Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Zaneta Kimber-Trojnar | -- | 1893 | 2023-08-15 20:39:51 | | | |
| 2 | Jessie Wu | + 4 word(s) | 1897 | 2023-08-16 03:29:53 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Trojnar, M.; Patro-Małysza, J.; Kimber-Trojnar, �.; Leszczyńska-Gorzelak, B.; Mosiewicz, J. Fatty Acid-Binding Protein 4. Encyclopedia. Available online: https://encyclopedia.pub/entry/48101 (accessed on 26 March 2026).
Trojnar M, Patro-Małysza J, Kimber-Trojnar �, Leszczyńska-Gorzelak B, Mosiewicz J. Fatty Acid-Binding Protein 4. Encyclopedia. Available at: https://encyclopedia.pub/entry/48101. Accessed March 26, 2026.
Trojnar, Marcin, Jolanta Patro-Małysza, Żaneta Kimber-Trojnar, Bożena Leszczyńska-Gorzelak, Jerzy Mosiewicz. "Fatty Acid-Binding Protein 4" Encyclopedia, https://encyclopedia.pub/entry/48101 (accessed March 26, 2026).
Trojnar, M., Patro-Małysza, J., Kimber-Trojnar, �., Leszczyńska-Gorzelak, B., & Mosiewicz, J. (2023, August 15). Fatty Acid-Binding Protein 4. In Encyclopedia. https://encyclopedia.pub/entry/48101
Trojnar, Marcin, et al. "Fatty Acid-Binding Protein 4." Encyclopedia. Web. 15 August, 2023.
Copy Citation
Type 2 diabetes mellitus (T2DM) represents a common metabolic disorder that is characterized by chronic hyperglycemia. For more than half a century, the link between insulin resistance and T2DM has been well recognized. Insulin resistance is not only the most powerful predictor of future development of T2DM, but it is also a therapeutic target. On the other hand, gestational diabetes mellitus (GDM) is one of the most common metabolic disorders of pregnancy and its incidence has considerably increased by 10–100% in the last 20 years. There is ample scientific evidence to suggest a link between the fatty acid-binding protein 4 (FABP4) and insulin resistance, gestational (GDM), and type 2 (T2DM) diabetes mellitus.
fatty acid-binding protein 4
insulin resistance
type 2 diabetes mellitus
1. Introduction
Fatty acid-binding protein 4 (FABP4) plays a crucial role in the regulation of lipid-mediated actions, such as the initialization of inflammation and oxidative stress processes [1]. The expression of this adipogenic protein can be induced by vascular endothelial growth factor (VEGF) signaling [2]. FABP4 increases the ciatriphosphate-binding cassette A1 pathway [3]. Emerging data suggests an essential involvement of FABP4 in endothelial dysfunction [2]. Exogenous FABP4 interferes with insulin stimulated production of nitric oxide in endothelial cells [4]. Cabré et al. [5] observed that increased FABP4 concentrations were associated with excessive oxidative stress and inflammatory markers in diabetes.
FABP4 negatively regulates PPARγ in macrophages and adipocytes, affecting adipocyte differentiation. Higher levels of FABP4 and lower levels of PPARγ in visceral adipose tissue, when compared with subcutaneous adipose tissue, suggest a causative link between FABP4 and the metabolic syndrome. It may also explain certain morphological and functional differences between the adipocytes found in these two types of fat tissue. Visceral preadipocytes are known to proliferate and differentiate into mature adipocytes less actively than those from subcutaneous fat. Visceral adipose tissue secretes more proinflammatory cytokines [6].
It has also been postulated that the transcription factor forkhead box protein O1 (FOXO1) is involved in lipid metabolism. The available scientific evidence suggests that metformin may have a protective effect against lipid accumulation in macrophages as it decreases FABP4 expression at the mRNA level. The exact mechanism is not yet fully understood; it may deal with decreases in transcription or by promotion of mRNA degradation. For this reason, some authors suggest that metformin should also be perceived as a therapeutic agent for the prevention and targeting of atherosclerosis in metabolic syndrome [7].
FABP4 contributes to the accumulation of short-chain free fatty acids and suppresses the activity of relevant proteins in the phosphatidylinositol 3′-kinase (PI3K)-AKT signal pathway. Accordingly, FABP4 inhibits the presence of glucose oxidation and glycolysis and decreases the uptake and utilization of glucose in human organs, such as in muscles and the liver [8]. It is known that FABP4 can bind various intracellular fatty acids and probably mediates intracellular lipid trafficking between cellular compartments [9]. It might also modulate the availability and composition of fatty acids in muscles and adipose tissues [10]. In the myocytes and adipose tissue in mice, improved glucose homeostasis after the ablation of FABP4 was documented [11].
Increased FABP4 production could contribute to macrophage activity, possibly by activation of inflammatory pathways, resulting in inflammation [12]. An alternative hypothesis, more in line with the published evidence, is that the elevated circulating FABP4 reflects the increased cellular production in both adipocytes and macrophages in response to greater lipid availability, with increased Kupffer cell production of FABP4 triggering an increased inflammatory response [12][13].
FABP4 plays a crucial role in mediating the endoplasmic reticulum stress observed in macrophages upon lipotoxic signal exposure, which contributes to atherosclerosis, inflammation, and perhaps plaque vulnerability [14]. High levels of FABP4 may contribute to adverse prognosis via the induction of endoplasmic reticulum stress in macrophages and upregulation of pro-inflammatory cytokine production. Indeed, FABP4 modulates inflammatory responses in macrophages through a positive feedback loop involving c-Jun NH2-terminal kinases and activator protein-1 [12][15]. FABP4 is also implicated in modulating the eicosanoid balance by affecting both cyclooxygenase 2 (COX2) activity and leukotriene A4 (LTA4) stability, and upregulates uncoupling protein 2 (UCP2); all these processes influence macrophage function and adipose tissue inflammation. FABP4 can also interact with HSL and Janus kinase 2 (JAK2) [16] (Figure 1). Two different mechanisms of the latter action have been proposed. The first one includes direct protein–protein interactions between FABP4 and HSL and JAK2 [17][18]. FABP4 and HSL may also interact indirectly via two protein kinases A and G pathways [19].
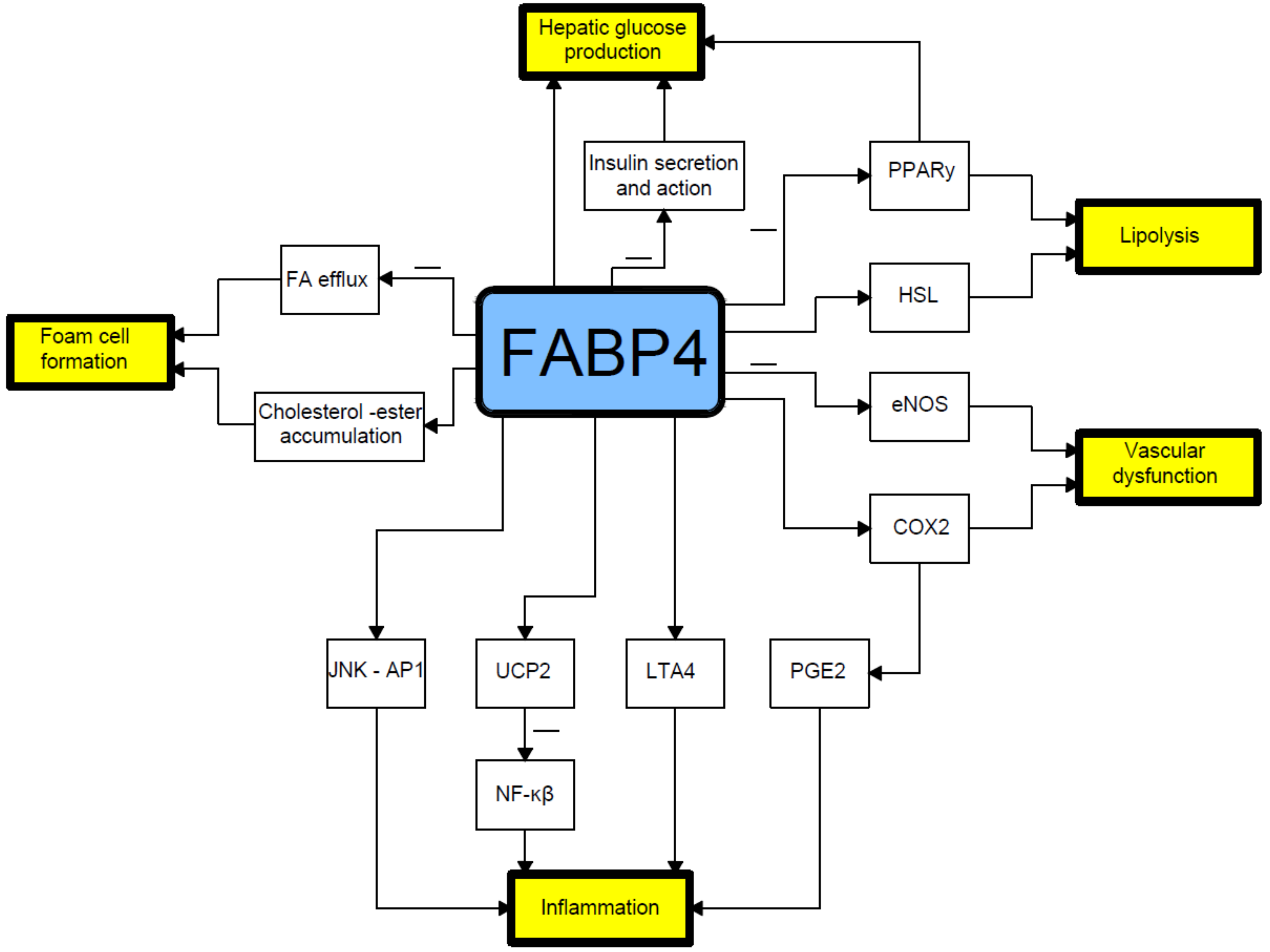
Figure 1. Relationship between fatty acid-binding protein 4 and pathophysiology of type 2 diabetes mellitus. COX2- cyclooxygenase-2; eNOS- endothelial nitric oxide synthase; FA- fatty acid; HSL- hormone sensitive lipase; JNK AP1-Jun N-terminal kinase-activator protein 1; LTA4-leukotriene A4; NF-κB-nuclear factor-kappa B; PGE2-prostaglandin E2; PPARγ–peroxisome proliferator-activated receptor γ; UCP2-uncoupling protein 2.
Several proinflammatory stimuli have been reported to upregulate FABP4 expression in macrophages, including oxidized low-density lipoproteins, toll-like receptor (TLR) agonists, and PPARγ agonists [20][21][22][23]. In this respect, lipopolysaccharide (LPS), a TLR4 ligand, stimulates FABP4 expression and the activated FABP4, reciprocally, enhances the LPS-TLR4 signaling-evoked JNK inflammatory pathways [15]. In addition, prolonged hyperglycemia has been shown to induce FABP4 expression in mesangial cells and trigger the release of proinflammatory cytokines [20][24] (Figure 2).
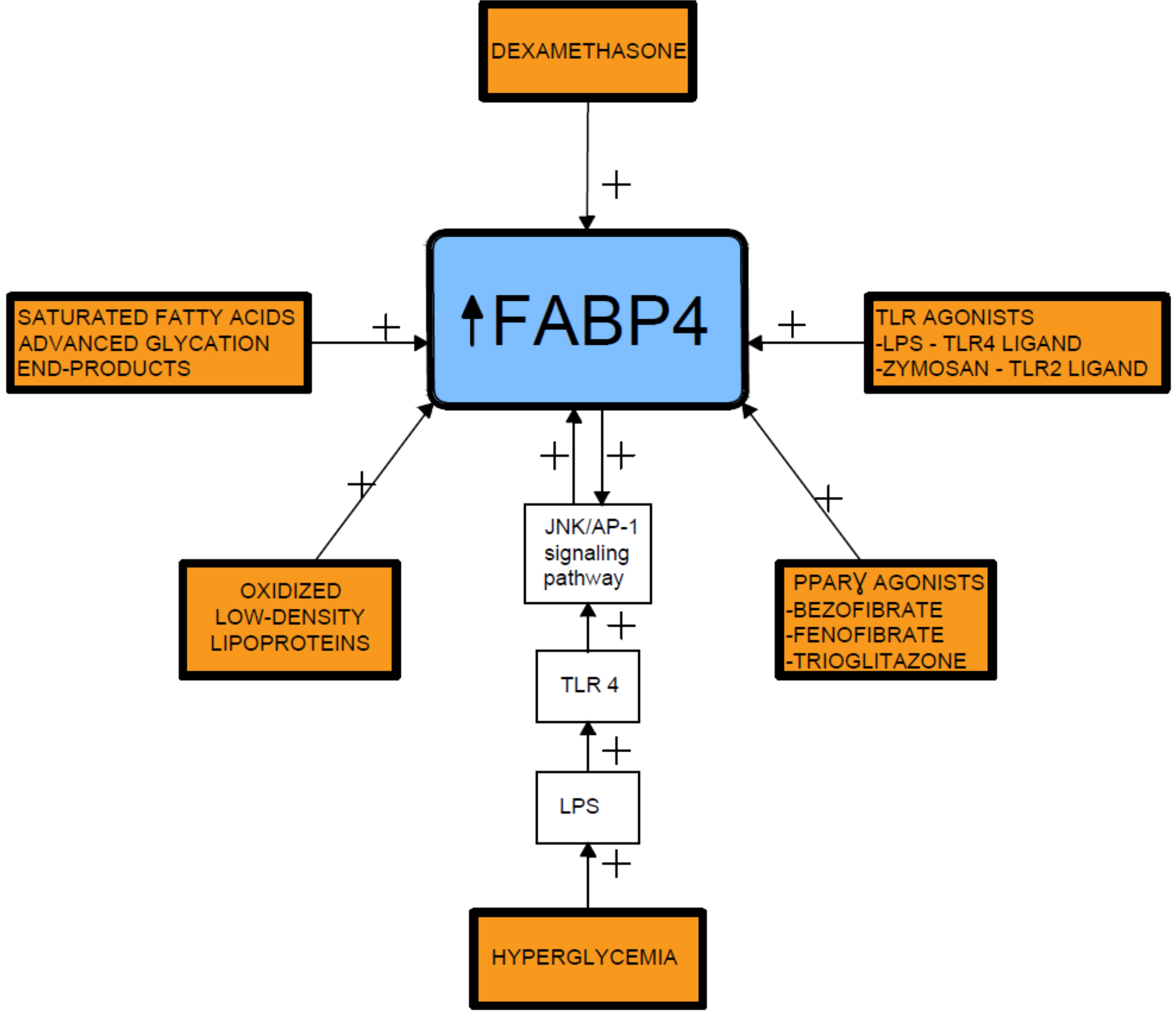
Figure 2. Fatty acid-binding protein 4 inducing factors. TLR- toll-like receptor; LPS- lipopolysaccharide; FABP4-fatty acid-binding protein 4.
2. Relationship between Fatty Acid-Binding Protein 4 (FABP4) and Insulin Resistance
Fatty acid-binding protein 4 (FABP4) is a cytoplasmic fatty acid chaperone clearly engaged in the onset of insulin resistance [6]. Studies in animal models suggest that FABP4 is important for glucose homeostasis [8]. Deletion of the FABP4 gene protected mice against insulin resistance as well as hyperinsulinemia associated with both diet-induced obesity and genetic obesity [8][11][13][25][26][27]. A reduced ability of adipocytes to take up and retain free fatty acids, leading to ectopic lipid accumulation, and abnormalities in the release of adipokines by adipocytes are critical factors for insulin resistance and the development of T2DM [27].
FABP4 was also detected in apoptotic granulosa cells in atretic antral follicles of the mouse ovary, which suggests a potential link to polycystic ovary syndrome (PCOS), which is known to be frequently associated with insulin resistance [28]. Expression of FABP4 mRNA in isolated granulosa cells was found to be higher in patients diagnosed with PCOS than in controls [29].
It has been proven that FABP4 is negatively correlated with the glucose-disposal rate (GDR) [30]. In non-DM subjects, serum FABP concentrations were negatively correlated with the mean rate of glucose infusion during the last 30 min of the clamp test, which reflects insulin sensitivity [31]. Nakamura et al. [6] also showed that circulating FABP4 concentrations were negatively correlated with GDR, which is a marker of insulin resistance in skeletal muscles in individuals with T2DM. On the contrary, FABP4 concentration was positively related to the insulinogenic index in non-diabetic participants. Wu et al. [32] reported that circulating FABP4 concentrations were correlated with glucose-stimulated insulin secretion in healthy controls.
It has been suggested that insulinotropic potential of FABP4 is similar to the effects of glucagon-like peptide-1 (GLP-1) [6]. To maintain glucose homeostasis, FABP4 may stimulate β cells and alter insulin secretion. Moreover, FABP4 presented a positive relationship with insulin secretion at an early stage in the non-diabetic group, which may be due to the fact that insulin secretion is damaged relatively early in T2DM.
Nakamura et al. [6] found the most pronounced negative correlation between FABP4 and GDR when compared to some other markers of insulin resistance or body composition in T2DM. FABP4 has been reported to have a negative correlation with GDR in type 1 diabetes mellitus, T2DM, and the controls of Asian Americans [33]. FABP4 represents an important molecule dealing with insulin resistance in T2DM.
3. Relationship between Fatty Acid-Binding Protein 4 and Diabetic Nephropathy
In the past decades, several biomarkers have emerged for the detection of early diabetic nephropathy (DN) besides the glomerular filtration rate (GFR) and urine albumin-to-creatinine ratio (UACR). Among them, FABP4 has attracted increased attention [34]. FABP4 was also reported at increased concentrations in nondiabetic as well as T2DM patients with end-stage renal disease [35]. Yeung et al. [36] reported that serum levels of FABP4 had a significantly inverse relationship with the estimated GFR (eGFR) and was independently associated with macrovascular complications and DN staging classified by albuminuria.
Cabré et al. [37] reported that FABP4 was independently associated with eGFR in T2DM patients with eGFR ≥ 60 mL/min/1.73 m2. Ni X et al. [34] found that serum FABP4 along with UACR or a panel of biomarkers might be more sensitive for the detection of early DN. Serum FABP4 had an inverse correlation with GFR and could be an independent predictor for early DN [34].
The mechanisms behind the elevation of FABP4 in patients with diabetic kidney disease are not yet fully understood. It is known that FABP4 is abundantly expressed in adipocytes, macrophages, and endothelial cells [34]. Firstly, it is suggested that, during the early stage of DN, the accumulation of active macrophages is more evident in the kidney because of the increased oxidative stress and chronic inflammation, which consequently induces greater expression of serum FABP4 [5][36]. Secondly, damage to glomeruli and tubulointerstitium might result in both decreased glomerular filtration and increased tubular reabsorption, leading to an increase in FABP4 in the circulation [38]. Okazaki et al. [39] reported that urinary excretion of FABP4 was associated with the progression of proteinuria and renal dysfunction in healthy subjects. The cited authors suggested that the urinary FABP4 reflects damage of the glomerular with the hypothesis proposed by Tanaka et al. [40] that the main source of the urinary FABP4 is derived from ectopic expression of glomerular FABP4 rather than increased adiposity and that locally increased FABP4 in the glomerulus affects renal dysfunction.
Toruner et al. [35] found that serum FABP4 levels were independently and positively associated with the albumin excretion rate in patients with T2DM, suggesting an involvement of the increased serum FABP4 levels in the occurrence and development of microalbuminuria among patients with T2DM. Furthermore, researchers from Hong Kong [36] also documented that among patients with diabetes mellitus, serum FABP4 levels were shown to be independently associated with the severity of nephropathy. Their findings raised the possibility that FABP4 might be used as a serum biomarker for stratifying nephropathy stages in patients with T2DM. The serum FABP4 level can be used as an indicator of microalbuminuria not only in diabetic patients with early stage disease, but also for hyperglycemic individuals before the onset of diabetes [41].
4. Relationship between Fatty Acid-Binding Protein 4 and Non-Alcoholic Fatty Liver Disease
There are multiple reports available that clearly link metabolic disorders, including insulin resistance and T2DM, to non-alcoholic fatty liver disease (NAFLD), which nowadays represents the most frequent liver disease worldwide. Its incidence has been estimated at 20–30% in the populations of Western countries. Approximately 70% of T2DM and obese subjects present some extent of NAFLD. This abnormal lipid accumulation in the liver is considered a significant causative factor of cancerogenesis resulting in hepatocellular carcinoma (HCC) [42].
At present, HCC represents the third leading cause of cancer-related mortality. Interestingly, except for the well-known risk factors of malignancy, such as viral hepatitis and alcohol, a higher incidence of HCC was observed in T2DM patients. In the analysis, obese subjects and patients with metabolic syndrome were also at a higher risk of developing cancer [42]. Additionally, HCC treatment outcomes of all those patients appeared worse with T2DM, representing a prognostic predictor of the increased risk of mortality [42].
In a study by Thompson et al., the FABP4 expression was indeed proven to be significantly increased in animal models of obesity promoted HCC [43]. These findings are consistent with the previous report, which concluded that PPARγ is considered a tumor suppressor gene, whereas FABP4 plays a role in tumorigenesis [6].
References
- Martínez-Micaelo, N.; Rodríguez-Calvo, R.; Guaita-Esteruelas, S.; Heras, M.; Girona, J.; Masana, L. Extracellular FABP4 uptake by endothelial cells is dependent on cytokeratin 1 expression. Biochim. Biophys. Acta Mol. Cell. Biol. Lipids 2018, 1864, 234–244.
- Chen, R.A.; Sun, X.M.; Yan, C.Y.; Liu, L.; Hao, M.W.; Liu, Q.; Jiao, X.Y.; Liang, Y.M. Hyperglycemia-induced PATZ1 negatively modulates endothelial vasculogenesis via repression of FABP4 signaling. Biochem. Biophys. Res. Commun. 2016, 477, 548–555.
- Li, L.; Lee, S.J.; Kook, S.Y.; Ahn, T.G.; Lee, J.Y.; Hwang, J.Y. Serum from pregnant women with gestational diabetes mellitus increases the expression of FABP4 mRNA in primary subcutaneous human pre-adipocytes. Obstet. Gynecol. Sci. 2017, 60, 274–282.
- Harjes, U.; Bridges, E.; McIntyre, A.; Fielding, B.A.; Harris, A.L. Fatty acid-binding protein 4, a point of convergence for angiogenic and metabolic signaling pathways in endothelial cells. J. Biol. Chem. 2014, 289, 23168–23176.
- Cabré, A.; Lázaro, I.; Girona, J.; Manzanares, J.M.; Marimón, F.; Plana, N.; Heras, M.; Masana, L. Fatty acid binding protein 4 is increased in metabolic syndrome and with thiazolidinedione treatment in diabetic patients. Atherosclerosis 2007, 195, e150–e158.
- Garin-Shkolnik, T.; Rudich, A.; Hotamisligil, G.S.; Rubinstein, M. FABP4 attenuates PPARγ and adipogenesis and is inversely correlated with PPARγ in adipose tissues. Diabetes 2014, 63, 900–911.
- Song, J.; Ren, P.; Zhang, L.; Wang, X.L.; Chen, L.; Shen, Y.H. Metformin reduces lipid accumulation in macrophages by inhibiting FOXO1-mediated transcription of fatty acid-binding protein 4. Biochem. Biophys. Res. Commun. 2010, 393, 89–94.
- Tu, W.J.; Guo, M.; Shi, X.D.; Cai, Y.; Liu, Q.; Fu, C.W. First-Trimester Serum Fatty Acid-Binding Protein 4 and Subsequent Gestational Diabetes Mellitus. Obstet. Gynecol. 2017, 130, 1011–1016.
- Coe, N.R.; Bernlohr, D.A. Physiological properties and functions of intracellular fatty acid-binding proteins. Biochim. Biophys. Acta 1998, 1391, 287–306.
- Maeda, K.; Cao, H.; Kono, K.; Gorgun, C.Z.; Furuhashi, M.; Uysal, K.T.; Cao, Q.; Atsumi, G.; Malone, H.; Krishnan, B.; et al. Adipocyte/macrophage fatty acid binding proteins control integrated metabolic responses in obesity and diabetes. Cell. Metab. 2005, 1, 107–119.
- Kralisch, S.; Klöting, N.; Ebert, T.; Kern, M.; Hoffmann, A.; Krause, K.; Jessnitzer, B.; Lossner, U.; Sommerer, I.; Stumvoll, M.; et al. Circulating adipocyte fatty acid-binding protein induces insulin resistance in mice in vivo. Obesity (Silver Spring) 2015, 23, 1007–1013.
- Li, S.; Bi, P.; Zhao, W.; Lian, Y.; Zhu, H.; Xu, D.; Ding, J.; Wang, Q.; Yin, C. Prognostic Utility of Fatty Acid-Binding Protein 4 in Patients with Type 2 Diabetes and Acute Ischemic Stroke. Neurotox. Res. 2018, 33, 309–315.
- Furuhashi, M.; Tuncman, G.; Görgün, C.Z.; Makowski, L.; Atsumi, G.; Vaillancourt, E.; Kono, K.; Babaev, V.R.; Fazio, S.; Linton, M.F.; et al. Treatment of diabetes and atherosclerosis by inhibiting fatty-acid-binding protein aP2. Nature 2007, 447, 959–965.
- Erbay, E.; Babaev, V.R.; Mayers, J.R.; Makowski, L.; Charles, K.N.; Snitow, M.E.; Fazio, S.; Wiest, M.M.; Watkins, S.M.; Linton, M.F.; et al. Reducing endoplasmic reticulum stress through a macrophage lipid chaperone alleviates atherosclerosis. Nat. Med. 2009, 15, 1383–1391.
- Hui, X.; Li, H.; Zhou, Z.; Lam, K.S.; Xiao, Y.; Wu, D.; Ding, K.; Wang, Y.; Vanhoutte, P.M.; Xu, A. Adipocyte fatty acid-binding protein modulates inflammatory responses in macrophages through a positive feedback loop involving c-Jun NH2-terminal kinases and activator protein-1. J. Biol. Chem. 2010, 285, 10273–10280.
- Hotamisligil, G.S.; Bernlohr, D.A. Metabolic functions of FABPs--mechanisms and therapeutic implications. Nat. Rev. Endocrinol. 2015, 11, 592–605.
- Smith, A.J.; Thompson, B.R.; Sanders, M.A.; Bernlohr, D.A. Interaction of the adipocyte fatty acid-binding protein with the hormone-sensitive lipase: regulation by fatty acids and phosphorylation. J. Biol. Chem. 2007, 282, 32424–32432.
- Thompson, B.R.; Mazurkiewicz-Muñoz, A.M.; Suttles, J.; Carter-Su, C.; Bernlohr, D.A. Interaction of adipocyte fatty acid-binding protein (AFABP) and JAK2: AFABP/aP2 as a regulator of JAK2 signaling. J. Biol. Chem. 2009, 284, 13473–13480.
- Furuhashi, M.; Saitoh, S.; Shimamoto, K.; Miura, T. Fatty Acid-Binding Protein 4 (FABP4): Pathophysiological Insights and Potent Clinical Biomarker of Metabolic and Cardiovascular Diseases. Clin. Med. Insights Cardiol. 2015, 8, 23–33.
- Li, H.; Luo, H.Y.; Liu, Q.; Xiao, Y.; Tang, L.; Zhong, F.; Huang, G.; Xu, J.M.; Xu, A.M.; Zhou, Z.G.; et al. Intermittent High Glucose Exacerbates A-FABP Activation and Inflammatory Response through TLR4-JNK Signaling in THP-1 Cells. J. Immunol. Res. 2018, 2018, 1319272.
- Fu, Y.; Luo, N.; Lopes-Virella, M.F.; Garvey, W.T. The adipocyte lipid binding protein (ALBP/aP2) gene facilitates foam cell formation in human THP-1 macrophages. Atherosclerosis 2002, 165, 259–269.
- Kazemi, M.R.; McDonald, C.M.; Shigenaga, J.K.; Grunfeld, C.; Feingold, K.R. Adipocyte fatty acid-binding protein expression and lipid accumulation are increased during activation of murine macrophages by toll-like receptor agonists. Arterioscler. Thromb. Vasc. Biol. 2005, 25, 1220–1224.
- Pelton, P.D.; Zhou, L.; Demarest, K.T.; Burris, T.P. PPARgamma activation induces the expression of the adipocyte fatty acid binding protein gene in human monocytes. Biochem. Biophys. Res. Commun. 1999, 261, 456–458.
- Yao, F.; Li, Z.; Ehara, T.; Yang, L.; Wang, D.; Feng, L.; Zhang, Y.; Wang, K.; Shi, Y.; Duan, H.; et al. Fatty Acid-Binding Protein 4 mediates apoptosis via endoplasmic reticulum stress in mesangial cells of diabetic nephropathy. Mol. Cell. Endocrinol. 2015, 411, 232–242.
- Nakamura, R.; Okura, T.; Fujioka, Y.; Sumi, K.; Matsuzawa, K.; Izawa, S.; Ueta, E.; Kato, M.; Taniguchi, S.I.; Yamamoto, K. Serum fatty acid-binding protein 4 (FABP4) concentration is associated with insulin resistance in peripheral tissues, A clinical study. PLoS ONE 2017, 12, e0179737.
- Zhang, Y.; Zhang, H.H.; Lu, J.H.; Zheng, S.Y.; Long, T.; Li, Y.T.; Wu, W.Z.; Wang, F. Changes in serum adipocyte fatty acid-binding protein in women with gestational diabetes mellitus and normal pregnant women during mid- and late pregnancy. J. Diabetes Investig. 2016, 7, 797–804.
- Xie, X.; Yi, Z.; Sinha, S.; Madan, M.; Bowen, B.P.; Langlais, P.; Ma, D.; Mandarino, L.; Meyer, C. Proteomics analyses of subcutaneous adipocytes reveal novel abnormalities in human insulin resistance. Obesity (Silver Spring) 2016, 24, 1506–1514.
- Ning, H.; Tao, H.; Weng, Z.; Zhao, X. Plasma fatty acid-binding protein 4 (FABP4) as a novel biomarker to predict gestational diabetes mellitus. Acta Diabetol. 2016, 53, 891–898.
- Nourani, M.R.; Owada, Y.; Kitanaka, N.; Sakagami, H.; Hoshi, H.; Iwasa, H.; Spener, F.; Kondo, H. Occurrence of immunoreactivity for adipocyte-type fatty acid binding protein in degenerating granulosa cells in atretic antral follicles of mouse ovary. J. Mol. Histol. 2005, 36, 491–497.
- Hsu, W.C.; Okeke, E.; Cheung, S.; Keenan, H.; Tsui, T.; Cheng, K.; King, G.L. A cross-sectional characterization of insulin resistance by phenotype and insulin clamp in East Asian Americans with type 1 and type 2 diabetes. PLoS ONE 2011, 6, e28311.
- Ota, H.; Furuhashi, M.; Ishimura, S.; Koyama, M.; Okazaki, Y.; Mita, T.; Fuseya, T.; Yamashita, T.; Tanaka, M.; Yoshida, H.; et al. Elevation of fatty acid-binding protein 4 is predisposed by family history of hypertension and contributes to blood pressure elevation. Am. J. Hypertens. 2012, 25, 1124–1130.
- Wu, L.E.; Samocha-Bonet, D.; Whitworth, P.T.; Fazakerley, D.J.; Turner, N.; Biden, T.J.; James, D.E.; Cantley, J. Identification of fatty acid binding protein 4 as an adipokine that regulates insulin secretion during obesity. Mol. Metab. 2014, 3, 465–473.
- Greco, E.A.; Francomano, D.; Fornari, R.; Marocco, C.; Lubrano, C.; Papa, V.; Wannenes, F.; di Luigi, L.; Donini, L.M.; Lenzi, A.; et al. Negative association between trunk fat, insulin resistance and skeleton in obese women. World J. Diabetes 2013, 4, 31–39.
- Ni, X.; Gu, Y.; Yu, H.; Wang, S.; Chen, Y.; Wang, X.; Yuan, X.; Jia, W. Serum Adipocyte Fatty Acid-Binding Protein 4 Levels Are Independently Associated with Radioisotope Glomerular Filtration Rate in Type 2 Diabetic Patients with Early Diabetic Nephropathy. Biomed. Res. Int. 2018, 2018, 4578140.
- Toruner, F.; Altinova, A.E.; Akturk, M.; Kaya, M.; Arslan, E.; Bukan, N.; Kan, E.; Yetkin, I.; Arslan, M. The relationship between adipocyte fatty acid binding protein-4, retinol binding protein-4 levels and early diabetic nephropathy in patients with type 2 diabetes. Diabetes Res. Clin. Pract. 2011, 91, 203–207.
- Yeung, D.C.; Xu, A.; Tso, A.W.; Chow, W.S.; Wat, N.M.; Fong, C.H.; Tam, S.; Sham, P.C.; Lam, K.S. Circulating levels of adipocyte and epidermal fatty acid-binding proteins in relation to nephropathy staging and macrovascular complications in type 2 diabetic patients. Diabetes Care 2009, 32, 132–134.
- Kralisch, S.; Stepan, H.; Kratzsch, J.; Verlohren, M.; Verlohren, H.J.; Drynda, K.; Lössner, U.; Blüher, M.; Stumvoll, M.; Fasshauer, M. Serum levels of adipocyte fatty acid binding protein are increased in gestational diabetes mellitus. Eur. J. Endocrinol. 2009, 160, 33–38.
- Cabré, A.; Lázaro, I.; Girona, J.; Manzanares, J.M.; Marimón, F.; Plana, N.; Heras, M.; Masana, L. Plasma fatty acid binding protein 4 is associated with atherogenic dyslipidemia in diabetes. J. Lipid Res. 2008, 49, 1746–1751.
- Okazaki, Y.; Furuhashi, M.; Tanaka, M.; Mita, T.; Fuseya, T.; Ishimura, S.; Watanabe, Y.; Hoshina, K.; Akasaka, H.; Ohnishi, H.; et al. Urinary excretion of fatty acid-binding protein 4 is associated with albuminuria and renal dysfunction. PLoS ONE 2014, 9, e115429.
- Tanaka, M.; Furuhashi, M.; Okazaki, Y.; Mita, T.; Fuseya, T.; Ohno, K.; Ishimura, S.; Yoshida, H.; Miura, T. Ectopic expression of fatty acid-binding protein 4 in the glomerulus is associated with proteinuria and renal dysfunction. Nephron. Clin. Pract. 2014, 128, 345–351.
- Hu, X.; Ma, X.; Luo, Y.; Xu, Y.; Xiong, Q.; Pan, X.; Bao, Y.; Jia, W. Contribution of serum adipocyte fatty acid-binding protein levels to the presence of microalbuminuria in a Chinese hyperglycemic population. J. Diabetes Investig. 2017, 8, 582–589.
- Scalera, A.; Tarantino, G. Could metabolic syndrome lead to hepatocarcinoma via non-alcoholic fatty liver disease? World J. Gastroenterol. 2014, 20, 9217–9228.
- Thompson, K.J.; Austin, R.G.; Nazari, S.S.; Gersin, K.S.; Iannitti, D.A.; McKillop, I.H. Altered fatty acid-binding protein 4 (FABP4) expression and function in human and animal models of hepatocellular carcinoma. Liver Int. 2018, 38, 1074–1083.
More
Information
Subjects:
Others
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
920
Revisions:
2 times
(View History)
Update Date:
16 Aug 2023
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





