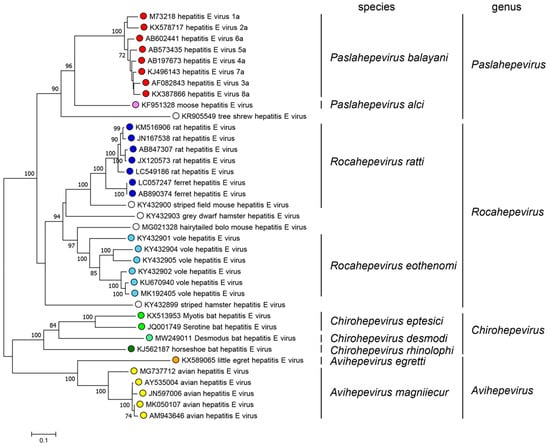
The HEV virus presents eight main genotypes within the
Paslahepevirus genus [4]. Genotypes 1 and 2 transmission is through the consumption of contaminated water by feces, mainly in developing countries. This situation has triggered zoonotic transmission by these genotypes, due to the contamination of the environment with the pathogen from animal feces or urine. As well as the transmission through the ingestion of raw or undercooked meat of the infected animal host, which predominates in genotypes 3 to 8
[5][6]. Within the RHEV, there are two main genotypes, HEV-C1 and HEV-C2, showing a divergence of 44%. In addition, two extra tentative genotypes (C3 and C4) have been proposed within RHEV species
[3].
According to the genomic sequence analysis, the HEV-A virus possesses a single- stranded RNA genome positive-sense approximately 7.2 kb in length. The genome encodes three open reading frames (ORFs) which participate in the viral replication and transcription
[8][9]. ORF1 contains a large polyprotein that is processed into several non- structural proteins, including RNA-dependent RNA polymerase (RdRp), helicase, and protease. ORF2 codifies a protein for the viral capsid, while ORF3 produces a minor protein responsible for the virion assembly and release. The genomic structure of HEV-A resembles other hepeviruses, like the RHEV
[10]. Recent studies have identified a new open reading frame (ORF4), located within the ORF1 region of the HEV-A genome. This ORF4 encodes a 20 kDa protein with 139–158 amino acids span. Despite its discovery, the role of the ORF4 in the HEV-A or RHEV viral replication and pathogenesis remain unclear
[11]. Investigating the ORFs of the RHEV virus is crucial to understand the genetic structure of the virus, the pathogenesis, replication, transcription and translation mechanisms, and how they adapt into the host to develop more effective strategies against this pathogen
[10].
3. Epidemiology and Geographical Distribution
3.1. Human Infections Associated with Rocahepevirus ratti
At this moment, up to 21 cases of HEV-C1 infection have been reported in humans through RNA detection in different samples. This data includes sixteen patients in Hong Kong, three patients in Spain, one patient in Central Africa, and one patient in France. RHEV initial detection occurred in rats from Germany and Vietnam
[3], but no human cases have been found in Germany
[12] or Hungary
[13] after investigation. In Spain, a comprehensive study conducted by Rivero-Juarez et al. revealed the presence of
Rocahepevirus ratti infection in three human patients.
In China, particularly in Hong Kong, three studies have been carried out to investigate the role of
Rocahepevirus ratti. The most recent of these studies was published in 2022 and focused on a cohort of 53 patients from the Microbiology Department of Queen Mary Hospital. The study was conducted between 1 August 2019 and 31 December 2020, and the results indicated a total of eight cases with positive RNA HEV-C1
[14]. The study was a continuation of a prior investigation, which analyzed 2201 sera from patients with liver function abnormalities between 1 January 2017 and 31 July 2019 (group 1), and 659 cases in a second group, composed of transplant recipients and patients with solid organ neoplasms, hematologic neoplasms, autoimmune disorders, and other immunosuppressive conditions between 1 January 2019 and 30 June 2019 (group 2). The previous study identified six cases of HEV-C1 RNA positivity in group 1 and one case in group 2, leading to a total of seven cases
[15]. Notably, the first confirmed human infection caused by RHEV, originating from rats, was identified in Hong Kong in 2017. The study examined a patient who was Anti-HEV IgG positive and had borderline Anti-HEV IgM results. This patient was found to have persistent Hepatitis E after receiving a liver transplant. For this research, a total of 518 solid organ transplant recipients, including kidney, liver, lung, and heart transplants, were evaluated. These recipients exhibited persistent hepatitis, defined as an elevation of alanine aminotransferase (ALT) greater than 1.5 times the upper limit of the reference level for a continuous period of more than six weeks, from 1 January 2014, or the date of transplant (whichever was later) until 31 December 2017
[16].
Recent studies have been conducted, revealing also the transmission of RHEV to humans in Europe. A recent study, conducted in Spain, examined 267 cases and evaluated two cohorts: one comprising 169 patients without HEV infection, and the other consisting of 98 patients diagnosed of HEV infection (either HEV RNA or HEV-IgM positive). In the first cohort, two cases of HEV-C1 RNA-positive samples were detected, whereas one case was found in the second cohort (HEV-IgM(+)). Therefore, the first RHEV recorded cases in Europe were these three human infections identified in Spain
[4]. A fourth European case was discovered in France thanks to metagenomics testing, revealing the presence of RHEV in liver/blood samples of a male who developed cirrhosis after resolving HBV infection (loss of HBsAg and non detectable DNA).
3.2. Animal Infections Associated with Rocahepevirus ratti
Regarding the discovery and dissemination of RHEV on a global scale, including regions such as Europe, Asia, America, and Africa, the Table 1 provides comprehensive data concerning the prevalence of cases, categorized by continents and individual countries.
Table 1. Global data of animal infections associated with Rocahepevirus ratti (N = 489).
| Continent |
Asia |
n |
Europe |
n |
America |
n |
Africa |
n |
| |
China |
159 |
Germany |
42 |
USA |
37 |
Kenya |
1 |
| |
Indonesia |
117 |
Czech Republic |
20 |
Canada |
21 |
|
|
| Japan |
14 |
Lithuania |
9 |
Brazil |
7 |
|
|
| |
Vietnam |
5 |
Romania |
9 |
|
|
|
|
| |
|
|
Belgium |
8 |
|
|
|
|
| |
|
|
Great Britain |
8 |
|
|
|
|
| |
|
|
Hungary |
7 |
|
|
|
|
| |
|
|
France |
5 |
|
|
|
|
Country
RHEV infections |
|
|
Austria |
4 |
|
|
|
|
| |
|
|
Spain |
4 |
|
|
|
|
| |
|
|
Switzerland |
4 |
|
|
|
|
| |
|
|
Denmark |
3 |
|
|
|
|
| |
|
|
Greece |
2 |
|
|
|
|
| |
|
|
Netherlands |
2 |
|
|
|
|
| |
|
|
Italy |
1 |
|
|
|
|
| Total infections |
|
295 |
|
128 |
|
65 |
|
1 |
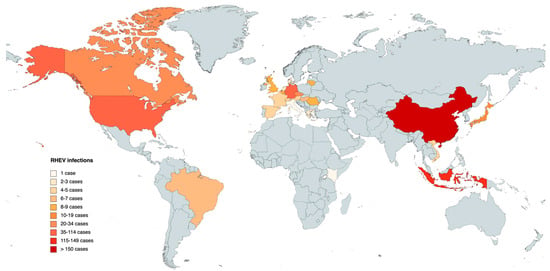
Until 2023, a cumulative total of 489 infections have been reported in different animals (mainly rodents), notably revealing Asia as the continent with the highest incidence of infected animals, accounting for a total of 295 documented instances. China emerges as the country with the highest reported cases within the Asian region. Europe, on the other hand, stands as the second-highest affected continent, with a combined total of 128 confirmed cases distributed across 15 different countries. In America, the presence of RHEV has only been detected in the United States, Canada, and Brazil, providing a combined total of 65 reported infections. In Africa, a solitary case has been identified, specifically in Kenya (Table 1).
The global map in Figure 2 corresponds to the data presented in Table 1, offering a comprehensive review of the results pertaining the different countries. The data are differentiated by RHEV animal infections, and the colored regions on the map represent the locations where detected animal infections have been reported.
Figure 2. Map of animal infections caused by Rocahepevirus ratti in different countries. The darker the color, the greater the number of infections.
3.3. Investigation of Rocahepevirus ratti in Wastewater Systems
Sewage surveillance is recognized as a powerful tool to gather information on the epidemiology of infectious diseases in the served population. The presence of RHEV has been explored in sewage treatment plants at different locations in Sweden, Italy, and Spain, allowing to get a better view of its epidemiology.
In Gothenburg (Sweden), a comprehensive study monitored influent and effluent wastewater for enteric viruses by qPCR, at the Rya treatment plant over a year. Seasonal variations in viral concentrations were observed in incoming wastewater, correlating with the number of diagnosed patients. Different HEV strains previously identified in drinking water, including two novel strains similar to those infecting rats and humans, were identified in the effluents
[17]Another study in Abruzzo (Italy), detected viral RNA of RHEV in 43.9% of sewage collected from 14 wastewater treatment plants, by using broadly reactive primers for hepevirus. These strains exhibited genetic variability and a clear geographic and wastewater treatment plant-related pattern. The study suggested that RHEV was a significant component of wastewater microbiota in the region
[18]Finally, a longitudinal study was carried out in Cordoba (southern Spain) from 2021 to 2023 using samples from patients with acute hepatitis, specimens from rodents, and wastewater to evaluate the correlation with human cases. The results showed that while RHEV was detected in almost all wastewater samples, there was no correlation between clinical cases and wastewater detection for both HEV and RHEV
[19]These studies highlight the importance of monitoring wastewater for RHEV. Untreated wastewater collects viruses excreted by both humans and synanthropic animals, including rodents, thereby providing a comprehensive overview of the viral strains circulating. Despite the reduction in viral concentrations during wastewater treatment, the presence of novel strains concerns the potential public health impact.
4. Pathogenesis and Diagnosis
Rocahepevirus ratti, like
Paslahepevirus balayani, can cause acute hepatitis, chronic hepatitis, and subclinical infection. HEV-C1 infections are self-limiting and less severe than HEV-A infections, characterized by lower mean peak ALT and bilirubin levels in those with intact immunity. In general, the criteria for inclusion in these investigations to identify RHEV infection include clinical and biological manifestations consistent with acute or chronic hepatitis, as well as an ALT level significantly greater than the upper limit of normal. Infections in immunocompromised individuals, on the other hand, are challenging, with 50% of HEV-C1 infections progressing to persistence, according to research by S. Sridhar et al.
[15]. This may be due to the fact that immunocompromised people might be more vulnerable to HEV-C1 infections. Not only was the viral pathogenicity examined, but meningoencephalitis was detected as a sequela in an immunocompromised patient after the hepatitis had entirely cured. Therefore, the correlation between RHEV infection and neurological symptoms must be studied. Subclinical HEV-C1 infection, without changes in hepatic function tests or clinical symptoms, is concerning over the safety of blood transfusions because the virus can be transmitted through contaminated blood samples due to the high viral load of HEV-C1 in plasma. RHEV’s viral tropism is oriented towards the liver, with a special affinity for liver cells. Virus replication occurs there, leading to liver damage and the release of liver enzymes into the blood, resulting in acute and chronic hepatitis in humans and animals. Recognizing the pathogenicity is crucial for the development of therapies for the prevention and treatment of hepatitis caused by this virus
[15].
Ferrets and rats have been proposed as candidate animal models to study RHEV pathogenesis. Ferrets exhibit three patterns of infection: subclinical infection, acute hepatitis, and persistent infection
[20]. Induced infections of RHEV on immunosuppressed rats have shown the effect of high-dose immunosuppression to induce chronic hepatitis and viral load suppression was observed with ribavirin treatment
[21].
Molecular biology techniques have facilitated the identification of RHEV RNA in liver, feces, and blood samples obtained from various animal and human species. Such techniques include RT-PCR amplification, Sanger DNA sequencing, and metagenomic tests involving next generation sequencing. The combination of these methods, along with detailed phylogenetic and sequence evaluations, have allowed sequencing of RHEV genomes
[4]. The first human RHEV infection was detected in a patient with chronic hepatitis, and this case was validated by RT-PCR and sequencing of plasma, stool, saliva, and liver tissue samples.
The most accurate method for detecting
Rocahepevirus ratti infection is the RT-PCR method by which viral genomic RNA can be identified. The sensitivity of the method depends on the design and selection of specific primers. The molecular diagnosis of HEV-A and RHEV may be challenging as several assays have shown low sensitivity for endemic genotypes in Europe and the number of genotypes/subtypes described is increasing, therefore, without an evaluation, their sensitivity in this context is unknown. Primers used to detect human HEV may not be able to recognize the RHEV genome in patient samples, limiting the sensitivity of PCR, and only the use of specific primers for RHEV has allowed its detection in different samples. It is important to note that the genetic diversity of HEV-C1 is still unclear, which could also limit the sensitivity of the techniques
[16].
5. Conclusions
An increasing number of cases of animals and humans infected with Rocahepevirus ratti are being detected. Some species of rats have been confirmed as natural reservoirs of RHEV, and they are considered a source of zoonotic infection. It is likely that more cases will be discovered as detection techniques improve. Understanding the virus evolution and diffusion is essential for the development of successful prevention and control measures, treatments, and vaccines. The information obtained from the phylogenetic analysis and genomic sequencing will help researchers track the development of new strains, contributing to epidemiological distribution maps. In addition, this information can also be used to develop diagnostic tests to accurately detect the virus in collected samples and evolve in the discovery of a more precise pathogenesis. These tests are needed to assist healthcare professionals in diagnosing RHEV infections and preventing the spread of the virus. This is a public health concern because it is a new pathogen that can be transmitted to other species and even to humans. Further research is needed to determine the exact route of transmission of the virus to humans, as it remains unknown.