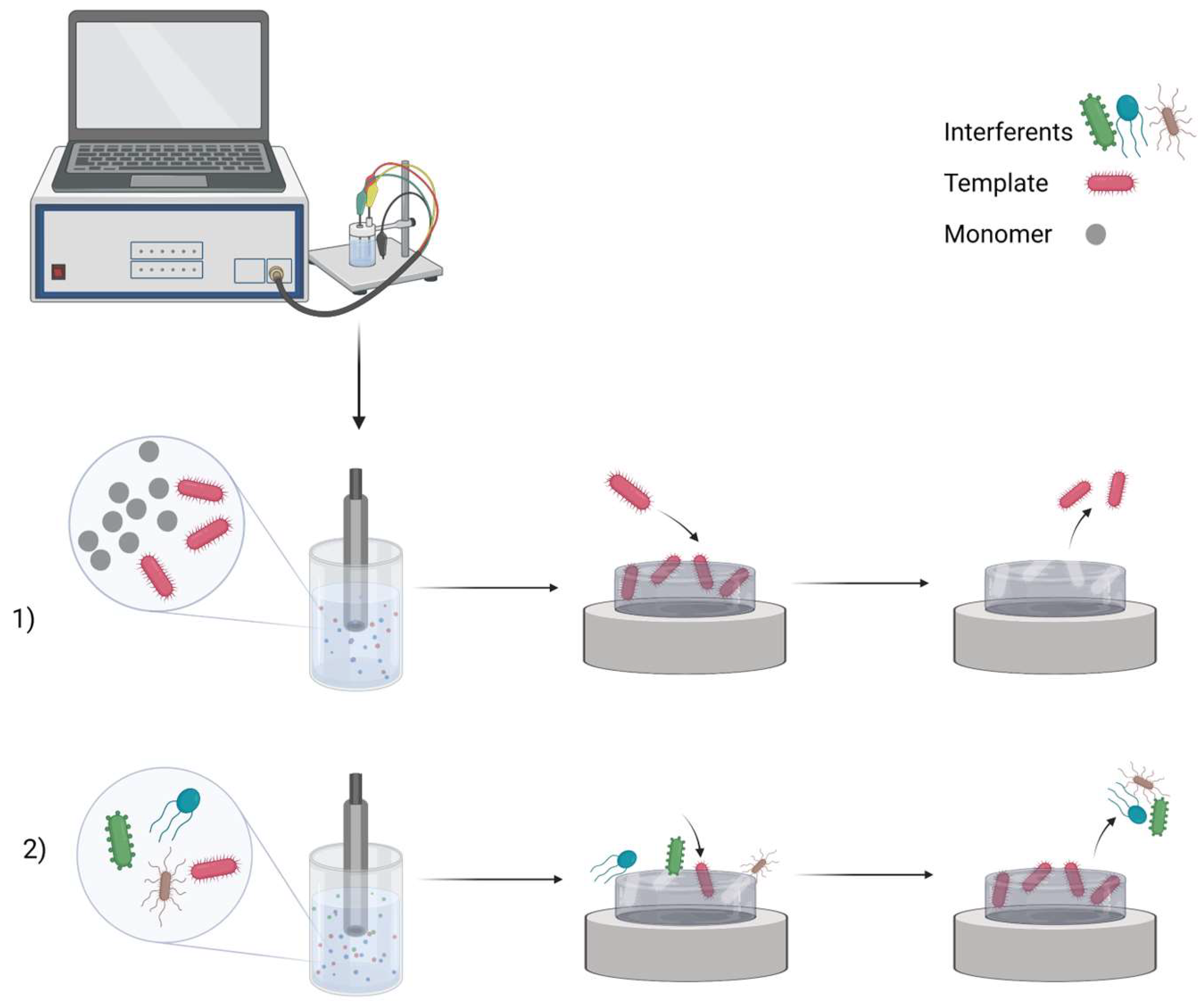
Electrochemical sensor design starts with the selection of particular electrode material. Pencil graphite electrode
[1], graphite electrode
[2], boron-doped nanocrystalline diamond
[2][3][4], platinum electrode
[5], gold of quartz crystal microbalance
[6][7][8] or surface plasmon resonance sensor
[9], etc. were used previously in the design of the electrochemical sensor. MIPs are formed from a solution containing functional monomers and an analyte, the so-called template molecule. Common imprinting techniques include bulk, particle, surface, and epitope imprinting (
Figure 1). By adjusting electrochemical parameters, electropolymerization has unique advantages over other types of polymerization methods in controlling film thickness and porosity. This method is also fast, easy, and affordable. After polymerization occurs, the template molecules are extracted from the polymer matrix to leave complementary cavities. Previous studies have demonstrated MIPs for low molecular weight molecules
[2][3][4][6][8][10][11] or large molecular weight objects (such as proteins
[5][12], DNA
[1], viruses , or bacteria
[13]). The development of MIPs for large objects might be somewhat challenging
[14] and extraction of the template molecule from the polymer matrix is considered to be one of the most complex processes.
2. HIV-1
Lu et al.
[15] developed a biosensor for the detection of protein gp41 related to HIV-1 based on imprinting of the epitope in PDA. Some advantages of epitope imprinting over whole protein imprinting include easier and cheaper template acquisition, more control in template conformation maintenance, easier template removal, and recognition site accessibility. The mixture of dopamine as a functional monomer and the synthetic peptide fragment 579–613 of gp41 as a template was coated on a quartz crystal microbalance chip. This QCM chip was able to bind the most templates when polymerized using 5 mg/mL of dopamine. Lower rebinding and subsequently lower sensitivity would result from a thicker film. The MIP was capable of recognizing not only the epitope template but also the entire gp41 macromolecule. The LOD of gp41 was reached at 2 ng/mL and was successfully used to monitor HIV-1 gp41 in human urine samples. This simple MIP method demonstrates how imprinted epitopes combined with CPs can be used for rapid biomolecular analysis.
Ma et al.
[16] developed a sensitive electrochemical MIP biosensor constructed with conducting polymer Ppy for the detection of HIV surface protein gp120. Through an imine bond formed between glutaraldehyde’s aldehyde groups and the amino groups of the protein, template molecules were covalently joined to the glassy carbon electrode surface. As the free gp120 protein is conformationally unstable, its detection can be challenging. Combining free gp120 with NBD-556 (inhibitor of the interaction between gp120 and receptor) in the MIP biosensor improves recognition, linear range, and LOD of 0.0003 ng/mL by limiting the gp120 conformation.
Even though routine HIV testing of blood products has become standard, the increased transmission of transfusion-related diseases in underdeveloped nations is a result of the absence of quick, accurate, and affordable diagnostics. MIP-based electrochemical sensors, including whole protein or its epitope imprinting, together with CP, can be used to detect biomarkers of early HIV infection.
3. COVID-19
Ratautaite et al.
[6] developed a sensor using molecular imprinting technology for the detection of SARS-CoV-2-S spike glycoprotein. Ppy was chosen as a CP film to entrap the template proteins. MIP (with imprinted template molecule cavities) and non-imprinted polymer (without imprinting) were synthesized on the Pt electrode. The results showed that the changes in MIP current are larger than in the non-imprinted polymer, and the sensor can be applied to the selective detection of imprinted SARS-CoV-2-S glycoprotein.
Ayankojo et al.
[17] designed an electrochemical MIP sensor for a quantitative study of the SARS-CoV-2 spike protein. The SARS-CoV-2 spike protein was imprinted in a thin aminophenyl boronic acid polymer film using a surface imprinting technique. Only in the presence of fluoride ions can 3-aminophenyl boronic acid be electropolymerized into a CP. Real patients’ nasopharyngeal samples were tested, and the LOD was 4.8 pg/mL. Furthermore, the proposed sensor was compatible with a portable potentiostat and can serve as a monitoring platform for COVID-19 patients for rapid and early diagnosis. The sensor demonstrated rapid diagnostic capability with a rebinding time of 15 min and a measurement duration of 5 min.
Also, electrochemical biosensing approach for detecting antibodies against SARS-CoV-2 spike proteins has been developed by immobilizing recombinant SARS-CoV-2 spike proteins on the surface of an AuE modified by a self-assembled monolayer. Cyclic voltammetry, differential pulse voltammetry, potential pulsed amperometry, and electrochemical impedance spectroscopy were chosen for the electrochemical evaluation
[18].
According to the discussed articles, MIP-based electrochemical biosensors are a potential diagnostic strategy to be considered for biomarkers of SARS-CoV-2 infection.
4. Dengue Virus
A MIP-based impedimetric biosensor for dengue virus detection was developed by Arshad et al.
[19]. It is a specific biomarker for dengue virus infection and was used as a template during the polymerization process to modify SPCE, which was subsequently coated with dopamine. To prepare the imprinting material, polysulfone nanofibers were made by electrospinning and later utilized as a support due to their substantial surface area and mechanical durability. NS1 concentrations as low as 0.3 ng/mL were selectively detected by the proposed sensor. According to the authors of the study, the goal of achieving a specific and sensitive analysis succeeded because of the dopamine ability to self-polymerize at room temperature. This feature helped them retain the template’s exact structure (NS1). In the final results, geometrically fit imprinted sites for specific detection of the target analyte were generated. Dengue virus has been detected in other studies using MIP technology with conducting
[20] and non-conducting polymers. Buensuceso et al.
[20] designed an MIP-based sensor on gold-coated QCM crystal for dengue NS1 detection using a terthiophene-based monomer (G03TCOOH) for epitope-imprinting. Terthiophene compounds have low oxidation potential, which makes them ideal for electrochemical functionalization and modification. A LOD of 0.056 μg/mL for NS1 protein was achieved, but the sensor was not tested in real-life samples. Overall, the sensor demonstrated long-term stability, high sensitivity, and selectivity.
In conclusion, both sensors showed satisfactory results for NS1 protein determination. However, the biosensor with PDA on the SPCE was more sensitive (LOD 0.3 ng/mL) than the epitope-imprinted sensor (LOD 0.056 μg/mL). This is probably due to the imprinting of the entire protein template and polysulfone nanofibers, which greatly increased the surface area. MIP-based biosensors can offer simple and cheap diagnostic devices instead of tourniquet tests that can be inaccurate or immunological-based tests that are time-consuming, laborious, expensive, and require complex equipment and highly qualified staff.
5. Hepatitis C Virus
Antipchik et al.
[21] published the first report on the development of an MIP-based electrochemical sensor for detecting the hepatitis C virus via its surface protein E2. Green fluorescent protein was used in conjunction with E2 (total MW of 54 kDa) to prevent protein agglomeration and stabilize its structure. The MIP was prepared by electrochemical surface imprinting the E2 template molecule into poly(m-phenylenediamine) on the SPE electrode. As the proteins are large with unstable conformation, MIP imprinting can have some challenges, such as permanent template entrapment, lower mass transfer, denaturation, and diffusion, which limits the availability of imprinted cavities. The type of imprinted polymer—surface imprinted polymer (SIP) technique—can be advantageous to overcome these challenges. The ability to detect both free antigen E2 and the entire virus particle via E2 is a clear benefit of this technique. A LOD of 0.46 pg/mL and 15 min detection time indicate that the biosensor could be used for early-stage or chronic hepatitis C detection.
Ghanbari and Roushani
[22] developed a novel biosensor for hepatitis C virus core antigen by electropolymerized dopamine around the aptamer (hepatitis C virus core antigen) complex on multi-walled carbon nanotubes-chitosan modified GCE. The improved properties of the MIP-aptamer dual recognition sensor include high sensitivity, low detection limit, high stability, and high selectivity. The results demonstrated that this biosensor could be used to detect HCV core antigens quantitatively in human serum. However, no selectivity studies have been reported. Chitosan is highly adhesive, water-permeable, membrane-forming, biocompatible, and prone to chemical modification due to its reactive hydroxyl and amino functional groups. The biosensor achieved a low LOD of 1.67 fg/mL and showed high stability.
In conclusion, the MIP-based sensor with E2 template poly(m-phenylenediamine) showed a sufficient LOD of 0.46 pg/mL, while the sensor that used the aptamer-antigen complex in the imprinting step had more sensitive results (LOD 1.67 fg/mL), likely due to the MWCN-Chi modification and aptamer affinity for the target protein. MIP-based electrochemical sensors have great potential for the diagnosis of hepatitis C infection.
6. Nosocomial Infections
Sharma et al.
[23] fabricated a simple MIP-based electrochemical sensor for
K. pneumonia bacteria (rod-shaped, 2 µm long, and 0.5 µm diameter) detection using a conducting polymer Ppy (a LOD of 1.352 CFU/mL). The oxidative polymerization reaction produced positively charged Ppy, which easily captured the negatively charged bacteria inside the polymer through weak electrostatic interactions; thus, the bacteria were easily removed from the polymer matrix by sonication and rinsed with deionized water. The electrochemical sensor was tested with five interferents: two other bacteria,
Lactobacillus and
E. coli, different ions (K
+, Mg
++) and molecules (urea, uric acid) that are present in human urine. As for testing interferences, no significant change was observed in DPV current peaks, though the peak was the lowest upon contact with
K. pneumonia bacteria showing the highest affinity to the sensor. The sensor was also tested in urine samples. Later, Pintavirooj et al.
[13] created a more sensitive MIP-based electrochemical biosensor consisting of three monomers to identify
K. pneumoniae, resulting in a high linear response with a lower LOD of 0.012 CFU/mL. Methyl methacrylate (MMA), acrylamide (AAM), and N-vinylpyrrolidone (NVP) at a ratio of 2:1:1 gave the best linearity results. The specificity for the target
K. pneumonia was the highest compared to the other two bacteria,
E. faecalis and
P. aeruginosa.
A simple MIP-based sensor consisting of a conducting Ppy layer on the ITO electrode showed efficient results for
K. pneumoniae detection (LOD 1.352 CFU/mL)
[23], but the more complex sensor consisting of three monomers mixture and graphene oxide combination on the gold SPE, it gives more sensitive detection (LOD 0.012 CFU/mL)
[13].
Sarabaegi and Roushani
[24] reported a sensor based on aptasensing and molecular imprinting for the detection of
P. aeruginosa bacteria. GCE was covered with AuNPs, and then the aptamer-
P. aeruginosa complex was immobilized on the electrode by PDA electropolymerization. The sensor showed excellent results in sensitivity and selectivity against
Shigella flexneri,
Salmonella enteritidis,
Escherichia coli, and
Klebsiella pneumonia. Results in real blood samples also showed a high (99–102%) recovery. With easy preparation, low cost, and high stability, this sensor can provide the detection of a variety of bacteria by imprinting linked aptamers, antibodies, or peptide fragments. Tokonami et al.
[25] constructed an MIP-based sensor for the detection of
P. aeruginosa using electropolymerized Ppy on the surface of Au-QCM. The characterization was obtained by di-electrophoresis. The overoxidation of Ppy allowed the formation of cavities that were shape-complementary to the template bacteria. Although the sensor had good selectivity, the LOD was significantly higher (10
3 CFU/mL) compared to the Sarabaegi and Roushani
[24] reported sensor (LOD 1 CFU/mL).
Liustrovaite et al.
[26] found that an SPCE electrode is more efficient than a Pt electrode for
L. monocytogenes bacteria (0.5–2 μm-long) detection resulting in a LOD of 70 CFU/mL. Since template extraction is challenging for MIP-based sensors, various extraction solutions (sulfuric acid, acetic acid, L-lysine, and trypsin) were tested. The team found that 10% acetic acid and proteolytic enzyme trypsin worked best to extract
L. monocytogenes from the Ppy film.
A summary of the MIP application for the detection of biomarkers of infectious diseases is given in Table 1.
Table 1. Summary of the electrochemical sensors based on the molecularly imprinted polymers for the detection of infectious diseases HIV-1, COVID-19, Dengue virus, hepatitis C virus, and nosocomial infections biomarkers.
From reviewed articles about inflammation, sepsis, COVID-19, HIV-1, dengue, Hepatitis C, and some nosocomial infection biomarker detection, it can be noted that MIP technology in electrochemical biosensors for infectious diseases is well applicable. This type of biosensor could offer more potential rapid clinical or point-of-care diagnostic tests that identify specific pieces of SARS-CoV-2, HIV, dengue, etc. MIPs open up the possibility of early detection of infectious COVID-19, HIV, dengue, sepsis, and other infectious diseases.