Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Satoru Matsuda | -- | 1832 | 2023-09-07 15:44:10 | | | |
| 2 | Peter Tang | Meta information modification | 1832 | 2023-09-08 04:30:46 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Ikeda, Y.; Taniguchi, K.; Sawamura, H.; Yoshikawa, S.; Tsuji, A.; Matsuda, S. APRO Family Proteins in Cancer Invasiveness. Encyclopedia. Available online: https://encyclopedia.pub/entry/48927 (accessed on 08 February 2026).
Ikeda Y, Taniguchi K, Sawamura H, Yoshikawa S, Tsuji A, Matsuda S. APRO Family Proteins in Cancer Invasiveness. Encyclopedia. Available at: https://encyclopedia.pub/entry/48927. Accessed February 08, 2026.
Ikeda, Yuka, Kurumi Taniguchi, Haruka Sawamura, Sayuri Yoshikawa, Ai Tsuji, Satoru Matsuda. "APRO Family Proteins in Cancer Invasiveness" Encyclopedia, https://encyclopedia.pub/entry/48927 (accessed February 08, 2026).
Ikeda, Y., Taniguchi, K., Sawamura, H., Yoshikawa, S., Tsuji, A., & Matsuda, S. (2023, September 07). APRO Family Proteins in Cancer Invasiveness. In Encyclopedia. https://encyclopedia.pub/entry/48927
Ikeda, Yuka, et al. "APRO Family Proteins in Cancer Invasiveness." Encyclopedia. Web. 07 September, 2023.
Copy Citation
The APRO family members may be involved in the regulation of cell growth, migration, and/or invasion. Although an APRO protein could suppress the invasiveness of several cancer cells, it has been reported that overexpression of the same APRO protein could also promote the invasiveness and/or metastasis of the same cancer cells. In general, the invasiveness of cancer cells might be associated with the function of matrix metalloproteinases (MMPs) as well as with the function of certain exosomes. However, it has been shown that exosomes involving particular APRO proteins, MMPs, and/or microRNA could contribute to the regulation of invasiveness.
APRO protein
microRNA
matrix metalloproteinase
tissue inhibitors of metalloproteinase
exosome
1. Introduction
The APRO family is composed of at least six distinct members in vertebrates, namely, Tob1, Tob2, BTG1, BTG2/TIS21/PC3, ANA/Tob5/BTG3, and PC3b [1]. The main characteristic of this family is the presence of a highly conserved, 110-amino-acid N-terminal region, designated as the APRO homology domain [1], which holds two highly homologous regions, designated Box-A and Box-B [1][2]. Box-A has been suggested to play an antiproliferative role [2]. All the APRO family members may be involved in the regulation of cell proliferation, and actually, have the potential to regulate tumor cell growth [1][3]. Tob1 was isolated as a protein associating with the ErbB2 receptor protein [3]. Subsequently, Tob2 was isolated on the basis of its similarity to Tob1 [4]. Other family members had been identified using several different strategies [5]. A significant association has been identified between the expression level of Tob1 and clinicopathological characteristics, including the depth of invasion and/or the lymph node metastasis stage [6]. The downregulated expression of Tob1 has been found in malignant gastric cancer, suggesting that the low expression of Tob1 may be an independent indicator of poor prognosis in gastric cancers [7]. Similarly, the downregulation of Tob1 may be associated with the shorter survival of gastric cancer patients [8]. Consistently, Tob1 overexpression could not only increase the expression of PTEN, but also regulate the downstream effectors in the PI3K/PTEN signaling pathway, leading to the suppression of cancer cell proliferation [9]. In addition, significant prognostic effects of the whole APRO family have been found in lung adenocarcinoma and ovarian, colorectal, and brain cancers, but not in squamous-cell lung carcinoma [10]. Thus, accumulating evidence has shown that APRO family proteins may function as a tumor suppressor.
2. Exosomes with APRO Proteins and/or Certain MicroRNAs May Contribute to Cancer Invasion
Exosomes are a class of extracellular membrane vesicles with a circular lipid bilayer ranging in diameter from about 30 to 150 nm [11], which are capable of mediating invasion and/or metastasis by transferring their contents, including proteins, lipids, and nucleic acids, to adjacent cells [12]. Exosomes are broadly distributed in blood cells, dendritic cells, tumor cells, and other cells [13], which can be used for diagnosis and/or prognosis within cancer patients [14]. Molecular machineries of prevailing biogenesis, cargo loading, and/or delivery of exosomes may be intricate and are still under investigation. Exosomes secreted from gastric cancer cells overexpressing Tob1 could induce autophagy of LC3-II accumulation in the gastric cancer cells [15], which may influence the proliferation, migration, and invasion of cancer cells [16]. It has been shown that exosomes containing the BTG1 protein are present in the pleural fluid obtained from patients suffering from mesothelioma, lung cancer, breast cancer, and ovarian cancer [17]. The BTG1 protein has been also identified in plasma exosomes. In addition, the plasma-exosome-derived BTG-1 levels have been related to tumor diameter, stage, tumor metastasis, the degree of tumor differentiation, and abnormal CEA levels, in accordance with previous findings of BTG-1 expression in other cancers [17]. Furthermore, a low number of plasma exosomes with low levels of BTG-1 have been observed in the poor differentiation group, suggesting that plasma-exosome-derived BTG-1 might be a potential biomarker for a prognosis in patients with non-small-cell lung cancer [17].
Noncoding RNAs in exosomes from a variety of cells have been shown to influence metastasis via various mechanisms [18]. In particular, the microRNAs (miRNAs) commonly detected in exosomes are single-strand, non-encoding RNAs with the length of about 20 nucleotides, which are usually found in eukaryotic organisms [19]. The miRNAs can affect the stability and/or translational efficacy of their target mRNAs, consequently resulting in decreased protein translation [20]. More than 1000 miRNAs have been shown to regulate a lot of biological processes including proliferation, migration, and/or differentiation in cells [21]. Interestingly, miR590 in the exosome exerts its effects through targeting Tob1 [22]. miR32 might regulate the invasion of cancer stem cells, as it is upregulated in colorectal cancer tissues compared to the adjacent normal tissues [23][24]. An exosome containing miR-135a-5p could activate MMP-7 to promote liver metastasis in colorectal cancer [25] (Figure 1).
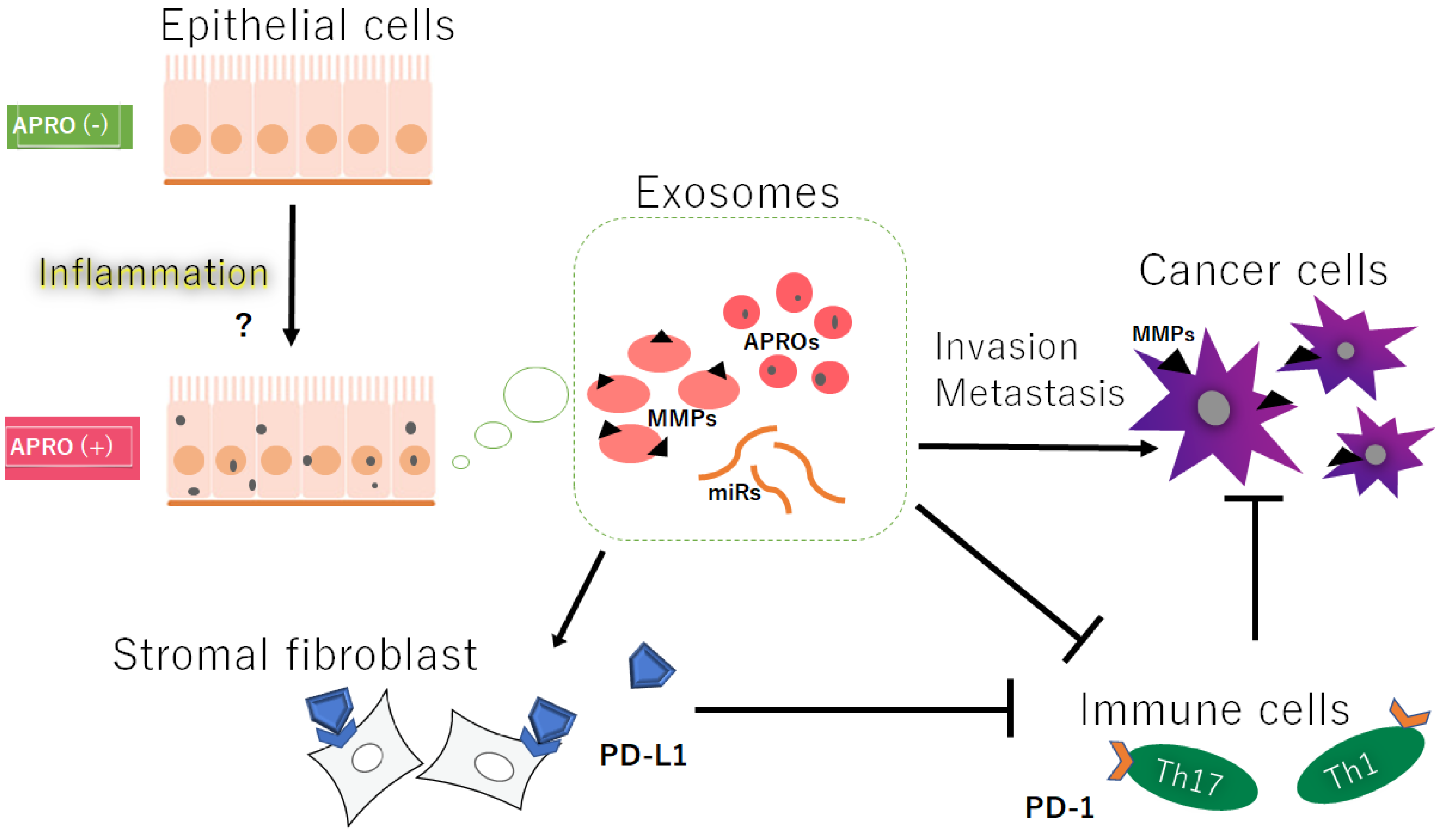
Figure 1. Hypothetical schematic image of the relationship among the APRO proteins (APROs), exosomes, immune cells, immune check point PD-L1 on stromal fibroblasts, and the cancer cells’ invasion/metastasis. Indicated molecules are examples. Arrowhead means stimulation, whereas hammerhead represents inhibition. Note that some critical pathways such as inflammation activation and/or cancer cell growth pathway have been omitted for clarity. Abbreviations: APROs—APRO family proteins; miRNAs—microRNAs; MMPs—matrix metalloproteinases; PD-L1—programmed cell-death ligand 1.
3. MMPs and TIMPs Could Be Also Involved in Cancer Invasion and Metastasis
MMPs are key players in matrix remodeling. Their function has been principally investigated in cancer biology; they are involved in different steps of cancer development, from local expansion by the proliferation of cancer cells to tissue invasion and/or metastasis through extracellular matrix degradation [26]. MMPs could promote cell migration and tumor invasion through the proteolytic degradation of the extracellular basement. MMPs are synthesized as pre-proMMPs, from which the signal peptide is removed during translation to produce mature proMMPs. MMP expression can be also affected by several hormones, growth factors, and/or cytokines [27]. A higher expression of MMPs has been revealed as a potential marker of higher invasiveness and/or worse prognosis in patients with various cancers. For example, ovarian hormones could affect the expression of several MMPs, which might participate in endometrial tissue remodeling during menstrual cycles [28]. Additionally, increases in estrogen and/or progesterone, as well as vascular endothelial growth factor (VEGF), during pregnancy could promote the expression of several MMPs, which might also facilitate the tissue invasion of cytotrophoblasts [29]. Exosomes from vascular smooth muscle cells (VSMCs) may be burdened with the MMP-2 protein and specific miRNAs for controlling cell adhesion and/or migration [30]. Furthermore, macrophage-derived exosomes could trigger the expression of MMP-2 in the VSMCs via JNK and p38 pathways [31]. Interestingly, the Box-A domain in the Tob1 protein may have protease activity, which activates the MMP-7 [32]. It has been discovered that MMPs exist in exosomes from various cell types and/or some body fluids [33].
The activity of MMPs could be regulated by endogenous tissue inhibitors of matrix metalloproteinases (TIMPs). In general, MMPs are regulated at multiple levels, including in mRNA expression, the activation of the proenzyme to the active form, and the counteracting actions of these TIMPs, which are specific for each MMP type. There are four homologous members in the TIMP family with a similar structure [34], which could also regulate remodeling and turnover of the extracellular matrix (ECM) during normal and/or pathological conditions [35]. The N-terminal domain of each TIMP protein holds the inhibitory activity for the wasting potential of the MMPs [36]. The role of TIMPs in the ECM turnover could be defined as the potential inhibition of MMPs with various efficacies. Increased MMP activity and/or decreased TIMP expression could lead to MMP/TIMP imbalance, which might result in various pathological conditions including cancer invasion and/or metastasis.
4. Activated MMPs and/or Certain MicroRNAs in Exosomes Could Contribute to the Enhanced Migration, Invasion, and/or Metastasis of Cancer Cells
In general, various MMPs are upregulated in various cancers and inflamed regions. Cancer progression could be a complex process, during which numerous cells, including malignant cells, inflammatory cells, and/or surrounding stromal cells, might communicate with each other in the microenvironment. MMPs may be involved in the remodeling of the extracellular matrix in the microenvironment to allow dissemination and/or metastasis of cancer cells [37]. Among them, MMP-2 and MMP-9 are the most distinctive MMPs characterized by a strong proteolytic activity in the extracellular matrix [38], which could be overexpressed in tumor cells and may be linked to risky metastasis and/or poor prognosis [39] (Figure 1). Exosomes could also regulate the migration of lung cancer cells into the rich vasculature by promoting MMP-2 expression [40]. In addition, exosomes could activate MMP-2 to enhance the invasiveness required for the first step in the metastasis of cancer cells [41].
MMPs might be crucial for ECM remodeling under the pathological conditions of cancers. MMPs are of crucial importance for the invasiveness of cancer cells. For a good invasion performance, cancer cells must adjust the activation rate of MMPs corresponding to the solidity of the surrounding ECM. Accordingly, their expression may correlate with metastatic potential and to a significant prognostic marker.
5. Activated MMPs Could Also Regulate the Responses of Immune Cells against Cancers
Cancers must evade antitumor immune responses to continue to grow. In fact, cancer cells can often escape from immune surveillance, which has been shown to be associated with various types of immune cells including Tregs and Th17 cells [42] (Figure 1). Therefore, immune responses against cancer have been revealed as a crucial issue in the treatment of cancer. Most tumor cells express antigens that can mediate recognition by host CD8+ T cells. Interestingly, high levels of MMP-9 detected in laryngeal cancer could play a critical role in the development of Treg cells, which have an ability to suppress the tumor-specific CD8+ T cells [43]. In addition, the increased production of MMP-7 might trigger an increase in the suppressive function of Treg cells [44]. Additionally, the expression of MMP-9 might be correlated with the markers of Th1 cells and/or T-cell exhaustion [45]. Furthermore, upregulated expressions of MMP-2 and MMP-9 may promote the migration and/or invasiveness of esophageal adenocarcinoma via the action of IL-17A, which is a proinflammatory cytokine secreted from Th17 cells [46]. Likewise, the MMP inhibitor may regulate the expression of TGF-β, thus reducing the number of Tregs [47]. Amazingly, the expression of MMP-7 caused by H. pylori infection could contribute to poor responses of the adaptive immune system characterized by insufficient Th1 and/or Th17 cells and the inappropriate activation of Treg cells [48][49]. Human chorionic gonadotropin (hCG), a hormone essential for pregnancy, is also ectopically expressed by a variety of cancers and is associated with a poor prognosis, which could induce the synthesis of MMP-2 and/or MMP-9, thereby increasing invasiveness in an MMP-dependent manner. The hCG could also upmodulate the secretion of TGFβ and IL-10, thereby inhibiting T-cell proliferation [50].
Consistently, the inhibition of MMP-2/MMP-9 may improve the efficacy of PD-1 or CTLA4 blockade therapy in the treatment of aggressive metastatic cancers [51]. The PD-1 or CTLA4 checkpoint blockade are dramatic therapies for several cancers that enhance antitumor immune activity. Immune checkpoints are diligently related to tumor immune escape, which may be related to the poor prognosis of some tumors in the survival analysis [52]. The PD-1 ligand is regulated through proteolytic cleavage by endogenous MMPs from stromal fibrocytes (Figure 1).
References
- Matsuda, S.; Rouault, J.; Magaud, J.; Berthet, C. In search of a function for the TIS21/PC3/BTG1/TOB family. FEBS Lett. 2001, 497, 67–72.
- Guéhenneux, F.; Duret, L.; Callanan, M.B.; Bouhas, R.; Hayette, S.; Berthet, C.; Samarut, C.; Rimokh, R.; Birot, A.M.; Wang, Q.; et al. Cloning of the mouse BTG3 gene and definition of a new gene family (the BTG family) involved in the negative control of the cell cycle. Leukemia 1997, 11, 370–375.
- Matsuda, S.; Kawamura-Tsuzuku, J.; Ohsugi, M.; Yoshida, M.; Emi, M.; Nakamura, Y.; Onda, M.; Yoshida, Y.; Nishiyama, A.; Yamamoto, T. Tob, a novel protein that interacts with p185erbB2, is associated with anti-proliferative activity. Oncogene 1996, 12, 705–713.
- Ikematsu, N.; Yoshida, Y.; Kawamura-Tsuzuku, J.; Ohsugi, M.; Onda, M.; Hirai, M.; Fujimoto, J.; Yamamoto, T. Tob2, a novel anti-proliferative Tob/BTG1 family member, associates with a component of the CCR4 transcriptional regulatory complex capable of binding cyclin-dependent kinases. Oncogene 1999, 18, 7432–7441.
- Buanne, P.; Corrente, G.; Micheli, L.; Palena, A.; Lavia, P.; Spadafora, C.; Lakshmana, M.K.; Rinaldi, A.; Banfi, S.; Quarto, M.; et al. Cloning of PC3B, a novel member of the PC3/BTG/TOB family of growth inhibitory genes, highly expressed in the olfactory epithelium. Genomics 2000, 68, 253–263.
- Zhang, S.Q.; Sun, K.K.; Wu, X.Y.; Zhong, N.; Zhao, H.; Li, D.C. Clinicopathological significance of cytoplasmic transducer of ErbB2. 1 expression in gastric cancer. Mol. Med. Rep. 2015, 12, 1177–1182.
- Yang, Y.; Shen, X.; Sun, K. Prognostic value of transducer of ErbB2.1 (TOB1) expression in patients with gastric cancer: Tissue microarray analysis. Int. J. Clin. Exp. Pathol. 2018, 11, 4060–4066.
- Guo, H.; Zhang, R.; Afrifa, J.; Wang, Y.; Yu, J. Decreased expression levels of DAL-1 and TOB1 are associated with clinicopathological features and poor prognosis in gastric cancer. Pathol. Res. Pract. 2019, 215, 152403.
- Jiao, Y.; Sun, K.K.; Zhao, L.; Xu, J.Y.; Wang, L.L.; Fan, S.J. Suppression of human lung cancer cell proliferation and metastasis in vitro by the transducer of ErbB-2.1 (TOB1). Acta Pharm. Sin. 2012, 33, 250–260.
- Bai, Y.; Qiao, L.; Xie, N.; Shi, Y.; Liu, N.; Wang, J. Expression and prognosis analyses of the Tob/BTG antiproliferative (APRO) protein family in human cancers. PLoS ONE 2017, 12, e0184902.
- Conde-Vancells, J.; Rodriguez-Suarez, E.; Embade, N.; Gil, D.; Matthiesen, R.; Valle, M.; Elortza, F.; Lu, S.C.; Mato, J.M.; Falcon-Perez, J.M. Characterization and comprehensive proteome profiling of exosomes secreted by hepatocytes. J. Proteome Res. 2008, 7, 5157–5166.
- Moradi-Chaleshtori, M.; Hashemi, S.M.; Soudi, S.; Bandehpour, M.; Mohammadi-Yeganeh, S. Tumor-derived exosomal microRNAs and proteins as modulators of macrophage function. J. Cell Physiol. 2019, 234, 7970–7982.
- Yu, Y.; Zhang, W.S.; Guo, Y.; Peng, H.; Zhu, M.; Miao, D.; Su, G. Engineering of exosome-triggered enzyme-powered DNA motors for highly sensitive fluorescence detection of tumor-derived exosomes. Biosens. Bioelectron. 2020, 167, 112482.
- Li, M.; Xia, B.; Wang, Y.; You, M.J.; Zhang, Y. Potential Therapeutic Roles of Exosomes in Multiple Myeloma: A Systematic Review. J. Cancer 2019, 10, 6154–6160.
- Wang, Y.; Song, Y.; Zhou, L.; Wang, M.; Wang, D.; Bai, J.; Fu, S.; Yu, J. The Overexpression of TOB1 Induces Autophagy in Gastric Cancer Cells by Secreting Exosomes. Dis. Markers 2022, 2022, 7925097.
- Zhu, H.; Yang, Y.; Wang, L.; Xu, X.; Wang, T.; Qian, H. Leptomycin B inhibits the proliferation, migration, and invasion of cultured gastric carcinoma cells. Biosci. Biotechnol. Biochem. 2020, 84, 290–296.
- Wan, L.; Chen, X.; Deng, J.; Zhang, S.; Tu, F.; Pei, H.; Hu, R.; Liu, J.; Yu, H. Plasma exosome-derived B-cell translation gene 1: A predictive marker for the prognosis in patients with non-small cell lung cancer. J. Cancer. 2021, 12, 1538–1547.
- Wang, D.; Zhang, W.; Zhang, C.; Wang, L.; Chen, H.; Xu, J. Exosomal non-coding RNAs have a significant effect on tumor metastasis. Mol. Nucleic Acids 2022, 29, 16–35.
- Bartel, D.P. MicroRNAs: Target recognition and regulatory functions. Cell 2009, 136, 215–233.
- Mohr, A.M.; Mott, J.L. Overview of microRNA biology. Semin. Liver Dis. 2015, 35, 3–11.
- Dexheimer, P.J.; Cochella, L. MicroRNAs: From Mechanism to Organism. Front Cell Dev. Biol. 2020, 8, 409.
- Liu, Q.; Gao, Q.; Zhang, Y.; Li, Z.; Mei, X. MicroRNA-590 promotes pathogenic Th17 cell differentiation through targeting Tob1 and is associated with multiple sclerosis. Biochem. Biophys. Res. Commun. 2017, 493, 901–908.
- Tang, G.; Wang, J.; Dong, W.; Dai, K.; Du, J. Exosomal miRNA Expression Profiling and the Roles of Exosomal miR-4741, miR-32, miR-3149, and miR-6727 on Gastric Cancer Progression. Biomed. Res. Int. 2022, 2022, 1263812.
- Wu, W.; Chen, Y.; Ye, S.; Yang, H.; Yang, J.; Quan, J. Transcription factor forkhead box K1 regulates miR-32 expression and enhances cell proliferation in colorectal cancer. Oncol. Lett. 2021, 21, 407.
- Sun, H.; Meng, Q.; Shi, C.; Yang, H.; Li, X.; Wu, S.; Familiari, G.; Relucenti, M.; Aschner, M.; Wang, X.; et al. Hypoxia-Inducible Exosomes Facilitate Liver-Tropic Premetastatic Niche in Colorectal Cancer. Hepatology 2021, 74, 2633–2651.
- De Almeida, L.G.N.; Thode, H.; Eslambolchi, Y.; Chopra, S.; Young, D.; Gill, S.; Devel, L.; Dufour, A. Matrix Metalloproteinases: From Molecular Mechanisms to Physiology, Pathophysiology, and Pharmacology. Pharmacol. Rev. 2022, 74, 712–768.
- Verma, R.P.; Hansch, C. Matrix metalloproteinases (MMPs): Chemical-biological functions and (Q)SARs. Bioorg. Med. Chem. 2007, 15, 2223–2268.
- Grzechocińska, B.; Dąbrowski, F.; Cyganek, A.; Panek, G.; Wielgoś, M. The role of metalloproteinases in endometrial remodelling during menstrual cycle. Ginekol. Pol. 2017, 88, 337–342.
- Duffy, D.M.; Stouffer, R.L. Luteinizing hormone acts directly at granulosa cells to stimulate periovulatory processes: Modulation of luteinizing hormone effects by prostaglandins. Endocrine 2003, 22, 249–256.
- Kapustin, A.N.; Shanahan, C.M. Emerging roles for vascular smooth muscle cell exosomes in calcification and coagulation. J. Physiol. 2016, 594, 2905–2914.
- Wang, Y.; Jia, L.; Xie, Y.; Cai, Z.; Liu, Z.; Shen, J.; Lu, Y.; Wang, Y.; Su, S.; Ma, Y.; et al. Involvement of macrophage-derived exosomes in abdominal aortic aneurysms development. Atherosclerosis 2019, 289, 64–72.
- Nakamura, R.; Konishi, M.; Taniguchi, M.; Hatakawa, Y.; Akizawa, T. The discovery of shorter synthetic proteolytic peptides derived from Tob1 protein. Peptides 2019, 116, 71–77.
- Shimoda, M. Extracellular vesicle-associated MMPs: A modulator of the tissue microenvironment. Adv. Clin. Chem. 2019, 88, 35–66.
- Kaur, N.; Sunil Kumar, B.V.; Mahajan, K.; Singh, S. Expression and characterization of tissue inhibitor of metalloproteinase 4 from complex canine mammary carcinomas. J. Immunoass. Immunochem. 2016, 37, 515–526.
- Gomis-Rüth, F.X.; Maskos, K.; Betz, M.; Bergner, A.; Huber, R.; Suzuki, K.; Yoshida, N.; Nagase, H.; Brew, K.; Bourenkov, G.P.; et al. Mechanism of inhibition of the human matrix metalloproteinase stromelysin-1 by TIMP-1. Nature 1997, 389, 77–81.
- Raeeszadeh-Sarmazdeh, M.; Greene, K.A.; Sankaran, B.; Downey, G.P.; Radisky, D.C.; Radisky, E.S. Directed evolution of the metalloproteinase inhibitor TIMP-1 reveals that its N- and C-terminal domains cooperate in matrix metalloproteinase recognition. J. Biol. Chem. 2019, 294, 9476–9488.
- Liao, W.T.; Ye, Y.P.; Deng, Y.J.; Bian, X.W.; Ding, Y.Q. Metastatic cancer stem cells: From the concept to therapeutics. Am. J. Stem. Cells 2014, 3, 46–62.
- Huang, Q.; Lan, F.; Wang, X.; Yu, Y.; Ouyang, X.; Zheng, F.; Han, J.; Lin, Y.; Xie, Y.; Xie, F.; et al. IL-1β-induced activation of p38 promotes metastasis in gastric adenocarcinoma via upregulation of AP-1/c-fos, MMP2 and MMP9. Mol. Cancer 2014, 13, 18.
- Roy, R.; Yang, J.; Moses, M.A. Matrix metalloproteinases as novel biomarkers and potential therapeutic targets in human cancer. J. Clin. Oncol. 2009, 27, 5287–5297.
- Wu, D.M.; Deng, S.H.; Liu, T.; Han, R.; Zhang, T.; Xu, Y. TGF-β-mediated exosomal lnc-MMP2–2 regulates migration and invasion of lung cancer cells to the vasculature by promoting MMP2 expression. Cancer Med. 2018, 7, 5118–5129.
- McCready, J.; Sims, J.D.; Chan, D.; Jay, D.G. Secretion of extracellular hsp90alpha via exosomes increases cancer cell motility: A role for plasminogen activation. BMC Cancer 2010, 10, 294.
- Srivastava, M.K.; Zhu, L.; Harris-White, M.; Huang, M.; St John, M.; Lee, J.M.; Salgia, R.; Cameron, R.B.; Strieter, R.; Dubinett, S.; et al. Targeting myeloid-derived suppressor cells augments antitumor activity against lung cancer. Immunotargets Ther. 2012, 2012, 7–12.
- Wang, B.Q.; Zhang, C.M.; Gao, W.; Wang, X.F.; Zhang, H.L.; Yang, P.C. Cancer-derived matrix metalloproteinase-9 contributes to tumor tolerance. J. Cancer Res. Clin. Oncol. 2011, 137, 1525–1533.
- Lee, S.; Yamamoto, S.; Srinivas, B.; Shimizu, Y.; Sada, N.; Yoshitome, K.; Ito, T.; Kumagai-Takei, N.; Nishimura, Y.; Otsuki, T. Increased production of matrix metalloproteinase-7 (MMP-7) by asbestos exposure enhances tissue migration of human regulatory T-like cells. Toxicology 2021, 452, 152717.
- Zeng, Y.; Gao, M.; Lin, D.; Du, G.; Cai, Y. Prognostic and Immunological Roles of MMP-9 in Pan-Cancer. Biomed Res. Int. 2022, 2022, 2592962.
- Liu, D.; Zhang, R.; Wu, J.; Pu, Y.; Yin, X.; Cheng, Y.; Wu, J.; Feng, C.; Luo, Y.; Zhang, J. Interleukin-17A promotes esophageal adenocarcinoma cell invasiveness through ROS-dependent, NF-κB-mediated MMP-2/9 activation. Oncol. Rep. 2017, 37, 1779–1785.
- Krstic, J.; Santibanez, J.F. Transforming growth factor-beta and matrix metalloproteinases: Functional interactions in tumor stroma-infiltrating myeloid cells. Sci. World J. 2014, 2014, 521754.
- Wilson, K.T.; Crabtree, J.E. Immunology of Helicobacter pylori: Insights into the failure of the immune response and perspectives on vaccine studies. Gastroenterology 2007, 133, 288–308.
- Ogden, S.R.; Noto, J.M.; Allen, S.S.; Patel, D.A.; Romero-Gallo, J.; Washington, M.K.; Fingleton, B.; Israel, D.A.; Lewis, N.D.; Wilson, K.T.; et al. Matrix metalloproteinase-7 and premalignant host responses in Helicobacter pylori-infected mice. Cancer Res. 2010, 70, 30–35.
- Khare, P.; Bose, A.; Singh, P.; Singh, S.; Javed, S.; Jain, S.K.; Singh, O.; Pal, R. Gonadotropin and tumorigenesis: Direct and indirect effects on inflammatory and immunosuppressive mediators and invasion. Mol. Carcinog. 2017, 56, 359–370.
- Ye, Y.; Kuang, X.; Xie, Z.; Liang, L.; Zhang, Z.; Zhang, Y.; Ma, F.; Gao, Q.; Chang, R.; Lee, H.-H.; et al. Small-molecule MMP2/MMP9 inhibitor SB-3CT modulates tumor immune surveillance by regulating PD-L1. Genome Med. 2020, 12, 83.
- Chen, Z.; Zhuo, S.; He, G.; Tang, J.; Hao, W.; Gao, W.Q.; Yang, K.; Xu, H. Prognosis and Immunotherapy Significances of a Cancer-Associated Fibroblasts-Related Gene Signature in Gliomas. Front Cell Dev. Biol. 2021, 9, 721897.
More
Information
Subjects:
Biochemistry & Molecular Biology
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
465
Revisions:
2 times
(View History)
Update Date:
08 Sep 2023
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No




