
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Abul Hossain | -- | 3831 | 2022-08-23 13:50:55 | | | |
| 2 | Peter Tang | Meta information modification | 3831 | 2022-08-24 02:44:12 | | |
Video Upload Options
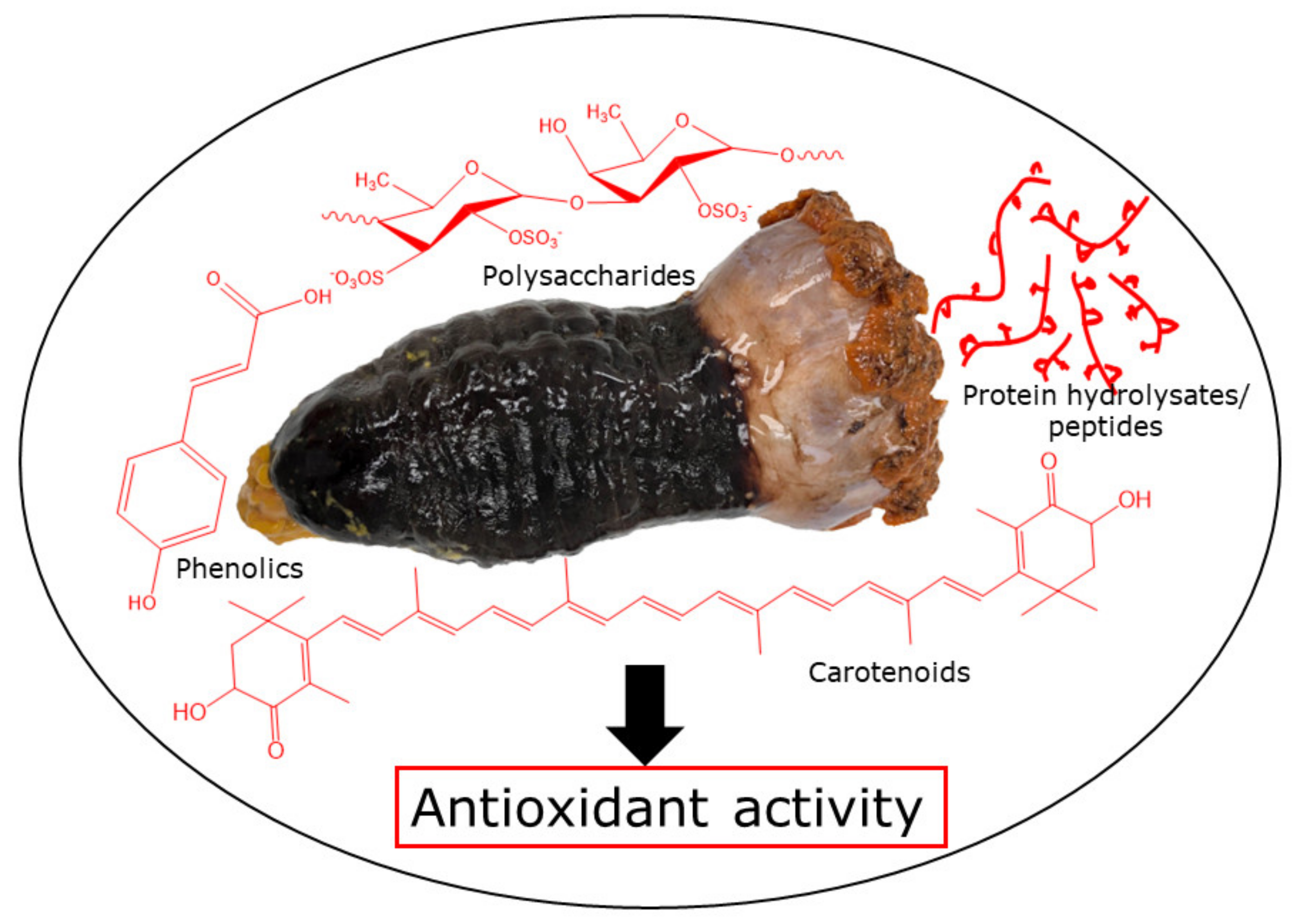
Sea cucumbers are considered a luxury food item and used locally in traditional medication due to their impressive nutritional profile and curative effects. Sea cucumbers contain a wide range of bioactive compounds, namely phenolics, polysaccharides, proteins (collagen and peptides), carotenoids, and saponins, demonstrating strong antioxidant and other activities. In particular, phenolic compounds, mainly phenolic acids and flavonoids, are abundant in this marine invertebrate and exhibit antioxidant activity. Protein hydrolysates and peptides obtained from sea cucumbers exhibit antioxidant potential, mainly dependent on the amino acid compositions and sequences as well as molecular weight, displayed for those of ≤20 kDa. Moreover, the antioxidant activity of sea cucumber polysaccharides, including fucosylated chondroitin sulfate and fucan, is a combination of numerous factors and is mostly associated with molecular weight, degree of sulfation, and type of major sugars. However, the activity of these bioactive compounds typically depends on the sea cucumber species, harvesting location, food habit, body part, and processing methods employed.
1. Introduction
2. Bioactive Compounds of Sea Cucumbers and Their Antioxidant Activity
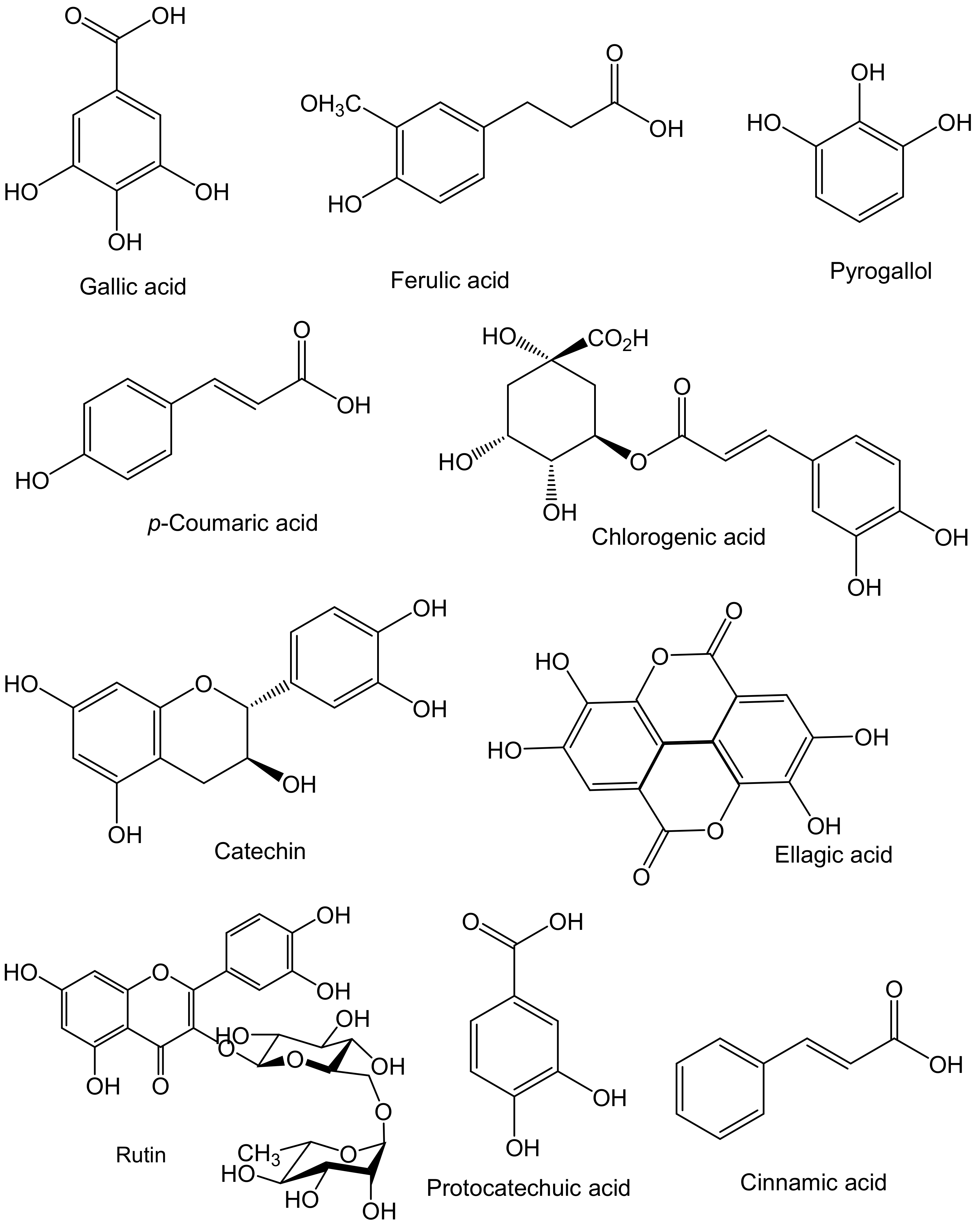
2.1. Antioxidant Potential of Sea Cucumber Phenolics and Their Beneficial Effects on Human Health
2.2. Antioxidant Potential of Protein Hydrolysates and Peptides and Their Health Benefits
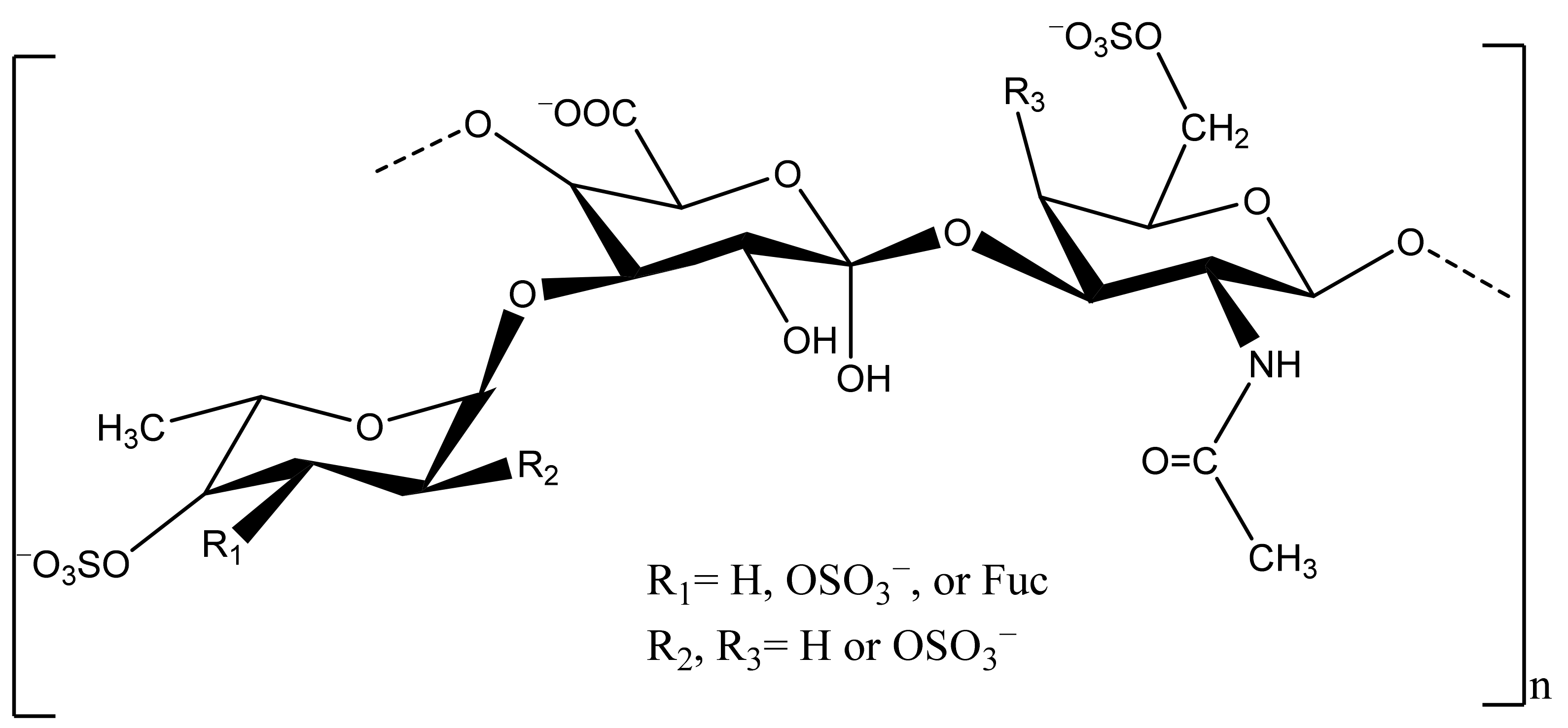
2.3. Antioxidant Potential of Sea Cucumber Polysaccharides
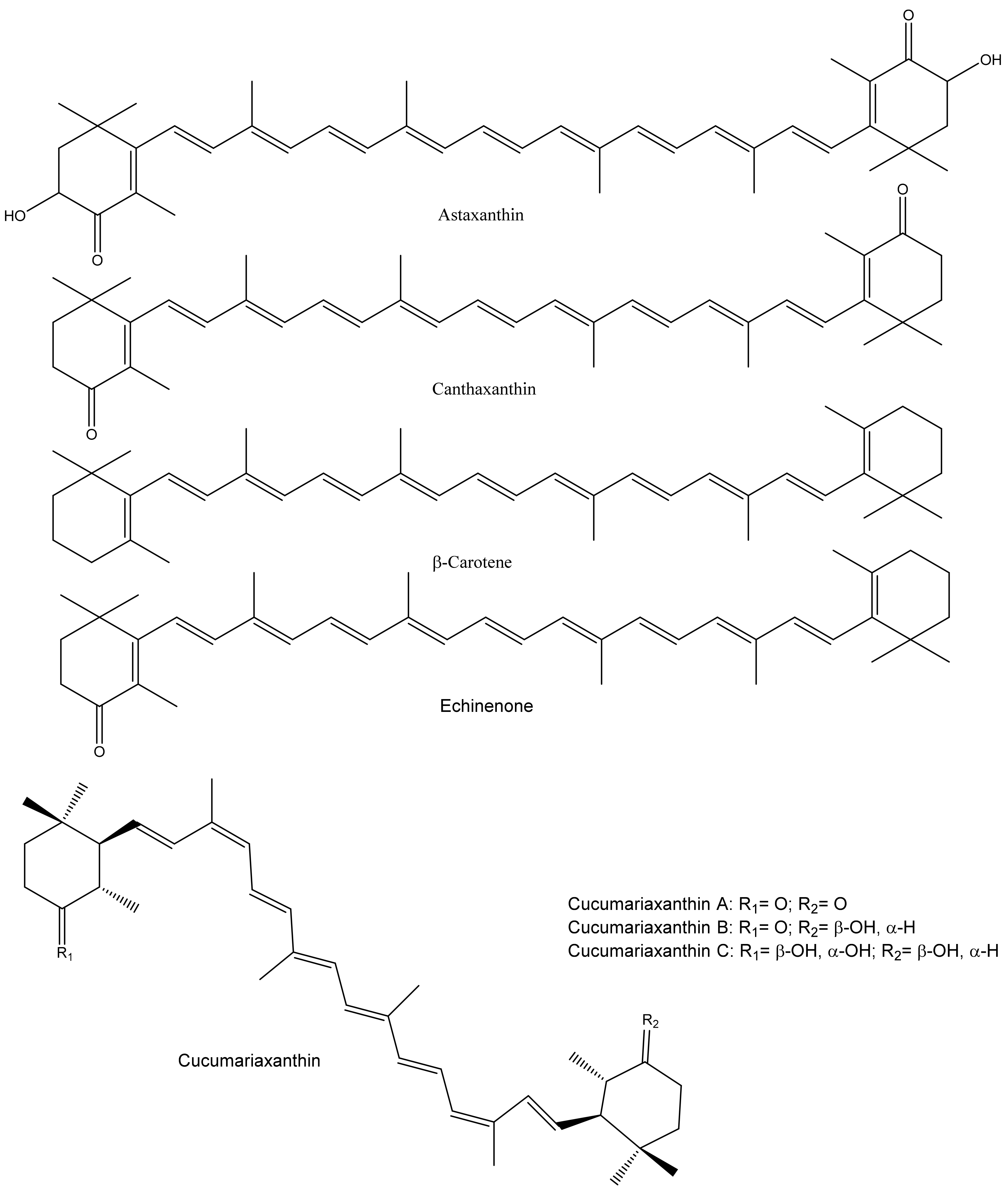
2.4. Antioxidant Potential of Carotenoids and Physiological Effects of PUFAs
2.5. Antioxidant Potential of Other Bioactive Compounds of Sea Cucumber
References
- Wen, J.; Hu, C.; Fan, S. Chemical composition and nutritional quality of sea cucumbers. J. Sci. Food Agric. 2010, 90, 2469–2474.
- Hossain, A.; Dave, D.; Shahidi, F. Northern sea cucumber (Cucumaria frondosa): A potential candidate for functional food, nutraceutical, and pharmaceutical sector. Mar. Drugs 2020, 18, 274.
- Gajdosechova, Z.; Palmer, C.H.; Dave, D.; Jiao, G.; Zhao, Y.; Tan, Z.; Chisholm, J.; Zhang, J.; Stefanova, R.; Hossain, A.; et al. Arsenic speciation in sea cucumbers: Identification and quantitation of water-extractable species. Environ. Pollut. 2020, 266, 1–10.
- Chen, S.; Xue, C.; Yin, L.; Tang, Q.; Yu, G.; Chai, W. Comparison of structures and anticoagulant activities of fucosylated chondroitin sulfates from different sea cucumbers. Carbohydr. Polym. 2011, 83, 688–696.
- Hossain, A.; Dave, D.; Shahidi, F. Effect of high-pressure processing (HPP) on phenolics of North Atlantic sea cucumber (Cucumaria frondosa). J. Agric. Food Chem. 2022, 70, 3489–3501.
- Bordbar, S.; Anwar, F.; Saari, N. High-value components and bioactives from sea cucumbers for functional foods—A review. Mar. Drugs 2011, 9, 1761–1805.
- Shahidi, F.; Naczk, M. Phenolics in Food and Nutraceuticals, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2004.
- Pereira, D.M.; Valentão, P.; Pereira, J.A.; Andrade, P.B. Phenolics: From Chemistry to Biology. Molecules 2009, 14, 2202–2211.
- Shahidi, F.; Zhong, Y. Measurement of antioxidant activity. J. Funct. Foods 2015, 18, 757–781.
- Hossain, A.; Moon, H.K.; Kim, J.-K. Antioxidant properties of Korean major persimmon (Diospyros kaki) leaves. Food Sci. Biotechnol. 2018, 27, 177–184.
- Shahidi, F.; Ho, C.T. Antioxidant measurement and applications: An overview. In Antioxidant Measurement and Applications; American Chemical Society: Washington, DC, USA, 2007; pp. 2–7.
- Shahidi, F.; Hossain, A. Bioactives in spices, and spice oleoresins: Phytochemicals and their beneficial effects in food preservation and health promotion. J. Food Bioact. 2018, 3, 8–75.
- Shahidi, F.; Hossain, A. Preservation of aquatic food using edible films and coatings containing essential oils: A review. Crit. Rev. Food Sci. Nutr. 2020, 62, 66–105.
- Balasundram, N.; Sundram, K.; Samman, S. Phenolic compounds in plants and agri-industrial by-products : Antioxidant activity, occurrence, and potential uses. Food Chem. 2006, 99, 191–203.
- Shahidi, F. Antioxidants: Principles and applications. In Handbook of Antioxidants for Food Preservation; Shahidi, F., Ed.; Woodhead Publishing: Cambridge, UK, 2015; pp. 1–14.
- Shahidi, F.; Ambigaipalan, P. Phenolics and polyphenolics in foods, beverages and spices: Antioxidant activity and health effects—A review. J. Funct. Foods 2015, 18, 820–897.
- Shahidi, F.; Varatharajan, V.; Oh, W.Y.; Peng, H. Phenolic compounds in agri-food by-products, their bioavailability and health effects. J. Food Bioact. 2019, 5, 57–119.
- Zhong, Y.; Khan, M.A.; Shahidi, F. Compositional characteristics and antioxidant properties of fresh and processed sea cucumber (Cucumaria frondosa). J. Agric. Food Chem. 2007, 55, 1188–1192.
- Ceesay, A.; Nor Shamsudin, M.; Aliyu-Paiko, M.; Ismail, I.S.; Nazarudin, M.F.; Mohamed Alipiah, N. Extraction and characterization of organ components of the Malaysian sea cucumber Holothuria leucospilota yielded bioactives exhibiting diverse properties. BioMed Res. Int. 2019, 2019, 2640684.
- Althunibat, O.Y.; Hashim, R.B.; Taher, M.; Daud, J.M.; Ikeda, M.-A.; Zali, B.I. In vitro antioxidant and antiproliferative activities of three Malaysian sea cucumber species. Eur. J. Sci. Res. 2009, 37, 376–387.
- Nobsathian, S.; Tuchinda, P.; Sobhon, P.; Tinikul, Y.; Poljaroen, J.; Tinikul, R.; Sroyraya, M.; Poomton, T.; Chaichotranunt, S. An antioxidant activity of the whole body of Holothuria scabra. Chem. Biol. Technol. Agric. 2017, 4, 17–21.
- Wulandari, D.A.; Gustini, N.; Murniasih, T.; Bayu, A.; Sari, M.; Syahputra, G.; Harahap, I.A.; Rasyid, A.; Moria, S.B.; Rahmawati, S.I.; et al. Nutritional value and biological activities of sea cucumber Holothuria scabra cultured in the open pond system. J. Aquat. Food Prod. Technol. 2022, 31, 599–614.
- Hossain, A.; Yeo, J.D.; Dave, D.; Shahidi, F. Phenolic compounds and antioxidant capacity of sea cucumber (Cucumaria frondosa) processing discards as affected by high-pressure processing (HPP). Antioxidants 2022, 11, 337.
- Mamelona, J.; Pelletier, É.; Girard-Lalancette, K.; Legault, J.; Karboune, S.; Kermasha, S. Quantification of phenolic contents and antioxidant capacity of Atlantic sea cucumber, Cucumaria frondosa. Food Chem. 2007, 104, 1040–1047.
- Mamelona, J.; Pelletier, É. Producing high antioxidant activity extracts from echinoderm by products by using pressured liquid extraction. Biotechnology 2010, 9, 523–528.
- Ridhowati, S.; Zakaria, F.R.; Syah, D.; Chasanah, E. Anticancer and antioxidant activities from sea cucumber (Stichopus variegatus) flour dried vacuum oven. Pertanika J. Trop. Agric. Sci. 2018, 41, 1125–1138.
- Husni, A.; Shin, I.S.; You, S.G.; Chung, D. Antioxidant properties of water and aqueous ethanol extracts and their crude saponin fractions from a far-eastern sea cucumber, Stichopus japonicus. Food Sci. Biotechnol. 2009, 18, 419–424.
- Safari, R.; Yaghoubzadeh, Z. Antioxidant activity of bioactive peptides extracted from sea cucumber (Holothuria leucospilata). Int. J. Pept. Res. Ther. 2020, 26, 2393–2398.
- Senadheera, T.R.L.; Dave, D.; Shahidi, F. Sea cucumber derived type I collagen: A comprehensive review. Mar. Drugs 2020, 18, 471.
- Senadheera, T.R.L.; Dave, D.; Shahidi, F. Antioxidant potential and physicochemical properties of protein hydrolysates from body parts of North Atlantic sea cucumber (Cucumaria frondosa). Food Prod. Process. Nutr. 2021, 3, 3.
- Shahidi, F.; Zhong, Y. Bioactive peptides. J. AOAC Int. 2008, 91, 914–931.
- Jin, H.-X.; Xu, H.-P.; Li, Y.; Zhang, Q.-W.; Xie, H. Preparation and evaluation of peptides with potential antioxidant activity by microwave assisted enzymatic hydrolysis of collagen from sea cucumber Acaudina molpadioides obtained from Zhejiang province in China. Mar. Drugs 2019, 17, 169.
- Yan, M.; Tao, H.; Qin, S. Effect of enzyme type on the antioxidant activities and functional properties of enzymatic hydrolysates from sea cucumber (Cucumaria frondosa) viscera. J. Aquat. Food Prod. Technol. 2016, 25, 940–952.
- Zhang, Y.; He, S.; Bonneil, É.; Simpson, B.K. Generation of antioxidative peptides from Atlantic sea cucumber using alcalase versus trypsin: In vitro activity, de novo sequencing, and in silico docking for in vivo function prediction. Food Chem. 2020, 306, 125581.
- Mamelona, J.; Saint-Louis, R.; Pelletier, É. Nutritional composition and antioxidant properties of protein hydrolysates prepared from echinoderm byproducts. Int. J. Food Sci. Technol. 2010, 45, 147–154.
- Wang, T.; Zheng, L.; Wang, S.; Zhao, M.; Liu, X. Anti-diabetic and anti-hyperlipidemic effects of sea cucumber (Cucumaria frondosa) gonad hydrolysates in type II diabetic rats. Food Sci. Hum. Wellness 2022, 11, 1614–1622.
- Tripoteau, L.; Bedoux, G.; Gagnon, J.; Bourgougnon, N. In vitro antiviral activities of enzymatic hydrolysates extracted from byproducts of the Atlantic holothurian Cucumaria frondosa. Process Biochem. 2015, 50, 867–875.
- Lin, L.; Yang, K.; Zheng, L.; Zhao, M.; Sun, W.; Zhu, Q.; Liu, S. Anti-aging effect of sea cucumber (Cucumaria frondosa) hydrolysate on fruit flies and d-galactose-induced aging mice. J. Funct. Foods 2018, 47, 11–18.
- Liu, X.; Sun, Z.; Zhang, M.; Meng, X.; Xia, X.; Yuan, W.; Xue, F.; Liu, C. Antioxidant and antihyperlipidemic activities of polysaccharides from sea cucumber Apostichopus japonicus. Carbohydr. Polym. 2012, 90, 1664–1670.
- Zhu, B.W.; Zhou, D.Y.; Li, T.; Yan, S.; Yang, J.F.; Li, D.M.; Dong, X.P.; Murata, Y. Chemical composition and free radical scavenging activities of a sulphated polysaccharide extracted from abalone gonad (Haliotis Discus Hannai Ino). Food Chem. 2010, 121, 712–718.
- Qin, Y.; Yuan, Q.; Zhang, Y.; Li, J.; Zhu, X.; Wen, J.; Liu, J.; Zhao, L.; Zhao, J.; Zhao, L. Enzyme-assisted extraction optimization, characterization and antioxidant activity of polysaccharides from sea cucumber Phyllophorus proteus. Molecules 2018, 23, 590.
- Xie, J.H.; Wang, Z.J.; Shen, M.Y.; Nie, S.P.; Gong, B.; Li, H.S.; Zhao, Q.; Li, W.J.; Xie, M.Y. Sulfated modification, characterization and antioxidant activities of polysaccharide from Cyclocarya paliurus. Food Hydrocoll. 2016, 53, 7–15.
- Simal-Gandara, J.; Gao, L.; Xu, C.; Tao, X.; Zuo, Z.; Ning, Z.; Wang, L.; Gao, N.; Zhao, J. Structure elucidation of fucan sulfate from sea cucumber Holothuria fuscopunctata through a bottom-up strategy and the antioxidant activity analysis. Int. J. Mol. Sci. 2022, 23, 4488.
- Li, Q.; Jiang, S.; Shi, W.; Qi, X.; Song, W.; Mou, J.; Yang, J. Structure characterization, antioxidant and immunoregulatory properties of a novel fucoidan from the sea cucumber Stichopus chloronotus. Carbohydr. Polym. 2020, 231, 115767.
- Guo, X.; Ye, X.; Sun, Y.; Wu, D.; Wu, N.; Hu, Y.; Chen, S. Ultrasound effects on the degradation kinetics, structure, and antioxidant activity of sea cucumber fucoidan. J. Agric. Food Chem. 2014, 62, 1088–1095.
- Li, R.; Huahua, Y.U.; Yang, Y.; Song, L.; Rong’e, X.; Xiaolin, C.; Li, P. Sulfated polysaccharides with antioxidant and anticoagulant activity from the sea cucumber Holothuria fuscogliva. Chin. J. Oceanol. Limnol. 2017, 35, 763–769.
- Mou, J.; Li, Q.; Qi, X.; Yang, J. Structural comparison, antioxidant and anti-inflammatory properties of fucosylated chondroitin sulfate of three edible sea cucumbers. Carbohydr. Polym. 2018, 185, 41–47.
- Yu, L.; Xue, C.; Chang, Y.; Xu, X.; Ge, L.; Liu, G.; Wang, Y. Structure elucidation of fucoidan composed of a novel tetrafucose repeating unit from sea cucumber Thelenota ananas. Food Chem. 2014, 146, 113–119.
- Zou, S.; Pan, R.; Dong, X.; He, M.; Wang, C. Physicochemical properties and antioxidant activities of two fucosylated chondroitin sulfate from sea cucumber Acaudina molpadioidea and Holothuria nobilis. Process Biochem. 2016, 51, 650–658.
- Wang, J.; Shi, S.; Li, F.; Du, X.; Kong, B.; Wang, H.; Xia, X. Physicochemical properties and antioxidant activity of polysaccharides obtained from sea cucumber gonads via ultrasound-assisted enzymatic techniques. LWT Food Sci. Technol. 2022, 160, 113307.
- Qi, H.; Ji, X.; Liu, S.; Feng, D.; Dong, X.; He, B.; Srinivas, J.; Yu, C. Antioxidant and anti-dyslipidemic effects of polysaccharidic extract from sea cucumber processing liquor. Electron. J. Biotechnol. 2017, 28, 1–6.
- Matsuno, T.; Tsushima, M. Comparative biochemical studies of carotenoids in sea cucumber. Comp. Biochem. Physiol. 1995, 111, 597–605.
- Tsushima, M.; Fujiwara, Y.; Matsuno, T. Novel marine di-Z-carotenoids: Cucumariaxanthins A, B, and C from the sea cucumber Cucumaria japonica. J. Nat. Prod. 1996, 59, 30–34.
- Maoka, T.; Nakachi, S.; Kobayashi, R.; Mori, M.; Sakagami, Y. A new carotenoid, 9Z,9′Z-tetrahydroastaxanthin, from the sea cucumber Plesiocolochirus minutus. Tetrahedron Lett. 2015, 56, 5954–5955.
- Zakharenko, A.; Romanchenko, D.; Thinh, P.D.; Pikula, K.; Thuy Hang, C.T.; Yuan, W.; Xia, X.; Chaika, V.; Chernyshev, V.; Zakharenko, S.; et al. Features and advantages of supercritical CO2 extraction of sea cucumber Cucumaria frondosa japonica semper, 1868. Molecules 2020, 25, 4088.
- Chasanah, E.; Fawzya, Y.N.; Tarman, K.; Januar, H.I.; Nursid, M. Fatty acid profile, carotenoid content, and in vitro anticancer Activity of Karimunjawa and Lampung sea cucumber. Squalen Bull. Mar. Fish. Postharvest Biotechnol. 2016, 11, 117.
- Young, A.J.; Lowe, G.L. Carotenoids—antioxidant properties. Antioxidants 2018, 7, 28.
- Althunibat, O.; Ridzwan, B.; Taher, M.; Daud, J.; Jauhari Arief Ichwan, S.; Qaralleh, H. Antioxidant and cytotoxic properties of two sea cucumbers, Holothuria edulis Lesson and Stichopus horrens Selenka. Acta Biol. Hung. 2013, 64, 10–20.
- Hawas, U.W.; Abou El-Kassem, L.T.; Shaher, F.M.; Ghandourah, M.; Al-Farawati, R. Sulfated triterpene glycosides from the Saudi red sea cucumber Holothuria atra with antioxidant and cytotoxic activities. Thalassas 2021, 37, 817–824.
- Ansharullah; Tamrin; Patadjai, A.B.; Asranuddin. Powder production of sea cucumber (Holothuria scabra): Effect of processing methods on the antioxidant activities and physico-chemical characteristics. IOP Conf. Ser. Earth Environ. Sci. 2020, 443, 331–334.
- Nugroho, A.; Harahap, I.A.; Ardiansyah, A.; Bayu, A.; Rasyid, A.; Murniasih, T.; Setyastuti, A.; Putra, M.Y. Antioxidant and antibacterial activities in 21 species of Indonesian sea cucumbers. J. Food Sci. Technol. 2022, 59, 239–248.
- Rasyid, A.; Putra, M.Y.; Yasman. Free radical scavenging activity of five selected sea cucumbers collected from Lampung waters, Indonesia. IOP Conf. Ser. Earth Environ. Sci. 2021, 890, 012014.
- Wulandari, D.A.; Murniasih, T.; Sari, M.; Syahputra, G.; Rasyid, A.; Septiana, E.; Untari, F.; Harahap, I.A.; Ardiansyah, A.; Gustini, N.; et al. Characterization, antioxidant and antibacterial activity of cultivated sea cucumbers from Bali, Indonesia. IOP Conf. Ser. Earth Environ. Sci. 2021, 744, 012102.
- Wu, F.-J.; Xue, Y.; Tang, Q.-J.; Xu, J.; Du, L.; Xue, C.-H.; Takahashi, K.; Wang, Y.-M. The protective effects of cerebrosides from sea cucumber and starfish on the oxidative damage in PC12 cells. J. Oleo Sci. 2013, 62, 717–727.





