Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Ramon Eritja | -- | 2118 | 2023-01-31 11:11:27 | | | |
| 2 | Sirius Huang | Meta information modification | 2118 | 2023-02-01 02:13:15 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Fàbrega, C.; Aviñó, A.; Navarro, N.; Jorge, A.F.; Grijalvo, S.; Eritja, R. Lipid-Oligonucleotide Conjugates and the Development of RNA-Based Therapeutics. Encyclopedia. Available online: https://encyclopedia.pub/entry/40658 (accessed on 24 May 2026).
Fàbrega C, Aviñó A, Navarro N, Jorge AF, Grijalvo S, Eritja R. Lipid-Oligonucleotide Conjugates and the Development of RNA-Based Therapeutics. Encyclopedia. Available at: https://encyclopedia.pub/entry/40658. Accessed May 24, 2026.
Fàbrega, Carme, Anna Aviñó, Natalia Navarro, Andreia F. Jorge, Santiago Grijalvo, Ramon Eritja. "Lipid-Oligonucleotide Conjugates and the Development of RNA-Based Therapeutics" Encyclopedia, https://encyclopedia.pub/entry/40658 (accessed May 24, 2026).
Fàbrega, C., Aviñó, A., Navarro, N., Jorge, A.F., Grijalvo, S., & Eritja, R. (2023, January 31). Lipid-Oligonucleotide Conjugates and the Development of RNA-Based Therapeutics. In Encyclopedia. https://encyclopedia.pub/entry/40658
Fàbrega, Carme, et al. "Lipid-Oligonucleotide Conjugates and the Development of RNA-Based Therapeutics." Encyclopedia. Web. 31 January, 2023.
Copy Citation
Antisense and small interfering RNA (siRNA) oligonucleotides have been recognized as powerful therapeutic compounds for targeting mRNAs and inducing their degradation. However, a major obstacle is that unmodified oligonucleotides are not readily taken up into tissues and are susceptible to degradation by nucleases. For these reasons, the design and preparation of modified DNA/RNA derivatives with better stability and an ability to be produced at large scale with enhanced uptake properties is of vital importance.
antisense oligonucleotides
siRNA
lipid-oligonucleotide conjugates
1. Introduction
Over the last decade, therapeutic oligonucleotides have gained momentum as an approach to drug development; consequently, there has been a large development of the field. Although the first oligonucleotide approved for therapeutic application in humans dates back to 1998 [1], the recognition of their full therapeutic potential started in 2016 with the authorization of Spinraza [2], for the treatment of Spinal muscular dystrophy, and Etiplersen [3], for the treatment of Duchenne muscular dystrophy. In both cases, the possibility of targeting a mutated gene through alternative splicing became a major success for a long-time dream. Since then, the list of oligonucleotides approved for human practices has reached the dozens, especially with the incorporation of siRNAs in the therapeutic arena [4].
Oligonucleotide therapeutics include antisense oligonucleotides (ASOs) [5], small interfering RNAs (siRNAs) [4], aptamers [6], microRNAs [7], and others [8]. ASOs are small single stranded nucleic acids that by complementarity, bind to a particular mRNA and form a hybrid molecule to modulate gene expression. They act through two mechanisms of action (a) by steric blockade at the ribosomes or (b) by recruiting RNase H enzyme that catalyzes the degradation of mRNA [5]. On the other hand, siRNAs consist of 21–23 mer RNA duplex formed by a sense and an antisense strand complementary to mRNA. The latter is responsible for the recruitment of the target transcript into the RNA-induced silencing complex (RISC) that leads to gene silencing [4]. Unmodified oligonucleotides are not readily taken up into tissues and are also susceptible to degradation by nucleases. For these reasons, the design and preparation of more stable modified DNA/RNA derivatives to improve the existing limitations, like inefficient delivery and mature to the position of clinical utility, is of key importance. Regarding the nuclease resistance, novel derivatives are being developed [9]. The delivery issue is being addressed by the following approaches: by encapsulation in nanomaterials such as solid lipid nanoparticles (SLNs) or by preparing novel oligonucleotide conjugates with selective targeting moieties [10]. The first FDA-approved siRNA, Onpattro [11], is the paradigm of the former; N-acetylgalactosamine (GalNAc) oligonucleotide conjugates [12] are the paradigm of the latter. GalNAc oligonucleotide-conjugates have been shown to be delivered to hepatocytes by binding to asyaloglycoprotein receptors [12]. The latest FDA-approved therapeutic siRNAs, including Givosiran [13], Lumasiran [14], Inclisiran [15], Vutrisiran [16], are based on this strategy. Importantly, Inclisiran (Leqvio®) exerts its therapeutic action within a twice-yearly administration regime [17] while most of the therapeutic siRNAs are administered monthly or every two months [13][14][16]. The large duration of the therapeutic effects of Inclisiran is not only due to a combination of the stability achieved by the modifications on the siRNAs and the efficacy in the delivery, but also to the efficient inhibition of the proprotein convertase subtilisin kexin type 9 (PCSK9) [15][17].
The success in exploring oligonucleotide conjugates for hepatic delivery has triggered an intense quest for oligonucleotide conjugates with tissue-selective targeting properties, particularly for extrahepatic delivery [18].
2. Early Developments in the Synthesis of Lipid-Oligonucleotide Conjugates
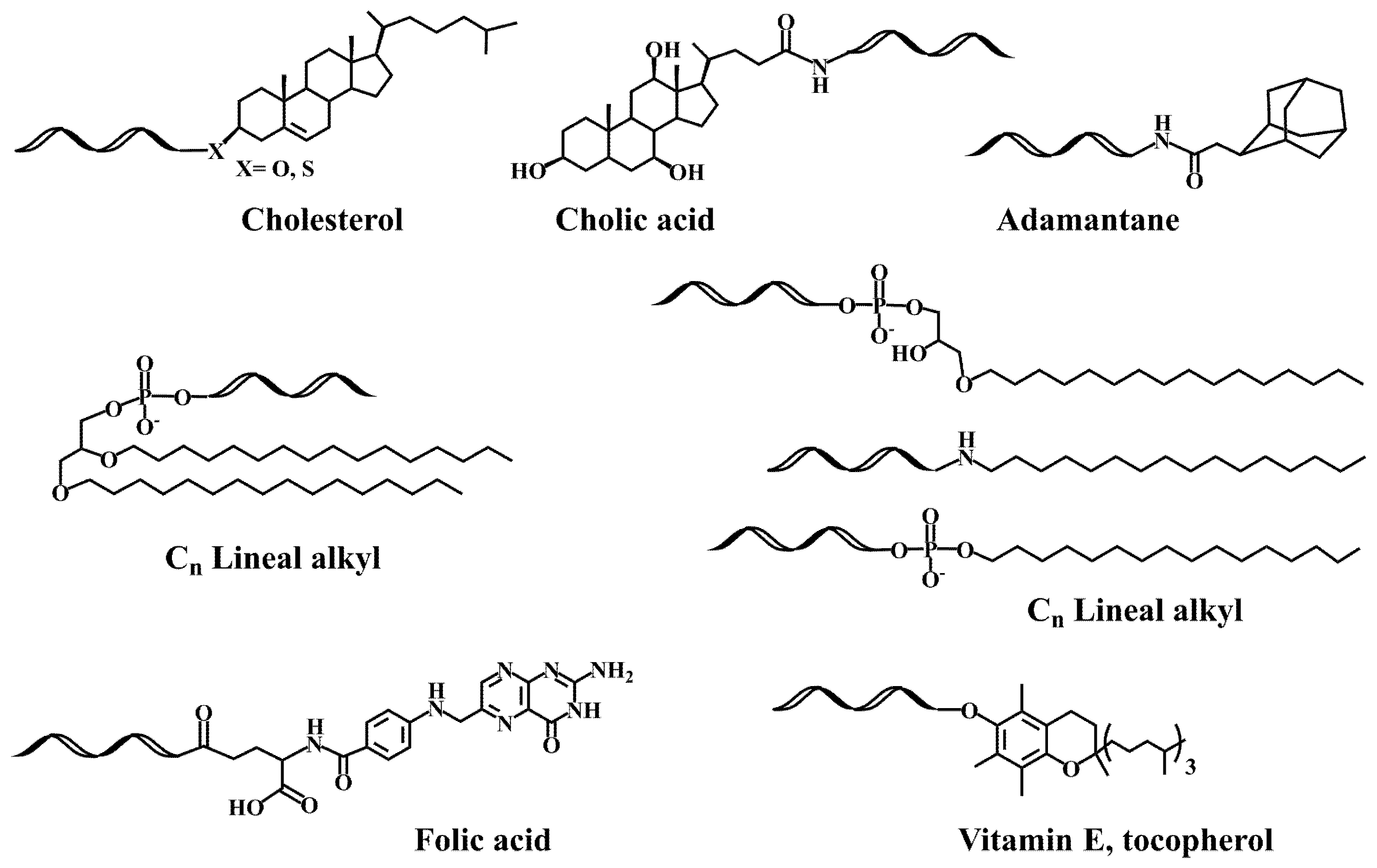
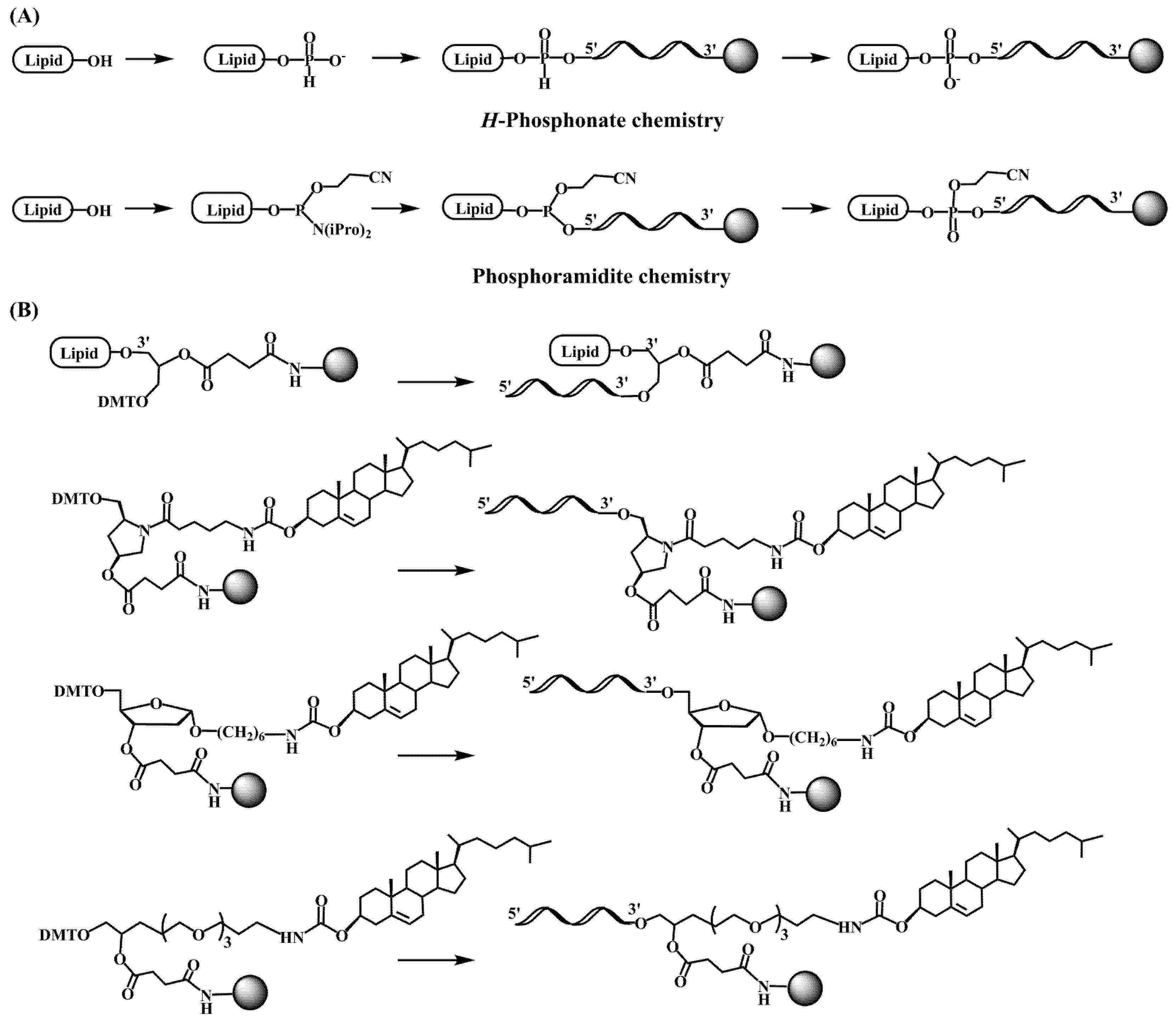
The pioneering works on the antiviral activity of oligonucleotides [19][20] stimulated the development on lipid-oligonucleotide conjugates as potential candidates for the inhibition of the human immunodeficiency virus (HIV-1) in cell culture. Cholesterol was first selected to enhance the interaction between oligonucleotides and cell membranes, which increases the antiviral activity of the oligomers [21][22]. Letsinger’s group designed a synthetic protocol based on the solid-phase oxidation of H-phosphonate dinucleotide intermediates with amino-functionalized cholesterol and catalyzation by carbon tetrachloride, which generated the desired cholesterol-oligonucleotides bond through a phosphoramidate link (Figure 1A) [21] or by direct coupling at the 5′-termini with the H-phosphonate derivative of cholesterol [22][23]. The H-phosphonate derivative of a diacylglycerol was also used for the incorporation of 1,2-di-O-hexadecyl-rac-glyceryl residue at the 5′-end of antiviral oligonucleotides [24]. Solution techniques using amino-lipids [25] or thiocholesterol [26] were also used for conjugation in order to generate physiologically-labile ester [25] or disulfide [26][27] bonds between the lipid and the oligonucleotide. These groundbreaking studies proved the utility of lipid-oligonucleotides by demonstrating that the lipid moiety enhances nuclease resistance and maintains or improves hybridization properties [24][28]. However, in some cases, antiviral properties groundbreaking antisense inhibition rules suggest other mechanisms, such as binding to viral and/or cell membranes [21][22][24].
The next step was the development of specific lipid-phosphoramidites and lipid-functionalized solid supports (Figure 1B). Due to the lability of the ester bonds to ammonia [29], they were replaced by ether, amide and urethane linkers. Several derivatives carrying ether and glyceryl ether bonds were developed by the group of Tom Brown, including 3′ and 5′-cholesteryl, 5′-(1,2-dihexadecylglyceryl), 3′ and 5′-hexadecyl, 5′-octadecyl and 5′-adamantyl [30] as well as vitamin E derivatives (Scheme 1) [31]. Other groups worked on new cholesterol derivatives containing aminodiols such as 3-amino-1,2-propanediol [28] and 3-aminopropylsolketal [32][33], in which the cholesterol moiety was linked to the amino group by reaction with cholesterol chloroformate generating an urethane bond stable to ammonia.
On the other hand, postsynthetic conjugation reactions between amino-oligonucleotides and carboxylic acid derivatives of lipids such as cholic acid, adamantane acetic acid and fatty acids were described [34][35][36]. A variation of this protocol implies the addition of 9-fluorenylmethoxycarbonyl (Fmoc)-protected amino linkers into oligonucleotides. After the assembly of the sequence, the Fmoc moiety can be removed generating a free amino group that reacts with cholesterol chloroformate followed by standard ammonia deprotection [37].
The availability of lipid-oligonucleotides allowed its preclinical evaluation demonstrating specific antisense activity, enhanced nuclease resistance and maintenance or improvement of hybridization properties [40][41]. Another interesting property is its capability to bind to serum proteins and lipoproteins, which is important to avoid renal clearance of oligonucleotides [42]. Several specific receptor-mediated uptake mechanisms have been described to explain selective uptake by hepatocytes via lipoprotein receptors [43]. Although lipid-oligonucleotide conjugates have interesting properties, none of them, except polyethyleneglycol (PEG) derivatives, have found their way to clinical studies. The synthesis of oligonucleotides functionalized with PEG has been described by the Erdmann and Bonora groups, being somehow similar to the methodology described herein for other lipid diols [44][45][46][47]. Pegaptanib (Macugen®) is a therapeutic oligonucleotide used for the treatment of aged-associated macular degeneration. This oligonucleotide is an aptamer constituted by 28 nucleotides and functionalized with PEG at the 5′-end and an inverted-T at the 3′-end to prevent degradation by nucleases [48]. This aptamer has a strong affinity to the vascular endothelial growth factor VEGF165 (Kd = 49 pM), inhibiting the binding of VEGF to its receptor, suppressing the VEGF-mediated angiogenesis and consequently lowering vascular permeability and inflammation [49].
3. Lipid-Oligonucleotide Conjugates and the Development of RNA-Based Therapeutics
The discovery of the RNA interference mechanisms provided a great resurgence in the area of therapeutic oligonucleotides. Soon after the work of Mello and Fire [50], it was established that the effector molecules of the RNA interference process were double stranded RNA molecules of 19–21 nucleotides. Then, synthetic oligonucleotides with chemical modifications were proved to improve the efficacy and the duration compared to natural substrates [51][52]. Later, the discovery of the microRNAs increased the therapeutic potential of oligonucleotides [53]. Cholesterol-siRNAs were developed and were demonstrated to be successful derivatives for the inhibition of lipoproteins [54]. Stable nucleic acid lipid nanoparticles (SNALP) and solid-lipid nanoparticles (SLN) were developed for the delivery of siRNAs [55][56] showing for the first time the in vivo inhibition of ApoB in non-human primates [55]. To expand the arsenal of available cationic lipids for siRNAs delivery, several lipids and lipoids libraries were screened, thereby generating lipids with high efficiency and less toxicity [57][58].
These studies led to the search for new hydrophobic molecules to enhance the cellular uptake of siRNAs [59][60][61][62]. Conjugation of amino-siRNAs with a small library of carboxyl-lipids including cholesterol, fatty-acids and bile acids resulted in hydrophobic siRNA derivatives that interact with lipoproteins [43]. The obtained hydrophobic (lipo-siRNA-protein) complexes were efficiently delivered to liver, gut and kidney by specific lipoprotein-mediated receptors. Inspired by these results, researchers studied a small lipid library including both neutral [38] and cationic lipids [39]. The study of TNF-alpha inhibition with and without lipofectamine proved that these lipid-siRNA conjugates carrying ammonia-resistant glycerol ether bonds were compatible with RNA interference mechanisms. A lipid carrying two linear hydrocarbon chains was the best derivative in terms of increasing cellular entrance (Scheme 1) [38]. These double-chain lipid-siRNA conjugates stimulated the formation of small vesicles that may explain the improved uptake properties [63][64]. In addition, a good correlation was found among cell lines expressing abundant CR3 receptors [63]. The vesicle formation properties and the enhanced binding of siRNAs carrying double-chain lipids to hydrophobic membranes has also been observed by several authors [65][66][67]. Furthermore, researchers found that the sonication of lipophilic siRNA in presence of serum enhanced the binding of lipophilic-siRNA to lipoproteins, resulting in a more efficient transfection [68].
In a different approach, cholesterol-conjugated single-stranded short RNA molecules, or antagomiRs, were successfully used to silence miRNA [69][70][71][72]. In addition, G-quadruplex-forming oligonucleotides carrying lipid moieties were found to increase their affinity for viral membrane proteins showing antiviral properties by inhibition of viral cell entry [73][74][75]. The most frequent methodology for the preparation of oligonucleotide-lipid conjugates is based on amide formation (Figure 2), but other reactions such as the copper catalyzed azide-alkyne cycloaddition (click chemistry) have also been reported [76].
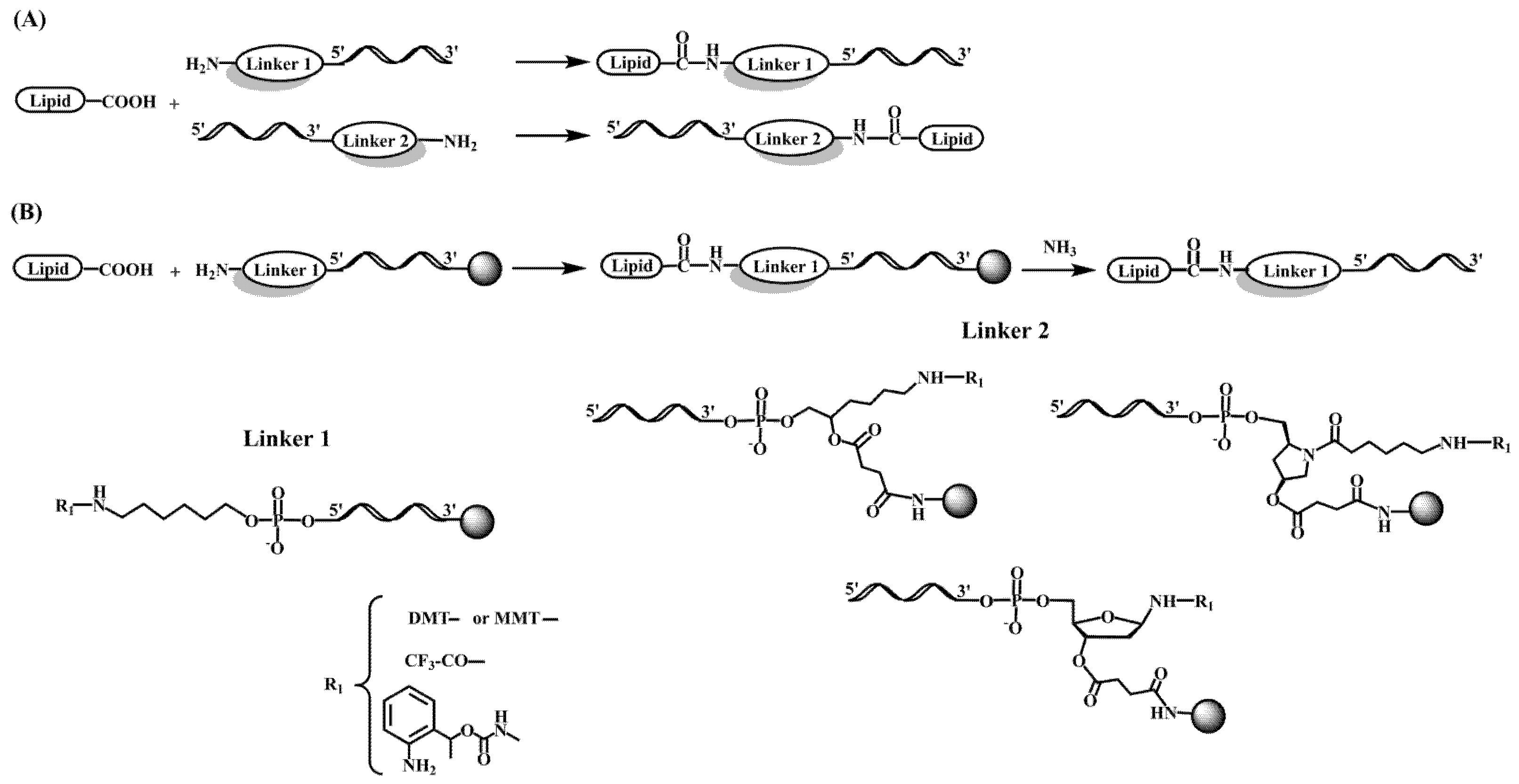
Figure 2. Chemical structure of several linker molecules connecting solid supports and lipids used for the preparation of lipid-oligonucleotide conjugates. (A) Lipid conjugation to the 5′ or 3′ end of an oligonucleotide in solution [34][35][36]. (B) Lipid conjugation to the 5′ or 3′ terminus of an oligonucleotide on a solid support [28][30].
Recently, studies have been addressed towards the application of lipid-oligonucleotides to transfect cells and tissues other than liver. Primary neurons are difficult to transfect with siRNAs because the unique nature of the blood brain barrier (BBB) and the difficulty of direct administration [77]. Alterman et al. found that cholesterol-tetraethyleneglycol functionalized siRNA at the passenger strand was efficiently internalized in primary cortical neurons, inducing a potent and specific silencing of huntingtin gene [78][79]. This potent silencing activity was maintained in vivo when injected into mouse brain [78]. Additionally, docosahexaenoic acid conjugation (Figure 3) was judged to increase further the distribution and the inhibitory properties of lipid-siRNAs when administered into the brain [80].
Another interesting property of lipid-siRNAs is the enhancement of siRNA loading into extracellular vesicles [81][82], which generates attractive nanoparticles for the delivery of therapeutic siRNAs. The best option in terms of higher loading and efficiency was the conjugation of vitamin E (Sheme 1) [82]. Recently, siRNAs were modified with vitamin E by a benzonorbonadiene linker, which releases active siRNAs when reacting with tetrazines [83].
Next, the distribution of siRNAs conjugated to a small library of complex lipids was analyzed, including saturated and unsaturated fatty acids, steroids and lipophilic vitamins with or without phosphocholine heads. The level of hydrophobicity is critical in order to define accumulation in the liver or in the kidney. In addition, it was shown that some lipid derivatives were able to accumulate in non-hepatic tissues such as lung, muscle, heart, adrenal glands and fat [84][85]. In more detailed studies, factors such as the chemical structure of the lipids [85], the phosphorothioate content [86], the presence of single-stranded phosphorothioate regions [87] or the valency of fatty acid modifications [88] were demonstrated to affect the pharmacokinetics, the extrahepatic distribution and the in vivo efficacy of lipid-siRNAs [89][90]. Recently, the in vivo properties of siRNA carrying 2′-O-hexadecyl (C16) moieties have been described (Figure 3). These lipophilic siRNAs can be delivered into the central nervous system, eye and lungs of rats and non-human primates, where they exert inhibitory properties for at least 3 months [91]. These results opened the possibility of using lipophilic siRNAs in the treatment of Alzheimer’s disease.
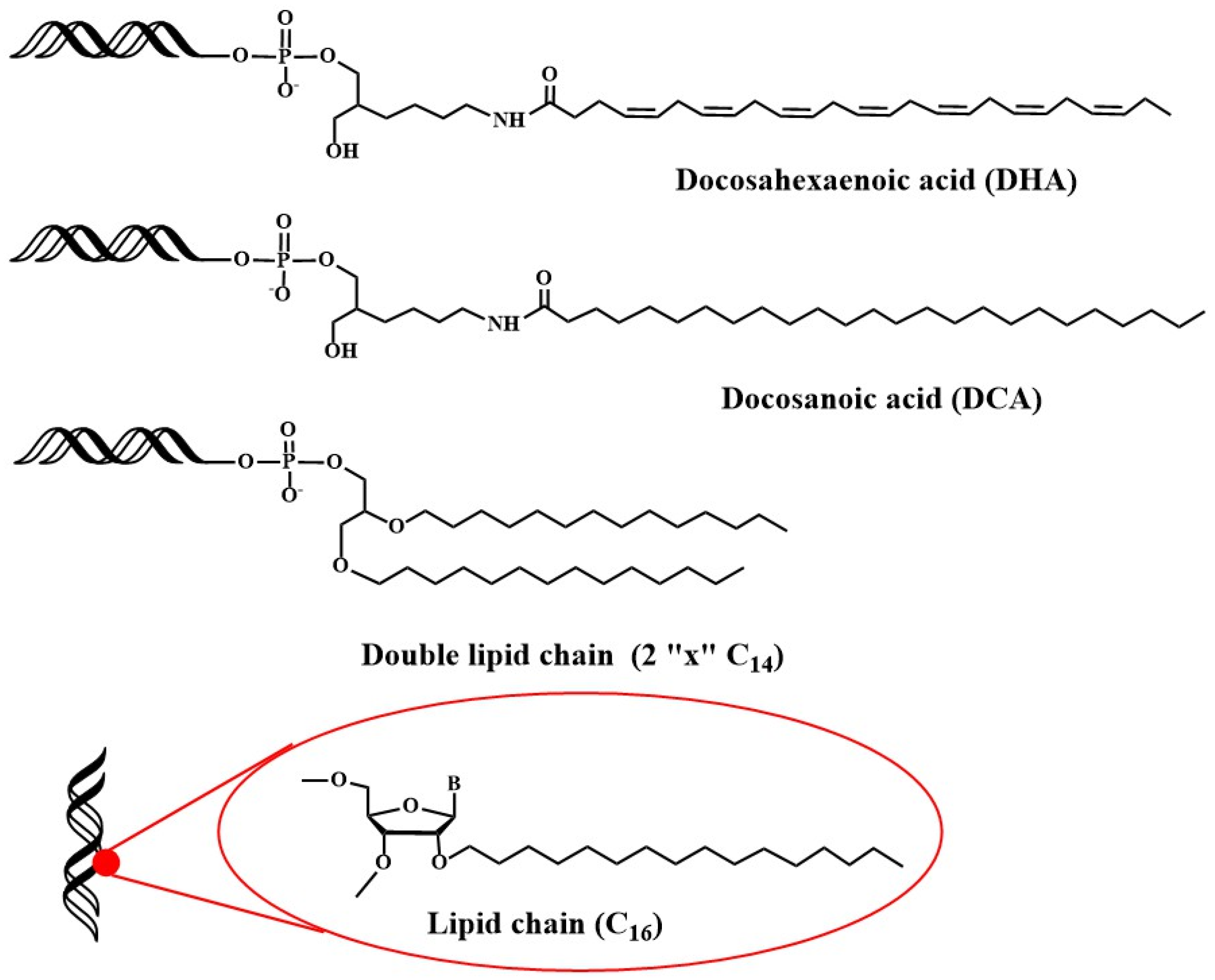
A systematic study on the effect of the conjugation of antisense oligonucleotides with fatty acids confirms its potential delivery to muscle and other extrahepatic tissues [92][93]. Moreover, palmitic acid-, tocopherol-, and cholesterol-conjugated (Scheme 1) antisense oligonucleotides were reported to increase protein binding and enhance intracellular uptake [94]. These properties were explored by several groups for the development of lipid-antisense oligonucleotides targeting the exon 51 of human Duchene Muscular Distrophy gene [95][96].
The development of mRNA vaccines, especially during the COVID-19 pandemic, triggered the interest in lipid nanoparticles for mRNA delivery. The approval of Onpattro for the treatment of transthyretin–mediated amyloidosis demonstrated the efficacy and safety of these non-viral vectors for siRNA delivery to liver [11]. This approval facilitated the rapid authorization of the two mRNA vaccines for SARS-CoV-2 [97][98]. The great potential of mRNA vaccines for cancer, infectious diseases and genetic disorders is stimulating the search for the next generation of lipid nanoparticles that would increase efficacy, degradability [99] and tissue-specificity properties [100].
References
- Belfort, R.; Goldstein, D.A.; Johnson, D.W.; Mansour, S.E.; Muccioli, C.; Sheppard, J.D.; Mora-Durate, J.; Chan, C.K.; Palestine, A.G.; Perez, J.E.; et al. A Randomized Controlled Clinical Trial of Intravitreous Fomivirsen for Treatment of Newly Diagnosed Peripheral Cytomegalovirus Retinitis in Patients with AIDS. Am. J. Ophthalmol. 2002, 133, 467–474.
- Finkel, R.S.; Mercuri, E.; Darras, B.T.; Connolly, A.M.; Kuntz, N.L.; Kirschner, J.; Chiriboga, C.A.; Saito, K.; Servais, L.; Tizzano, E. Nusinersen versus Sham Control in Infantile-Onset Spinal Muscular Atrophy. N. Engl. J. Med. 2017, 377, 1723–1732.
- Charleston, J.S.; Schnell, F.J.; Dworzak, J.; Donoghue, C.; Lewis, S.; Chen, L.; Young, G.D.; Milici, A.J.; Voss, J.; DeAlwis, U. Eteplirsen Treatment for Duchenne Muscular Dystrophy: Exon Skipping and Dystrophin Production. Neurology 2018, 90, e2146–e2154.
- Hu, B.; Zhong, L.; Weng, Y.; Peng, L.; Huang, Y.; Zhao, Y.; Liang, X.-J. Therapeutic SiRNA: State of the Art. Signal. Transduct. Target. Ther. 2020, 5, 101.
- Crooke, S.T.; Liang, X.-H.; Baker, B.F.; Crooke, R.M. Antisense Technology: A Review. J. Biol. Chem. 2021, 296, 100416.
- Mallikaratchy, P. Evolution of Complex Target SELEX to Identify Aptamers against Mammalian Cell-Surface Antigens. Molecules 2017, 22, 215.
- Li, Z.; Rana, T.M. Therapeutic Targeting of MicroRNAs: Current Status and Future Challenges. Nat. Rev. Drug Discov. 2014, 13, 622–638.
- Smith, C.I.E.; Zain, R. Therapeutic Oligonucleotides: State of the Art. Annu. Rev. Pharmacol. Toxicol. 2019, 59, 605–630.
- Khvorova, A.; Watts, J.K. The Chemical Evolution of Oligonucleotide Therapies of Clinical Utility. Nat. Biotechnol. 2017, 35, 238–248.
- Grijalvo, S.; Alagia, A.; Jorge, A.F.; Eritja, R. Covalent Strategies for Targeting Messenger and Non-Coding RNAs: An Updated Review on SiRNA, MiRNA and AntimiR Conjugates. Genes 2018, 9, 74.
- Adams, D.; Gonzalez-Duarte, A.; O’Riordan, W.D.; Yang, C.-C.; Ueda, M.; Kristen, A.V.; Tournev, I.; Schmidt, H.H.; Coelho, T.; Berk, J.L. Patisiran, an RNAi Therapeutic, for Hereditary Transthyretin Amyloidosis. N. Engl. J. Med. 2018, 379, 11–21.
- Nair, J.K.; Willoughby, J.L.S.; Chan, A.; Charisse, K.; Alam, M.R.; Wang, Q.; Hoekstra, M.; Kandasamy, P.; Kel’in, A.V.; Milstein, S. Multivalent N-Acetylgalactosamine-Conjugated SiRNA Localizes in Hepatocytes and Elicits Robust RNAi-Mediated Gene Silencing. J. Am. Chem. Soc. 2014, 136, 16958–16961.
- Balwani, M.; Sardh, E.; Ventura, P.; Peiró, P.A.; Rees, D.C.; Stölzel, U.; Bissell, D.M.; Bonkovsky, H.L.; Windyga, J.; Anderson, K.E. Phase 3 Trial of RNAi Therapeutic Givosiran for Acute Intermittent Porphyria. N. Engl. J. Med. 2020, 382, 2289–2301.
- Garrelfs, S.F.; Frishberg, Y.; Hulton, S.A.; Koren, M.J.; O’Riordan, W.D.; Cochat, P.; Deschênes, G.; Shasha-Lavsky, H.; Saland, J.M.; van’t Hoff, W.G. Lumasiran, an RNAi Therapeutic for Primary Hyperoxaluria Type 1. N. Engl. J. Med. 2021, 384, 1216–1226.
- Ray, K.K.; Wright, R.S.; Kallend, D.; Koenig, W.; Leiter, L.A.; Raal, F.J.; Bisch, J.A.; Richardson, T.; Jaros, M.; Wijngaard, P.L.J. Two Phase 3 Trials of Inclisiran in Patients with Elevated LDL Cholesterol. N. Engl. J. Med. 2020, 382, 1507–1519.
- Adams, D.; Tournev, I.L.; Taylor, M.S.; Coelho, T.; Planté-Bordeneuve, V.; Berk, J.L.; González-Duarte, A.; Gillmore, J.D.; Low, S.-C.; Sekijima, Y. Efficacy and Safety of Vutrisiran for Patients with Hereditary Transthyretin-Mediated Amyloidosis with Polyneuropathy: A Randomized Clinical Trial. Amyloid 2022, 1–9.
- Kosmas, C.E.; Muñoz Estrella, A.; Sourlas, A.; Silverio, D.; Hilario, E.; Montan, P.D.; Guzman, E. Inclisiran: A New Promising Agent in the Management of Hypercholesterolemia. Diseases 2018, 6, 63.
- Hawner, M.; Ducho, C. Cellular Targeting of Oligonucleotides by Conjugation with Small Molecules. Molecules 2020, 25, 5963.
- Zamecnik, P.C.; Stephenson, M.L. Inhibition of Rous Sarcoma Virus Replication and Cell Transformation by a Specific Oligodeoxynucleotide. Proc. Natl. Acad. Sci. USA 1978, 75, 280–284.
- Smith, C.C.; Aurelian, L.; Reddy, M.P.; Miller, P.S.; Ts’o, P.O. Antiviral Effect of an Oligo (Nucleoside Methylphosphonate) Complementary to the Splice Junction of Herpes Simplex Virus Type 1 Immediate Early Pre-MRNAs 4 and 5. Proc. Natl. Acad. Sci. USA 1986, 83, 2787–2791.
- Letsinger, R.L.; Zhang, G.R.; Sun, D.K.; Ikeuchi, T.; Sarin, P.S. Cholesteryl-Conjugated Oligonucleotides: Synthesis, Properties, and Activity as Inhibitors of Replication of Human Immunodeficiency Virus in Cell Culture. Proc. Natl. Acad. Sci. USA 1989, 86, 6553–6556.
- Stein, C.A.; Pal, R.; DeVico, A.L.; Hoke, G.; Mumbauer, S.; Kinstler, O.; Sarngadharan, M.G.; Letsinger, R.L. Mode of Action of 5’-Linked Cholesteryl Phosphorothioate Oligodeoxynucleotides in Inhibiting Syncytia Formation and Infection by HIV-1 and HIV-2 in Vitro. Biochemistry 1991, 30, 2439–2444.
- Krieg, A.M.; ToNkINsoN, J.; MATsoN, S.; ZHAo, Q.; SAxoN, M.; Zhang, L.-M.; Bhanja, U.; Yakubov, L.; Stein, C.A. Modification of Antisense Phosphodiester Oligodeoxynucleotides by a 5’cholesteryl Moiety Increases Cellular Association and Improves Efficacy. Proc. Natl. Acad. Sci. USA 1993, 90, 1048–1052.
- Shea, R.G.; Marsters, J.C.; Bischofberger, N. Synthesis, Hybridization Properties and Antiviral Activity of Lipid-Oligodeoxynucleotide Conjugates. Nucleic Acids Res. 1990, 18, 3777–3783.
- Svinarchuk, F.P.; Konevetz, D.A.; Pliasunova, O.A.; Pokrovsky, A.G.; Vlassov, V. V Inhibition of HIV Proliferation in MT-4 Cells by Antisense Oligonucleotide Conjugated to Lipophilic Groups. Biochimie 1993, 75, 49–54.
- Oberhauser, B.; Wagner, E. Effective Incorporation of 2’-O-Methyl-Oligoribonuclectides into Liposomes and Enhanced Cell Association through Modification with Thiocholesterol. Nucleic Acids Res. 1992, 20, 533–538.
- Boutorine, A.S.; Kostina, E. V Reversible Covalent Attachment of Cholesterol to Oligodeoxyribonucleotides for Studies of the Mechanisms of Their Penetration into Eucaryotic Cells. Biochimie 1993, 75, 35–41.
- Gryaznov, S.M.; Lloyd, D.H. Modulation of Oligonucleotide Duplex and Triplex Stability via Hydrophobic Interactions. Nucleic Acids Res. 1993, 21, 5909–5915.
- Polushin, N.N.; Cohen, J.S. Antisense Pro-Drugs: 5’-Ester Oligodeoxynucleotides. Nucleic Acids Res. 1994, 22, 5492–5496.
- MacKellar, C.; Graham, D.; Will, D.W.; Burgess, S.; Brown, T. Synthesis and Physical Properties of Anti-HIV Antisense Oligonucleotides Bearing Terminal Lipophilic Groups. Nucleic Acids Res. 1992, 20, 3411–3417.
- Will, D.W.; Brown, T. Attachment of Vitamin E Derivatives to Oligonucleotides during Solid-Phase Synthesis. Tetrahedron Lett. 1992, 33, 2729–2732.
- Vu, H.; Singh, P.; Lewis, L.; Zendegui, J.G.; Jayaraman, K. Synthesis of Cholesteryl Supports and Phosphoramidite for Automated DNA Synthesis of Triple-Helix Forming Oligonucleotides (TFOs). Nucleosides Nucleotides 1993, 12, 853–864.
- Vu, H.; Hill, T.S.; Jayaraman, K. Synthesis and Properties of Cholesteryl-Modified Triple-Helix Forming Oligonucleotides Containing a Triglycyl Linker. Bioconjug. Chem. 1994, 5, 666–668.
- Manoharan, M.; Johnson, L.K.; Bennett, C.F.; Vickers, T.A.; Ecker, D.J.; Cowsert, L.M.; Freier, S.M.; Cook, P.D. Cholic Acid-Oligonucleotide Conjugates for Antisense Applications. Bioorg. Med. Chem. Lett. 1994, 4, 1053–1060.
- Manoharan, M.; Tivel, K.L.; Cook, P.D. Lipidic Nucleic Acids. Tetrahedron Lett. 1995, 36, 3651–3654.
- Li, S.; Deshmukh, H.M.; Huang, L. Folate-Mediated Targeting of Antisense Oligodeoxynucleotides to Ovarian Cancer Cells. Pharm. Res. 1998, 15, 1540–1545.
- Marasco, C.J., Jr.; Angelino, N.J.; Paul, B.; Dolnick, B.J. A Simplified Synthesis of Acridine and/or Lipid Containing Oligodeoxynucleotides. Tetrahedron Lett. 1994, 35, 3029–3032.
- Grijalvo, S.; Ocampo, S.M.; Perales, J.C.; Eritja, R. Synthesis of Lipid–Oligonucleotide Conjugates for RNA Interference Studies. Chem. Biodivers. 2011, 8, 287–299.
- Grijalvo, S.; Ocampo, S.M.; Perales, J.C.; Eritja, R. Synthesis of Oligonucleotides Carrying Amino Lipid Groups at the 3′-End for RNA Interference Studies. J. Org. Chem. 2010, 75, 6806–6813.
- Manoharan, M. Oligonucleotide Conjugates as Potential Antisense Drugs with Improved Uptake, Biodistribution, Targeted Delivery, and Mechanism of Action. Antisense Nucleic Acid Drug Dev. 2002, 12, 103–128.
- Crooke, S.T.; Graham, M.J.; Zuckerman, J.E.; Brooks, D.; Conklin, B.S.; Cummins, L.L.; Greig, M.J.; Guinosso, C.J.; Kornbrust, D.; Manoharan, M. Pharmacokinetic Properties of Several Novel Oligonucleotide Analogs in Mice. J. Pharmacol. Exp. Ther. 1996, 277, 923–937.
- De Smidt, P.C.; Le Doan, T.; de Falco, S.; van Berkel, T.J.C. Association of Antisense Oligonucleotides with Lipoproteins Prolongs the Plasma Half-Life and Modifies the Tissue Distribution. Nucleic Acids Res. 1991, 19, 4695–4700.
- Wolfrum, C.; Shi, S.; Jayaprakash, K.N.; Jayaraman, M.; Wang, G.; Pandey, R.K.; Rajeev, K.G.; Nakayama, T.; Charrise, K.; Ndungo, E.M.; et al. Mechanisms and Optimization of in Vivo Delivery of Lipophilic SiRNAs. Nat. Biotechnol. 2007, 25, 1149–1157.
- Jäschke, A.; Fürste, J.P.; Cech, D.; Erdmann, V.A. Automated Incorporation of Polyethylene Glycol into Synthetic Oligonucleotides. Tetrahedron Lett. 1993, 34, 301–304.
- Jäschke, A.; Fürste, J.P.; Nordhoff, E.; Hillenkamp, F.; Cech, D.; Erdmann, V.A. Synthesis and Properties of Oligodeoxyribonucleotide—Polyethylene Glycol Conjugates. Nucleic Acids Res. 1994, 22, 4810–4817.
- Bonora, G.M.; Ivanova, E.; Zarytova, V.; Burcovich, B.; Veronese, F.M. Synthesis and Characterization of High-Molecular Mass Polyethylene Glycol-Conjugated Oligonucleotides. Bioconjug. Chem. 1997, 8, 793–797.
- Burcovich, B.; Veronese, F.M.; Zarytova, V.; Bonora, G.M. Branched Polyethylene Glycol (BPEG) Conjugated Antisense Oligonucleotides. Nucleosides Nucleotides 1998, 17, 1567–1570.
- Gragoudas, E.S.; Adamis, A.P.; Cunningham Jr, E.T.; Feinsod, M.; Guyer, D.R. Pegaptanib for Neovascular Age-Related Macular Degeneration. N. Engl. J. Med. 2004, 351, 2805–2816.
- Ng, E.W.M.; Shima, D.T.; Calias, P.; Cunningham, E.T.; Guyer, D.R.; Adamis, A.P. Pegaptanib, a Targeted Anti-VEGF Aptamer for Ocular Vascular Disease. Nat. Rev. Drug Discov. 2006, 5, 123–132.
- Fire, A.; Xu, S.; Montgomery, M.K.; Kostas, S.A.; Driver, S.E.; Mello, C.C. Potent and Specific Genetic Interference by Double-Stranded RNA in Caenorhabditis Elegans. Nature 1998, 391, 806–811.
- Elbashir, S.M.; Harborth, J.; Lendeckel, W.; Yalcin, A.; Weber, K.; Tuschl, T. Duplexes of 21-Nucleotide RNAs Mediate RNA Interference in Cultured Mammalian Cells. Nature 2001, 411, 494–498.
- Dong, Y.; Siegwart, D.J.; Anderson, D.G. Strategies, Design, and Chemistry in SiRNA Delivery Systems. Adv. Drug Deliv. Rev. 2019, 144, 133–147.
- Ha, M.; Kim, V.N. Regulation of MicroRNA Biogenesis. Nat. Rev. Mol. Cell Biol. 2014, 15, 509–524.
- Soutscheck, J.; Akinc, A.; Bramlage, B.; Charisse, K.; Constien, R.; Donoghue, M.; Elbashir, S.; Geick, A.; Hadwiger, P.; Harborth, J.; et al. Therapeutic Silencing of an Endogenous Gene by Systemic Administration of Modified SiRNAs. Nature 2004, 432, 173–178.
- Zimmermann, T.S.; Lee, A.C.; Akinc, A.; Bramlage, B.; Bumcrot, D.; Fedoruk, M.N.; Harborth, J.; Heyes, J.A.; Jeffs, L.B.; John, M.; et al. RNAi-Mediated Gene Silencing in Non-Human Primates. Nature 2006, 441, 111–114.
- Heyes, J.; Palmer, L.; Bremner, K.; MacLachlan, I. Cationic Lipid Saturation Influences Intracellular Delivery of Encapsulated Nucleic Acids. J. Control. Release 2005, 107, 276–287.
- Akinc, A.; Zumbuehl, A.; Goldberg, M.; Leshchiner, E.S.; Busini, V.; Hossain, N.; Bacallado, S.A.; Nguyen, D.N.; Fuller, J.; Alvarez, R. A Combinatorial Library of Lipid-like Materials for Delivery of RNAi Therapeutics. Nat. Biotechnol. 2008, 26, 561–569.
- Semple, S.C.; Akinc, A.; Chen, J.; Sandhu, A.P.; Mui, B.L.; Cho, C.K.; Sah, D.W.Y.; Stebbing, D.; Crosley, E.J.; Yaworski, E. Rational Design of Cationic Lipids for SiRNA Delivery. Nat. Biotechnol. 2010, 28, 172–176.
- Ueno, Y.; Kawada, K.; Naito, T.; Shibata, A.; Yoshikawa, K.; Kim, H.-S.; Wataya, Y.; Kitade, Y. Synthesis and Silencing Properties of SiRNAs Possessing Lipophilic Groups at Their 3′-Termini. Bioorg. Med. Chem. 2008, 16, 7698–7704.
- Lorenz, C.; Hadwiger, P.; John, M.; Vornlocher, H.-P.; Unverzagt, C. Steroid and Lipid Conjugates of SiRNAs to Enhance Cellular Uptake and Gene Silencing in Liver Cells. Bioorg. Med. Chem. Lett. 2004, 14, 4975–4977.
- Nishina, T.; Numata, J.; Nishina, K.; Yoshida-Tanaka, K.; Nitta, K.; Piao, W.; Iwata, R.; Ito, S.; Kuwahara, H.; Wada, T. Chimeric Antisense Oligonucleotide Conjugated to α-Tocopherol. Mol. Ther. Acids 2015, 4, e220.
- Aviñó, A.; Ocampo, S.M.; Perales, J.C.; Eritja, R. Synthesis and in Vitro Inhibition Properties of SiRNA Conjugates Carrying Acridine and Quindoline Moieties. Chem. Biodivers. 2012, 9, 557–566.
- Ugarte-Uribe, B.; Grijalvo, S.; Busto, J.V.; Martín, C.; Eritja, R.; Goñi, F.M.; Alkorta, I. Double-Tailed Lipid Modification as a Promising Candidate for Oligonucleotide Delivery in Mammalian Cells. Biochim. Biophys. Acta (BBA)-General Subj. 2013, 1830, 4872–4884.
- Ugarte-Uribe, B.; Grijalvo, S.; Pertíñez, S.N.; Busto, J.V.; Martín, C.; Alagia, A.; Goñi, F.M.; Eritja, R.; Alkorta, I. Lipid-Modified Oligonucleotide Conjugates: Insights into Gene Silencing, Interaction with Model Membranes and Cellular Uptake Mechanisms. Bioorg. Med. Chem. 2017, 25, 175–186.
- Ries, O.; Löffler, P.M.G.; Vogel, S. Convenient Synthesis and Application of Versatile Nucleic Acid Lipid Membrane Anchors in the Assembly and Fusion of Liposomes. Org. Biomol. Chem. 2015, 13, 9673–9680.
- Liu, H.; Zhu, Z.; Kang, H.; Wu, Y.; Sefan, K.; Tan, W. DNA-based Micelles: Synthesis, Micellar Properties and Size-dependent Cell Permeability. Chem. Eur. J. 2010, 16, 3791–3797.
- Patwa, A.; Gissot, A.; Bestel, I.; Barthélémy, P. Hybrid Lipid Oligonucleotide Conjugates: Synthesis, Self-Assemblies and Biomedical Applications. Chem. Soc. Rev. 2011, 40, 5844–5854.
- Grijalvo, S.; Ocampo, S.M.; Perales, J.C.; Eritja, R. Preparation of Lipid-Conjugated SiRNA Oligonucleotides for Enhanced Gene Inhibition in Mammalian Cells. Methods Mol. Biol. 2021, 2282, 119–136.
- Campbell, A.; Morris, G.; Heller, J.P.; Langa, E.; Brindley, E.; Worm, J.; Jensen, M.A.; Miller, M.T.; Henshall, D.C.; Reschke, C.R. Antagomir-Mediated Suppression of MicroRNA-134 Reduces Kainic Acid-Induced Seizures in Immature Mice. Sci. Rep. 2021, 11, 340.
- Haftmann, C.; Riedel, R.; Porstner, M.; Wittmann, J.; Chang, H.-D.; Radbruch, A.; Mashreghi, M.-F. Direct Uptake of Antagomirs and Efficient Knockdown of MiRNA in Primary B and T Lymphocytes. J. Immunol. Methods 2015, 426, 128–133.
- Maschmeyer, P.; Petkau, G.; Siracusa, F.; Zimmermann, J.; Zügel, F.; Kühl, A.A.; Lehmann, K.; Schimmelpfennig, S.; Weber, M.; Haftmann, C. Selective Targeting of Pro-Inflammatory Th1 Cells by MicroRNA-148a-Specific Antagomirs in Vivo. J. Autoimmun. 2018, 89, 41–52.
- Velu, C.S.; Grimes, H.L. Utilizing AntagomiR (Antisense MicroRNA) to Knock down MicroRNA in Murine Bone Marrow Cells. Methods Mol. Biol. 2012, 928, 185–195.
- Koutsoudakis, G.; Paris de León, A.; Herrera, C.; Dorner, M.; Pérez-Vilaró, G.; Lyonnais, S.; Grijalvo, S.; Eritja, R.; Meyerhans, A.; Mirambeau, G. Oligonucleotide-Lipid Conjugates Forming G-Quadruplex Structures Are Potent and Pangenotypic Hepatitis C Virus Entry Inhibitors in Vitro and Ex Vivo. Antimicrob. Agents Chemother. 2017, 61, e02354-16.
- Musumeci, D.; Montesarchio, D. Synthesis of a Cholesteryl-HEG Phosphoramidite Derivative and Its Application to Lipid-Conjugates of the Anti-HIV 5’TGGGAG3’Hotoda’s Sequence. Molecules 2012, 17, 12378–12392.
- Wolfe, J.L.; Goodchild, J. Modulation of Tetraplex Formation by Chemical Modifications of a G4-Containing Phosphorothioate Oligonucleotide. J. Am. Chem. Soc. 1996, 118, 6301–6302.
- Godeau, G.; Staedel, C.; Barthéleémy, P. Lipid-Conjugated Oligonucleotides via “Click Chemistry” Efficiently Inhibit Hepatitis C Virus Translation. J. Med. Chem. 2008, 51, 4374–4376.
- Terstappen, G.C.; Meyer, A.H.; Bell, R.D.; Zhang, W. Strategies for Delivering Therapeutics across the Blood–Brain Barrier. Nat. Rev. Drug Discov. 2021, 20, 362–383.
- Alterman, J.F.; Hall, L.M.; Coles, A.H.; Hassler, M.R.; Didiot, M.-C.; Chase, K.; Abraham, J.; Sottosanti, E.; Johnson, E.; Sapp, E. Hydrophobically Modified SiRNAs Silence Huntingtin MRNA in Primary Neurons and Mouse Brain. Mol. Ther. Acids 2015, 4, e266.
- Ly, S.; Navaroli, D.M.; Didiot, M.-C.; Cardia, J.; Pandarinathan, L.; Alterman, J.F.; Fogarty, K.; Standley, C.; Lifshitz, L.M.; Bellve, K.D. Visualization of Self-Delivering Hydrophobically Modified SiRNA Cellular Internalization. Nucleic Acids Res. 2017, 45, 15–25.
- Nikan, M.; Osborn, M.F.; Coles, A.H.; Godinho, B.M.D.C.; Hall, L.M.; Haraszti, R.A.; Hassler, M.R.; Echeverria, D.; Aronin, N.; Khvorova, A. Docosahexaenoic Acid Conjugation Enhances Distribution and Safety of SiRNA upon Local Administration in Mouse Brain. Mol. Ther. Acids 2016, 5, e344.
- Haraszti, R.A.; Miller, R.; Didiot, M.-C.; Biscans, A.; Alterman, J.F.; Hassler, M.R.; Roux, L.; Echeverria, D.; Sapp, E.; DiFiglia, M. Optimized Cholesterol-SiRNA Chemistry Improves Productive Loading onto Extracellular Vesicles. Mol. Ther. 2018, 26, 1973–1982.
- Biscans, A.; Haraszti, R.A.; Echeverria, D.; Miller, R.; Didiot, M.-C.; Nikan, M.; Roux, L.; Aronin, N.; Khvorova, A. Hydrophobicity of Lipid-Conjugated SiRNAs Predicts Productive Loading to Small Extracellular Vesicles. Mol. Ther. 2018, 26, 1520–1528.
- Zhang, X.; Gubu, A.; Xu, J.; Yan, N.; Su, W.; Feng, D.; Wang, Q.; Tang, X. Tetrazine-Induced Bioorthogonal Activation of Vitamin E-Modified SiRNA for Gene Silencing. Molecules 2022, 27, 4377.
- Biscans, A.; Coles, A.; Haraszti, R.; Echeverria, D.; Hassler, M.; Osborn, M.; Khvorova, A. Diverse Lipid Conjugates for Functional Extra-Hepatic SiRNA Delivery in Vivo. Nucleic Acids Res. 2019, 47, 1082–1096.
- Osborn, M.F.; Khvorova, A. Improving SiRNA Delivery in Vivo through Lipid Conjugation. Nucleic Acid Ther. 2018, 28, 128–136.
- Biscans, A.; Caiazzi, J.; Davis, S.; McHugh, N.; Sousa, J.; Khvorova, A. The Chemical Structure and Phosphorothioate Content of Hydrophobically Modified SiRNAs Impact Extrahepatic Distribution and Efficacy. Nucleic Acids Res. 2020, 48, 7665–7680.
- Ly, S.; Echeverria, D.; Sousa, J.; Khvorova, A. Single-Stranded Phosphorothioated Regions Enhance Cellular Uptake of Cholesterol-Conjugated SiRNA but Not Silencing Efficacy. Mol. Ther. Acids 2020, 21, 991–1005.
- Biscans, A.; Coles, A.; Echeverria, D.; Khvorova, A. The Valency of Fatty Acid Conjugates Impacts SiRNA Pharmacokinetics, Distribution, and Efficacy in Vivo. J. Control. Release 2019, 302, 116–125.
- Osborn, M.F.; Coles, A.H.; Biscans, A.; Haraszti, R.A.; Roux, L.; Davis, S.; Ly, S.; Echeverria, D.; Hassler, M.R.; Godinho, B.M.D.C. Hydrophobicity Drives the Systemic Distribution of Lipid-Conjugated SiRNAs via Lipid Transport Pathways. Nucleic Acids Res. 2019, 47, 1070–1081.
- Hassler, M.R.; Turanov, A.A.; Alterman, J.F.; Haraszti, R.A.; Coles, A.H.; Osborn, M.F.; Echeverria, D.; Nikan, M.; Salomon, W.E.; Roux, L. Comparison of Partially and Fully Chemically-Modified SiRNA in Conjugate-Mediated Delivery in Vivo. Nucleic Acids Res. 2018, 46, 2185–2196.
- Brown, K.M.; Nair, J.K.; Janas, M.M.; Anglero-Rodriguez, Y.I.; Dang, L.T.H.; Peng, H.; Theile, C.S.; Castellanos-Rizaldos, E.; Brown, C.; Foster, D. Expanding RNAi Therapeutics to Extrahepatic Tissues with Lipophilic Conjugates. Nat. Biotechnol. 2022, 40, 1500–1508.
- Østergaard, M.E.; Jackson, M.; Low, A.; Chappell, A.E.; Lee, R.G.; Peralta, R.Q.; Yu, J.; Kinberger, G.A.; Dan, A.; Carty, R. Conjugation of Hydrophobic Moieties Enhances Potency of Antisense Oligonucleotides in the Muscle of Rodents and Non-Human Primates. Nucleic Acids Res. 2019, 47, 6045–6058.
- Prakash, T.P.; Mullick, A.E.; Lee, R.G.; Yu, J.; Yeh, S.T.; Low, A.; Chappell, A.E.; Østergaard, M.E.; Murray, S.; Gaus, H.J. Fatty Acid Conjugation Enhances Potency of Antisense Oligonucleotides in Muscle. Nucleic Acids Res. 2019, 47, 6029–6044.
- Wang, S.; Allen, N.; Prakash, T.P.; Liang, X.; Crooke, S.T. Lipid Conjugates Enhance Endosomal Release of Antisense Oligonucleotides Into Cells. Nucleic Acid Ther. 2019, 29, 245–255.
- Relizani, K.; Echevarría, L.; Zarrouki, F.; Gastaldi, C.; Dambrune, C.; Aupy, P.; Haeberli, A.; Komisarski, M.; Tensorer, T.; Larcher, T. Palmitic Acid Conjugation Enhances Potency of Tricyclo-DNA Splice Switching Oligonucleotides. Nucleic Acids Res. 2022, 50, 17–34.
- Marchesi, E.; Cortesi, R.; Preti, L.; Rimessi, P.; Sguizzato, M.; Bovolenta, M.; Perrone, D. Antisense Oligonucleotides Conjugated with Lipophilic Compounds: Synthesis and In Vitro Evaluation of Exon Skipping in Duchenne Muscular Dystrophy. Int. J. Mol. Sci. 2022, 23, 4270.
- Anderson, E.J.; Rouphael, N.G.; Widge, A.T.; Jackson, L.A.; Roberts, P.C.; Makhene, M.; Chappell, J.D.; Denison, M.R.; Stevens, L.J.; Pruijssers, A.J. Safety and Immunogenicity of SARS-CoV-2 MRNA-1273 Vaccine in Older Adults. N. Engl. J. Med. 2020, 383, 2427–2438.
- Polack, F.P.; Thomas, S.J.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Perez, J.L.; Marc, G.P.; Moreira, E.D.; Zerbini, C. Safety and Efficacy of the BNT162b2 MRNA Covid-19 Vaccine. N. Engl. J. Med. 2020, 383, 2603–2615.
- Hou, X.; Zaks, T.; Langer, R.; Dong, Y. Lipid Nanoparticles for MRNA Delivery. Nat. Rev. Mater. 2021, 37, 237–246.
- Cheng, Q.; Wei, T.; Farbiak, L.; Johnson, L.T.; Dilliard, S.A.; Siegwart, D.J. Selective Organ Targeting (SORT) Nanoparticles for Tissue-Specific MRNA Delivery and CRISPR–Cas Gene Editing. Nat. Nanotechnol. 2020, 15, 313–320.
More
Information
Subjects:
Chemistry, Organic
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.9K
Entry Collection:
Organic Synthesis
Revisions:
2 times
(View History)
Update Date:
01 Feb 2023
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No