Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Tarek Bismar | -- | 1712 | 2022-05-12 16:33:41 | | | |
| 2 | Amina Yu | + 1 word(s) | 1713 | 2022-05-13 04:37:41 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Bismar, T.; Khosh Kish, E.; Choudhry, M.; Gamallat, Y.; M. Buharideen, S.; , . Proto-Oncogene ETS-Related Gene in Prostate Cancer. Encyclopedia. Available online: https://encyclopedia.pub/entry/22881 (accessed on 14 May 2026).
Bismar T, Khosh Kish E, Choudhry M, Gamallat Y, M. Buharideen S, . Proto-Oncogene ETS-Related Gene in Prostate Cancer. Encyclopedia. Available at: https://encyclopedia.pub/entry/22881. Accessed May 14, 2026.
Bismar, Tarek, Ealia Khosh Kish, Muhammad Choudhry, Yaser Gamallat, Sabrina M. Buharideen, . "Proto-Oncogene ETS-Related Gene in Prostate Cancer" Encyclopedia, https://encyclopedia.pub/entry/22881 (accessed May 14, 2026).
Bismar, T., Khosh Kish, E., Choudhry, M., Gamallat, Y., M. Buharideen, S., & , . (2022, May 12). Proto-Oncogene ETS-Related Gene in Prostate Cancer. In Encyclopedia. https://encyclopedia.pub/entry/22881
Bismar, Tarek, et al. "Proto-Oncogene ETS-Related Gene in Prostate Cancer." Encyclopedia. Web. 12 May, 2022.
Copy Citation
The ETS-related gene (ERG) is proto-oncogene that is classified as a member of the ETS transcription factor family, which has been found to be consistently overexpressed in about half of the patients with clinically significant prostate cancer (PCa). The overexpression of ERG can mostly be attributed to the fusion of the ERG and transmembrane serine protease 2 (TMPRSS2) genes, and this fusion is represented about 85% of all gene fusions observed in prostate cancer. Clinically, individuals with ERG gene fusion are mostly documented to have advanced tumor stages, increased mortality, and higher rates of metastasis in non-surgical cohorts.
gene fusion
prostate cancer
tumorigenesis
1. Structural Characteristics and Allosteric Autoinhibition of ERG
The ERG transcription factor has a primary structure that consists of 486 amino acids and a corresponding molecular weight of 54kDa [1]. A distinguishing characteristic of ETS family proteins is the presence of a DNA-binding domain called the ETS DNA binding domain (EBD). This EBD domain is made of 85 amino acids and contains 3 alpha-helices that are further supported by a 4-stranded anti-parallel beta sheet [2]. The EBD domain plays a critical role in DNA recognition as well as in AP-1 and co-activator recruitment [3]. Within the EBD, there are three highly conserved tryptophan residues that serve as a hydrophobic core to facilitate the helix–turn–helix binding domain in proteins [4]. ERG analysis by means peptide sequencing has also predicted the presence of phosphorylation sites for protein kinase C and a pointed (PNT) domain in the N-terminus [1]. The PNT domains are a part of a larger sterile alpha motif (SAM) family that is involved in many diverse protein–protein interactions that can allow for self-association [5]. This PNT domain has also been shown to facilitate the heterodimerization of ERG with other proteins, including other members of the ETS family, DNA-dependent kinases, AP-1 complex, and the androgen receptor (AR) [6].
2. The Function of ETS-Related Gene (ERG) in Normal Cell Types
ERG has a multitude of physiological functions that differ based on the type of cells or the organism’s developmental stage. During embryogenesis, ERG has been observed to be highly expressed in the mesoderm and endothelium, where it plays a crucial role in vasculogenesis and in the development of bone [7]. In adults, it regulates vascular homeostasis and angiogenesis by activating the transcription of endothelial specific genes, such as vascular endothelial (VE)-cadherin, an adhesion molecule that promotes vascular stability by maintaining and controlling endothelial cell contact [8]. In addition to this, VE-cadherin also plays a central role in cell proliferation and apoptosis and modulates endothelial growth factor receptor functions [9]. Other endothelial genes that are positively regulated by ERG include the vascular endothelial growth factor (VEGF), von Willebrand factor, and endoglin, which are all involved in endothelial cell differentiation and angiogenesis [4][10].
3. Prominent TMPRSS2-ERG Gene Fusion Found in PCa
In prostate cancer cells, a surprisingly common occurrence involves the fusion of ERG to TMPRSS2, which forms the fusion product of TMPRSS2-ERG. The most common mechanism by which these two genes fuse involves the deletion of intronic sequences on the long arm of chromosome 21 via an intron deletion between TMPRSS2 and ERG on chromosome 21q22.2-3 (Figure 1). This fusion mechanism has been identified as being prevalent in approximately 50% of prostate cancer patients [11]. The frequent occurrence of this fusion protein can be attributed to the presence of a homogenous deletion site that is present between ERG and TMPRSS2 [12]. Moreover, this deletion site is separated into two different classifications according to various start sites. In both of the deletion products, the 5′ end of the TMPRSS2 gene has been ligated to the 3′ end of ERG. TMPRSS2-ERG fusion results in ERG overexpression due to the androgen responsive promoter of the TMPSS2 gene allowing for the constitutive transcription of ERG, which has been shown to be correlated with increased cell proliferation, cell invasion, angiogenesis, and invasiveness in PCa cells [13][14]. In addition, this TMPRSS2-ERG fusion enhances the transcription and activates downstream oncogenes [15].
4. Functional ERG Overexpression in Prostate Cancer Cells
In prostate cancer cells, ERG overexpression increases the rate of epithelial to mesenchymal transitions via the EMT pathway, enhancing the ability of PCa cells to invade and metastasize. ERG achieves this by upregulating matrix metalloproteinases (MMPs), CXCR4, and Osteopontin (OPN), which have been correlated with higher rates of cell invasion and metastasis among patients [16]. Additionally, the TMPRSS2-ERG pathway reveals the epithelial to mesenchymal transition via the ZEB1/ZEB2 axis in PCa [17]. The constitutive expression of ERG also hyperactivates the inflammatory pathway in PCa cells by binding to Toll-like receptor 4; this activates the NF-kb pathway, increasing the transcription of target genes such as TNFA, IL6, BCLXL, BCL2, BCLXS, XIAP, and VEGF [18][19]. These proteins trigger tumor growth and progression by enhancing cell proliferation, survival, and angiogenesis. Tumor growth is further accentuated by the activation of the EZH2 promoter by ERG. This relieves the epigenetic inhibition of tumor suppressor genes such as NKX3.1, resulting in the constitutive expression of the TMPRSS2-ERG fusion gene. In addition to its role in regulating tumor cell invasion and proliferation, ERG also plays an important role in negatively regulating tumor cell differentiation by inhibiting the transcription of genes such as KLK3/PSA and SLC45A3/Prostein [20]. Altogether, the overexpression of ERG in prostate cancer is a key modulator of tumor progression and aggressiveness, as it can regulate the transcription of the proteins that mediate inflammation, cell invasion, differentiation, and oncogenesis [21].
5. TMPRSS2-ERG Fusion Upregulates CXCR4 Enhances Tumor Adhesion and Aggregation
Previous ones have been concluded that there are eight ERG/Ets factor binding sites near the promoter of chemokine receptor type 4 (CXCR4) (Figure 1) and that TMPRSS2-ERG expression enhances the function and expression of CXCR4 [22][23]. CXCL12 is a known CXCR4 receptor ligand, and the interactions between the two functions enhance tumor aggressiveness and increase the ability of cancer cells to adhere to the extracellular matrix [24][25]. The CXCR4/CXCL12 axis has been shown to increase MMP expression, which promotes cell migration and growth in PCa [22]. An analysis of PCa demographics revealed relatively higher CXCR4 expression during PCa progression and in metastatic bone tissue compared to benign PCa cells that were reported. In vitro, a direct correlation between TMPRSS-ERG and CXCR4 was observed when ERG was knocked down [25].
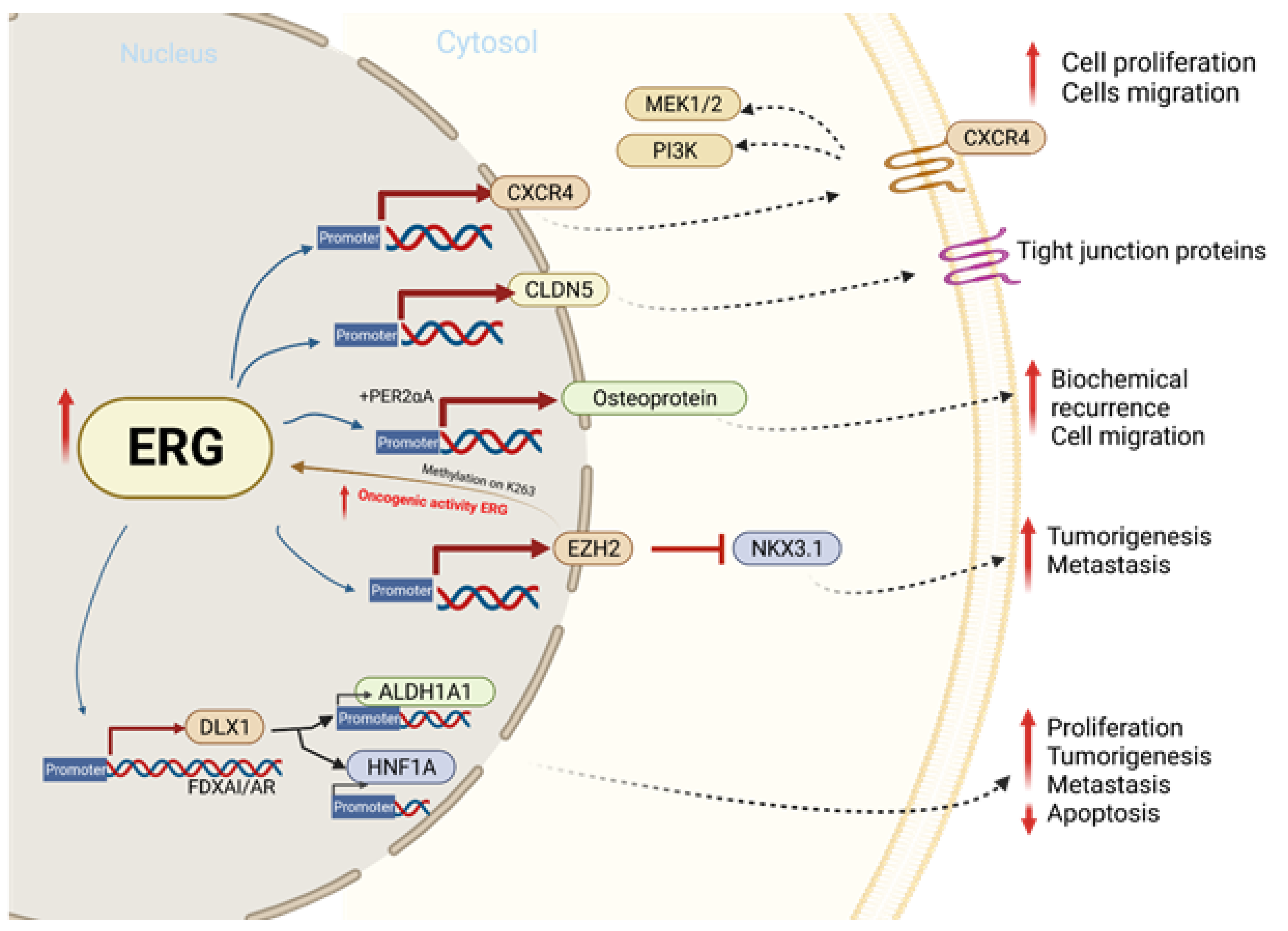
Figure 1. The molecular mechanism involved in ERG overexpression and related outcomes in tumor cell proliferation, migration, invasion, and metastasis.
6. TMPRSS2-ERG Binds to EMT Key Regulators ZEB1/ZEB2
Prior one was showed that TMPRSS2-ERG can directly bind to the Zinc finger e-box binding homeobox 1 (ZEB1) promoter and binds to ZEB2 modulators such as ILIR2 and SPINT1 and increases their overall expression [26]. ZEB1 and ZEB2 are members of the ZEB family of transcriptional factors and are key regulators in the EMT and disease progression pathways [27]. Furthermore, ZEB1 knockdown revealed a significant decline in the migration and invasion capacity of TMPRSS2-ERG-expressing cells.
7. EZH2 Enhances ERG Oncogenic Activity
It appears that the enhancer of zeste jomolog 2 (EZH2), which is a histone H3K27 methyltransferase, catalyzes the methylation of ERG at the lysine 263 residue. This interaction promotes the translocation and DNA binding of ERG in the nucleus, which results in the enhancement of the oncogenic activity of ERG [28][29]. It has been shown that the methylation of ERG at the lysine362 residue is associated with metastatic properties and increased tumorigenic characteristics in cell lines [28]. Further, these interactions seem to enhance the progression of PCa from non-invasive lesions to invasive adenocarcinomas in Erg/Pten mice [28].
8. ERG and Tight Junction Protein CLDN5
Recently it has been shown that ERG is positively correlated with the expression of an important tight junction protein known as Claudin 5 (CLDN5); it has been observed that there are two major ERG binding sites near the promoter of the CLDN5 gene, making ERG a direct transcriptional regulator of CLDN5. Moreover, CLDN5 is known as a member of the 24 tetraspan transmembrane protein family and one of the major components of the tight junction strands that are responsible for regulating barrier functions [30]. This protein is involved in many of the processes related to vascular homeostasis. Research has shown that the knockout of ERG expression in mice resulted in increased endothelial cell permeability due to decreased CLDN5 expression [31]. Interestingly, CLDN5 upregulation results in decreased the cell migration and invasion ability of lung cancer cells due to the decreased permeability of the cell membrane due to the enhancement of the CLDN5 tight junctions [32][33]. In addition to ERG’s function in maintaining vascular homeostasis, it also plays a critical role in maintaining the population of hematopoietic stem cells (HSC) by regulating their differentiation. A recent study demonstrated that ERG is necessary for arresting HSCs in a dormant G0 phase by analyzing the number of cells within each phase of the cell cycle. CLDN5 has been implicated as a negative regulator of many biological processes such as angiogenesis, cell migration, and vascular permeability; alternatively, it has also been shown to be a positive regulator of tight junction assembly, cell population differentiation, protein binding, and endothelial barrier development [32][34][35]. Furthermore, TNF-alpha has been classified as a direct inhibitor of ERG and CLDN5 by extension [30].
9. ERG Upregulates Distal-Less Homeobox-1(DLX1)
It is thought that ERG upregulates distal-less homeobox 1 (DLX1) by interacting with enhanced bound AR and FOXA1 [36]. To support this hypothesis, it has been observed that when ERG and therefore DLX1 transcription is inhibited via BET inhibitors, there is a significant reduction in the oncogenic effects of DLX1 [36]. DLX is a transcriptional factor and is part of the homeobox-containing family [37]. Several malignancies, such as those including the prostate, have been linked to the deregulation of the homeobox gene, and therefore, DLX1 has been validated as a potential PCa biomarker [36][38].
10. GSK3B and WEE1 Induces TMPRSS2-ERG Degradation
Hong et al. [39] revealed that the glycogen synthase kinase 3 beta (GSK3B) and WEE1 induce TMPRSS2-ERG degradation via the dual phosphorylation of ERG threonine-187 and tyrosine-190. Such phosphorylation allows for the recognition and degradation of the ERG oncoprotein by the E3 ubiquitin ligase FBW7. GSK3B and WEE1 have been found to be associated with the DNA damage that is induced proteasomal degradation in PCa [40]. The relationship between TMPRSS2-ERG and PTEN has been described previously, but it is interesting to note that this degradation pathway is eradicated for PTEN loss or GSK3B inactivation. This has further been implicated with the growth of chemoresistant PCa cell lines in culture and in mice.
References
- Adamo, P.; Ladomery, M.R. The oncogene ERG: A key factor in prostate cancer. Oncogene 2016, 35, 403–414.
- Babal, Y.; Kandemir, B.; Kurnaz, I. Gene Regulatory Network of ETS Domain Transcription Factors in Different Stages of Glioma. J. Pers. Med. 2021, 11, 138.
- Parolia, A.; Cieslik, M.; Chu, S.-C.; Xiao, L.; Ouchi, T.; Zhang, Y.; Wang, X.; Vats, P.; Cao, X.; Pitchiaya, S.; et al. Distinct structural classes of activating FOXA1 alterations in advanced prostate cancer. Nature 2019, 571, 413–418.
- Majesky, M.W. Vascular Development. Arterioscler. Thromb. Vasc. Biol. 2018, 38, e17–e24.
- Vivekanand, P. Lessons from Drosophila Pointed, an ETS family transcription factor and key nuclear effector of the RTK signaling pathway. Genesis 2018, 56, e23257.
- Pham, D.; Moseley, C.E.; Gao, M.; Savic, D.; Winstead, C.J.; Sun, M.; Kee, B.L.; Myers, R.M.; Weaver, C.T.; Hatton, R.D. Batf Pioneers the Reorganization of Chromatin in Developing Effector T Cells via Ets1-Dependent Recruitment of Ctcf. Cell Rep. 2019, 29, 1203–1220.e7.
- Vasuri, F.; Valente, S.; Motta, I.; Degiovanni, A.; Ciavarella, C.; Pasquinelli, G. ETS-Related Gene Expression in Healthy Femoral Arteries With Focal Calcifications. Front. Cell Dev. Biol. 2021, 9, 623782.
- Tharakan, B.; Hunter, F.A.; Muthusamy, S.; Randolph, S.; Byrd, C.; Rao, V.N.; Reddy, E.S.P.; Childs, E.W. ETS-Related Gene Activation Preserves Adherens Junctions and Permeability in Microvascular Endothelial Cells. Shock 2021, 57, 309–315.
- Neal, A.; Nornes, S.; Louphrasitthiphol, P.; Sacilotto, N.; Preston, M.D.; Fleisinger, L.; Payne, S.; De Val, S. ETS factors are required but not sufficient for specific patterns of enhancer activity in different endothelial subtypes. Dev. Biol. 2021, 473, 1–14.
- Looney, A.P.; Han, R.; Stawski, L.; Marden, G.; Iwamoto, M.; Trojanowska, M. Synergistic Role of Endothelial ERG and FLI1 in Mediating Pulmonary Vascular Homeostasis. Am. J. Respir. Cell Mol. Biol. 2017, 57, 121–131.
- Lorenzin, F.; Demichelis, F. Past, Current, and Future Strategies to Target ERG Fusion-Positive Prostate Cancer. Cancers 2022, 14, 1118.
- Kohaar, I.; Li, Q.; Chen, Y.; Ravindranath, L.; Young, D.; Ali, A.; Sesterhenn, I.A.; Rosner, I.L.; Cullen, J.; Srivastava, S.; et al. Association of germline genetic variants with TMPRSS2-ERG fusion status in prostate cancer. Oncotarget 2020, 11, 1321–1333.
- Kobelyatskaya, A.; Pudova, E.; Snezhkina, A.; Fedorova, M.; Pavlov, V.; Guvatova, Z.; Savvateeva, M.; Melnikova, N.; Dmitriev, A.; Trofimov, D.; et al. Impact TMPRSS2–ERG Molecular Subtype on Prostate Cancer Recurrence. Life 2021, 11, 588.
- Giunchi, F.; Massari, F.; Altimari, A.; Gruppioni, E.; Nobili, E.; Fiorentino, M.; Ardizzoni, A. Dual TMPRSS2:ERG Fusion in a Patient with Lung and Prostate Cancers. Diagnostics 2020, 10, 1109.
- Yamoah, K.; Lal, P.; Awasthi, S.; Naghavi, A.O.; Rounbehler, R.J.; Gerke, T.; Berglund, A.E.; Pow-Sang, J.M.; Schaeffer, E.M.; Dhillon, J.; et al. TMPRSS2-ERG fusion impacts anterior tumor location in men with prostate cancer. Prostate 2020, 81, 109–117.
- Fang, L.; Li, D.; Yin, J.; Pan, H.; Ye, H.; Bowman, J.; Capaldo, B.; Kelly, K. TMPRSS2-ERG promotes the initiation of prostate cancer by suppressing oncogene-induced senescence. Cancer Gene Ther. 2022, 1–14.
- Sakamoto, K.; Endo, K.; Sakamoto, K.; Kayamori, K.; Ehata, S.; Ichikawa, J.; Ando, T.; Nakamura, R.; Kimura, Y.; Yoshizawa, K.; et al. EHF suppresses cancer progression by inhibiting ETS1-mediated ZEB expression. Oncogenesis 2021, 10, 26.
- Greulich, B.M.; Plotnik, J.P.; Jerde, T.J.; Hollenhorst, P.C. Toll-like receptor 4 signaling activates ERG function in prostate cancer and provides a therapeutic target. NAR Cancer 2021, 3, zcaa046.
- Tsourlakis, M.C.; Khosrawi, P.; Weigand, P.; Kluth, M.; Hube-Magg, C.; Minner, S.; Koop, C.; Graefen, M.; Heinzer, H.; Wittmer, C.; et al. VEGFR-1 Overexpression Identifies a Small Subgroup of Aggressive Prostate Cancers in Patients Treated by Prostatectomy. Int. J. Mol. Sci. 2015, 16, 8591–8606.
- Sun, C.; Dobi, A.; Mohamed, A.; Li, H.; Thangapazham, R.L.; Furusato, B.; Shaheduzzaman, S.; Tan, S.H.; Vaidyanathan, G.; Whitman, E.; et al. TMPRSS2-ERG fusion, a common genomic alteration in prostate cancer activates C-MYC and abrogates prostate epithelial differentiation. Oncogene 2008, 27, 5348–5353.
- Vestweber, D. VE-cadherin: The major endothelial adhesion molecule controlling cellular junctions and blood vessel formation. Arterioscler. Thromb. Vasc. Biol. 2008, 28, 223–232.
- Cai, J.; Kandagatla, P.; Singareddy, R.; Kropinski, A.; Sheng, S.; Cher, M.L.; Chinni, S.R. Androgens Induce Functional CXCR4 through ERG Factor Expression in TMPRSS2-ERG Fusion-Positive Prostate Cancer Cells. Transl. Oncol. 2010, 3, 195–203.
- Clézardin, P.; Coleman, R.; Puppo, M.; Ottewell, P.; Bonnelye, E.; Paycha, F.; Confavreux, C.B.; Holen, I. Bone metastasis: Mechanisms, therapies, and biomarkers. Physiol. Rev. 2021, 101, 797–855.
- Sun, Y.X.; Fang, M.; Wang, J.; Cooper, C.R.; Pienta, K.J.; Taichman, R.S. Expression and activation of alpha v beta 3 integrins by SDF-1/CXC12 increases the aggressiveness of prostate cancer cells. Prostate 2007, 67, 61–73.
- Singareddy, R.; Semaan, L.; Conley-LaComb, M.K.; John, J.S.; Powell, K.; Iyer, M.; Smith, D.; Heilbrun, L.K.; Shi, N.; Sakr, W.; et al. Transcriptional Regulation of CXCR4 in Prostate Cancer: Significance of TMPRSS2-ERG Fusions. Mol. Cancer Res. 2013, 11, 1349–1361.
- Zarrabi, A.; Hashemi, F.; Hashemi, F.; Zabolian, A.; Banihashemi, S.M.; Moghadam, S.S.; Hushmandi, K.; Samarghandian, S.; Ashrafizadeh, M.; Khan, H. Role of ZEB Family Members in Proliferation, Metastasis, and Chemoresistance of Prostate Cancer Cells: Revealing Signaling Networks. Curr. Cancer Drug Targets 2021, 21, 749–767.
- Wang, J.; Farkas, C.; Benyoucef, A.; Carmichael, C.; Haigh, K.; Wong, N.; Huylebroeck, D.; Stemmler, M.P.; Brabletz, S.; Brabletz, T.; et al. Interplay between the EMT transcription factors ZEB1 and ZEB2 regulates hematopoietic stem and progenitor cell differentiation and hematopoietic lineage fidelity. PLOS Biol. 2021, 19, e3001394.
- Zoma, M.; Curti, L.; Shinde, D.; Albino, D.; Mitra, A.; Sgrignani, J.; Mapelli, S.N.; Sandrini, G.; Civenni, G.; Merulla, J.; et al. EZH2-induced lysine K362 methylation enhances TMPRSS2-ERG oncogenic activity in prostate cancer. Nat. Commun. 2021, 12, 4147.
- Margueron, R.; Reinberg, D. The Polycomb complex PRC2 and its mark in life. Nature 2011, 469, 343–349.
- Yuan, L.; Le Bras, A.; Sacharidou, A.; Itagaki, K.; Zhan, Y.; Kondo, M.; Carman, C.V.; Davis, G.E.; Aird, W.C.; Oettgen, P. ETS-related Gene (ERG) Controls Endothelial Cell Permeability via Transcriptional Regulation of the Claudin 5 (CLDN5) Gene. J. Biol. Chem. 2012, 287, 6582–6591.
- Liu, F.; Liu, Q.; Yuan, F.; Guo, S.; Liu, J.; Sun, Z.; Gao, P.; Wang, Y.; Yan, S.; Liu, J. Erg mediates downregulation of claudin-5 in the brain endothelium of a murine experimental model of cerebral malaria. FEBS Lett. 2019, 593, 2585–2595.
- Ma, S.-C.; Li, Q.; Peng, J.-Y.; Zhouwen, J.-L.; Diao, J.-F.; Niu, J.-X.; Wang, X.; Guan, X.-D.; Jia, W.; Jiang, W.-G. Claudin-5 regulates blood-brain barrier permeability by modifying brain microvascular endothelial cell proliferation, migration, and adhesion to prevent lung cancer metastasis. CNS Neurosci. Ther. 2017, 23, 947–960.
- Kind, S.; Büscheck, F.; Höflmayer, D.; Hube-Magg, C.; Kluth, M.; Tsourlakis, M.C.; Steurer, S.; Clauditz, T.S.; Luebke, A.M.; Burandt, E.; et al. Claudin-1 upregulation is associated with favorable tumor features and a reduced risk for biochemical recurrence in ERG-positive prostate cancer. World J. Urol. 2019, 38, 2185–2196.
- Tian, R.; Luo, Y.; Liu, Q.; Cai, M.; Li, J.; Sun, W.; Wang, J.; He, C.; Liu, Y.; Liu, X. The effect of claudin-5 overexpression on the interactions of claudin-1 and -2 and barrier function in retinal cells. Curr. Mol. Med. 2014, 14, 1226–1237.
- Kluger, M.S.; Clark, P.R.; Tellides, G.; Gerke, V.; Pober, J.S. Claudin-5 Controls Intercellular Barriers of Human Dermal Microvascular but Not Human Umbilical Vein Endothelial Cells. Arter. Thromb. Vasc. Biol. 2013, 33, 489–500.
- Goel, S.; Bhatia, V.; Kundu, S.; Biswas, T.; Carskadon, S.; Gupta, N.; Asim, M.; Morrissey, C.; Palanisamy, N.; Ateeq, B. Transcriptional network involving ERG and AR orchestrates Distal-less homeobox-1 mediated prostate cancer progression. Nat. Commun. 2021, 12, 5325.
- Cani, A.K.; Hu, K.; Liu, C.J.; Siddiqui, J.; Zheng, Y.; Han, S.; Nallandhighal, S.; Hovelson, D.H.; Xiao, L.; Pham, T.; et al. Development of a Whole-urine, Multiplexed, Next-generation RNA-sequencing Assay for Early Detection of Aggressive Prostate Cancer. Eur. Urol. Oncol. 2021.
- Van Neste, L.; Hendriks, R.J.; Dijkstra, S.; Trooskens, G.; Cornel, E.B.; Jannink, S.A.; de Jong, H.; Hessels, D.; Smit, F.P.; Melchers, W.J.G.; et al. Detection of High-grade Prostate Cancer Using a Urinary Molecular Biomarker–Based Risk Score. Eur. Urol. 2016, 70, 740–748.
- Hong, Z.; Zhang, W.; Ding, D.; Huang, Z.; Yan, Y.; Cao, W.; Pan, Y.; Hou, X.; Weroha, S.J.; Karnes, R.J.; et al. DNA Damage Promotes TMPRSS2-ERG Oncoprotein Destruction and Prostate Cancer Suppression via Signaling Converged by GSK3beta and WEE1. Mol. Cell 2020, 79, 1008–1023.
- Strittmatter, B.G.; Jerde, T.J.; Hollenhorst, P.C. Ras/ERK and PI3K/AKT signaling differentially regulate oncogenic ERG medi-ated transcription in prostate cells. PLoS Genet. 2021, 17, e1009708.
More
Information
Subjects:
Biochemistry & Molecular Biology
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
814
Revisions:
2 times
(View History)
Update Date:
13 May 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





