
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Wenxin Tian | -- | 686 | 2023-07-06 17:00:08 | | | |
| 2 | Wenxin Tian | + 1425 word(s) | 2111 | 2023-07-10 10:46:02 | | |
Video Upload Options
Vanadium redox flow batteries (VRFBs) are promising candidates for large-scale energy storage, and the electrolyte plays a critical role in chemical–electrical energy conversion. However, the operating temperature of VRFBs is limited to 10–40 °C because of the stability of the electrolyte. To overcome this, various chemical species are added, but the progress and mechanism have not been summarized and discussed yet.
1. Introduction
VRFBs, first proposed by Skyllas-Kazacos in 1986 [19], consist of three key factors: electrodes, an ion exchange membrane, and electrolytes [20–23]. Among them, the electrolyte, as the core part of the vanadium battery system, greatly affects the energy density and overall performance of the battery. It can generally be divided into positive and negative electrolytes, which correspond to the sulfuric acid solutions of the V(IV)/V(V) and V(II)/V(III) redox couples in Equations (1)–(3) [24], respectively. The same elements at different oxidation states can be converted to one another at the electrodes, achieving the chemical–electrical energy conversion, as shown in Figure 1.
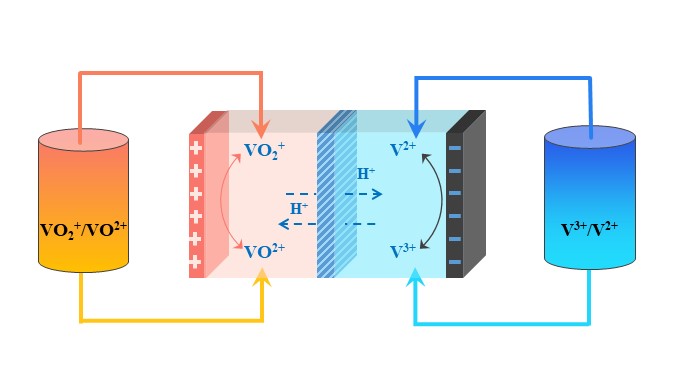
Figure 1. Vanadium redox flow battery schematic.
The vanadium electrolyte is generally prepared through the methods of physical dissolution, chemical reduction, electrolysis, and chemistry–electrolysis coupling [25] Among them, the chemistry–electrolysis coupling is the dominant method, which takes high-purity V2O5 as the raw material and adds reducing agents such as H2C2O4 [26], SO2, [27], and elemental sulfur to the sulfuric acid to prepare the V(IV) electrolyte, and then reduces it by electrolysis to obtain the V(III) electrolyte. If the operating temperature of the vanadium electrolyte is higher than 40 °C or lower than 10 °C, both the electrolyte stability and energy density of vanadium batteries will decrease, accompanied by capacity loss and battery failure [28]. To solve this problem, additives are added to the electrolyte [29] to improve its stability and optimize the electrochemical kinetics, so as to expand the operating temperature range and raise the energy density of the VRFB. For example, the introduction of ammonium dihydrogen phosphate [30] and acidic amino acid [31] as additives can enhance the high-temperature stability of electrolytes, and ammonium and α-lactose monohydrate [32] have been used to improve the low-temperature stability of electrolytes. Additionally, additives such as polyacrylic acid (PAA) [33] can strengthen electrochemical mass transfer.
Additives in vanadium electrolytes, generally classified as inorganics, organics, and compounds, exhibit different microscopic mechanisms, including complexation, electrostatic repulsion, and growth inhibition. Specifically, while complexation improves anti-precipitation properties by changing the distribution of electron clouds, electrostatic repulsion reduces the agglomeration of V(V) ions by enhancing the dispersion effect of V(V) ions from each other. Growth inhibition, on the other hand, lowers the size of settled particles by impeding the growth kinetics of V2O5. Furthermore, the electrochemical mass transfer enhancers in vanadium electrolytes mainly perform hydrophilic modification to enhance electrochemical kinetics, such as MSA, which can be adsorbed on the electrodes to increase the active sites. This adsorption behavior can promote redox reaction kinetics in the interface and optimize the electrochemical performance of the VRFB. Therefore, the introduction of additives can effectively increase the operating temperature range of the vanadium electrolyte, providing effective technical support for the large-scale application of the VRFB.
2. The Function Mechanisms of Additives
The function mechanisms of additives involved in electrolyte performance improvement are still under investigation. Because additives are mainly classified into stabilizing agents, including complexing agents and electrostatic repulsion agents, and growth inhibitors and electrochemical enhancers, the stabilizing mechanisms and enhancement mechanisms will be discussed separately. Whereas the principal stabilizing mechanisms involve complexation, electrostatic repulsion, and growth inhibition, the enhancement mechanism of electrochemical mass transfer mainly involves additives’ hydrophilic modification of the electrode.
3.1.1. Complexation
Complexation is the foremost stabilizing mechanism of additives, and is applied to both inorganic and organic additives. As illustrated in Figure 2, ions (Cl−, H2PO4−) or functional groups (-COOH, -NH2, -OH, -SO3H) carrying lone-pair electrons are capable of coordinating with hydrated vanadium ions in the electrolyte to form a more stable intermediate with V–O–S, V–O–P, V–O–Cl, V–O–N, and so on, thereby effectively reducing the formation of V–O–V and inhibiting V2O5 precipitation. Subsequently, the electron density of vanadium will increase and the local positive charge of vanadium will decrease [50]. Furthermore, the reaction barrier of forming V–O–X (X is the core element of additives) is generally lower than that of forming V–O–V from V2O5, which considerably reduces the generation selectivity of V2O5 precipitation [51,52].
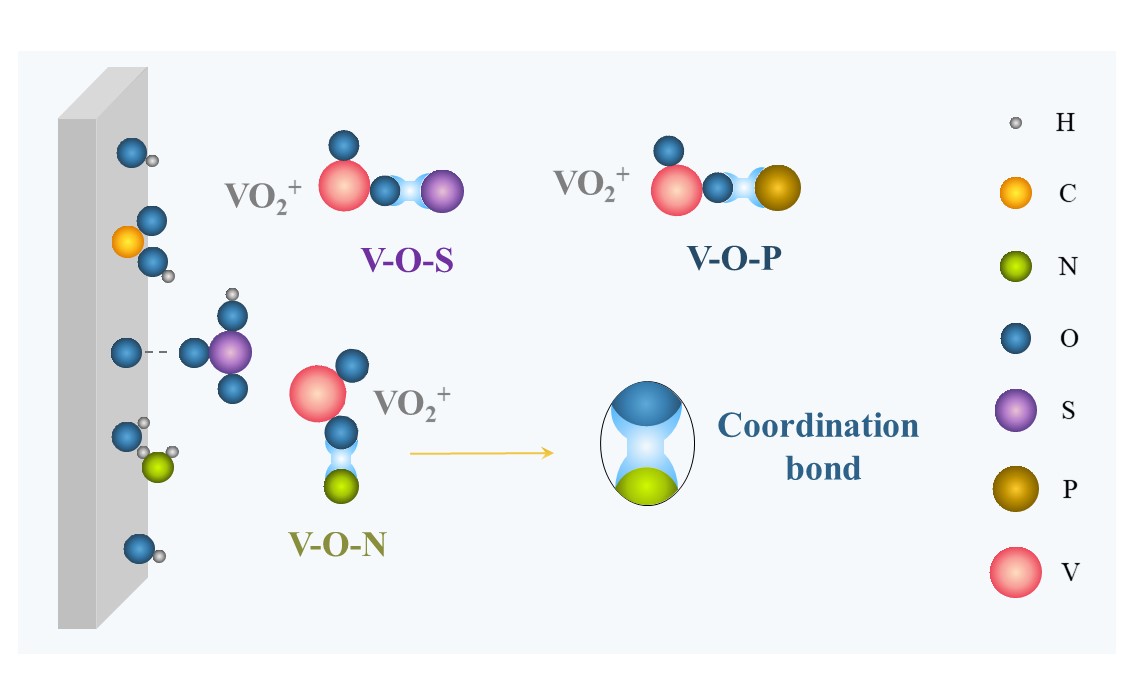
Figure 2. Schematic of complexation.
In addition, a large number of studies have shown that complexation behaviors can be impacted by the geometries of hydrated vanadium ions in different valences, the synergistic effect of additives, and other supplementary factors. For one, the geometries of hydrated ions of vanadium in diverse valences are different and the complexing capacity is positively correlated with the stability of the additives to vanadium ions in all valences. Clarifying the complexation of vanadium ions with additives in each valence is necessary for both stabilization maximization and additive selection. For another, adopting synergistic effects can intensify the stabilization effect and maintain a balance between various ions, such as phosphate and ammonium. Finally, other supplementary factors can be involved, including introducing double additives to form a competing relationship, modifying the electrodes of VRFBs, integrating a thermally regenerative electrochemical cycle (TREC) into the VRFBs, and so on. Researchers should be aware of the above-mentioned considerations when selecting additives.
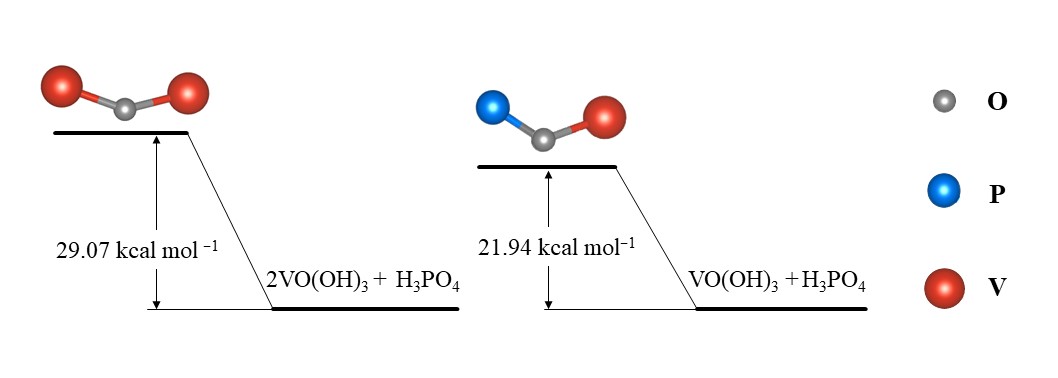
Taking H3PO4 as an example, the transformation path of V(V) in phosphates-added electrolytes is: [VO2(H2O)2]+ → [VO(OH)2(H2O)]+ → VO(OH)3 [53]. The VO(OH)3 intermediate can form a compound containing a V–O–P bond with H3PO4. Moreover, the activation energy of this reaction is generally lower than that of the formation of the V–O–V bond, which could effectively avoid V2O5 precipitation, as presented in Figure 3. Furthermore, the dominant form of the anions is H2PO4− after phosphate additives are added to the positive electrolyte [51]. In this case, due to the partial dimerization of V(V), the sulfate is coordinated to two oxygen atoms in a bridging or bidentate coordination. This can be explained by the coordination of H3PO4 or by the rearrangement of the complexation pattern due to dimerization.
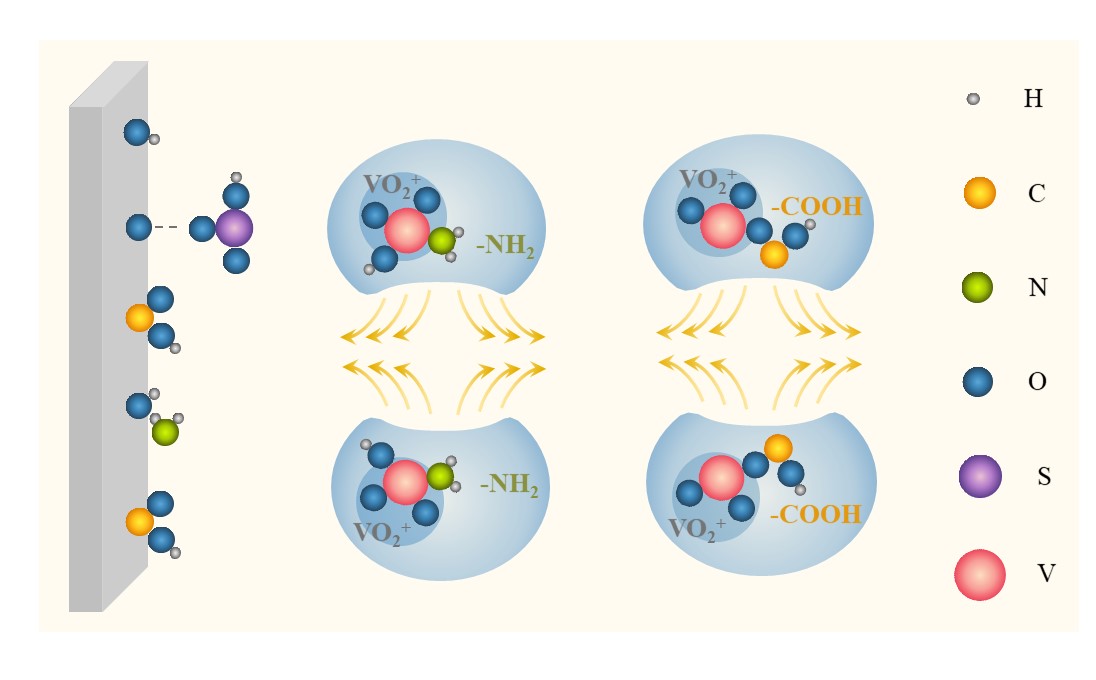
2.1.2. Electrostatic Repulsion
2.1.3. Growth Inhibition
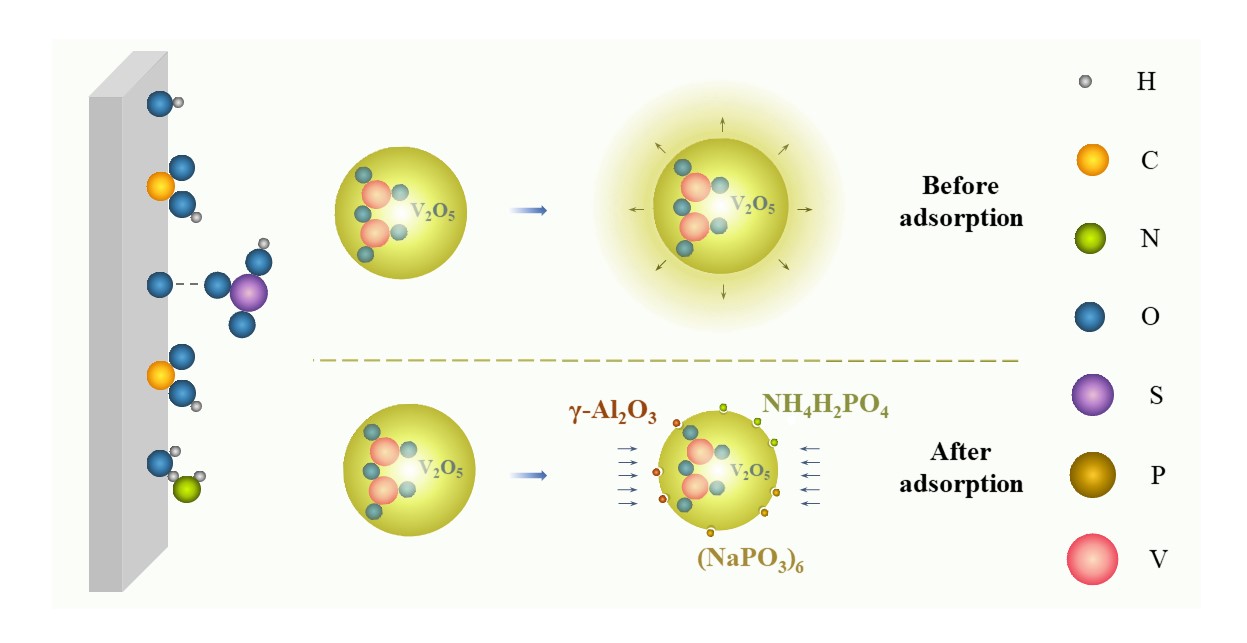
Figure 6. Schematic of growth inhibition.
2.2. Electrochemical Mass Transfer Enhancement Mechanism
As Figure 7 shows, the Helmholtz model depicts how the opposite charges can form a set of polar plates in an electrolyte due to mutual attraction in the electrode–electrolyte interface, further forming a capacitor to store energy, which is called the electric double layer. The whole process of redox reaction is divided into a reaction region and a transfer region, whose rates are co-controlled by electron transfer and migrating mass transfer. The high charge gradient in the region of the electric double layer reduces the mass transfer rate of the solid–liquid interface and accelerates the reaction rate. Therefore, in the transfer region, the rate of transfer usually slows down, thus limiting the electrochemical performance, including energy efficiency, capacity retention rate, and the properties of charging and discharging. Accordingly, the mass transfer of electric double layers becomes an important speed-controlled step of electrochemical kinetics.
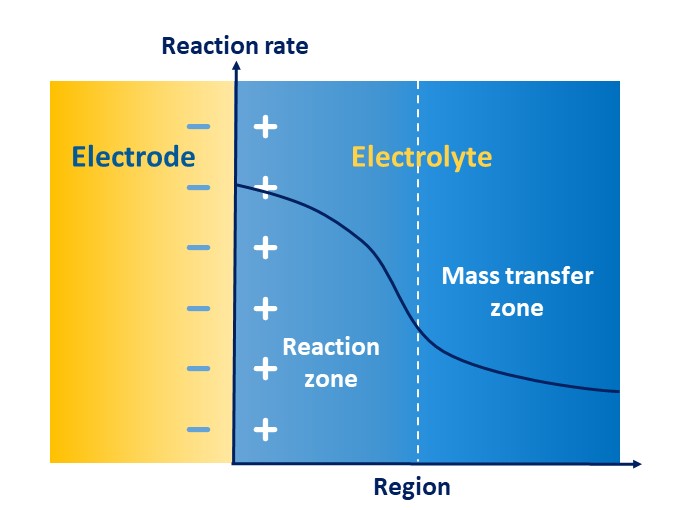
Figure 7. Schematic of the mass transfer of electric double layers.
The foremost mechanism of enhancing electrochemical mass transfer is that additives can be adsorbed on the surface of the electrode to promote active sites to form a “hydrophilic modification” to electrodes, mainly including hydroxyl, the sulfonic group, pyridyl, and other hydrophilic functional groups, or certain ions [32,57], as illustrated in Figure 8. This modifying behavior to electrodes can activate the interfacial activity between electrodes and the electrolyte, accelerating both the redox reaction of vanadium ions in all valences and the migration mass transfer. Alternatively, additives, such as taurine, MSA, PPS, benzoyl peroxide, and so on, can reduce the overpotential of the VRFB to facilitate the migrating mass transfer, reducing the resistance of the electric double layer and enhancing the kinetics of redox reactions [58–62].
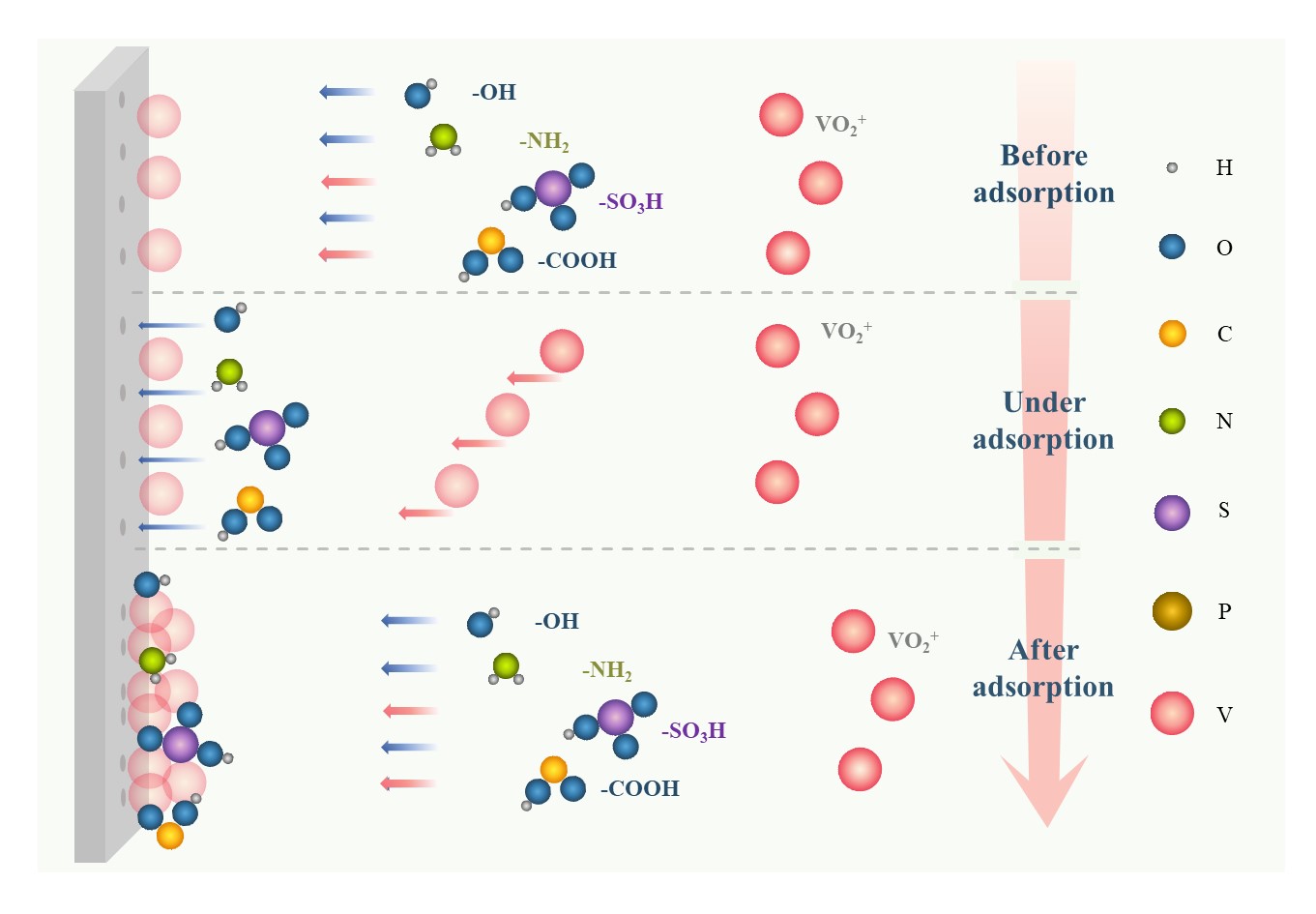
Figure 8. Schematic of the mechanism of electrochemical mass transfer enhancers.





