
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Grazia Maugeri | -- | 1985 | 2024-03-07 13:17:52 | | | |
| 2 | Peter Tang | Meta information modification | 1985 | 2024-03-08 02:49:45 | | |
Video Upload Options
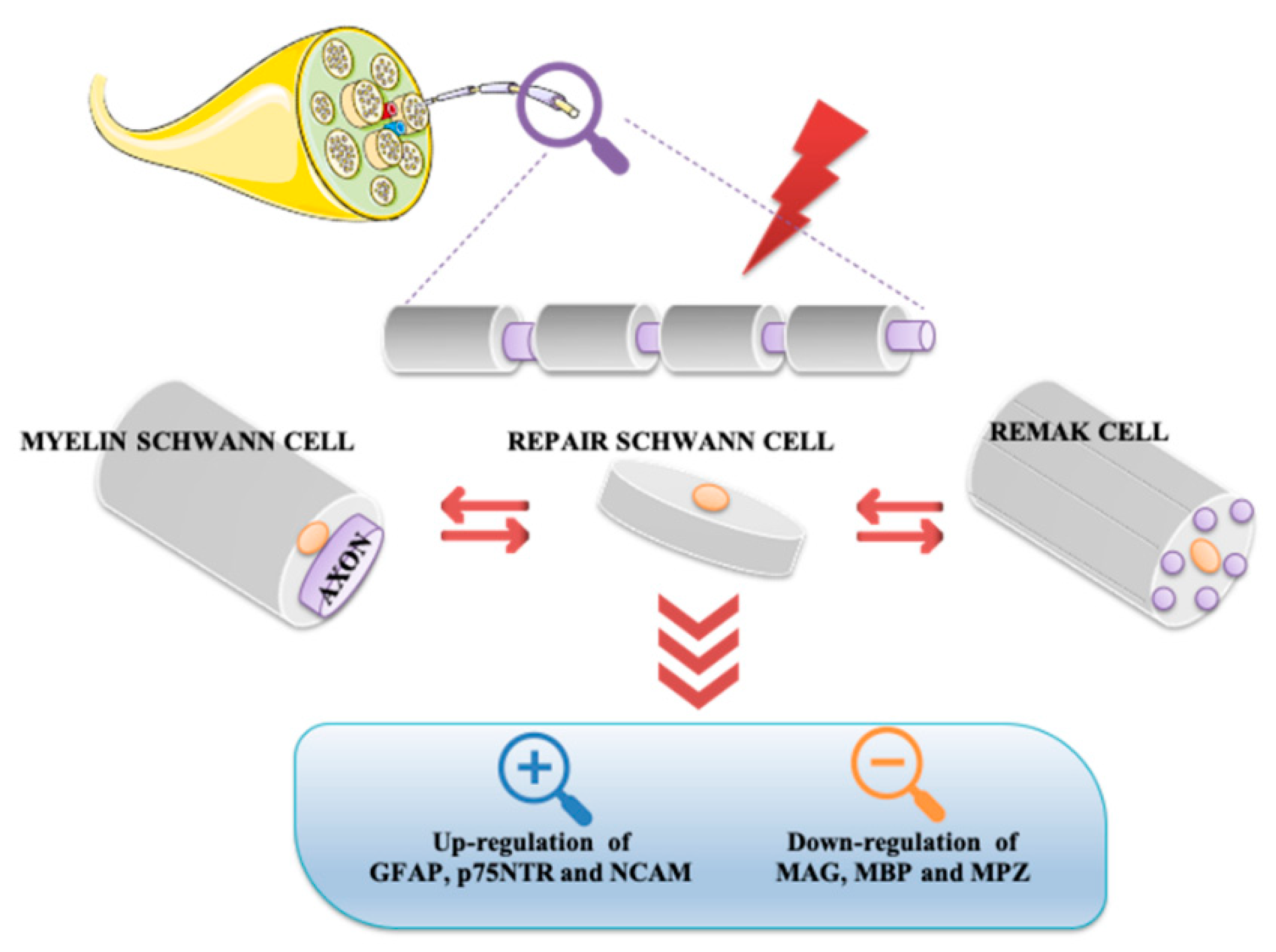
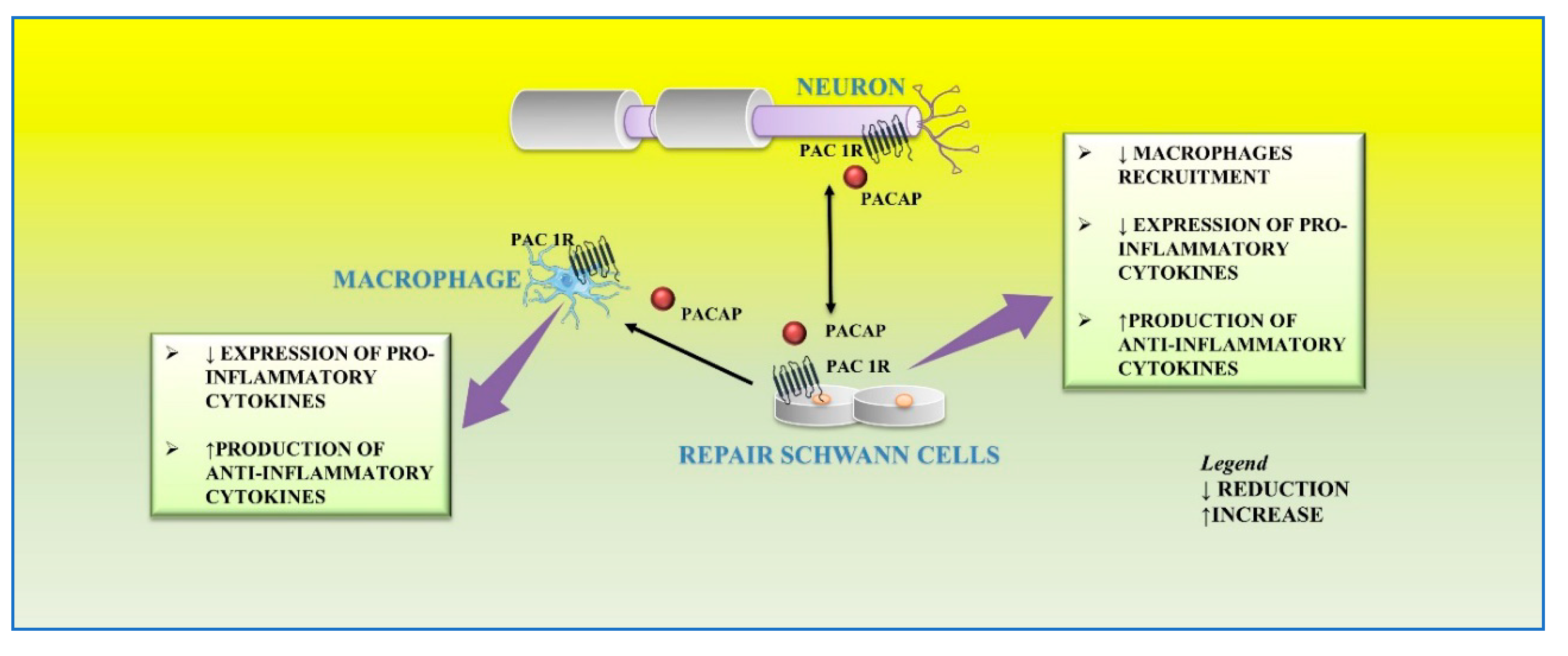
Schwann cells, the most abundant glial cells of the peripheral nervous system, represent the key players able to supply extracellular microenvironment for axonal regrowth and restoration of myelin sheaths on regenerating axons. Following nerve injury, Schwann cells respond adaptively to damage by acquiring a new phenotype. In particular, some of them localize in the distal stump to form the Bungner band, a regeneration track in the distal site of the injured nerve, whereas others produce cytokines involved in recruitment of macrophages infiltrating into the nerve damaged area for axonal and myelin debris clearance. Several neurotrophic factors, including pituitary adenylyl cyclase-activating peptide (PACAP), promote survival and axonal elongation of injured neurons.
1. Introduction
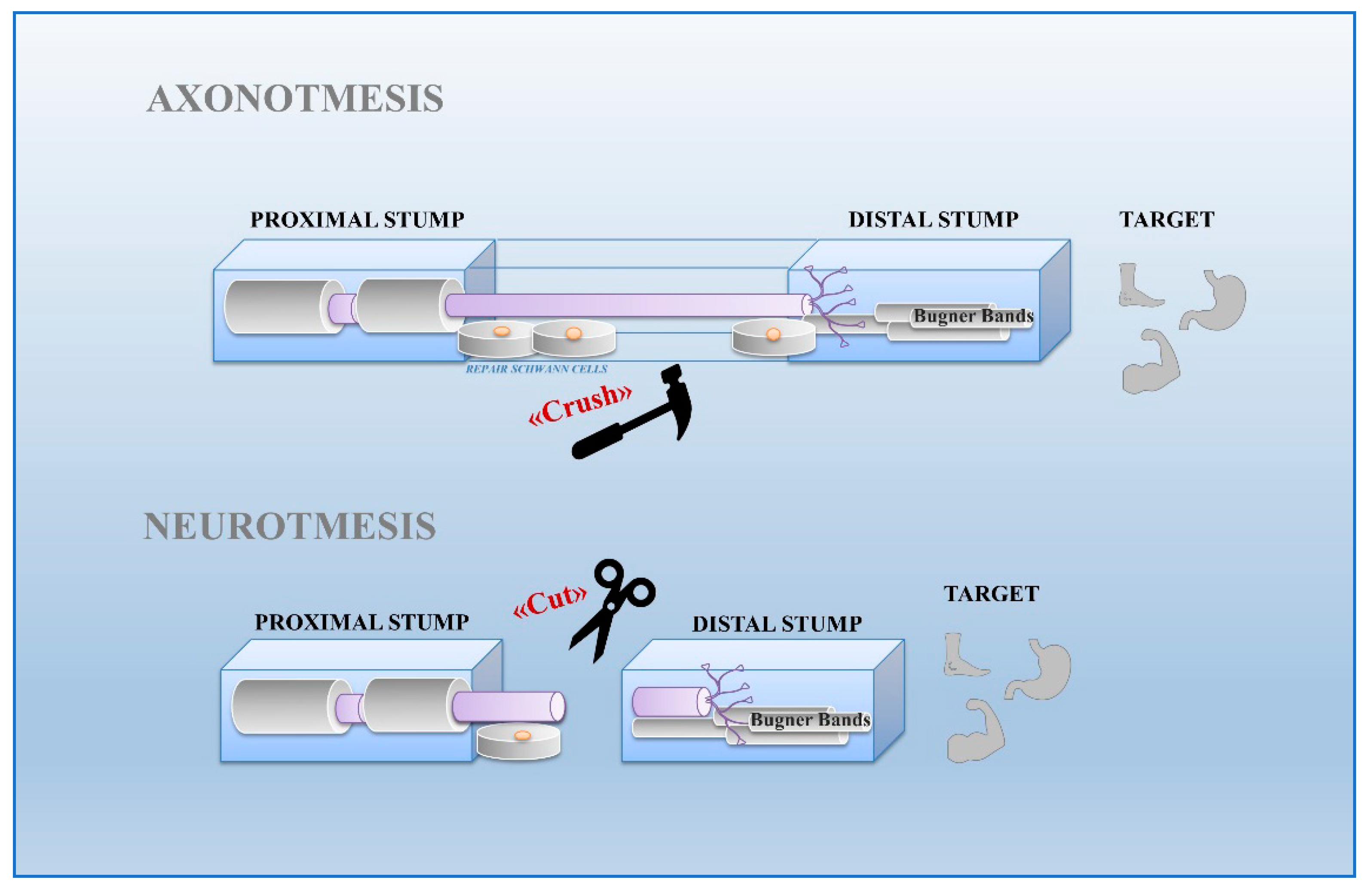
2. Schwann Cells and Nerve Injury
3. Effect of PACAP on Schwann Cells during Nerve Injury
References
- Jessen, K.R.; Mirsky, R. The repair Schwann cell and its function in regenerating nerves. J. Physiol. 2016, 594, 3521–3531.
- Jessen, K.R.; Mirsky, R. The Success and Failure of the Schwann Cell Response to Nerve Injury. Front. Cell. Neurosci. 2019, 13, 33.
- Miyata, A.; Arimura, A.; Dahl, R.R.; Minamino, N.; Uehara, A.; Jiang, L.; Culler, M.D.; Coy, D.H. Isolation of a novel 38 residue-hypothalamic polypeptide which stimulates adenylate cyclase in pituitary cells. Biochem. Biophys. Res. Commun. 1989, 164, 567–574.
- Rasband, M.N. Glial Contributions to Neural Function and Disease. Mol. Cell. Proteom. 2016, 15, 355–361.
- Brown, A.M.; Evans, R.D.; Black, J.; Ransom, B.R. Schwann cell glycogen selectively supports myelinated axon function. Ann. Neurol. 2012, 72, 406–418.
- Voas, M.G.; Glenn, T.D.; Raphael, A.R.; Talbot, W.S. Schwann cells inhibit ectopic clustering of axonal sodium channels. J. Neurosci. 2009, 29, 14408–14414.
- Scheib, J.; Höke, A. Advances in peripheral nerve regeneration. Nat. Rev. Neurol. 2013, 9, 668–676.
- Ioghen, O.; Manole, E.; Gherghiceanu, M.; Popescu, B.O.; Ceafalan, L.C. Non-Myelinating Schwann Cells in Health and Disease. Schwann Cells 2020.
- Brosius Lutz, A.; Barres, B.A. Contrasting the glial response to axon injury in the central and peripheral nervous systems. Dev. Cell 2014, 28, 7–17.
- Pereira, J.A.; Lebrun-Julien, F.; Suter, U. Molecular mechanisms regulating myelination in the peripheral nervous system. Trends Neurosci. 2012, 35, 123–134.
- Topilko, P.; Schneider-Maunoury, S.; Levi, G.; Baron-Van Evercooren, A.; Chennoufi, A.B.; Seitanidou, T.; Babinet, C.; Charnay, P. Krox-20 controls myelination in the peripheral nervous system. Nature 1994, 371, 796–799.
- Han, H.; Myllykoski, M.; Ruskamo, S.; Wang, C.; Kursula, P. Myelin-specific proteins: A structurally diverse group of membrane-interacting molecules. Biofactors 2013, 39, 233–241.
- Li, C.; Tropak, M.B.; Gerial, R.; Clapoff, S.; Abramow-Newerly, W.; Trapp, B.; Peterson, A.; Roder, J. Myelination in the absence of myelin-associated glycoprotein. Nature 1994, 369, 747–750.
- Hsieh, S.T.; Kidd, G.J.; Crawford, T.O.; Xu, Z.; Lin, W.M.; Trapp, B.D.; Cleveland, D.W.; Griffin, J.W. Regional modulation of neurofilament organization by myelination in normal axons. J. Neurosci. 1994, 14, 6392–6401.
- Boerboom, A.; Dion, V.; Chariot, A.; Franzen, R. Molecular mechanisms involved in schwann cell plasticity. Front. Mol. Neurosci. 2017, 10, 38.
- Seddon, H.J. Three types of nerve injury. Brain 1943, 66, 237–288.
- Burnett, M.G.; Zager, E.L. Pathophysiology of peripheral nerve injury: A brief review. Neurosurg. Focus 2004, 5, 1–7.
- Martini, R.; Fischer, S.; Lopez-Vales, R.; David, S. Interactions between Schwann cells and macrophages in injury and inherited demyelinating disease. Glia 2008, 56, 1566–1577.
- Rotshenker, S. Wallerian degeneration: The innate-immune response to traumatic nerve injury. J. Neuroinflammation 2011, 8, 109.
- Grothe, C.; Haastert, K.; Jungnickel, J. Physiological function and putative therapeutic impact of the FGF-2 system in peripheral nerve regeneration—Lessons from in vivo studies in mice and rats. Brain Res. Rev. 2006, 51, 293–299.
- Fontana, X.; Hristova, M.; Da Costa, C.; Patodia, S.; Thei, L.; Makwana, M.; Spencer-Dene, B.; Latouche, M.; Mirsky, R.; Jessen, K.R.; et al. c-Jun in Schwann cells promotes axonal regeneration and motoneuron survival via paracrine signaling. J. Cell Biol. 2012, 198, 127–141.
- Brushart, T.M.; Aspalter, M.; Griffin, J.W.; Redett, R.; Hameed, H.; Zhou, C.; Wright, M.; Vyas, A.; Höke, A. Schwann cell phenotype is regulated by axon modality and central-peripheral location and persists in vitro. Exp. Neurol. 2013, 247, 272–281.
- Woodley, P.K.; Min, Q.; Li, Y.; Mulvey, N.F.; Parkinson, D.B.; Dun, X.P. Distinct VIP and PACAP Functions in the Distal Nerve Stump During Peripheral Nerve Regeneration. Front. Neurosci. 2019, 13, 1326.
- Zhou, X.; Rodriguez, W.I.; Casillas, R.A.; Ma, V.; Tam, J.; Hu, Z.; Lelievre, V.; Chao, A.; Waschek, J.A. Axotomy-induced changes in pituitary adenylate cyclase activating polypeptide (PACAP) and PACAP receptor gene expression in the adult rat facial motor nucleus. J. Neurosci. Res. 1999, 57, 953–961.
- Armstrong, B.D.; Hu, Z.; Abad, C.; Yamamoto, M.; Rodriguez, W.I.; Cheng, J.; Tam, J.; Gomariz, R.P.; Patterson, P.H.; Waschek, J.A. Lymphocyte regulation of neuropeptide gene expression after neuronal injury. J. Neurosci. Res. 2003, 74, 240–247.
- Navarro, X.; Vivo, M.; Valero-Cabre, A. Neural plasticity after peripheral nerve injury and regeneration. Prog. Neurobiol. 2007, 82, 163–201.
- Reimer, M.; Moller, K.; Sundler, F.; Hannibal, J.; Fahrenkrug, J.; Kanje, M. Increased expression, axonal transport and release of pituitary adenylate cyclase-activating polypeptide in the cultured rat vagus nerve. Neuroscience 1999, 88, 213–222.
- Armstrong, B.D.; Abad, C.; Chhith, S.; Cheung-Lau, G.; Hajji, O.E.; Nobuta, H.; Waschek, J.A. Impaired nerve regeneration and enhanced neuroinflammatory response in mice lacking pituitary Adenylyl cyclase activating peptide. Neuroscience 2008, 151, 63–73.
- Zigmond, R.E.; Echevarria, F.D. Macrophage biology in the peripheral nervous system after injury. Prog. Neurobiol. 2019, 173, 102–121.
- Maurel, P.; Salzer, J.L. Axonal regulation of Schwann cell proliferation and survival and the initial events of myelination requires PI 3-kinase activity. J. Neurosci. 2000, 20, 4635–4645.
- Harrisingh, M.C.; Perez-Nadales, E.; Parkinson, D.B.; Malcolm, D.S.; Mudge, A.W.; Lloyd, A.C. The Ras/Raf/ERK signalling pathway drives Schwann cell dedifferentiation. EMBO J. 2004, 23, 3061–3071.
- Musumeci, G.; Leggio, G.M.; Marzagalli, R.; Al-Badri, G.; Drago, F.; Castorina, A. Identification of Dysregulated microRNA Networks in Schwann Cell-Like Cultures Exposed to Immune Challenge: Potential Crosstalk with the Protective VIP/PACAP Neuropeptide System. Int. J. Mol. Sci. 2018, 19, 981.
- Lee, H.; Park, K.; Soo, J.; Lee, K.; Lee, S.J. Vasoactive intestinal peptide inhibits toll-like receptor 3-induced nitric oxide production in Schwann cells and subsequent sensory neuronal cell death in vitro. J. Neurosci. Res. 2009, 87, 171–178.
- Vincze, A.; Reglodi, D.; Helyes, Z.; Hashimoto, H.; Shintani, N.; Abraham, H. Role of endogenous pituitary adenylate cyclase activating polypeptide (PACAP) in myelination of the rodent brain: Lessons from PACAP-deficient mice. Int. J. Dev. Neurosci. 2011, 29, 923–935.
- Castorina, A. Multiple Actions of Pituitary Adenylate Cyclase-Activating Polypeptide (PACAP) in Schwann Cell Biology. In Pituitary Adenylate Cyclase Activating Polypeptide—PACAP. Current Topics in Neurotoxicity; Reglodi, D., Tamas, A., Eds.; Springer: Cham, Switzerlnad, 2016; Volume 11.
- Castorina, A.; Scuderi, S.; D’Amico, A.G.; Drago, F.; D’Agata, V. PACAP and VIP increase the expression of myelin-related proteins in rat schwannoma cells: Involvement of PAC1/VPAC2 receptor-mediated activation of PI3K/Akt signaling pathways. Exp. Cell Res. 2014, 322, 108–121.
- Vallejo, M. PACAP signaling to dream: A camp-dependent pathway that regulates cortical astrogliogenesis. Mol. Neurobiol. 2009, 39, 90–100.
- Li, Y.; Tennekoon, G.I.; Birnbaum, M.; Marchionni, M.A.; Rutkowski, J.L. Neuregulin signaling through a PI3K/Akt/Bad pathway in Schwann cell survival. Mol. Cell. Neurosci. 2001, 17, 761–767.
- Liang, G.; Cline, G.W.; Macica, C.M. IGF-1 stimulates de novo fatty acid biosynthesis by Schwann cells during myelination. Glia 2007, 55, 632–641.
- Akassoglou, K.; Kombrinck, K.W.; Degen, J.L.; Strickland, S. Tissue plasminogen activator-mediated fibrinolysis protects against axonal degeneration and demyelination after sciatic nerve injury. J. Cell Biol. 2000, 149, 1157–1166.
- Castorina, A.; Tiralongo, A.; Giunta, S.; Carnazza, M.L.; Rasi, G.; D’Agata, V. PACAP and VIP prevent apoptosis in schwannoma cells. Brain Res. 2008, 1241, 29–35.
- Baskozos, G.; Sandy-Hindmarch, O.; Clark, A.J.; Windsor, K.; Karlsson, P.; Weir, G.A.; McDermott, L.A.; Burchall, J.; Wiberg, A.; Furniss, D.; et al. Molecular and cellular correlates of human nerve regeneration: ADCYAP1/PACAP enhance nerve outgrowth. Brain 2020, 143, 2009–2026.
- Geuna, S.; Raimondo, S.; Fregnan, F.; Haastert-Talini, K.; Grothe, C. In vitro models for peripheral nerve regeneration. Eur. J. Neurosci. 2016, 43, 287–296.
- Stassart, R.M.; Fledrich, R.; Velanac, V.; Brinkmann, B.G.; Schwab, M.H.; Meijer, D.; Sereda, M.W.; Nave, K.A. A role for Schwann cell-derived neuregulin-1 in remyelination. Nat. Neurosci. 2013, 16, 48–54.





