
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Chuin Hau Teo | -- | 1712 | 2023-05-25 00:55:18 | | | |
| 2 | Lindsay Dong | Meta information modification | 1712 | 2023-05-25 03:08:29 | | |
Video Upload Options
There are significant gender differences in the relationship between cortisol and depression. While the results may vary based on the age group and source of the samples, several mixed-gender studies show that there appears to be heightened serum cortisol levels in depressed males compared to depressed females. There are also differences noticed in salivary cortisol reactivity, with male children having increased reactivity, though these differences seem to diminish with age. Significant gender differences can also be seen in glucocorticoid and mineralocorticoid receptor expression. These differences are found both in expression levels as well as epigenetic regulation depending on the disorder and on the brain region.
1. Introduction
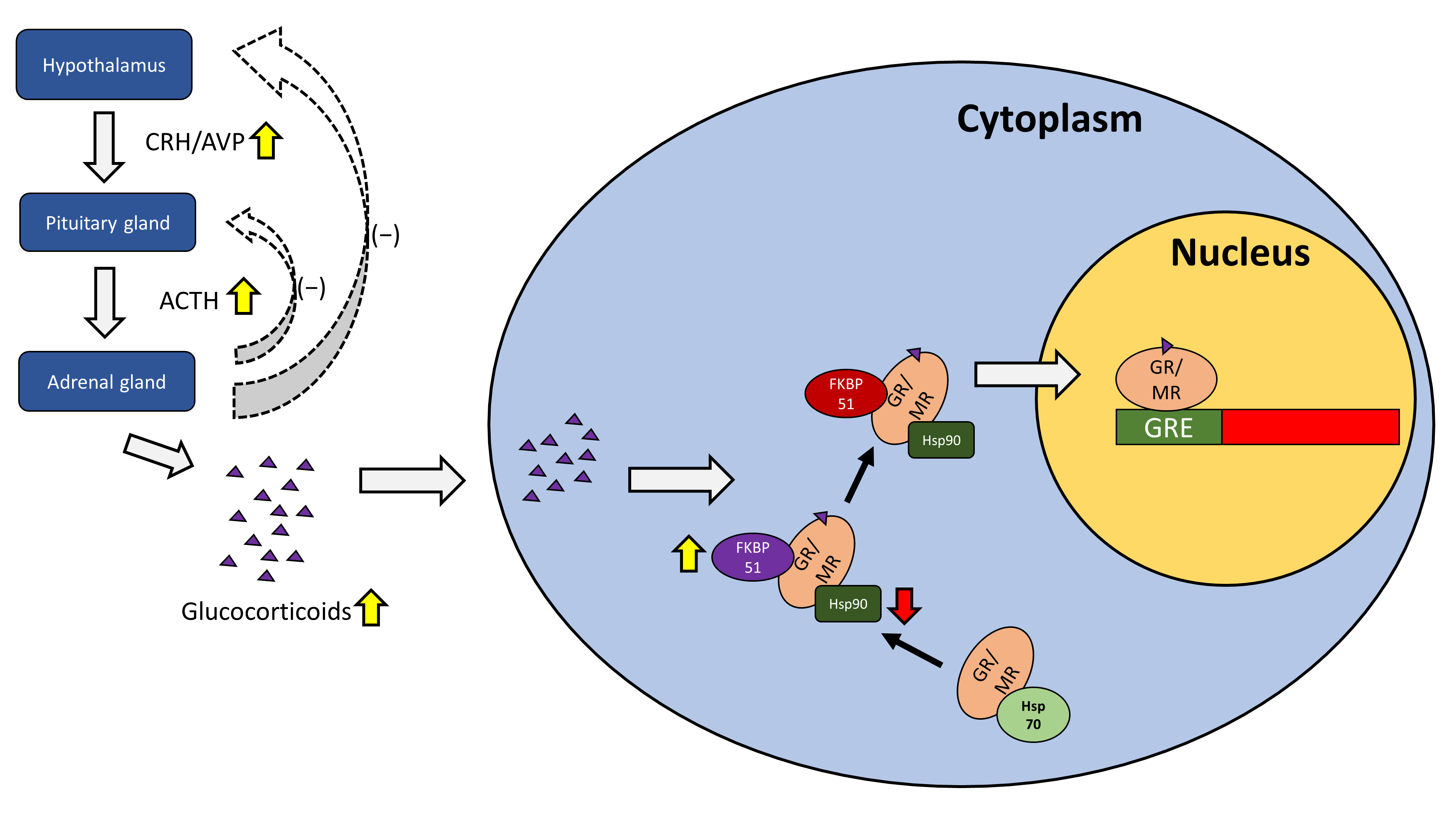
2. Structure and Function of Glucocorticoid and Mineralocorticoid Receptors in the HPA Axis
3. Gender Differences in Glucocorticoid and Mineralocorticoid Receptors in Mental Health
3.1. Gene Expression of Glucocorticoid Receptor- and Mineralocorticoid Receptor-Related Genes
3.2. Gender Differences in Animal Studies of Glucocorticoid and Mineralocorticoid Receptor Expression
References
- Adam, E.K.; Quinn, M.E.; Tavernier, R.; McQuillan, M.T.; Dahlke, K.A.; Gilbert, K.E. Diurnal cortisol slopes and mental and physical health outcomes: A systematic review and meta-analysis. Psychoneuroendocrinology 2017, 83, 25–41.
- Hidalgo, V.; Pulopulos, M.M.; Puig-Perez, S.; Montoliu, T.; Salvador, A. Diurnal cortisol secretion and health-related quality of life in healthy older people. Int. J. Psychophysiol. 2021, 166, 127–133.
- Biliaminu, S.A.; Saka, M.J.; Sanni, E.O.; Imran, J.; Oluwatosin, I.O.; Dane, S. Gender-related Differences in Correlations among BMI, Salivary Testosterone and Cortisol and Depression and Alexithymia Scores in University Students. J. Res. Med. Dent. Sci. 2020, 8, 152–157.
- Altemus, M. Sex differences in depression and anxiety disorders: Potential biological determinants. Horm. Behav. 2006, 50, 534–538.
- McEwen, B.S. Glucocorticoids, depression, and mood disorders: Structural remodeling in the brain. Metabolism 2005, 54, 20–23.
- Kumar, R.; Thompson, E.B. Gene regulation by the glucocorticoid receptor: Structure:function relationship. J. Steroid Biochem. Mol. Biol. 2005, 94, 383–394.
- Trapp, T.; Holsboer, F. Heterodimerization between mineralocorticoid and glucocorticoid receptors increases the functional diversity of corticosteroid action. Trends Pharmacol. Sci. 1996, 17, 145–149.
- Oldehinkel, A.J.; Bouma, E.M.C. Sensitivity to the depressogenic effect of stress and HPA-axis reactivity in adolescence: A review of gender differences. Neurosci. Biobehav. Rev. 2011, 35, 1757–1770.
- Medina, A.; Seasholtz, A.F.; Sharma, V.; Burke, S.; Bunney, W., Jr.; Myers, R.M.; Schatzberg, A.; Akil, H.; Watson, S.J. Glucocorticoid and mineralocorticoid receptor expression in the human hippocampus in major depressive disorder. J. Psychiatr. Res. 2013, 47, 307–314.
- Gehring, U. The structure of glucocorticoid receptors. J. Steroid Biochem. Mol. Biol. 1993, 45, 183–190.
- Duma, D.; Jewell, C.M.; Cidlowski, J.A. Multiple glucocorticoid receptor isoforms and mechanisms of post-translational modification. J. Steroid Biochem. Mol. Biol. 2006, 102, 11–21.
- Oakley, R.H.; Cidlowski, J.A. The biology of the glucocorticoid receptor: New signaling mechanisms in health and disease. J. Allergy Clin. Immunol. 2013, 132, 1033–1044.
- Cao-Lei, L.; Suwansirikul, S.; Jutavijittum, P.; Mériaux, S.B.; Turner, J.D.; Muller, C.P. Glucocorticoid receptor gene expression and promoter CpG modifications throughout the human brain. J. Psychiatr. Res. 2013, 47, 1597–1607.
- Wang, Q.; Van Heerikhuize, J.; Aronica, E.; Kawata, M.; Seress, L.; Joels, M.; Swaab, D.F.; Lucassen, P.J. Glucocorticoid receptor protein expression in human hippocampus; stability with age. Neurobiol. Aging 2013, 34, 1662–1673.
- Wang, Q.; Verweij, E.; Krugers, H.; Joels, M.; Swaab, D.F.; Lucassen, P. Distribution of the glucocorticoid receptor in the human amygdala; changes in mood disorder patients. Brain Struct. Funct. 2014, 219, 1615–1626.
- Koning, A.-S.C.A.M.; Buurstede, J.C.; van Weert, L.T.C.M.; Meijer, O.C. Glucocorticoid and Mineralocorticoid Receptors in the Brain: A Transcriptional Perspective. J. Endocr. Soc. 2019, 3, 1917–1930.
- Rogerson, F.M.; Brennan, F.E.; Fuller, P.J. Mineralocorticoid receptor binding, structure and function. Mol. Cell. Endocrinol. 2004, 217, 203–212.
- Reul, J.M.H.M.; Gesing, A.; Droste, S.; Stec, I.S.M.; Weber, A.; Bachmann, C.; Bilang-Bleuel, A.; Holsboer, F.; Linthorst, A.C.E. The brain mineralocorticoid receptor: Greedy for ligand, mysterious in function. Eur. J. Pharmacol. 2000, 405, 235–249.
- Roy, B.; Dunbar, M.; Shelton, R.C.; Dwivedi, Y. Identification of microRNA-124-3p as a putative epigenetic signature of major depressive disorder. Neuropsychopharmacology 2017, 42, 864–875.
- Wang, S.-S.; Mu, R.-H.; Li, C.-F.; Dong, S.-Q.; Geng, D.; Liu, Q.; Yi, L.-T. microRNA-124 targets glucocorticoid receptor and is involved in depression-like behaviors. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2017, 79, 417–425.
- Farrell, C.; Doolin, K.; O’Leary, N.; Jairaj, C.; Roddy, D.; Tozzi, L.; Morris, D.; Harkin, A.; Frodl, T.; Nemoda, Z. DNA methylation differences at the glucocorticoid receptor gene in depression are related to functional alterations in hypothalamic–pituitary–adrenal axis activity and to early life emotional abuse. Psychiatry Res. 2018, 265, 341–348.
- Plieger, T.; Felten, A.; Splittgerber, H.; Duke, É.; Reuter, M. The role of genetic variation in the glucocorticoid receptor (NR3C1) and mineralocorticoid receptor (NR3C2) in the association between cortisol response and cognition under acute stress. Psychoneuroendocrinology 2018, 87, 173–180.
- Rao, S.; Yao, Y.; Ryan, J.; Li, T.; Wang, D.; Zheng, C.; Xu, Y.; Xu, Q. Common variants in FKBP5 gene and major depressive disorder (MDD) susceptibility: A comprehensive meta-analysis. Sci. Rep. 2016, 6, 32687.
- Wang, H.; Ba, Y.; Han, W.; Zhang, H.; Zhu, L.; Jiang, P. Association of heat shock protein polymorphisms with patient susceptibility to coronary artery disease comorbid depression and anxiety in a Chinese population. PeerJ 2021, 9, e11636.
- Tatro, E.T.; Everall, I.P.; Masliah, E.; Hult, B.J.; Lucero, G.; Chana, G.; Soontornniyomkij, V.; Achim, C.L. Differential expression of immunophilins FKBP51 and FKBP52 in the frontal cortex of HIV-infected patients with major depressive disorder. J. Neuroimmune Pharmacol. 2009, 4, 218–226.
- Sarubin, N.; Hilbert, S.; Naumann, F.; Zill, P.; Wimmer, A.-M.; Nothdurfter, C.; Rupprecht, R.; Baghai, T.C.; Bühner, M.; Schüle, C. The sex-dependent role of the glucocorticoid receptor in depression: Variations in the NR3C1 gene are associated with major depressive disorder in women but not in men. Eur. Arch. Psychiatry Clin. Neurosci. 2017, 267, 123–133.
- Klok, M.D.; Giltay, E.J.; Van der Does, A.J.W.; Geleijnse, J.M.; Antypa, N.; Penninx, B.W.J.H.; de Geus, E.J.C.; Willemsen, G.; Boomsma, D.I.; van Leeuwen, N.; et al. A common and functional mineralocorticoid receptor haplotype enhances optimism and protects against depression in females. Transl. Psychiatry 2011, 1, e62.
- Vinkers, C.H.; Joëls, M.; Milaneschi, Y.; Gerritsen, L.; Kahn, R.S.; Penninx, B.W.J.H.; Boks, M.P.M. Mineralocorticoid receptor haplotypes sex-dependently moderate depression susceptibility following childhood maltreatment. Psychoneuroendocrinology 2015, 54, 90–102.
- Webster, M.J.; Knable, M.B.; O’Grady, J.; Orthmann, J.; Weickert, C.S. Regional specificity of brain glucocorticoid receptor mRNA alterations in subjects with schizophrenia and mood disorders. Mol. Psychiatry 2002, 7, 985–994.
- Bonapersona, V.; Damsteegt, R.; Adams, M.L.; van Weert, L.T.; Meijer, O.C.; Joëls, M.; Sarabdjitsingh, R.A. Sex-Dependent modulation of acute stress reactivity after early life stress in mice: Relevance of mineralocorticoid receptor expression. Front. Behav. Neurosci. 2019, 13, 181.
- Madison, F.N.; Kesner, A.J.; Alward, B.A.; Ball, G.F. Sex differences in hippocampal mineralocorticoid and glucocorticoid receptor mRNA expression in response to acute mate pair separation in zebra finches (Taeniopygia guttata). Hippocampus 2018, 28, 698–706.
- Ramos-Ortolaza, D.L.; Doreste-Mendez, R.J.; Alvarado-Torres, J.K.; Torres-Reveron, A. Ovarian hormones modify anxiety behavior and glucocorticoid receptors after chronic social isolation stress. Behav. Brain Res. 2017, 328, 115–122.
- Boero, G.; Pisu, M.G.; Biggio, F.; Muredda, L.; Carta, G.; Banni, S.; Paci, E.; Follesa, P.; Concas, A.; Porcu, P. Impaired glucocorticoid-mediated HPA axis negative feedback induced by juvenile social isolation in male rats. Neuropharmacology 2018, 133, 242–253.





