1000/1000
Hot
Most Recent

The Vitaceae Juss., in the basal lineages of Rosids, contains sixteen genera and 950 species, mainly of tropical lianas. The family has been divided in five tribes: Ampelopsideae, Cisseae, Cayratieae, Parthenocisseae and Viteae. Seed shape is variable in this family and is described based on the comparison of bi-dimensional seed images with geometric models. Ten morphological types are described in the Vitaceae. Seven of them are general and three specific. Among the general types, three are shared with the Arecaceae and correspond to geometric figures well described (lenses, superellipses and elongated waterdrops). Four additional groups include waterdrops, normal or rounded, heart curves, normal or rounded, elongated heart curves and other elongated curves, respectively. Finally, the three specific types correspond to heart curves of the Cayratia and Pseudocayratia types, heart curves of the Squared Heart Curve (SqHC) type of Ampelocissus and Ampelopsis, and Elongated Superellipse-Heart Curves (ESHCs), frequent in Tetrastigma species and observed also in Cissus species and R. rhomboidea. All these groups are defined by geometric models obtained by the representation of algebraic equations. Modifications in the equations result in models adjusting to the shape of seeds for each species.
| Tribe | Genera |
| I. Ampelopsideae (47) | Ampelopsis Michx. (18)Nekemias Raf. (9) |
| Rhoicissus Planch. (14) | |
| Clematicissus Planch. (6) | |
| II. Cisseae (300) | Cissus L. (300) |
| III. Cayratieae (368) | Cayratia Juss. (25) |
| Causonis Raf. (30) | |
| Acareosperma Gagnep. (1) | |
| Afrocayratia (7) | |
| Cyphostemma (Planch.) Alston (200) | |
| Pseudocayratia J. Wen,L.M.Lu and Z.D. Chen (5) | |
| Tetrastigma (Miq.) Planch. (100) | |
| IV. Parthenocisseae (16) | Parthenocissus Planch. (14) |
| Yua C.L.Li (2) | |
| V. Viteae (190) | Ampelocissus Planch. (115) |
| Vitis L. (75) |
Cissus is the largest genus in the family with 300 species of complex classification [8]. Cyphostemma is second, with 200 species of an interesting diversity in their range of distribution as well as in growth habits (vines and lianas, herbs, stem succulents and a tree) [9]. Vitis has seventy-five inter-fertile wild species distributed in three continents under subtropical, Mediterranean and continental-temperate climatic conditions. Vitis vinifera L. is the species with highest economic importance in the family with some taxonomic uncertainty about the differentiation between V. vinifera L. subsp. vinifera and V. vinifera L. subsp. sylvestris (Willd.) Hegi [10][11]. Thousands of cultivars of V. vinifera are used worldwide in Viticulture. Species of other genera are widely cultivated, such as Parthenocissus quinquefolia (L.) Planch., the Virginia creeper, in temperate areas, and Cissus incisa Des Moul., the grape ivy, in tropical areas. Species of the genus Tetrastigma are the only host plants for the parasitic plant Rafflesia arnoldii R.Br., Rafflesiaceae, which is native only to a few areas within the Malay Archipelago [12].
| Tribe (Species Observed/Total) |
Genera (Species Observed/Total) |
Species (References for the Images) |
|---|---|---|
| I. Ampelopsideae (15/47) |
Ampelopsis Michx. (13/18) |
Ampelopsis aconitifolia [33], A. arborea [34], A. bodinieri [33], A. cantoniensis [35][33], A. cordata [36], A. chaffanjoni [33], A. delavayana [35], A. denudata [29], A. glandulosa [37], A. grossedentata [35], A. humulifolia [33], A. japonica [33], A. megalophylla [35][33] |
| Rhoicissus Planch. (2/14) |
Rhoicissus revoilii [35], R. rhomboidea [35] | |
| II. Cisseae (33/300) |
Cissus L. (33/300) |
Cissus antarctica [35], C. aralioides [32][38], C. barbeyana [38], C. bosseri [38], C. cactiformis [38], C. campestris [35][39][40], C. cornifolia [38], C. descoingsii, [35][39], C. diffusiflora [38], C. elongata [38], C. erosa [41], C. floribunda [38], C. fuliginea [35], C. granulosa [35], C. hastata [38], C. hypoglauca [35], C. integrifolia [38][40], C. leucophlea [38], C. penninervis [35], C. petiolata [38], C. pileata [38], C. populnea [38], C. quadrangularis [42], C. reniformis [35][39], C. repens [38], C. sciaphila [38], C. smithiana [38], C. sterculiifolia [35], C. subtetragona [38], C. trianae [35], C. tuberosa [40], C. verticillata [35][39][40][43], C. willardii [40], |
| III. Cayratieae (40/365) |
Causonis Raf. (1/9) | Causonis sp. [44] |
| Cayratia Juss. (7/60) |
Cayratia cheniana [44], C. geniculata [35], C. imerinensis [45], C. japonica [35][46], C. oligocarpa [35], C. saponaria [35], C. sp. [African, [44]] | |
| Cyphostemma (Planch.) Alston (3/200) |
Cyphostemma elephantopus [47], C. laza [35], C. junceum [35] | |
| Pseudocayratia J. Wen, L.M.Lu and Z.D.Chen (3/5) |
Pseudocayratia dichromocarpa [48], P. pengiana [48], P. speciosa [48][49] | |
| Tetrastigma (Miq.) Planch. (26/100) |
Tetrastigma campylocarpum [50], T. cauliflorum [50], T. caudatum [50], T. delavayi [50], T. dichotomum [49], T. formosanum [50], T. harmandi [35], T. hemsleyanum [35][50], T. henryi [50], T. hypoglaucum [50], T. jinghongense [50], T. kwangsiense [29][35], T. lanceolarium [29], T. laoticum [50], T. obovatum [49][50], T. obtectum [49][50], T. pachyllylum [50], T. pedunculare [35][49][50], T. petraeum [50], T. retinervum [50], T. rumicispermum [35][49][50], T. serrulatum [50], T. sichouense [50], T. thorsborneorum [50], T. triphyllum [35][50], T. xishuangbannaense [35][50] | |
| IV. Parthenocisseae (11/16) |
Parthenocissus Planch. (9/14) |
Parthenocissus dalzielii [33], P. heptaphylla [35], P. henryana [33], P. heterophylla [33], P. himalayana [51], P. laetevirens [33], P. quinquefolia [52], P. tricuspidata [33][34][43], P. vitacea [35] |
| Yua C.L.Li (2/2) | Yua austro-orientalis [35], Y. chinensis [35] | |
| V. Viteae (32/190) |
Ampelocissus Planch. (13/115) |
Ampelocissus acapulcensis [29], A. bombycina [29], A. bravoi [40], A. cavicaulis [29], A. erdvendbergiana [29], A. grantii [29], A. javalensis [29][40], A. latifolia [29], A. macrocirrha [29], A. martinii [40], A. obtusata [29], A. ochracea [29], A. robinsonii [29] |
| Vitis L. (19/75) |
Vitis aestivalis [53], V. amurensis [43][54], V. brandoniana [52], V. cinerea [55], V. eolabrusca [52], V. flexuosa [52], V. grayensis [56], V. labrusca [43][52][57], V. lanatoides [56], V. latisulcata [56], V. palmata [58], V. pseudorotundifolia [52], V. rostrata [52], V. rotundifolia [35][39][52], V. rupestris [43], V. tiliifolia [59], V. tsoi [35][39], V. vulpina [57][60], V. wilsoniae [35][39] |
Table 3. A summary of groups based on morphological seed types for the analysed species in the Vitaceae. The number of cases found in each group is given between dashes.
| Group (Geometric Model) | Examples |
|---|---|
| Group I (Lenses)-3- | Cissus quadrangularis [42], C. sterculiifolia [35], Tetrastigma petraeum [50] |
| Group II (Superellipses)-7- | Ampelocissus bravoi [40], C. reniformis [35][39], Cyphostemma elephantopus [47], C. laza [35], Tetrastigma campylocarpum [50], T. caudatum [50], T. henryi [50] |
| Group III (Elongated water drops)-15- | Ampelopsis arborea [34], Cayratia imerinensis [45], Cissus aralioides [32][38], C. cornifolia [38], C. erosa [41], C. integrifolia [38][40], C. petiolata [38], C. pileata [38], C. populnea [38], C. verticillata [35][39][40][43], C. sciaphila [38], C. smithiana [38], C. willardii [40], Cyphostemma junceum [35], V. vulpina [57][60] |
| Group IV (Water drops, normal or rounded)-14- | Ampelopsis bodinieri [33], A. glandulosa [33][37], A. humulifolia [33], Cayratia cheniana [44], Cissus campestris [35][39][40], C. fuliginea [35], C. tuberosa [40], C. granulosa [35], Parthenocissus dalzielii [33], Tetrastigma triphyllum [35][50], Vitis amurensis [43][54], V. labrusca [43][52][57], V. palmata [58], V. rupestris [43] |
| Group V (Heart curves normal or rounded)-19- | Ampelopsis aconitifolia [33], A. chaffanjoni [33], A. cordata [36], A. japonica [33], Parthenocissus heptaphylla [35], P. heterophylla [33], P. henryana [33], P. himalayana [50][51], P. quinquefolia [52], P. vitacea [35], P. tricuspidata [33][34][43], Rhoicissus revoilii [35], T. lanceolarium [29], Vitis cinerea [55], V. flexuosa [52], V. lanatoides [56], V. latisulcata [56], V. tsoi [35][39], V. wilsoniae [35][39] |
| Group VI (Elongated Heart curves)-6- | Ampelocissus acapulcensis [29], Cissus oligocarpa [35]. V. eolabrusca [52], V. grayensis [56], V. pseudorotundifolia [52], V. tiliifolia [59] |
| Group VII (Other elongated types)-11- | Ampelopsis megalophylla [35][33], Causonis sp. [44], Cayratia saponaria [35], Cissus trianae [35], C. hypoglauca [35], Parthenocissus laetevirens [33], T. hypoglaucum [50] Vitis aestivalis [53], V. rotundifolia [35][39][52], Yua austro-orientalis [35], Y. chinensis [35] |
| Group VIII (Heart curves of the Cayratia and Pseudocayratia types)-7- | Cayratia japonica [35][46], Cayratia sp. [African, [44]], Pseudocayratia dichromocarpa [48], P. pengiana [48], P. speciosa [48][50], Tetrastigma formosanum [49], T. pedunculare [35][49][50] |
| Group IX (Heart curves of the SqHC type of Ampelocissus and Ampelopsis)-15- | Ampelocissus bombycina [29], A. cavicaulis [29], A. erdvendbergiana [29], A. grantii [29], A. javalensis [29][40], A. latifolia [29], A. macrocirrha [29], A. martinii [40], A. obtusata [29], A. ochracea [29], A. robinsonii [29], Ampelopsis cantoniensis [35][33], A. delavayana [35], A. denudata [29], A. grossedentata [35] |
| Group X Elongated Superellipse-heart curves-16- | Cissus elongata [38], C. penninervis [35], Rhoicissus rhomboidea [35], Tetrastigma hemsleyanum [35][50], T. jinghongense [50], T. laoticum [50], T. cauliflorum [50], T. dichotomum [49], T. harmandi [35], T. pachyllylum [50], T. kwangsiense [29][35], T. obovatum [49][50], T. obtectum [49][50], T. retinervum [49], T. serrulatum [50], T. sichouense [50] |
| Undefined-18- | Cayratia geniculata [35], Cissus antarctica [35], C. barbeyana [38], C. bosseri [38], C. cactiformis [38], C. descoingsii, [35][39], C. diffusiflora [38], C. floribunda [38], C. hastata [38], C. leucophlea [38], C. repens [38], C. subtetragona [38], T. delavayi [50], T. rumicispermum [35][49][50], T. thorsborneorum [50], T. xishuangbannaense [35][50], V. brandoniana [52], V. rostrata [52] |
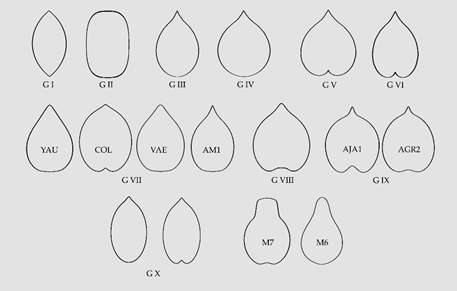
Figure 1. The images of seeds of Cissus sterculiifolia [35], Tetrastigma petraeum [49] and Cissus quadrangularis [42] resemble lenses of different proportions.
In general, the distribution of morphological types is not in close agreement with the current taxonomic classification; nevertheless, some results may be summarized in this aspect. First, the seeds of the Elongated Superellipse-Heart Curves (ESHCs) type (Group X) are more frequent in Tetrastigma and have been observed in Rhoicissus and Cissus, but not in species of other genera. While many seeds in species of Ampelopsis, Parthenocissus and Vitis share the typical shapes of water drop and heart curves, the squared heart curve (SqHC) type (Group IX) has been predominantly observed in Ampelocissus and Ampelopsis. A number of species remain undefined due to one of these two reasons: First, their irregular seed shape making difficult the identification of an adequate model (Cayratia geniculata, Cissus antarctica) and, second, the seed images have geometric shapes but the identification of the model with the corresponding equation is pending (Tetrastigma delavayi, T. rumicispermum). In addition, further work will be done on the seeds of Vitis species.