
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | yicong cai | -- | 1344 | 2023-03-01 07:01:06 | | | |
| 2 | Catherine Yang | Meta information modification | 1344 | 2023-03-01 08:41:06 | | |
Video Upload Options
The complex structure of the blood–brain barrier (BBB), which blocks nearly all large biomolecules, hinders drug delivery to the brain and drug assessment, thus decelerating drug development. Conventional in vitro models of BBB cannot mimic some crucial features of BBB in vivo including a shear stress environment and the interaction between different types of cells. There is a great demand for a new in vitro platform of BBB that can be used for drug delivery studies.
1. Structure of BBB and Substance Transportation
1.1. Cellular Structure of BBB
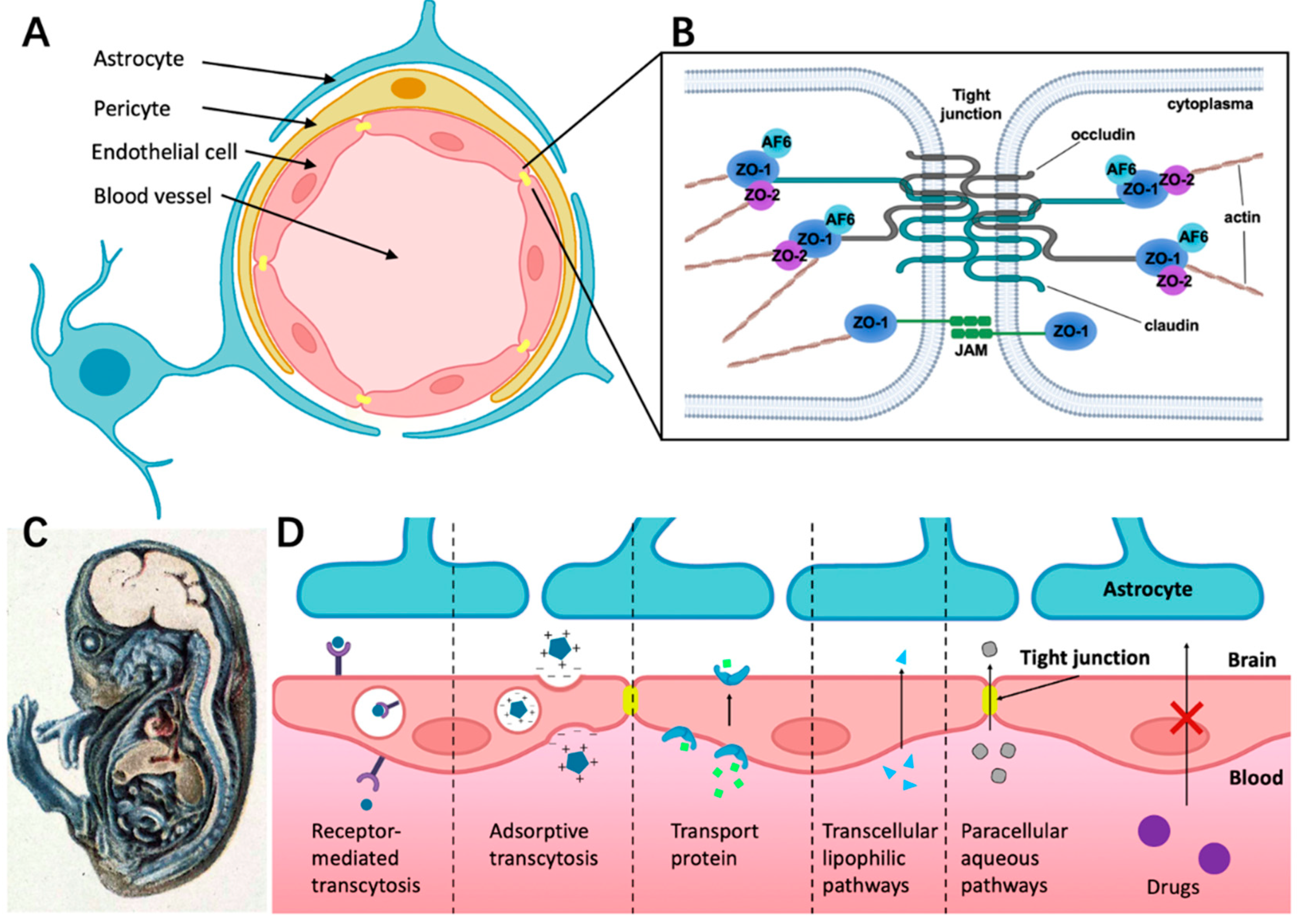
1.2. Structure Foundation of Tight Junctions (TJs)
1.3. Different Ways for Substances Crossing the BBB under Physiological Conditions
2. Ultrasound-Driven Microbubbles Reversibly Open BBB
3. BBB on chip (BOC)
To overcome the major drawbacks of the in vivo model, such as high consumption, ethical issues and large biological differences. There is an urgent need for a preclinical drug screening platform. In recent decades, great efforts have been made to recreate the in vivo environment in vitro. For example, the Transwell model is considered to be a simple way to recreate the BBB in vitro, allowing co-culture of different cell types within the BBB [33]. However, the Transwell model is too simple and static. It lacks mechanical stimuli that are critical for BBB integration [34]. Therefore, a new method was presented, the BOC, a new fabrication led by microfabrication and microfluidics, is an attractive way to reconstruct the BBB in vitro due to its controllability, transparency and ease of resistance measurement and permeability assessment. In addition, it can provide a repeatable and stable environment for drug screening, which could accelerate drug development [35]. Notably, a valid in vitro model must replicate the vital features of the BBB, including physiological and biological properties, and the BOC is considered a potential solution [36].
References
- Wolburg, H.; Lippoldt, A. Tight junctions of the blood–brain barrier: Development, composition and regulation. Vasc. Pharmacol. 2002, 38, 323–337.
- Ridet, J.; Privat, A.; Malhotra, S.; Gage, F. Reactive astrocytes: Cellular and molecular cues to biological function. Trends Neurosci. 1997, 20, 570–577.
- Sofroniew, M.V. Molecular dissection of reactive astrogliosis and glial scar formation. Trends Neurosci. 2009, 32, 638–647.
- Hirschi, K.K.; D’Amore, P.A. Pericytes in the microvasculature. Cardiovasc. Res. 1996, 32, 687–698.
- Sixt, M.; Engelhardt, B.; Pausch, F.; Hallmann, R.; Wendler, O.; Sorokin, L.M. Endothelial cell laminin isoforms, laminins 8 and 10, play decisive roles in T cell recruitment across the blood–brain barrier in experimental autoimmune encephalomyelitis. J. Cell Biol. 2001, 153, 933–946.
- Thomsen, M.S.; Routhe, L.J.; Moos, T. The vascular basement membrane in the healthy and pathological brain. J. Cereb. Blood Flow Metab. 2017, 37, 3300–3317.
- Weksler, B.; Romero, I.A.; Couraud, P.-O. The hCMEC/D3 cell line as a model of the human blood brain barrier. Fluids Barriers CNS 2013, 10, 16.
- Zakharova, M.; Palma do Carmo, M.A.; van der Helm, M.W.; Le-The, H.; de Graaf, M.N.S.; Orlova, V.; van den Berg, A.; van der Meer, A.D.; Broersen, K.; Segerink, L.I. Multiplexed blood-brain barrier organ-on-chip. Lab Chip 2020, 20, 3132–3143.
- Biemans, E.A.; Jäkel, L.; de Waal, R.M.; Kuiperij, H.B.; Verbeek, M.M. Limitations of the hCMEC/D3 cell line as a model for Aβ clearance by the human blood-brain barrier. J. Neurosci. Res. 2017, 95, 1513–1522.
- Ahn, S.I.; Sei, Y.J.; Park, H.-J.; Kim, J.; Ryu, Y.; Choi, J.J.; Sung, H.-J.; MacDonald, T.J.; Levey, A.I.; Kim, Y. Microengineered human blood–brain barrier platform for understanding nanoparticle transport mechanisms. Nat. Commun. 2020, 11, 175.
- Man, S.; Ubogu, E.E.; Williams, K.A.; Tucky, B.; Callahan, M.K.; Ransohoff, R.M. Human brain microvascular endothelial cells and umbilical vein endothelial cells differentially facilitate leukocyte recruitment and utilize chemokines for T cell migration. Clin. Dev. Immunol. 2008, 2008, 384982.
- Maoz, B.M.; Herland, A.; FitzGerald, E.A.; Grevesse, T.; Vidoudez, C.; Pacheco, A.R.; Sheehy, S.P.; Park, T.-E.; Dauth, S.; Mannix, R. A linked organ-on-chip model of the human neurovascular unit reveals the metabolic coupling of endothelial and neuronal cells. Nat. Biotechnol. 2018, 36, 865–874.
- Kim, D.; Eom, S.; Park, S.M.; Hong, H.; Kim, D.S. A collagen gel-coated, aligned nanofiber membrane for enhanced endothelial barrier function. Sci. Rep. 2019, 9, 14915.
- Juang, E.K.; De Cock, I.; Keravnou, C.; Gallagher, M.K.; Keller, S.B.; Zheng, Y.; Averkiou, M. Engineered 3D Microvascular Networks for the Study of Ultrasound-Microbubble-Mediated Drug Delivery. Langmuir 2019, 35, 10128–10138.
- Qian, T.; Maguire, S.E.; Canfield, S.G.; Bao, X.; Olson, W.R.; Shusta, E.V.; Palecek, S.P. Directed differentiation of human pluripotent stem cells to blood-brain barrier endothelial cells. Sci. Adv. 2017, 3, e1701679.
- Wang, Y.I.; Abaci, H.E.; Shuler, M.L. Microfluidic blood-brain barrier model provides in vivo-like barrier properties for drug permeability screening. Biotechnol. Bioeng. 2017, 114, 184–194.
- Lippmann, E.S.; Azarin, S.M.; Kay, J.E.; Nessler, R.A.; Wilson, H.K.; Al-Ahmad, A.; Palecek, S.P.; Shusta, E.V. Derivation of blood-brain barrier endothelial cells from human pluripotent stem cells. Nat. Biotechnol. 2012, 30, 783–791.
- Andreone, B.J.; Chow, B.W.; Tata, A.; Lacoste, B.; Ben-Zvi, A.; Bullock, K.; Deik, A.A.; Ginty, D.D.; Clish, C.B.; Gu, C. Blood-brain barrier permeability is regulated by lipid transport-dependent suppression of caveolae-mediated transcytosis. Neuron 2017, 94, 581–594.e585.
- Matter, K.; Balda, M.S. Signalling to and from tight junctions. Nat. Rev. Mol. Cell Biol. 2003, 4, 225–237.
- Saunders, N.R.; Dreifuss, J.J.; Dziegielewska, K.M.; Johansson, P.A.; Habgood, M.D.; Mollgard, K.; Bauer, H.C. The rights and wrongs of blood-brain barrier permeability studies: A walk through 100 years of history. Front. Neurosci. 2014, 8, 404.
- Tashima, T. Smart strategies for therapeutic agent delivery into brain across the blood–brain barrier using receptor-mediated transcytosis. Chem. Pharm. Bull. 2020, 68, 316–325.
- Hervé, F.; Ghinea, N.; Scherrmann, J.-M. CNS delivery via adsorptive transcytosis. AAPS J. 2008, 10, 455–472.
- Cockerill, I.; Oliver, J.A.; Xu, H.; Fu, B.M.; Zhu, D. Blood-Brain Barrier Integrity and Clearance of Amyloid-β from the BBB. Adv. Exp. Med. Biol. 2018, 1097, 261–278.
- Chen, Y.; Liu, L. Modern methods for delivery of drugs across the blood-brain barrier. Adv. Drug Deliv. Rev. 2012, 64, 640–665.
- Abbott, N.J.; Romero, I.A. Transporting therapeutics across the blood-brain barrier. Mol. Med. Today 1996, 2, 106–113.
- Joseph, E.; Saha, R.N. Advances in brain targeted drug delivery: Nanoparticulate systems. J. PharmaSciTech 2013, 3, 1–8.
- Mitragotri, S. Healing sound: The use of ultrasound in drug delivery and other therapeutic applications. Nat. Rev. Drug Discov. 2005, 4, 255–260.
- Kooiman, K.; Roovers, S.; Langeveld, S.A.G.; Kleven, R.T.; Dewitte, H.; O’Reilly, M.A.; Escoffre, J.M.; Bouakaz, A.; Verweij, M.D.; Hynynen, K.; et al. Ultrasound-Responsive Cavitation Nuclei for Therapy and Drug Delivery. Ultrasound Med. Biol. 2020, 46, 1296–1325.
- Ho, Y.J.; Huang, C.C.; Fan, C.H.; Liu, H.L.; Yeh, C.K. Ultrasonic technologies in imaging and drug delivery. Cell Mol. Life Sci. 2021, 78, 6119–6141.
- Presset, A.; Bonneau, C.; Kazuyoshi, S.; Nadal-Desbarats, L.; Mitsuyoshi, T.; Bouakaz, A.; Kudo, N.; Escoffre, J.-M.; Sasaki, N. Endothelial cells, first target of drug delivery using microbubble-assisted ultrasound. Ultrasound Med. Biol. 2020, 46, 1565–1583.
- De Cock, I.; Zagato, E.; Braeckmans, K.; Luan, Y.; de Jong, N.; De Smedt, S.C.; Lentacker, I. Ultrasound and microbubble mediated drug delivery: Acoustic pressure as determinant for uptake via membrane pores or endocytosis. J. Control. Release 2015, 197, 20–28.
- Park, Y.C.; Zhang, C.; Kim, S.; Mohamedi, G.; Beigie, C.; Nagy, J.O.; Holt, R.G.; Cleveland, R.O.; Jeon, N.L.; Wong, J.Y. Microvessels-on-a-Chip to Assess Targeted Ultrasound-Assisted Drug Delivery. ACS Appl. Mater Interfaces 2016, 8, 31541–31549.
- Christian Weidenfeller, Clive N. Svendsen, Eric V. Shusta Differentiating embryonic neural progenitor cells induce blood–brain barrier properties. Journal of Neurochemistry 2006, 101, Pages 555-565.
- Ross Booth and Hanseup Kim Characterization of a microfluidic in vitro model of the blood-brain barrier (μBBB). lab on chip 2012, 12, 1784-1792.
- Paola De Stefanoa, Elena Bianchi, and Gabriele Dubini The impact of microfluidics in high-throughput drug-screening applications. Biomicrofluidics 2022, Volume 16, 15-23.
- Ben M. Maoz Brain-on-a-Chip: Characterizing the next generation of advanced in vitro platforms for modeling the central nervous system. APL Bioengineering 2021, Volume 5, Issue 3 , null.





