
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Caroline S. Stokes | -- | 2736 | 2024-02-21 09:53:11 | | | |
| 2 | Lindsay Dong | Meta information modification | 2736 | 2024-02-23 02:34:00 | | |
Video Upload Options
A large body of research shows an association between higher body weight and low vitamin D status, as assessed using serum 25-hydroxyvitamin D concentrations. Vitamin D can be metabolised in adipose tissue and has been reported to influence gene expression and modulate inflammation and adipose tissue metabolism in vitro. However, the exact metabolism of vitamin D in adipose tissue is currently unknown. White adipose tissue expresses the vitamin D receptor and hydroxylase enzymes, substantially involved in vitamin D metabolism and efficacy. The distribution and concentrations of the generated vitamin D compounds in adipose tissue, however, are largely unknown. Closing this knowledge gap could help to understand whether the different vitamin D compounds have specific health effects in the setting of adiposity.
1. Introduction
1.1. Vitamin D Metabolism
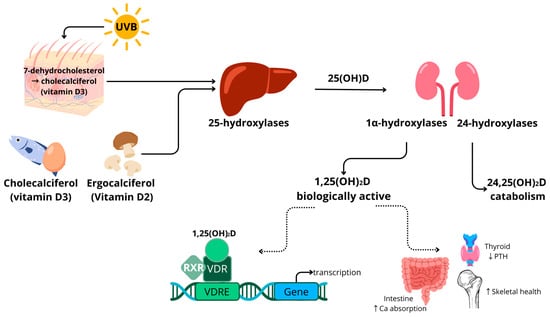
1.2. Vitamin D in Adiposity
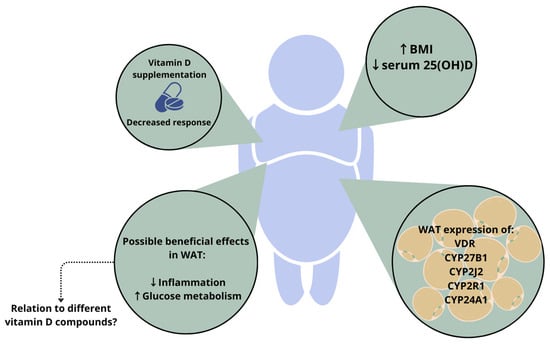
2. Relevance of Different Vitamin D Compounds
2.1. Free Vitamin D Compound
2.2. 1,25-Dihydroxyvitamin D (1,25(OH)2D)
2.3. 24,25-Dihydroxyvitamin D (24,25(OH)2D)
2.4. C-3 Epimers of Vitamin D Compounds
References
- Holick, M.F. Vitamin D and bone health. J. Nutr. 1996, 126, 1159S–1164S.
- Holick, M.F. The role of vitamin D for bone health and fracture prevention. Curr. Osteoporos. Rep. 2006, 4, 96–102.
- Bouillon, R.; Manousaki, D.; Rosen, C.; Trajanoska, K.; Rivadeneira, F.; Richards, J.B. The health effects of vitamin D supplementation: Evidence from human studies. Nat. Rev. Endocrinol. 2022, 18, 96–110.
- Holick, M.F. Deficiency of sunlight and vitamin D. BMJ 2008, 336, 1318–1319.
- Cashman, K.D. Vitamin D deficiency: Defining, prevalence, causes, and strategies of addressing. Calcif. Tissue Int. 2020, 106, 14–29.
- Holick, M.F.; Binkley, N.C.; Bischoff-Ferrari, H.A.; Gordon, C.M.; Hanley, D.A.; Heaney, R.P.; Murad, M.H.; Weaver, C.M. Evaluation, treatment, and prevention of vitamin D deficiency: An Endocrine Society clinical practice guideline. J. Clin. Endocrinol. Metab. 2011, 96, 1911–1930.
- Dissanayake, H.A.; de Silva, N.L.; Sumanatilleke, M.; de Silva, S.D.N.; Gamage, K.K.K.; Dematapitiya, C.; Kuruppu, D.C.; Ranasinghe, P.; Pathmanathan, S.; Katulanda, P. Prognostic and therapeutic role of vitamin D in COVID-19: Systematic review and meta-analysis. J. Clin. Endocrinol. Metab. 2022, 107, 1484–1502.
- Shah, K.; Saxena, D.; Mavalankar, D. Vitamin D supplementation, COVID-19 and disease severity: A meta-analysis. QJM Int. J. Med. 2021, 114, 175–181.
- Bikle, D.; Bouillon, R.; Thadhani, R.; Schoenmakers, I. Vitamin D metabolites in captivity? Should we measure free or total 25 (OH) D to assess vitamin D status? J. Steroid Biochem. Mol. Biol. 2017, 173, 105–116.
- Binkley, N.; Borchardt, G.; Siglinsky, E.; Krueger, D. Does vitamin D metabolite measurement help predict 25 (OH) D change following vitamin D supplementation? Endocr. Pract. 2017, 23, 432–441.
- Makris, K.; Sempos, C.; Cavalier, E. The measurement of vitamin D metabolites part II—The measurement of the various vitamin D metabolites. Hormones 2020, 19, 97–107.
- Wildman, R.P.; Muntner, P.; Reynolds, K.; McGinn, A.P.; Rajpathak, S.; Wylie-Rosett, J.; Sowers, M.R. The obese without cardiometabolic risk factor clustering and the normal weight with cardiometabolic risk factor clustering: Prevalence and correlates of 2 phenotypes among the US population (NHANES 1999–2004). Arch. Intern. Med. 2008, 168, 1617–1624.
- Noubiap, J.J.; Nansseu, J.R.; Lontchi-Yimagou, E.; Nkeck, J.R.; Nyaga, U.F.; Ngouo, A.T.; Tounouga, D.N.; Tianyi, F.L.; Foka, A.J.; Ndoadoumgue, A.L. Geographic distribution of metabolic syndrome and its components in the general adult population: A meta-analysis of global data from 28 million individuals. Diabetes Res. Clin. Pract. 2022, 188, 109924.
- Rao, D.S.; Raghuramulu, N. Food chain as origin of vitamin D in fish. Comp. Biochem. Physiol. Part A Physiol. 1996, 114, 15–19.
- Atsuko, T.; Toshio, O.; Makoto, T.; Tadashi, K. Possible origin of extremely high contents of vitamin D3 in some kinds of fish liver. Comp. Biochem. Physiol. Part A Physiol. 1991, 100, 483–487.
- Uwitonze, A.M.; Razzaque, M.S. Role of magnesium in vitamin D activation and function. J. Osteopath. Med. 2018, 118, 181–189.
- Risco, F.; Traba, M. Influence of magnesium on the in vitro synthesis of 24, 25-dihydroxyvitamin D3 and 1 alpha, 25-dihydroxyvitamin D3. Magnes. Res. 1992, 5, 5–14.
- Saponaro, F.; Saba, A.; Zucchi, R. An update on vitamin D metabolism. Int. J. Mol. Sci. 2020, 21, 6573.
- Plum, L.A.; DeLuca, H.F. Vitamin D, disease and therapeutic opportunities. Nat. Rev. Drug Discov. 2010, 9, 941–955.
- Carlberg, C. Vitamin D and Its Target Genes. Nutrients 2022, 14, 1354.
- Goltzman, D.; Mannstadt, M.; Marcocci, C. Physiology of the calcium-parathyroid hormone-vitamin D axis. Vitam. D Clin. Med. 2018, 50, 1–13.
- Pereira-Santos, M.; Costa, P.d.F.; Assis, A.d.; Santos, C.d.S.; Santos, D.d. Obesity and vitamin D deficiency: A systematic review and meta-analysis. Obes. Rev. 2015, 16, 341–349.
- Zakharova, I.; Klimov, L.; Kuryaninova, V.; Nikitina, I.; Malyavskaya, S.; Dolbnya, S.; Kasyanova, A.; Atanesyan, R.; Stoyan, M.; Todieva, A. Vitamin D insufficiency in overweight and obese children and adolescents. Front. Endocrinol. 2019, 10, 103.
- Abbas, M.A. Physiological functions of Vitamin D in adipose tissue. J. Steroid Biochem. Mol. Biol. 2017, 165, 369–381.
- Drincic, A.T.; Armas, L.A.; Van Diest, E.E.; Heaney, R.P. Volumetric dilution, rather than sequestration best explains the low vitamin D status of obesity. Obesity 2012, 20, 1444–1448.
- Blum, M.; Dolnikowski, G.; Seyoum, E.; Harris, S.S.; Booth, S.L.; Peterson, J.; Saltzman, E.; Dawson-Hughes, B. Vitamin D3 in fat tissue. Endocrine 2008, 33, 90–94.
- Himbert, C.; Ose, J.; Delphan, M.; Ulrich, C.M. A systematic review of the interrelation between diet-and surgery-induced weight loss and vitamin D status. Nutr. Res. 2017, 38, 13–26.
- Li, J.; Byrne, M.E.; Chang, E.; Jiang, Y.; Donkin, S.S.; Buhman, K.K.; Burgess, J.R.; Teegarden, D. 1α, 25-Dihydroxyvitamin D hydroxylase in adipocytes. J. Steroid Biochem. Mol. Biol. 2008, 112, 122–126.
- Nimitphong, H.; Holick, M.F.; Fried, S.K.; Lee, M.-J. 25-hydroxyvitamin D3 and 1, 25-dihydroxyvitamin D3 promote the differentiation of human subcutaneous preadipocytes. PLoS ONE 2012, 7, e52171.
- Wamberg, L.; Christiansen, T.; Paulsen, S.; Fisker, S.; Rask, P.; Rejnmark, L.; Richelsen, B.; Pedersen, S. Expression of vitamin D-metabolizing enzymes in human adipose tissue—The effect of obesity and diet-induced weight loss. Int. J. Obes. 2013, 37, 651–657.
- Jonas, M.I.; Kuryłowicz, A.; Bartoszewicz, Z.; Lisik, W.; Jonas, M.; Kozniewski, K.; Puzianowska-Kuznicka, M. Vitamin D receptor gene expression in adipose tissue of obese individuals is regulated by miRNA and correlates with the pro-inflammatory cytokine level. Int. J. Mol. Sci. 2019, 20, 5272.
- Elkhwanky, M.S.; Kummu, O.; Piltonen, T.T.; Laru, J.; Morin-Papunen, L.; Mutikainen, M.; Tavi, P.; Hakkola, J. Obesity represses CYP2R1, the vitamin D 25-hydroxylase, in the liver and extrahepatic tissues. JBMR Plus 2020, 4, e10397.
- Ochs-Balcom, H.M.; Chennamaneni, R.; Millen, A.E.; Shields, P.G.; Marian, C.; Trevisan, M.; Freudenheim, J.L. Vitamin D receptor gene polymorphisms are associated with adiposity phenotypes. Am. J. Clin. Nutr. 2011, 93, 5–10.
- Ruiz-Ojeda, F.J.; Anguita-Ruiz, A.; Leis, R.; Aguilera, C.M. Genetic factors and molecular mechanisms of vitamin D and obesity relationship. Ann. Nutr. Metab. 2018, 73, 89–99.
- Tobias, D.K.; Luttmann-Gibson, H.; Mora, S.; Danik, J.; Bubes, V.; Copeland, T.; LeBoff, M.S.; Cook, N.R.; Lee, I.-M.; Buring, J.E. Association of Body Weight With Response to Vitamin D Supplementation and Metabolism. JAMA Netw. Open 2023, 6, e2250681.
- van den Heuvel, E.G.; Lips, P.; Schoonmade, L.J.; Lanham-New, S.A.; van Schoor, N.M. Comparison of the effect of daily vitamin D2 and vitamin D3 supplementation on serum 25-hydroxyvitamin D concentration (total 25 (OH) D, 25 (OH) D2 and 25 (OHD3) and importance of body mass index: A systematic review and meta-analysis. Adv. Nutr. 2023.
- Liu, J.; Fox, C.S.; Hickson, D.A.; May, W.D.; Hairston, K.G.; Carr, J.J.; Taylor, H.A. Impact of abdominal visceral and subcutaneous adipose tissue on cardiometabolic risk factors: The Jackson Heart Study. J. Clin. Endocrinol. Metab. 2010, 95, 5419–5426.
- Fox, C.S.; Massaro, J.M.; Hoffmann, U.; Pou, K.M.; Maurovich-Horvat, P.; Liu, C.-Y.; Vasan, R.S.; Murabito, J.M.; Meigs, J.B.; Cupples, L.A. Abdominal visceral and subcutaneous adipose tissue compartments: Association with metabolic risk factors in the Framingham Heart Study. Circulation 2007, 116, 39–48.
- Cancello, R.; Clement, K. Is obesity an inflammatory illness? Role of low-grade inflammation and macrophage infiltration in human white adipose tissue. BJOG Int. J. Obstet. Gynaecol. 2006, 113, 1141–1147.
- Eckel, R.H.; Alberti, K.G.; Grundy, S.M.; Zimmet, P.Z. The metabolic syndrome. Lancet 2010, 375, 181–183.
- van Etten, E.; Mathieu, C. Immunoregulation by 1, 25-dihydroxyvitamin D3: Basic concepts. J. Steroid Biochem. Mol. Biol. 2005, 97, 93–101.
- Charoenngam, N.; Holick, M.F. Immunologic effects of vitamin D on human health and disease. Nutrients 2020, 12, 2097.
- Lei, X.; Zhou, Q.; Wang, Y.; Fu, S.; Li, Z.; Chen, Q. Serum and supplemental vitamin D levels and insulin resistance in T2DM populations: A meta-analysis and systematic review. Sci. Rep. 2023, 13, 12343.
- Farahmand, M.A.; Daneshzad, E.; Fung, T.T.; Zahidi, F.; Muhammadi, M.; Bellissimo, N.; Azadbakht, L. What is the impact of vitamin D supplementation on glycemic control in people with type-2 diabetes: A systematic review and meta-analysis of randomized controlled trails. BMC Endocr. Disord. 2023, 23, 15.
- Nykjaer, A.; Dragun, D.; Walther, D.; Vorum, H.; Jacobsen, C.; Herz, J.; Melsen, F.; Christensen, E.I.; Willnow, T.E. An endocytic pathway essential for renal uptake and activation of the steroid 25-(OH) vitamin D3. Cell 1999, 96, 507–515.
- Bikle, D.D. The free hormone hypothesis: When, why, and how to measure the free hormone levels to assess vitamin D, thyroid, sex hormone, and cortisol status. JBMR Plus 2021, 5, e10418.
- Mendel, C.M. The free hormone hypothesis: A physiologically based mathematical model. Endocr. Rev. 1989, 10, 232–274.
- Bikle, D.D.; Malmstroem, S.; Schwartz, J. Current controversies: Are free vitamin metabolite levels a more accurate assessment of vitamin D status than total levels? Endocrinol. Metab. Clin. 2017, 46, 901–918.
- Schwartz, J.; Lai, J.; Lizaola, B.; Kane, L.; Weyland, P.; Terrault, N.; Stotland, N.; Bikle, D. Variability in free 25 (OH) vitamin D levels in clinical populations. J. Steroid Biochem. Mol. Biol. 2014, 144, 156–158.
- Marques-Pamies, M.; López-Molina, M.; Pellitero, S.; Santillan, C.S.; Martínez, E.; Moreno, P.; Tarascó, J.; Granada, M.L.; Puig-Domingo, M. Differential behavior of 25 (OH) D and f25 (OH) D3 in patients with morbid obesity after bariatric surgery. Obes. Surg. 2021, 31, 3990–3995.
- Karlsson, T.; Osmancevic, A.; Jansson, N.; Hulthén, L.; Holmäng, A.; Larsson, I. Increased vitamin D-binding protein and decreased free 25 (OH) D in obese women of reproductive age. Eur. J. Nutr. 2014, 53, 259–267.
- Brommage, R.; Deluca, H.F. Evidence that 1, 25-dihydroxyvitamin D3 is the physiologically active metabolite of vitamin D3. Endocr. Rev. 1985, 6, 491–511.
- Nimitphong, H.; Guo, W.; Holick, M.F.; Fried, S.K.; Lee, M.J. Vitamin D inhibits adipokine production and inflammatory signaling through the vitamin D receptor in human adipocytes. Obesity 2021, 29, 562–568.
- Karkeni, E.; Bonnet, L.; Marcotorchino, J.; Tourniaire, F.; Astier, J.; Ye, J.; Landrier, J.-F. Vitamin D limits inflammation-linked microRNA expression in adipocytes in vitro and in vivo: A new mechanism for the regulation of inflammation by vitamin D. Epigenetics 2018, 13, 156–162.
- Marcotorchino, J.; Tourniaire, F.; Landrier, J.-F. Vitamin D, adipose tissue, and obesity. Horm. Mol. Biol. Clin. Investig. 2013, 15, 123–128.
- Mutt, S.J.; Karhu, T.; Lehtonen, S.; Lehenkari, P.; Carlberg, C.; Saarnio, J.; Sebert, S.; Hyppönen, E.; Järvelin, M.R.; Herzig, K.H. Inhibition of cytokine secretion from adipocytes by 1, 25-dihydroxyvitamin D3 via the NF-κB pathway. FASEB J. 2012, 26, 4400–4407.
- Wamberg, L.; Cullberg, K.; Rejnmark, L.; Richelsen, B.; Pedersen, S. Investigations of the anti-inflammatory effects of vitamin D in adipose tissue: Results from an in vitro study and a randomized controlled trial. Horm. Metab. Res. 2013, 45, 456–462.
- Trayhurn, P.; Bing, C.; Wood, I.S. Adipose tissue and adipokines—Energy regulation from the human perspective. J. Nutr. 2006, 136, 1935S–1939S.
- Sahu, B.; Bal, N.C. Adipokines from white adipose tissue in regulation of whole body energy homeostasis. Biochimie 2023, 204, 92–107.
- Gruzdeva, O.; Borodkina, D.; Uchasova, E.; Dyleva, Y.; Barbarash, O. Leptin resistance: Underlying mechanisms and diagnosis. Diabetes Metab. Syndr. Obes. Targets Ther. 2019, 12, 191–198.
- Izquierdo, A.G.; Crujeiras, A.B.; Casanueva, F.F.; Carreira, M.C. Leptin, obesity, and leptin resistance: Where are we 25 years later? Nutrients 2019, 11, 2704.
- Prolo, P.; Wong, M.-L.; Licinio, J. Leptin. Int. J. Biochem. Cell Biol. 1998, 30, 1285–1290.
- Paul, R.F.; Hassan, M.; Nazar, H.S.; Gillani, S.; Afzal, N.; Qayyum, I. Effect of body mass index on serum leptin levels. J. Ayub Med. Coll. Abbottabad 2011, 23, 40–43.
- Kumar, R.; Mal, K.; Razaq, M.K.; Magsi, M.; Memon, M.K.; Memon, S.; Afroz, M.N.; Siddiqui, H.F.; Rizwan, A. Association of leptin with obesity and insulin resistance. Cureus 2020, 12, e12178.
- Wang, Z.V.; Scherer, P.E. Adiponectin, the past two decades. J. Mol. Cell Biol. 2016, 8, 93–100.
- Fang, H.; Judd, R.L. Adiponectin regulation and function. Compr. Physiol. 2011, 8, 1031–1063.
- Lorente-Cebrián, S.; Eriksson, A.; Dunlop, T.; Mejhert, N.; Dahlman, I.; Åström, G.; Sjölin, E.; Wåhlén, K.; Carlberg, C.; Laurencikiene, J. Differential effects of 1α, 25-dihydroxycholecalciferol on MCP-1 and adiponectin production in human white adipocytes. Eur. J. Nutr. 2012, 51, 335–342.
- de Souza, W.N.; Norde, M.M.; Oki, É.; Rogero, M.M.; Marchioni, D.M.; Fisberg, R.M.; Martini, L.A. Association between 25-hydroxyvitamin D and inflammatory biomarker levels in a cross-sectional population-based study, São Paulo, Brazil. Nutr. Res. 2016, 36, 1–8.
- Zhang, M.; Gao, Y.; Tian, L.; Zheng, L.; Wang, X.; Liu, W.; Zhang, Y.; Huang, G. Association of serum 25-hydroxyvitamin D3 with adipokines and inflammatory marker in persons with prediabetes mellitus. Clin. Chim. Acta 2017, 468, 152–158.
- Lwow, F.; Bohdanowicz-Pawlak, A. Vitamin D and selected cytokine concentrations in postmenopausal women in relation to metabolic disorders and physical activity. Exp. Gerontol. 2020, 141, 111107.
- O’Sullivan, A.; Gibney, M.J.; Connor, A.O.; Mion, B.; Kaluskar, S.; Cashman, K.D.; Flynn, A.; Shanahan, F.; Brennan, L. Biochemical and metabolomic phenotyping in the identification of a vitamin D responsive metabotype for markers of the metabolic syndrome. Mol. Nutr. Food Res. 2011, 55, 679–690.
- Al-Sofiani, M.E.; Jammah, A.; Racz, M.; Khawaja, R.A.; Hasanato, R.; El-Fawal, H.A.; Mousa, S.A.; Mason, D.L. Effect of vitamin D supplementation on glucose control and inflammatory response in type II diabetes: A double blind, randomized clinical trial. Int. J. Endocrinol. Metab. 2015, 13, e22604.
- Wagner, D.; Hanwell, H.E.; Schnabl, K.; Yazdanpanah, M.; Kimball, S.; Fu, L.; Sidhom, G.; Rousseau, D.; Cole, D.E.; Vieth, R. The ratio of serum 24, 25-dihydroxyvitamin D3 to 25-hydroxyvitamin D3 is predictive of 25-hydroxyvitamin D3 response to vitamin D3 supplementation. J. Steroid Biochem. Mol. Biol. 2011, 126, 72–77.
- Dugar, A.; Hoofnagle, A.N.; Sanchez, A.P.; Ward, D.M.; Corey-Bloom, J.; Cheng, J.H.; Ix, J.H.; Ginsberg, C. The Vitamin D Metabolite Ratio (VMR) is a Biomarker of Vitamin D Status That is Not Affected by Acute Changes in Vitamin D Binding Protein. Clin. Chem. 2023, 69, 718–723.
- Ginsberg, C.; Hoofnagle, A.N.; Katz, R.; Becker, J.O.; Kritchevsky, S.B.; Shlipak, M.G.; Sarnak, M.J.; Ix, J.H. The vitamin D metabolite ratio is independent of vitamin D binding protein concentration. Clin. Chem. 2021, 67, 385–393.
- Herrmann, M.; Farrell, C.-J.L.; Pusceddu, I.; Fabregat-Cabello, N.; Cavalier, E. Assessment of vitamin D status–a changing landscape. Clin. Chem. Lab. Med. (CCLM) 2017, 55, 3–26.
- Schorr, P.; Kovacevic, B.; Volmer, D.A. Overestimation of 3α-over 3β-25-Hydroxyvitamin D3 Levels in Serum: A Mechanistic Rationale for the Different Mass Spectral Properties of the Vitamin D Epimers. J. Am. Soc. Mass Spectrom. 2021, 32, 1116–1125.
- Chen, Y.-C.; He, Y.-Y.; Li, Y.-M.; Wu, B.-T.; Yang, Y.-W.; Feng, J.-F. The importance of analyzing the serum C3-epimer level for evaluating vitamin D storage in some special populations. Eur. Rev. Med. Pharmacol. Sci. 2022, 26, 5334–5343.
- Bailey, D.; Veljkovic, K.; Yazdanpanah, M.; Adeli, K. Analytical measurement and clinical relevance of vitamin D3 C3-epimer. Clin. Biochem. 2013, 46, 190–196.
- Hazell, T.J.; Gallo, S.; Berzina, l.; Vanstone, C.A.; Rodd, C.; Weiler, H.A. Plasma 25-hydroxyvitamin D, more so than its epimer, has a linear relationship to leaner body composition across infancy in healthy term infants. Appl. Physiol. Nutr. Metab. 2014, 39, 1137–1143.
- Chailurkit, L.; Aekplakorn, W.; Ongphiphadhanakul, B. Serum C3 epimer of 25-hydroxyvitamin D and its determinants in adults: A national health examination survey in Thais. Osteoporos. Int. 2015, 26, 2339–2344.
- Moin, A.S.M.; Sathyapalan, T.; Atkin, S.L.; Butler, A.E. Inflammatory markers in non-obese women with polycystic ovary syndrome are not elevated and show no correlation with vitamin D metabolites. Nutrients 2022, 14, 3540.





