
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Zhongkui Hong | -- | 2914 | 2024-02-27 19:55:09 | | | |
| 2 | Lindsay Dong | + 3 word(s) | 2917 | 2024-02-28 06:33:10 | | |
Video Upload Options
HMG-CoA (3-hydroxy-3-methyl-glutaryl-coenzyme A) reductase inhibitors, commonly known as statins, are the primary treatment choice for cardiovascular diseases, which stand as the leading global cause of mortality. Statins also offer various pleiotropic effects, including improved endothelial function, anti-inflammatory properties, reduced oxidative stress, anti-thrombotic effects, and the stabilization of atherosclerotic plaques. However, the usage of statins can be accompanied by a range of adverse effects, such as the development of type 2 diabetes mellitus, muscular symptoms, liver toxicity, kidney diseases, cataracts, hemorrhagic strokes, and psychiatric complications. These issues are referred to as statin-associated symptoms (SAS) and are relatively infrequent in clinical trials, making it challenging to attribute them to statin use definitively. Therefore, these symptoms can lead to significant problems, necessitating dose adjustments or discontinuation of statin therapy.
1. Introduction
2. Pleiotropic Benefits of Statin
2.1. Enhancing Endothelial Function
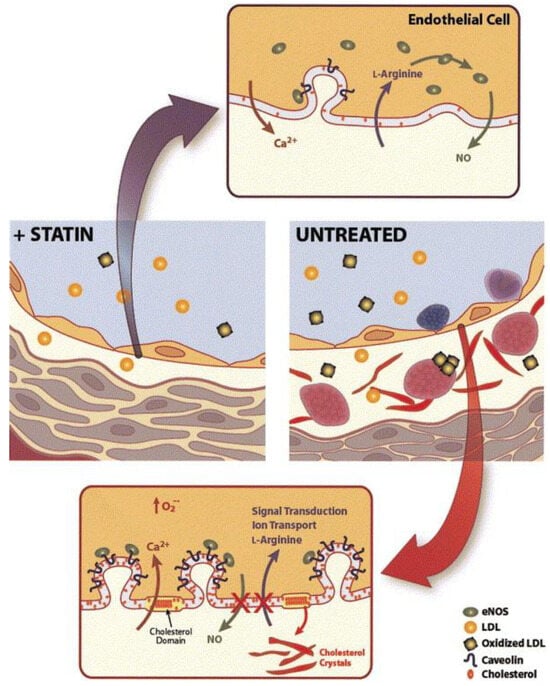
2.2. Plaque Stabilization
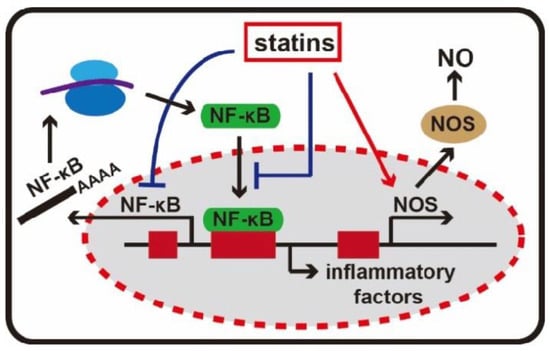
2.3. Anti-Inflammatory Effects
2.4. Immunomodulatory Effects
2.5. Anti-Thrombotic Effects
2.6. Reduced Oxidative Stress
2.7. Protection from High-Decibel Noise-Inducing Hearing Loss
2.8. Enhance Responses to Immune Checkpoint Blockade in Cancer Models
3. Adverse Effects of Statin Therapy
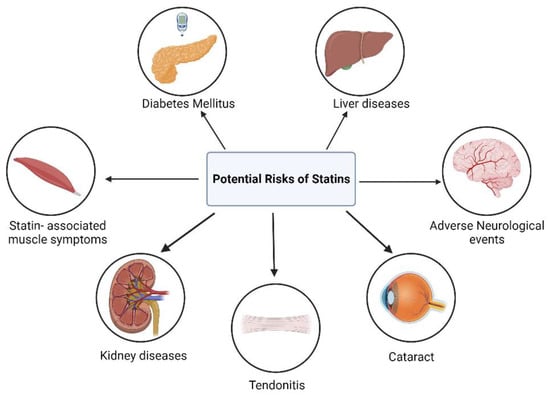
3.1. Myopathy and Rhabdomyolysis
3.2. Diabetes Mellitus
3.3. Liver Diseases
3.4. Adverse Neurological Events
3.5. Cataract
3.6. Kidney Diseases
3.7. Tendonitis and Tendon Rupture
References
- Cardiovascular Diseases (CVDs); World Health Organization: Geneva, Switzerland, 2021.
- Tsao, C.W.; Aday, A.W.; Almarzooq, Z.I.; Alonso, A.; Beaton, A.Z.; Bittencourt, M.S.; Boehme, A.K.; Buxton, A.E.; Carson, A.P.; Commodore-Mensah, Y. Heart disease and stroke statistics—2022 update: A report from the American Heart Association. Circulation 2022, 145, e153–e639.
- Benjamin, E.J.; Virani, S.S.; Callaway, C.W.; Chamberlain, A.M.; Chang, A.R.; Cheng, S.; Chiuve, S.E.; Cushman, M.; Delling, F.N.; Deo, R. Heart disease and stroke statistics—2018 update: A report from the American Heart Association. Circulation 2018, 137, e67–e492.
- Kannel, W.B.; Dawber, T.R.; Kagan, A.; Revotskie, N.; STOKES, J., III. Factors of risk in the development of coronary heart disease—Six-year follow-up experience: The Framingham Study. Ann. Intern. Med. 1961, 55, 33–50.
- Stary, H.C.; Chandler, A.B.; Glagov, S.; Guyton, J.R.; Insull, W., Jr.; Rosenfeld, M.E.; Schaffer, S.A.; Schwartz, C.J.; Wagner, W.D.; Wissler, R.W. A definition of initial, fatty streak, and intermediate lesions of atherosclerosis. A report from the Committee on Vascular Lesions of the Council on Arteriosclerosis, American Heart Association. Circulation 1994, 89, 2462–2478.
- Gofman, J.W.; Delalla, O.; Glazier, F.; Freeman, N.K.; Lindgren, F.T.; Nichols, A.V.; Strisower, B.; Tamplin, A.R. The serum lipoprotein transport system in health, metabolic disorders, atherosclerosis and coronary heart disease. J. Clin. Lipidol. 2007, 1, 104–141.
- Libby, P. The changing landscape of atherosclerosis. Nature 2021, 592, 524–533.
- Vega, G.L.; Grundy, S.M. Influence of lovastatin therapy on metabolism of low density lipoproteins in mixed hyperlipidaemia. J. Intern. Med. 1991, 230, 341–350.
- Goldstein, J.L.; Brown, M.S. A century of cholesterol and coronaries: From plaques to genes to statins. Cell 2015, 161, 161–172.
- Goldstein, J.L.; Brown, M.S. Familial hypercholesterolemia: Identification of a defect in the regulation of 3-hydroxy-3-methylglutaryl coenzyme A reductase activity associated with overproduction of cholesterol. Proc. Natl. Acad. Sci. USA 1973, 70, 2804–2808.
- Manzoni, M.; Rollini, M. Biosynthesis and biotechnological production of statins by filamentous fungi and application of these cholesterol-lowering drugs. Appl. Microbiol. Biotechnol. 2002, 58, 555–564.
- Xie, X.; Tang, Y. Efficient synthesis of simvastatin by use of whole-cell biocatalysis. Appl. Environ. Microbiol. 2007, 73, 2054–2060.
- Murphy, C.; Deplazes, E.; Cranfield, C.G.; Garcia, A. The role of structure and biophysical properties in the pleiotropic effects of statins. Int. J. Mol. Sci. 2020, 21, 8745.
- Istvan, E.S.; Deisenhofer, J. Structural mechanism for statin inhibition of HMG-CoA reductase. Science 2001, 292, 1160–1164.
- Ziaeian, B.; Fonarow, G.C. Statins and the prevention of heart disease. JAMA Cardiol. 2017, 2, 464.
- Baigent, C.; Blackwell, L.; Emberson, J.; Holland, L.; Reith, C.; Bhala, N.; Peto, R.; Barnes, E.; Keech, A.; Simes, J. Efficacy and safety of more intensive lowering of LDL cholesterol: A meta-analysis of data from 170,000 participants in 26 randomized trials. Lancet 2010, 376, 1670–1681.
- Baigent, C.; Keech, A.; Kearney, P.M.; Blackwell, L.; Buck, G.; Pollicino, C.; Kirby, A.; Sourjina, T.; Peto, R.; Collins, R.; et al. Efficacy and safety of cholesterol-lowering treatment: Prospective meta-analysis of data from 90,056 participants in 14 randomised trials of statins. Lancet 2005, 366, 1267–1278.
- Ridker, P.M.; Danielson, E.; Fonseca, F.A.; Genest, J.; Gotto Jr, A.M.; Kastelein, J.J.; Koenig, W.; Libby, P.; Lorenzatti, A.J.; MacFadyen, J.G. Rosuvastatin to prevent vascular events in men and women with elevated C-reactive protein. N. Engl. J. Med. 2008, 359, 2195–2207.
- Ridker, P.M.; Rifai, N.; Pfeffer, M.A.; Sacks, F.M.; Moye, L.A.; Goldman, S.; Flaker, G.C.; Braunwald, E. Inflammation, pravastatin, and the risk of coronary events after myocardial infarction in patients with average cholesterol levels. Circulation 1998, 98, 839–844.
- Grundy, S.M.; Stone, N.J.; Bailey, A.L.; Beam, C.; Birtcher, K.K.; Blumenthal, R.S.; Braun, L.T.; De Ferranti, S.; Faiella-Tommasino, J.; Forman, D.E. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA guideline on the management of blood cholesterol: Executive summary: A report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J. Am. Coll. Cardiol. 2019, 73, 3168–3209.
- Muscoli, S.; Ifrim, M.; Russo, M.; Candido, F.; Sanseviero, A.; Milite, M.; Di Luozzo, M.; Marchei, M.; Sangiorgi, G.M. Current Options and Future Perspectives in the Treatment of Dyslipidemia. J. Clin. Med. 2022, 11, 4716.
- Kosmas, C.E.; Pantou, D.; Sourlas, A.; Papakonstantinou, E.J.; Uceta, R.E.; Guzman, E. New and emerging lipid-modifying drugs to lower LDL cholesterol. Drugs Context 2021, 10, 1–22.
- Laufs, U.; La Fata, V.; Plutzky, J.; Liao, J.K. Upregulation of endothelial nitric oxide synthase by HMG CoA reductase inhibitors. Circulation 1998, 97, 1129–1135.
- Ota, H.; Eto, M.; Kano, M.R.; Kahyo, T.; Setou, M.; Ogawa, S.; Iijima, K.; Akishita, M.; Ouchi, Y. Induction of endothelial nitric oxide synthase, SIRT1, and catalase by statins inhibits endothelial senescence through the Akt pathway. Arterioscler. Thromb. Vasc. Biol. 2010, 30, 2205–2211.
- Dichtl, W.; Dulak, J.; Frick, M.; Alber, H.F.; Schwarzacher, S.P.; Ares, M.P.; Nilsson, J.; Pachinger, O.; Weidinger, F. HMG-CoA reductase inhibitors regulate inflammatory transcription factors in human endothelial and vascular smooth muscle cells. Arterioscler. Thromb. Vasc. Biol. 2003, 23, 58–63.
- Kuwahara, N.; Sasaki, S.; Kobara, M.; Nakata, T.; Tatsumi, T.; Irie, H.; Narumiya, H.; Hatta, T.; Takeda, K.; Matsubara, H. HMG-CoA reductase inhibition improves anti-aging klotho protein expression and arteriosclerosis in rats with chronic inhibition of nitric oxide synthesis. Int. J. Cardiol. 2008, 123, 84–90.
- Mason, R.P.; Walter, M.F.; Jacob, R.F. Effects of HMG-CoA reductase inhibitors on endothelial function: Role of microdomains and oxidative stress. Circulation 2004, 109, II-34–II-41.
- Hattori, K.; Ozaki, Y.; Ismail, T.F.; Okumura, M.; Naruse, H.; Kan, S.; Ishikawa, M.; Kawai, T.; Ohta, M.; Kawai, H. Impact of statin therapy on plaque characteristics as assessed by serial OCT, grayscale and integrated backscatter–IVUS. JACC Cardiovasc. Imaging 2012, 5, 169–177.
- Nissen, S.E.; Tuzcu, E.M.; Schoenhagen, P.; Brown, B.G.; Ganz, P.; Vogel, R.A.; Crowe, T.; Howard, G.; Cooper, C.J.; Brodie, B. Effect of intensive compared with moderate lipid-lowering therapy on progression of coronary atherosclerosis: A randomized controlled trial. JAMA 2004, 291, 1071–1080.
- Nissen, S.E.; Nicholls, S.J.; Sipahi, I.; Libby, P.; Raichlen, J.S.; Ballantyne, C.M.; Davignon, J.; Erbel, R.; Fruchart, J.C.; Tardif, J.-C. Effect of very high-intensity statin therapy on regression of coronary atherosclerosis: The ASTEROID trial. JAMA 2006, 295, 1556–1565.
- Nicholls, S.J.; Ballantyne, C.M.; Barter, P.J.; Chapman, M.J.; Erbel, R.M.; Libby, P.; Raichlen, J.S.; Uno, K.; Borgman, M.; Wolski, K. Effect of two intensive statin regimens on progression of coronary disease. N. Engl. J. Med. 2011, 365, 2078–2087.
- Komukai, K.; Kubo, T.; Kitabata, H.; Matsuo, Y.; Ozaki, Y.; Takarada, S.; Okumoto, Y.; Shiono, Y.; Orii, M.; Shimamura, K. Effect of atorvastatin therapy on fibrous cap thickness in coronary atherosclerotic plaque as assessed by optical coherence tomography: The EASY-FIT study. J. Am. Coll. Cardiol. 2014, 64, 2207–2217.
- Lee, S.-E.; Chang, H.-J.; Sung, J.M.; Park, H.-B.; Heo, R.; Rizvi, A.; Lin, F.Y.; Kumar, A.; Hadamitzky, M.; Kim, Y.J. Effects of statins on coronary atherosclerotic plaques: The PARADIGM study. JACC Cardiovasc. Imaging 2018, 11, 1475–1484.
- Zhang, Q.; Dong, J.; Yu, Z. Pleiotropic use of Statins as non-lipid-lowering drugs. Int. J. Biol. Sci. 2020, 16, 2704–2711.
- Albert, M.A.; Danielson, E.; Rifai, N.; Ridker, P.M.; Investigators, P.; Investigators, P. Effect of statin therapy on C-reactive protein levels: The pravastatin inflammation/CRP evaluation (PRINCE): A randomized trial and cohort study. JAMA 2001, 286, 64–70.
- Subramanian, S.; Emami, H.; Vucic, E.; Singh, P.; Vijayakumar, J.; Fifer, K.M.; Alon, A.; Shankar, S.S.; Farkouh, M.; Rudd, J.H. High-dose atorvastatin reduces periodontal inflammation: A novel pleiotropic effect of statins. J. Am. Coll. Cardiol. 2013, 62, 2382–2391.
- Wiklund, O.; Mattsson-Hulten, L.; Hurt-Camejo, E.; Oscarsson, J. Effects of simvastatin and atorvastatin on inflammation markers in plasma. J. Intern. Med. 2002, 251, 338–347.
- Jain, M.K.; Ridker, P.M. Anti-inflammatory effects of statins: Clinical evidence and basic mechanisms. Nat. Rev. Drug Discov. 2005, 4, 977–987.
- Kwak, B.; Mulhaupt, F.; Myit, S.; Mach, F. Statins as a newly recognized type of immunomodulator. Nat. Med. 2000, 6, 1399–1402.
- Hakamada-Taguchi, R.; Uehara, Y.; Kuribayashi, K.; Numabe, A.; Saito, K.; Negoro, H.; Fujita, T.; Toyo-oka, T.; Kato, T. Inhibition of hydroxymethylglutaryl-coenzyme a reductase reduces Th1 development and promotes Th2 development. Circ. Res. 2003, 93, 948–956.
- Ali, F.Y.; Armstrong, P.C.; Dhanji, A.-R.A.; Tucker, A.T.; Paul-Clark, M.J.; Mitchell, J.A.; Warner, T.D. Antiplatelet actions of statins and fibrates are mediated by PPARs. Arterioscler. Thromb. Vasc. Biol. 2009, 29, 706–711.
- Haramaki, N.; Ikeda, H.; Takenaka, K.; Katoh, A.; Sugano, R.; Yamagishi, S.-i.; Matsuoka, H.; Imaizumi, T. Fluvastatin alters platelet aggregability in patients with hypercholesterolemia: Possible improvement of intraplatelet redox imbalance via HMG-CoA reductase. Arterioscler. Thromb. Vasc. Biol. 2007, 27, 1471–1477.
- Pignatelli, P.; Carnevale, R.; Pastori, D.; Cangemi, R.; Napoleone, L.; Bartimoccia, S.; Nocella, C.; Basili, S.; Violi, F. Immediate antioxidant and antiplatelet effect of atorvastatin via inhibition of Nox2. Circulation 2012, 126, 92–103.
- Ridker, P.M.; Pradhan, A.; MacFadyen, J.G.; Libby, P.; Glynn, R.J. Cardiovascular benefits and diabetes risks of statin therapy in primary prevention: An analysis from the JUPITER trial. Lancet 2012, 380, 565–571.
- Joseph, P.; Glynn, R.; Lonn, E.; Ramasundarahettige, C.; Eikelboom, J.; MacFadyen, J.; Ridker, P.; Yusuf, S. Rosuvastatin for the prevention of venous thromboembolism: A pooled analysis of the HOPE-3 and JUPITER randomized controlled trials. Cardiovasc. Res. 2022, 118, 897–903.
- Wassmann, S.; Laufs, U.; Bäumer, A.T.; Müller, K.; Ahlbory, K.; Linz, W.; Itter, G.; Rösen, R.; Böhm, M.; Nickenig, G. HMG-CoA reductase inhibitors improve endothelial dysfunction in normocholesterolemic hypertension via reduced production of reactive oxygen species. Hypertension 2001, 37, 1450–1457.
- Aviram, M.; Hussein, O.; Rosenblat, M.; Schlezinger, S.; Hayek, T.; Keidar, S. Interactions of platelets, macrophages, and lipoproteins in hypercholesterolemia: Antiatherogenic effects of HMG-CoA reductase inhibitor therapy. J. Cardiovasc. Pharmacol. 1998, 31, 39–45.
- Wassmann, S.; Laufs, U.; Müller, K.; Konkol, C.; Ahlbory, K.; Bäumer, A.T.; Linz, W.; Böhm, M.; Nickenig, G. Cellular antioxidant effects of atorvastatin in vitro and in vivo. Arterioscler. Thromb. Vasc. Biol. 2002, 22, 300–305.
- Takemoto, M.; Node, K.; Nakagami, H.; Liao, Y.; Grimm, M.; Takemoto, Y.; Kitakaze, M.; Liao, J.K. Statins as antioxidant therapy for preventing cardiac myocyte hypertrophy. J. Clin. Investig. 2001, 108, 1429–1437.
- Dépreux, F.; Czech, L.; Young, H.; Richter, C.-P.; Zhou, Y.; Whitlon, D.S. Statins protect mice from high-decibel noise-induced hearing loss. Biomed. Pharmacother. 2023, 163, 114674.
- Richter, C.-P.; Young, H.; Richter, S.V.; Smith-Bronstein, V.; Stock, S.R.; Xiao, X.; Soriano, C.; Whitlon, D.S. Fluvastatin protects cochleae from damage by high-level noise. Sci. Rep. 2018, 8, 3033.
- Matsuzaki, M.; Kita, T.; Mabuchi, H.; Matsuzawa, Y.; Nakaya, N.; Oikawa, S.; Saito, Y.; Sasaki, J.; Shimamoto, K.; Itakura, H. Large Scale Cohort Study of the Relationship between Serum Cholesterol Concentration and Coronary Events with Low-Dose Simvastatin Therapy in Japanese Patients with Hypercholesterolemia Primary Prevention Cohort Study of the Japan Lipid Intervention Trial (J-LIT). Circ. J. 2002, 66, 1087–1095.
- Newman, T.B.; Hulley, S.B. Carcinogenicity of lipid-lowering drugs. JAMA 1996, 275, 55–60.
- Law, M.; Rudnicka, A.R. Statin safety: A systematic review. Am. J. Cardiol. 2006, 97, S52–S60.
- Cheeley, M.K.; Saseen, J.J.; Agarwala, A.; Ravilla, S.; Ciffone, N.; Jacobson, T.A.; Dixon, D.L.; Maki, K.C. NLA scientific statement on statin intolerance: A new definition and key considerations for ASCVD risk reduction in the statin intolerant patient. J. Clin. Lipidol. 2022, 16, 361–375.
- Stroes, E.S.; Thompson, P.D.; Corsini, A.; Vladutiu, G.D.; Raal, F.J.; Ray, K.K.; Roden, M.; Stein, E.; Tokgözoğlu, L.; Nordestgaard, B.G. Statin-associated muscle symptoms: Impact on statin therapy—European Atherosclerosis Society consensus panel statement on assessment, aetiology and management. Eur. Heart J. 2015, 36, 1012–1022.
- Ruscica, M.; Reiner, Z.; Sirtori, C.R. Can we further optimize statin therapy to increase tolerability? Expert. Opin. Drug Discov. 2019, 14, 843–847.
- Sattar, N.; Preiss, D.; Murray, H.M.; Welsh, P.; Buckley, B.M.; de Craen, A.J.; Seshasai, S.R.K.; McMurray, J.J.; Freeman, D.J.; Jukema, J.W. Statins and risk of incident diabetes: A collaborative meta-analysis of randomised statin trials. Lancet 2010, 375, 735–742.
- Rajpathak, S.N.; Kumbhani, D.J.; Crandall, J.; Barzilai, N.; Alderman, M.; Ridker, P.M. Statin therapy and risk of developing type 2 diabetes: A meta-analysis. Diabetes Care 2009, 32, 1924–1929.
- Chogtu, B.; Magazine, R.; Bairy, K. Statin use and risk of diabetes mellitus. World J. Diabetes 2015, 6, 352.
- Miller, P.E.; Martin, S.S. Approach to statin use in 2016: An update. Curr. Atheroscler. Rep. 2016, 18, 20.
- Pastori, D.; Pani, A.; Di Rocco, A.; Menichelli, D.; Gazzaniga, G.; Farcomeni, A.; D’Erasmo, L.; Angelico, F.; Del Ben, M.; Baratta, F. Statin liver safety in non-alcoholic fatty liver disease: A systematic review and metanalysis. Br. J. Clin. Pharmacol. 2022, 88, 441–451.
- Ward, N.C.; Watts, G.F.; Eckel, R.H. Statin toxicity: Mechanistic insights and clinical implications. Circ. Res. 2019, 124, 328–350.
- Reuben, A.; Koch, D.G.; Lee, W.M. Drug-induced acute liver failure: Results of a US multicenter, prospective study. Hepatology 2010, 52, 2065–2076.
- Vergouwen, M.D.; De Haan, R.J.; Vermeulen, M.; Roos, Y.B. Statin treatment and the occurrence of hemorrhagic stroke in patients with a history of cerebrovascular disease. Stroke 2008, 39, 497–502.
- Amarenco, P. For the Stroke Prevention by Aggressive Reduction in Cholesterol Levels (SPARCL) Investigators: High-dose atorvastatin after stroke or transient ischemic attack. N. Engl. J. Med. 2006, 355, 549–559.
- Morofuji, Y.; Nakagawa, S.; Ujifuku, K.; Fujimoto, T.; Otsuka, K.; Niwa, M.; Tsutsumi, K. Beyond lipid-lowering: Effects of statins on cardiovascular and cerebrovascular diseases and cancer. Pharmaceuticals 2022, 15, 151.
- Ribe, A.R.; Vestergaard, C.H.; Vestergaard, M.; Pedersen, H.S.; Prior, A.; Lietzen, L.W.; Brynningsen, P.K.; Fenger-Grøn, M. Statins and risk of intracerebral hemorrhage in individuals with a history of stroke. Stroke 2020, 51, 1111–1119.
- Hackam, D.G.; Woodward, M.; Newby, L.K.; Bhatt, D.L.; Shao, M.; Smith, E.E.; Donner, A.; Mamdani, M.; Douketis, J.D.; Arima, H. Statins and intracerebral hemorrhage: Collaborative systematic review and meta-analysis. Circulation 2011, 124, 2233–2242.
- Swiger, K.J.; Manalac, R.J.; Blumenthal, R.S.; Blaha, M.J.; Martin, S.S. Statins and cognition: A systematic review and meta-analysis of short-and long-term cognitive effects. Mayo Clin. Proc. 2013, 88, 1213–1221.
- Richardson, K.; Schoen, M.; French, B.; Umscheid, C.A.; Mitchell, M.D.; Arnold, S.E.; Heidenreich, P.A.; Rader, D.J.; Degoma, E.M. Statins and cognitive function: A systematic review. Ann. Intern. Med. 2013, 159, 688–697.
- Kirby, T. Cataracts produced by triparanol.(MER-29). Trans. Am. Ophthalmol. Soc. 1967, 65, 494.
- Dormuth, C.R.; Hemmelgarn, B.R.; Paterson, J.M.; James, M.T.; Teare, G.F.; Raymond, C.B.; Lafrance, J.-P.; Levy, A.; Garg, A.X.; Ernst, P. Use of high potency statins and rates of admission for acute kidney injury: Multicenter, retrospective observational analysis of administrative databases. BMJ 2013, 346, f880.
- Kearns, M.C.; Singh, V.K. Bilateral patellar tendon rupture associated with statin use. J. Surg. Case Rep. 2016, 2016, rjw072.
- Beri, A.; Dwamena, F.C.; Dwamena, B.A. Association between statin therapy and tendon rupture: A case-control study. J. Cardiovasc. Pharmacol. 2009, 53, 401–404.





