of Apoptosis
4. Anti-Inflammatory Response
5. Inhibition of Tumor Metastasis
6. Antioxidant Effect
References
- Arthur A. Spector; Hee-Yong Kim; Discovery of essential fatty acids. J. Lipid Res.. 2015, 56, 11-21.
- Toshimi Ogawa; Kento Sawane; Kouta Ookoshi; Ryuta Kawashima; Supplementation with Flaxseed Oil Rich in Alpha-Linolenic Acid Improves Verbal Fluency in Healthy Older Adults. Nutr.. 2023, 15, 1499.
- Marija Takic; Biljana Pokimica; Gordana Petrovic-Oggiano; Tamara Popovic; Effects of Dietary α-Linolenic Acid Treatment and the Efficiency of Its Conversion to Eicosapentaenoic and Docosahexaenoic Acids in Obesity and Related Diseases. Mol.. 2022, 27, 4471.
- Isabelle M. Berquin; Iris J. Edwards; Yong Q. Chen; Multi-targeted therapy of cancer by omega-3 fatty acids. Cancer Lett.. 2008, 269, 363-377.
- Kyu-Bong Kim; Yoon A. Nam; Hyung Sik Kim; A. Wallace Hayes; Byung-Mu Lee; α-Linolenic acid: Nutraceutical, pharmacological and toxicological evaluation. Food Chem. Toxicol.. 2014, 70, 163-178.
- Burdge, G.C.; Calder, P.C. Conversion of alpha-linolenic acid to longer-chain polyunsaturated fatty acids in human adults. Reprod. Nutr. Dev.. 2005, 45, 581–597.
- Aliza H. Stark; Ram Reifen; Michael A. Crawford; Past and Present Insights on Alpha-linolenic Acid and the Omega-3 Fatty Acid Family. Crit. Rev. Food Sci. Nutr.. 2015, 56, 2261-2267.
- Fereidoon Shahidi; Priyatharini Ambigaipalan; Omega-3 Polyunsaturated Fatty Acids and Their Health Benefits. Annu. Rev. Food Sci. Technol.. 2018, 9, 345-381.
- Qianghua Yuan; Fan Xie; Wei Huang; Mei Hu; Qilu Yan; Zemou Chen; Yan Zheng; Li Liu; The review of alpha‐linolenic acid: Sources, metabolism, and pharmacology. Phytotherapy Res.. 2021, 36, 164-188.
- John Parker; Amanda N. Schellenberger; Amy L. Roe; Hellen Oketch-Rabah; Angela I. Calderón; Therapeutic Perspectives on Chia Seed and Its Oil: A Review. Planta Medica. 2018, 84, 606-612.
- Natacha Porta; Béatrice Bourgois; Claude Galabert; Cécile Lecointe; Pierre Cappy; Régis Bordet; Louis Vallée; Stéphane Auvin; Anticonvulsant effects of linolenic acid are unrelated to brain phospholipid cell membrane compositions. Epilepsia. 2009, 50, 65-71.
- Federica Laguzzi; Agneta Åkesson; Matti Marklund; Frank Qian; Bruna Gigante; Traci M. Bartz; Julie K. Bassett; Anna Birukov; Hannia Campos; Yoichiro Hirakawa; Fumiaki Imamura; Susanne Jäger; Maria Lankinen; Rachel A. Murphy; Mackenzie Senn; Toshiko Tanaka; Nathan Tintle; Jyrki K. Virtanen; Kazumasa Yamagishi; Matthew Allison; Ingeborg A. Brouwer; Ulf De Faire; Gudny. Eiriksdottir; Luigi Ferrucci; Nita G. Forouhi; Johanna M. Geleijnse; Allison M Hodge; Hitomi Kimura; Markku Laakso; Ulf Risérus; Anniek C. van Westing; Stefania Bandinelli; Ana Baylin; Graham G. Giles; Vilmundur Gudnason; Hiroyasu Iso; Rozenn N. Lemaitre; Toshiharu Ninomiya; Wendy S. Post; Bruce M. Psaty; Jukka T. Salonen; Matthias B. Schulze; Michael Y. Tsai; Matti Uusitupa; Nicholas J. Wareham; Seung-Won Oh; Alexis C. Wood; William S. Harris; David Siscovick; Dariush Mozaffarian; Karin Leander; Fatty Acids and Outcomes Research Consortium (FORCE); Role of Polyunsaturated Fat in Modifying Cardiovascular Risk Associated With Family History of Cardiovascular Disease: Pooled De Novo Results From 15 Observational Studies. Circ.. 2024, 149, 305-316.
- Evan C. Lien; Matthew G. Vander Heiden; A framework for examining how diet impacts tumour metabolism. Nat. Rev. Cancer. 2019, 19, 651-661.
- Susan E. Steck; E. Angela Murphy; Dietary patterns and cancer risk. Nat. Rev. Cancer. 2019, 20, 125-138.
- Hemant Poudyal; Sunil K. Panchal; Leigh C. Ward; Lindsay Brown; Effects of ALA, EPA and DHA in high-carbohydrate, high-fat diet-induced metabolic syndrome in rats. J. Nutr. Biochem.. 2013, 24, 1041-1052.
- Jeong-Eun Choi; Yongsoon Park; EPA and DHA, but not ALA, have antidepressant effects with 17β-estradiol injection via regulation of a neurobiological system in ovariectomized rats. J. Nutr. Biochem.. 2017, 49, 101-109.
- Laura E Voorrips; Henny Am Brants; Alwine Fm Kardinaal; Gerrit J Hiddink; Piet A van Den Brandt; R Alexandra Goldbohm; Intake of conjugated linoleic acid, fat, and other fatty acids in relation to postmenopausal breast cancer: the Netherlands Cohort Study on Diet and Cancer. Am. J. Clin. Nutr.. 2002, 76, 873-882.
- Asmaa S Abdelhamid; Tracey J Brown; Julii S Brainard; Priti Biswas; Gabrielle C Thorpe; Helen J Moore; Katherine Ho Deane; Fai K AlAbdulghafoor; Carolyn D Summerbell; Helen V Worthington; Fujian Song; Lee Hooper; Omega-3 fatty acids for the primary and secondary prevention of cardiovascular disease. Cochrane Database Syst. Rev.. 2018, 7, CD003177.
- B. Dierberger; M. Schäch; I. Anadere; M. Brändle; R. Jacob; Effect of a diet rich in linseed oil on complex viscosity and blood pressure in spontaneously hypertensive rats (SHR). Basic Res. Cardiol.. 1991, 86, 561-566.
- Yuanyuan Ding; Yuejin Wang; Chaomei Li; Yongjing Zhang; Shiling Hu; Jiapan Gao; Rui Liu; Hongli An; α-Linolenic acid attenuates pseudo-allergic reactions by inhibiting Lyn kinase activity. Phytomedicine. 2020, 80, 153391.
- 21. Hu, F.B.; Stampfer, M.J.; Manson, J.E.; Rimm, E.B.; Wolk, A.; Colditz, G.A.; Hennekens, C.H.; Willett, W.C. Dietary intake of alpha-linolenic acid and risk of fatal ischemic heart disease among women. Am. J. Clin. Nutr.. 1999, 69, 890–897.
- Alan A. Hennessy; Paul R. Ross; Gerald F. Fitzgerald; Catherine Stanton; Sources and Bioactive Properties of Conjugated Dietary Fatty Acids. Lipids. 2016, 51, 377-397.
- Youjin Kim; Jasminka Z. Ilich; Implications of dietary α-linolenic acid in bone health. Nutr.. 2011, 27, 1101-1107.
- Mihir Parikh; Thane G. Maddaford; J. Alejandro Austria; Michel Aliani; Thomas Netticadan; Grant N. Pierce; Dietary Flaxseed as a Strategy for Improving Human Health. Nutr.. 2019, 11, 1171.
- Aleix Sala-Vila; Jennifer Fleming; Penny Kris-Etherton; Emilio Ros; Impact of α-Linolenic Acid, the Vegetable ω-3 Fatty Acid, on Cardiovascular Disease and Cognition. Adv. Nutr. Int. Rev. J.. 2022, 13, 1584-1602.
- Aliza H Stark; Michael A Crawford; Ram Reifen; Update on alpha-linolenic acid. Nutr. Rev.. 2008, 66, 326-332.
- Yuejin Wang; Yuanyuan Ding; Chaomei Li; Jiapan Gao; Xiaodong Wang; Hongli An; Alpha-linolenic acid inhibits IgE-mediated anaphylaxis by inhibiting Lyn kinase and suppressing mast cell activation. Int. Immunopharmacol.. 2021, 103, 108449.
- H M Kaplan; M Deger; K E Erdogan; T Ates; E Demir; Alpha-linolenic acid protects against methotrexate-induced nephrotoxicity in mouse kidney cells.. Eur. Rev. Med. Pharmacol. Sci.. 2023, 27, 11103-11108.
- Sana Noreen; Tabussam Tufail; Huma Bader Ul Ain; Chinaza Godswill Awuchi; Pharmacological, nutraceutical, and nutritional properties of flaxseed (Linum usitatissimum): An insight into its functionality and disease mitigation. Food Sci. Nutr.. 2023, 11, 6820-6829.
- Hyuna Sung; Jacques Ferlay; Rebecca L. Siegel; Mathieu Laversanne; Isabelle Soerjomataram; Ahmedin Jemal; Freddie Bray; Msc Freddie Bray Bsc; Jacques Ferlay Me; Msc Isabelle Soerjomataram Md; Lindsey A. Torre Msph; DVM Ahmedin Jemal PhD; Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA: A Cancer J. Clin.. 2021, 71, 209-249.
- Wei Cao; Hong-Da Chen; Yi-Wen Yu; Ni Li; Wan-Qing Chen; Changing profiles of cancer burden worldwide and in China: a secondary analysis of the global cancer statistics 2020. Chin. Med J.. 2021, 134, 783-791.
- Maria Chiara Mentella; Franco Scaldaferri; Caterina Ricci; Antonio Gasbarrini; Giacinto Abele Donato Miggiano; Cancer and Mediterranean Diet: A Review. Nutr.. 2019, 11, 2059.
- Emeline Dierge; Yvan Larondelle; Olivier Feron; Cancer diets for cancer patients: Lessons from mouse studies and new insights from the study of fatty acid metabolism in tumors. Biochim.. 2020, 178, 56-68.
- Irene Caffa; Vanessa Spagnolo; Claudio Vernieri; Francesca Valdemarin; Pamela Becherini; Min Wei; Sebastian Brandhorst; Chiara Zucal; Else Driehuis; Lorenzo Ferrando; Francesco Piacente; Alberto Tagliafico; Michele Cilli; Luca Mastracci; Valerio G. Vellone; Silvano Piazza; Anna Laura Cremonini; Raffaella Gradaschi; Carolina Mantero; Mario Passalacqua; Alberto Ballestrero; Gabriele Zoppoli; Michele Cea; Annalisa Arrighi; Patrizio Odetti; Fiammetta Monacelli; Giulia Salvadori; Salvatore Cortellino; Hans Clevers; Filippo De Braud; Samir G. Sukkar; Alessandro Provenzani; Valter D. Longo; Alessio Nencioni; Fasting-mimicking diet and hormone therapy induce breast cancer regression. Nat.. 2020, 583, 620-624.
- Salvatore Cortellino; Alessandro Raveane; Claudia Chiodoni; Gloria Delfanti; Federica Pisati; Vanessa Spagnolo; Euplio Visco; Giuseppe Fragale; Federica Ferrante; Serena Magni; Fabio Iannelli; Federica Zanardi; Giulia Casorati; Francesco Bertolini; Paolo Dellabona; Mario P. Colombo; Claudio Tripodo; Valter D. Longo; Fasting renders immunotherapy effective against low-immunogenic breast cancer while reducing side effects. Cell Rep.. 2022, 40, 111256.
- Naama Kanarek; Heather R. Keys; Jason R. Cantor; Caroline A. Lewis; Sze Ham Chan; Tenzin Kunchok; Monther Abu-Remaileh; Elizaveta Freinkman; Lawrence D. Schweitzer; David M. Sabatini; Histidine catabolism is a major determinant of methotrexate sensitivity. Nat.. 2018, 559, 632-636.
- Pablo Sierra Gonzalez; James O’prey; Simone Cardaci; Valentin J. A. Barthet; Jun-Ichi Sakamaki; Florian Beaumatin; Antonia Roseweir; David M. Gay; Gillian Mackay; Gaurav Malviya; Elżbieta Kania; Shona Ritchie; Alice D. Baudot; Barbara Zunino; Agata Mrowinska; Colin Nixon; Darren Ennis; Aoisha Hoyle; David Millan; Iain A. McNeish; Owen J. Sansom; Joanne Edwards; Kevin M. Ryan; Mannose impairs tumour growth and enhances chemotherapy. Nat.. 2018, 563, 719-723.
- 38. Horia, E.; Watkins, B.A. Comparison of stearidonic acid and alpha-linolenic acid on PGE2 production and COX-2 protein levels in MDA-MB-231 breast cancer cell cultures. J. Nutr. Biochem. . 2005, 16, 184–192.
- Shih-Chung Wang; Hai-Lun Sun; Yi-Hsuan Hsu; Shu-Hui Liu; Chong-Kuei Lii; Chia-Han Tsai; Kai-Li Liu; Chin-Shiu Huang; Chien-Chun Li; α-Linolenic acid inhibits the migration of human triple-negative breast cancer cells by attenuating Twist1 expression and suppressing Twist1-mediated epithelial-mesenchymal transition. Biochem. Pharmacol.. 2020, 180, 114152.
- Masataka Okuno; Takuji Tanaka; Chihito Komaki; Seisuke Nagase; Yoshimune Shiratori; Yasutoshi Muto; Kenta Kajiwara; Toshio Maki; Hisataka Moriwaki; Suppressive effect of low amounts of safflower and perilla oils on diethylnitrosamine‐induced hepatocarcinogenesis in male F344 rats. Nutr. Cancer. 1998, 30, 186-193.
- Shu Feng; Xingming Xie; Chaochun Chen; Shi Zuo; Xueke Zhao; Haiyang Li; Alpha-linolenic acid inhibits hepatocellular carcinoma cell growth through Farnesoid X receptor/β-catenin signaling pathway. Nutr. Metab.. 2022, 19, 1-10.
- John P. Chamberland; Hyun-Seuk Moon; Down-regulation of malignant potential by alpha linolenic acid in human and mouse colon cancer cells. Fam. Cancer. 2014, 14, 25-30.
- MarÍA JosÉ GonzÁLez-FernÁNdez; Ignacio Ortea; José Luis Guil-Guerrero; α-Linolenic and γ-linolenic acids exercise differential antitumor effects on HT-29 human colorectal cancer cells. Toxicol. Res.. 2020, 9, 474-483.
- Kyung Suk Park; Joo Weon Lim; Hyeyoung Kim; Inhibitory Mechanism of Omega‐3 Fatty Acids in Pancreatic Inflammation and Apoptosis. Ann. New York Acad. Sci.. 2009, 1171, 421-427.
- Ze-Bin Dai; Xiao-Li Ren; Yi-Lang Xue; Ya Tian; Bing-Bing He; Chang-Long Xu; Bo Yang; Association of Dietary Intake and Biomarker of α-Linolenic Acid With Incident Colorectal Cancer: A Dose-Response Meta-Analysis of Prospective Cohort Studies. Front. Nutr.. 2022, 9, 948604.
- Ana Calado; Pedro Miguel Neves; Teresa Santos; Paula Ravasco; The Effect of Flaxseed in Breast Cancer: A Literature Review. Front. Nutr.. 2018, 5, 4.
- Idris Adewale Ahmed; Maryam Abimbola Mikail; Mohammad Rais Mustafa; Muhammad Ibrahim; Rozana Othman; Lifestyle interventions for non-alcoholic fatty liver disease. Saudi J. Biol. Sci.. 2019, 26, 1519-1524.
- Michael A. Tsoukas; Byung-Joon Ko; Theodore R. Witte; Fadime Dincer; W. Elaine Hardman; Christos S. Mantzoros; Dietary walnut suppression of colorectal cancer in mice: Mediation by miRNA patterns and fatty acid incorporation. J. Nutr. Biochem.. 2015, 26, 776-783.
- Maree T. Brinkman; Margaret R. Karagas; Michael S. Zens; Alan R. Schned; Raoul C. Reulen; Maurice P. Zeegers; Intake of α-linolenic acid and other fatty acids in relation to the risk of bladder cancer: results from the New Hampshire case–control study. Br. J. Nutr.. 2011, 106, 1070-1077.
- Jinfeng Dai; Junhui Shen; Wensheng Pan; Shengrong Shen; Undurti N Das; Effects of polyunsaturated fatty acids on the growth of gastric cancer cells in vitro. Lipids Heal. Dis.. 2013, 12, 71-15.
- Ji Hoon Yu; Sin‐Gun Kang; Un‐Young Jung; Chul‐Ho Jun; Hyeyoung Kim; Effects of Omega‐3 Fatty Acids on Apoptosis of Human Gastric Epithelial Cells Exposed to Silica‐Immobilized Glucose Oxidase. Ann. New York Acad. Sci.. 2009, 1171, 359-364.
- Jingjing Li; Zhennan Gu; Yong Pan; Shunhe Wang; Haiqin Chen; Hao Zhang; Wei Chen; Yong Q. Chen; Dietary supplementation of α-linolenic acid induced conversion of n-3 LCPUFAs and reduced prostate cancer growth in a mouse model. Lipids Heal. Dis.. 2017, 16, 1-9.
- Hyun-Seuk Moon; Saime Batirel; Christos S. Mantzoros; Alpha linolenic acid and oleic acid additively down-regulate malignant potential and positively cross-regulate AMPK/S6 axis in OE19 and OE33 esophageal cancer cells. Metab.. 2014, 63, 1447-1454.
- 54. Kliewer, S.A.; Sundseth, S.S.; Jones, S.A.; Brown, P.J.; Wisely, G.B.; Koble, C.S.; Devchand, P.; Wahli, W.; Willson, T.M.; Len-hard, J.M.; et al. Fatty acids and eicosanoids regulate gene expression through direct interactions with peroxisome proliferator-activated re-ceptors alpha and gamma. Proc. Natl. Acad. Sci. USA. 1997, 94, 4318–4323.
- Lijun Yang; Jianlin Yuan; Liwen Liu; Changhong Shi; Longxin Wang; Feng Tian; Fei Liu; He Wang; Chen Shao; Qiang Zhang; Zhinan Chen; Weijun Qin; Weihong Wen; α-linolenic acid inhibits human renal cell carcinoma cell proliferation through PPAR-γ activation and COX-2 inhibition. Oncol. Lett.. 2013, 6, 197-202.
- Rashmi Deshpande; Prakash Mansara; Ruchika Kaul-Ghanekar; Alpha-linolenic acid regulates Cox2/VEGF/MAP kinase pathway and decreases the expression of HPV oncoproteins E6/E7 through restoration of p53 and Rb expression in human cervical cancer cell lines. Tumor Biol.. 2015, 37, 3295-3305.
- Mayra Montecillo-Aguado; Belen Tirado-Rodriguez; Sara Huerta-Yepez; The Involvement of Polyunsaturated Fatty Acids in Apoptosis Mechanisms and Their Implications in Cancer. Int. J. Mol. Sci.. 2023, 24, 11691.
- Z.Y. Chen; N.W. Istfan; Docosahexaenoic acid is a potent inducer of apoptosis in HT-29 colon cancer cells. Prostaglandins, Leukot. Essent. Fat. Acids. 2000, 63, 301-308.
- Sun En Lee; Joo Weon Lim; Hyeyoung Kim; Activator Protein‐1 Mediates Docosahexaenoic Acid‐induced Apoptosis of Human Gastric Cancer Cells. Ann. New York Acad. Sci.. 2009, 1171, 163-169.
- Hilde Heimli; Camilla Giske; Soheil Naderi; Christian A. Drevon; Kristin Hollung; Eicosapentaenoic acid promotes apoptosis in Ramos cells via activation of caspase‐3 and ‐9. Lipids. 2002, 37, 797-802.
- 62. Deshpande, R.; Mansara, P.; Suryavanshi, S.; Kaul-Ghanekar, R. Alpha-linolenic acid regulates the growth of breast and cervical cancer cell lines through regulation of NO release and in-duction of lipid peroxidation. J. Mol. Biochem. 2013, 2, 6–17.
- Subhadeep Roy; Atul Kumar Rawat; Shreesh Raj Sammi; Uma Devi; Manjari Singh; Swetlana Gautam; Rajnish Kumar Yadav; Jitendra Kumar Rawat; Lakhveer Singh; Mohd. Nazam Ansari; Abdulaziz S. Saeedan; Rakesh Pandey; Dinesh Kumar; Gaurav Kaithwas; Alpha-linolenic acid stabilizes HIF-1 α and downregulates FASN to promote mitochondrial apoptosis for mammary gland chemoprevention. Oncotarget. 2017, 8, 70049-70071.
- Ji‐Yoon Kim; Han Deuk Park; Eunju Park; Jeong‐Woo Chon; Yoo Kyoung Park; Growth‐Inhibitory and Proapoptotic Effects of Alpha‐Linolenic Acid on Estrogen‐Positive Breast Cancer Cells. Ann. New York Acad. Sci.. 2009, 1171, 190-195.
- Alberto Mantovani; Paola Allavena; Antonio Sica; Frances Balkwill; Cancer-related inflammation. Nat.. 2008, 454, 436-444.
- Eran Elinav; Roni Nowarski; Christoph A. Thaiss; Bo Hu; Chengcheng Jin; Richard A. Flavell; Inflammation-induced cancer: crosstalk between tumours, immune cells and microorganisms. Nat. Rev. Cancer. 2013, 13, 759-771.
- Muhammad Shahid Nadeem; Vikas Kumar; Fahad A. Al-Abbasi; Mohammad Amjad Kamal; Firoz Anwar; Risk of colorectal cancer in inflammatory bowel diseases. Semin. Cancer Biol.. 2019, 64, 51-60.
- Ana Elisa Valencise Quaglio; Thais Gagno Grillo; Ellen Cristina Souza De Oliveira; Luiz Claudio Di Stasi; Ligia Yukie Sassaki; Gut microbiota, inflammatory bowel disease and colorectal cancer. World J. Gastroenterol.. 2022, 28, 4053-4060.
- Nuoxi Fan; Jennifer L. Fusco; Daniel W. Rosenberg; Antioxidant and Anti-Inflammatory Properties of Walnut Constituents: Focus on Personalized Cancer Prevention and the Microbiome. Antioxidants. 2023, 12, 982.
- Maria Azrad; Chelsea Turgeon; Wendy Demark-Wahnefried; Current Evidence Linking Polyunsaturated Fatty Acids with Cancer Risk and Progression. Front. Oncol.. 2013, 3, 224.
- Meng Wang; Xiao-Jing Zhang; Chunyan Yan; Chengwei He; Peng Li; Meiwan Chen; Huanxing Su; Jian-Bo Wan; Preventive effect of α-linolenic acid-rich flaxseed oil against ethanol-induced liver injury is associated with ameliorating gut-derived endotoxin-mediated inflammation in mice. J. Funct. Foods. 2016, 23, 532-541.
- Jacqueline K. Innes; Philip C. Calder; Omega-6 fatty acids and inflammation. Prostaglandins, Leukot. Essent. Fat. Acids. 2018, 132, 41-48.
- Tse-Hung Huang; Pei-Wen Wang; Shih-Chun Yang; Wei-Ling Chou; Jia-You Fang; Cosmetic and Therapeutic Applications of Fish Oil’s Fatty Acids on the Skin. Mar. Drugs. 2018, 16, 256.
- D. Williams; M. Verghese; L.T. Walker; J. Boateng; L. Shackelford; C.B. Chawan; Flax seed oil and flax seed meal reduce the formation of aberrant crypt foci (ACF) in azoxymethane-induced colon cancer in Fisher 344 male rats. Food Chem. Toxicol.. 2007, 45, 153-159.
- Patricia S Steeg; Tumor metastasis: mechanistic insights and clinical challenges. Nat. Med.. 2006, 12, 895-904.
- Scott Valastyan; Robert A. Weinberg; Tumor Metastasis: Molecular Insights and Evolving Paradigms. Cell. 2011, 147, 275-292.
- Marianela Vara-Messler; Maria E. Pasqualini; Andrea Comba; Renata Silva; Carola Buccellati; Annalisa Trenti; Lucia Trevisi; Aldo R. Eynard; Angelo Sala; Chiara Bolego; Mirta A. Valentich; Increased dietary levels of α-linoleic acid inhibit mammary tumor growth and metastasis. Eur. J. Nutr.. 2015, 56, 509-519.
- MarijaDragan Jelic; AljosaD Mandic; SlobodanM Maricic; BranislavaU Srdjenovic; Oxidative stress and its role in cancer. J. Cancer Res. Ther.. 2021, 17, 22-28.
- Simone Reuter; Subash C. Gupta; Madan M. Chaturvedi; Bharat B. Aggarwal; Oxidative stress, inflammation, and cancer: How are they linked?. Free. Radic. Biol. Med.. 2010, 49, 1603-1616.
- Leslie Couëdelo; Benjamin Buaud; Hélène Abrous; Ikram Chamekh-Coelho; Didier Majou; Carole Boué-Vaysse; Effect of increased levels of dietary α-linolenic acid on the n-3 PUFA bioavailability and oxidative stress in rat. Br. J. Nutr.. 2021, 127, 1320-1333.
- Jin Hyang Song; Teruo Miyazawa; Enhanced level of n-3 fatty acid in membrane phospholipids induces lipid peroxidation in rats fed dietary docosahexaenoic acid oil. Atheroscler.. 2001, 155, 9-18.
- Jyoti Sharma; Ritu Singh; P. K. Goyal; Chemomodulatory Potential of Flaxseed Oil Against DMBA/Croton Oil–Induced Skin Carcinogenesis in Mice. Integr. Cancer Ther.. 2015, 15, 358-367.
- Joël R. Drevet; The antioxidant glutathione peroxidase family and spermatozoa: A complex story. Mol. Cell. Endocrinol.. 2006, 250, 70-79.
- Hao Han; Fubin Qiu; Haifeng Zhao; Haiying Tang; Xiuhua Li; Dongxing Shi; Dietary flaxseed oil improved western-type diet-induced atherosclerosis in apolipoprotein-E knockout mice. J. Funct. Foods. 2018, 40, 417-425.
- Xiaoyan Sun; Juntong Yu; Yong Wang; Jianming Luo; Guangwen Zhang; Xichun Peng; Flaxseed oil ameliorates aging in d‐galactose induced rats via altering gut microbiota and mitigating oxidative damage. J. Sci. Food Agric.. 2022, 102, 6432-6442.





