
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Timothy Omara | -- | 3485 | 2023-04-28 11:15:22 | | | |
| 2 | Timothy Omara | Meta information modification | 3485 | 2023-04-28 11:20:07 | | | | |
| 3 | Timothy Omara | + 37 word(s) | 3522 | 2023-04-28 12:23:49 | | | | |
| 4 | Lindsay Dong | Meta information modification | 3522 | 2023-05-03 03:35:43 | | | | |
| 5 | Timothy Omara | -268 word(s) | 3254 | 2023-05-05 12:33:00 | | |
Video Upload Options
Cyanobacteria, algal blooms and cyanotoxins have become common environmental enigmas in marine, freshwater and estuarine ecosystems. In East African lakes, mainly Microcystis, Arthrospira, Dolichospermum, Planktolyngbya and Anabaenopsis species of cyanobacteria have been responsible for the production of anatoxin-a, homoanatoxin-a, microcystins, cylindrospermopsin and nodularin. Microcystins and anatoxin-a have been implicated as the proximal cause of indiscriminate fish deaths and epornitic mortality of lesser flamingos
1. Introduction
2. Overview of cyanobacteria, harmful algal blooms and cyanotoxins in East African lacustrine ecosystems
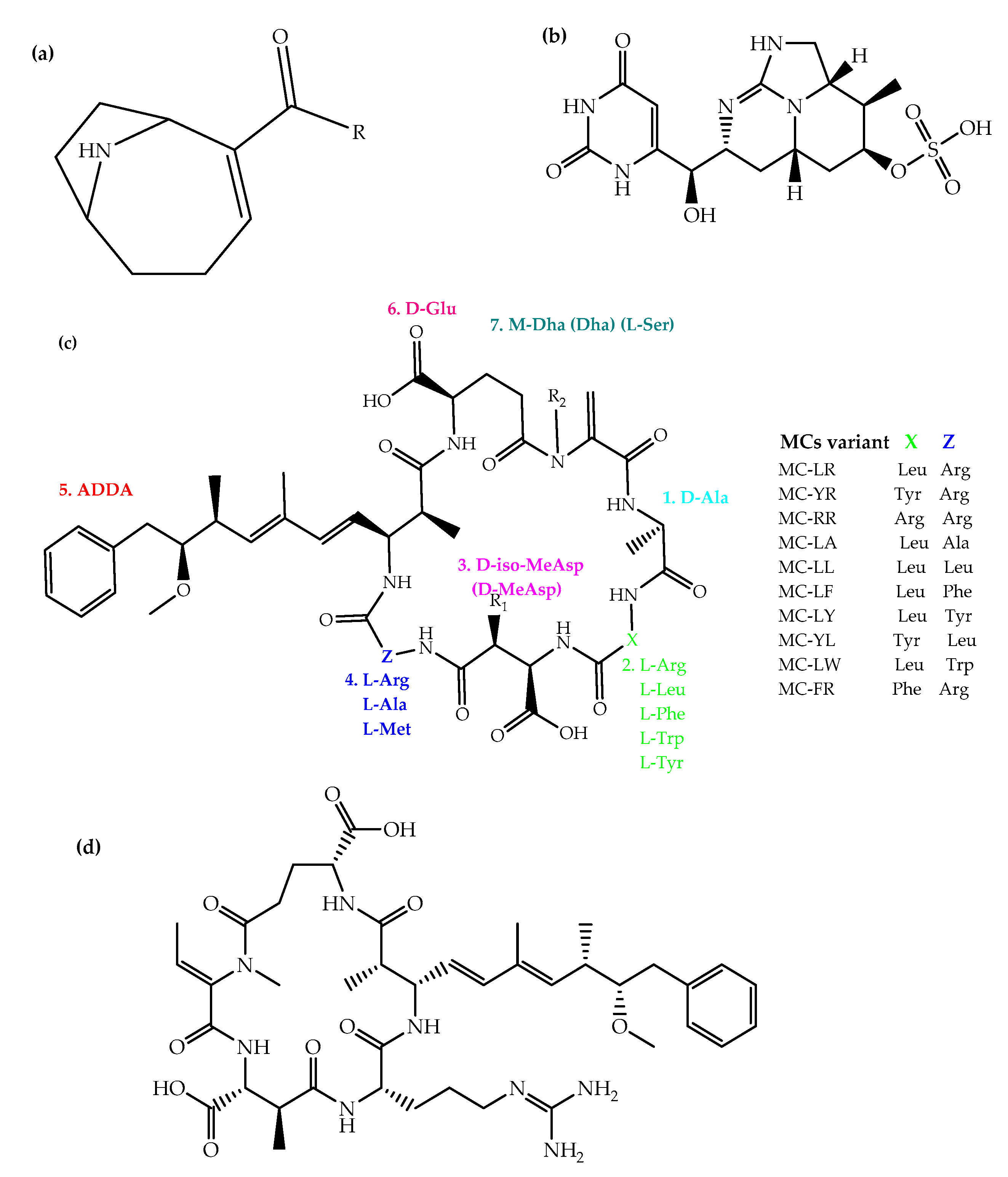
3. Toxicity, Human and Ecological Health Implications of Cyanotoxins in EAC Lakes
3.1. MCs
3.2. Anatoxin-a
3.3. Homoanatoxin-a
3.4. Cylindrospermopsin
3.5. Nodularins
In conclusion, CYB, algal blooms and cyanotoxins have increased in East African lacustrine ecosystems. Dolichospermum, Microcystis, Arthrospira, Planktolyngbya and Anabaenopsis species are the major groups of CYB prevalent in EAC lakes. The only direct ecological effects of cyanotoxins in EAC lakes is indiscriminate fish deaths and mass die-offs of lesser flamingos. With the unequivocal increase in climate change and variability, it is inferred that CYBHAB and cyanotoxins will increase in frequency and severity. This calls for urgent action to mitigate nutrient-rich pollutants loading into water resources and the expansion of CYBHAB from eutrophic lakes to the surrounding marine environments. The (eco)toxicological relevance of co-production of phycotoxins should be assessed in the EAC because such exposure may amplify the toxicological outcomes in aquatic biota and humans. As some CYB encountered in EAC lakes produce other cyanotoxins (such as β-N-methylamino-L-alanine and saxitoxins), studies targeting these cyanobacterial metabolites should be initiated. While there are no reports of cyanotoxin poisoning of humans in the EAC, future studies should examine the risk of hepatocellular cancer, the ingestion of CYB and mycotoxin-contaminated water and foods, and hepatitis virus, which were earlier linked to increased primary liver cancer cases in Asia. Another potential relationship with microplastics should be assessed because they are known to accumulate toxins and amplify their toxicity.
References
- Band-Schmidt, C.; Durán-Riveroll, L.; Bustillos-Guzmán, J.; Leyva-Valencia, I.; López-Cortés, D.; Núñez-Vázquez, E.J.; Hernández-Sandoval, F.; Ramírez-Rodríguez, D. Paralytic Toxin Producing Dinoflagellates in Latin America: Ecology and Physiology. Front. Mar. Sci. 2019, 6, 42.
- Klijnstra, M.D.; Faassen, E.J.; Gerssen, A. A Generic LC-HRMS Screening Method for Marine and Freshwater Phycotoxins in Fish, Shellfish, Water, and Supplements. Toxins 2021, 13, 823.
- Ndlela, L.L.; Oberholster, P.J.; Van Wyk, J.H.; Cheng, P.H. An overview of cyanobacterial bloom occurrences and research in Africa over the last decade. Harmful Algae 2016, 60, 11–26.
- Reinl, K.L.; Brookes, J.D.; Carey, C.C.; Harris, T.D.; Ibelings, B.W.; Morales-Williams, A.M.; Yokota, K.; Zhan, Q. Cyanobacterial blooms in oligotrophic lakes: Shifting the high-nutrient paradigm. Freshw. Biol. 2021, 66, 1846–1859.
- Kat, M. Special meeting on causes, dynamics and effects of exceptional marine blooms and related events. Int. Counc. Explor. Sea C 1984, 3.
- Jones, M.R.; Pinto, E.; Torres, M.A.; Dörr, F.; Mazur-Marzec, H.; Szubert, K.; Tartaglione, L.; Dell’Aversano, C.; Miles, C.O.; Beach, D.G. CyanoMetDB, a comprehensive public database of secondary metabolites from cyanobacteria. Water Res. 2021, 196, 117017.
- Gerssen, A.; Gago-Martinez, A. Emerging marine biotoxins. Toxins 2019, 11, 314.
- Lihepanyama, D.L.; Ndakidemi, P.A.; Treydte, A.C. Spatio–TemporalWater Quality Determines Algal Bloom Occurrence and Possibly Lesser Flamingo (Phoeniconaias minor) Presence in Momella Lakes, Tanzania. Water 2022, 14, 3532.
- Olokotum, M.; Mitroi, V.; Troussellier, M.; Semyaloa, R.; Bernarde, C.; Montuelle, B.; Okello, W.; Quiblier, C.; Humbert, J.-F. A review of the socioecological causes and consequences of cyanobacterial blooms in Lake Victoria. Harmful Algae 2020, 96, 101829.
- Saulnier-Talbot, É.; Gregory-Eaves, I.; Simpson, K.; Efitre, J.; Nowlan, T.; Taranu, Z.; Chapman, L. Small Changes in Climate Can Profoundly Alter the Dynamics and Ecosystem Services of Tropical Crater Lakes. PLoS ONE 2014, 9, e86561.
- Cocquyt, C.; Plisnier, P.-D.; Mulimbwa, N.; Nshombo, M. Unusual massive phytoplankton bloom in the oligotrophic Lake Tanganyika. Plant Ecol. Evol. 2021, 154, 351–361.
- Ehrenfels, B.; Bartosiewicz, M.; Mbonde, A.; Baumann, K.; Dinkel, C.; Junker, J.; Kamulali, T.; Kimirei, I.; Niederdorfer, R.; Odermatt, D.; et al. Diazotrophic Cyanobacteria are Associated With a Low Nitrate Resupply to Surface Waters in Lake Tanganyika. Front. Environ. Sci 2021, 9, 716765.
- Sarmento, H.; Darchambeau, F.; Descy, J. Phytoplankton of Lake Kivu; Descy, J.P., Darchambeau, F., Schmid, M., Eds.; Lake Kivu. Aquatic Ecology Series; Springer: Dordrecht, The Netherlands, 2012; Volume 5, pp. 67–83.
- Rugema, E.; Darchambeau, F.; Sarmento, H.; Stoyneva-Gärtner, M.; Leitao, M.; Thiery, W.; Latli, A.; Descy, J.P. Long-term change of phytoplankton in Lake Kivu: The rise of the greens. Freshw. Biol. 2019, 64, 1940–1955.
- Ballot, A.; Krienitz, L.; Kotut, K.; Wiegand, C.; Metcalf, J.S.; Codd, G.A.; Pflugmacher, S. Cyanobacteria and cyanobacterial toxins in three alkaline Rift Valley lakes of Kenya—Lakes Bogoria, Nakuru and Elmenteita. J. Plankton Res. 2004, 26, 925–935.
- Ballot, A.; Krienitz, L.; Kotut, K.; Wiegand, C.; Pflugmacher, S. Cyanobacteria and cyanobacterial toxins in the crater lakes Sonachi and Simbi, Kenya. Harmful Algae 2005, 4, 139–150.
- Sitoki, L.; Kurmayer, R.; Rott, E. Spatial variation of phytoplankton composition, biovolume, and resulting microcystin concentrations in the Nyanza Gulf (L. Victoria, Kenya). Hydrobiologia 2012, 691, 109–122.
- Githukia, C.; Onyango, D.; Lusweti, D.; Ramkat, R.; Kowenje, C.; Miruka, J.; Lung’ayia, H.; Orina, P. An Analysis of Knowledge, Attitudes and Practices of Communities in Lake Victoria, Kenya on Microcystin Toxicity. Open J. Ecol. 2022, 12, 198–210.
- Roegner, A.; Sitoki, L.; Weirich, C.; Corman, J.; Owage, D.; Umami, M.; Odada, E.; Miruka, J.; Ogari, Z.; Smith, W.; et al. Harmful Algal Blooms Threaten the Health of Peri-Urban Fisher Communities: A Case Study in Kisumu Bay, Lake Victoria, Kenya. Expo. Health 2020, 12, 835–848.
- Raffoul, Melissa H., "Assessing the potential health risk of cyanobacteria harmful algal blooms andcyanotoxins in Lake Naivasha, Kenya" (2012). Electronic Thesis and Dissertation Repository. 828.https://ir.lib.uwo.ca/etd/828
- Roegner, A. F., J. R. Corman, L. M. Sitoki, Z. A. Kwena, Z. Ogari, J. B. Miruka, A. Xiong, C. Weirich, C. M. Aura, and T. R. Miller Impacts of algal blooms and microcystins in fish on small-scale fishers in Winam Gulf, Lake Victoria: implications for health and livelihood. Ecology and Society 2023, 28, 49, https://doi.org/10.5751/ES-13860-280149.
- Hamisi, M.; Lugomela, C.; Lyimo, T.; Bergman, B.; Díez, B. Plankton composition, biomass, phylogeny and toxin genes in Lake Big Momela, Tanzania. Afr. J. Aquat. Sci. 2017, 42, 109–121.
- Lugomela, C.; Pratap, H.B.; Mgaya, Y.D. Cyanobacteria Blooms: A Possible Cause of Mass Mortality of Lesser Flamingos in Lake Manyara and Lake Big Momela, Tanzania. Harmful Algae 2006, 5, 534–541.
- Nonga, H.E.; Mdegela, R.H.; Sandvik, M.; Lie, E.; Miles, C.O.; Skaare, J.U. Cyanobacteria and cyanobacterial toxins in the alkaline-saline Lakes Natron and Momela, Tanzania. Tanzan. Vet. Assoc. Proc. 2017, 32, 108–116.
- Miles, C.O.; Sandvik, M.; Nonga, H.E.; Rundberget, T.; Wilkins, A.L.; Rise, F.; Ballot, A. Identification of microcystins in a Lake Victoria cyanobacterial bloom using LC-MS with thiol derivatization. Toxicon 2013, 70, 21–31.
- Mchau, G.; Machunda, R.; Kimanya, M.; Makule, E.; Gong, Y.; Mpolya, E.; Meneely, J.; Elliott, C.; Greer, B. First Report of the Co-occurrence of Cylindrospermopsin, Nodularin and Microcystins in the Freshwaters of Lake Victoria, Tanzania. Expo. Health 2021, 13, 185–194.
- Busobozi, E. Eutrophication in Ugandan Crater Lakes. A Case Study of Six Crater Lakes Located in Kabarole District Western Uganda. Master’s Thesis, University of Canterbury Christchurch, Christchurch, New Zealand, 2017.
- Nankabirwa, A.; De Crop, W.; Van der Meeren, T.; Cocquyt, C.; Plisnier, P.-D.; Balirwa, J.; Verschuren, D. Phytoplankton communities in the crater lakes of western Uganda, and their indicator species in relation to lake trophic status. Ecol. Indic. 2019, 107, 105563.
- Stoyneva-Gärtner, M.P.; Morana, C.; Borges, A.V.; Okello, W.; Bouillon, S.; Deirmendjian, L.; Lambert, T.; Roland, F.; Nankabirwa, A.; Nabafu, E.; et al. Diversity and ecology of phytoplankton in Lake Edward (East Africa): Present status and long-term changes. J. Great Lakes Res. 2020, 46, 741–751.
- Ganf, G.G. Phytoplankton Biomass and Distribution in a Shallow Eutrophic Lake (Lake George, Uganda). Oecologia 1974, 16, 9–29.
- Okello, W.; Ostermaier, V.; Portmann, C.; Gademann, K.; Kurmayer, R. Spatial isolation favours the divergence in microcystin net production by Microcystis in Ugandan freshwater lakes. Water Res. 2010, 44, 2803–2814.
- Kamanyi, J.R.; Ogwang, O.; Twongo, E. Plankton identified in stomach contents of Oreochromis niloticus (Pisces, Cichlidae) and the water system of Lakes Edward, George, and Kazinga channel—Uganda. Afr. J. Trop. Hydrobiol. Fish 1996, 7, 49–54.
- Nyakoojo, C.; Byarujali, S.M. An ecological study of two shallow, equatorial lakes: Lake Mburo and Lake Kachera, Uganda. Afr. J. Ecol. 2010, 48, 860–864.
- Kayiira, D. Algal Community of Lake Mburo and Murchison Bay, Lake Victoria. Master’s Thesis, Makerere University, Kampala, Uganda, 2007.
- Byarujali, S.M. Phytoplankton production in L. Mburo—Western Uganda. In Proceedings of the First Conference on Ecology and Sustainable Natural Resource Management for Development, Mweya, Queen Elizabeth National Park, Uganda, 27 February–3 March 1995; pp. 284–290.
- Nyakairu, G.; Nagawa, C.; Mbabazi, J. Assessment of cyanobacteria toxins in freshwater fish: A case study of Murchison Bay (Lake Victoria) and Lake Mburo, Uganda. Toxicon 2010, 55, 939–946.
- Semyalo, R.; Rohrlack, T.; Naggawa, C.; Nyakairu, G.W. Microcystin concentrations in Nile Tilapia (Oreochromis niloticus) caught from Murchison Bay, L. Victoria and Lake Mburo: Uganda. Hydrobiologia 2009, 638, 235–244.
- Mukankomeje, R.; Plisnier, P.-D.; Descy, J.-P.; Massaut, L. Lake Muzahi, Rwanda: Limnological features and phytoplankton production. Hydrobiologia 1993, 257, 107–120.
- Mukankomeje, R.; Laviolette, F.; Descy, J.-P. Régime alimentaire de Tilapia, Oreochromis niloticus, du Lac Muhazi (Rwanda). Ann. De Limnol. 1994, 30, 297–312.
- Fazi, S.; Butturini, A.; Tassi, F.; Amalfitano, S.; Venturi, S.; Vazquez, E.; Clokie, M.; Wanjala, S.W.; Pacini, N.; Harper, D.M. Biogeochemistry and biodiversity in a network of saline–alkaline lakes: Implications of ecohydrological connectivity in the Kenyan Rift Valley. Ecohydrol. Hydrobiol. 2018, 18, 96–106.
- Rastogi, R.; Madamwar, D.; Incharoensakdi, A. Bloom Dynamics of Cyanobacteria and Their Toxins: Environmental Health Impacts and Mitigation Strategies. Front. Microbiol 2015, 6, 1254.
- Diez-Quijada, L.; Benítez-González, M.; Puerto, M.; Jos, A.; Cameán, A.M. Immunotoxic Effects Induced by Microcystins and Cylindrospermopsin: A Review. Toxins 2021, 13, 711.
- International Agency for Research on Cancer (IARC). Microcystin-LR. IARC Monographs on the Identification of Carcinogenic Hazards to Humans. Available online: https://monographs.iarc.who.int/list-of-classifications (accessed on 2 December 2022).
- Otero, P.; Silva, M. The role of toxins: Impact on human health and aquatic environments. In the Pharmacological Potential of Cyanobacteria; Academic Press: Cambridge, MA, USA, 2022; pp. 173–199.
- WHO. Cyanobacterial Toxins: Microcystins. Background Document for Development of WHO Guidelines for Drinking-Water Quality and Guidelines for Safe Recreational Water Environments; World Health Organization: Geneva, Switzerland, 2020; WHO/HEP/ECH/WSH/2020.6; Available online: https://apps.who.int/iris/bitstream/handle/10665/338066/WHO-HEP-ECH-WSH-2020.6-eng.pdf (accessed on 2 December 2022).
- Codd, G.A.; Metcalf, J.S.; Morrison, L.F.; Krienitz, L.; Ballot, A.; Pflugmacher, S.; Wiegand, C.; Kotut, K. Susceptibility of flamingos to cyanobacterial toxins via feeding. Vet. Rec. 2003, 152, 722–723.
- Nowicka-Krawczyk, P.; Mühlsteinová, R.; Hauer, T. Detailed characterization of the Arthrospira type species separating commercially grown taxa into the new genus Limnospira (Cyanobacteria). Sci. Rep. 2019, 9, 36831.
- Fox, D.L.; McBeth, J.W. Some dietary carotenoids and blood-carotenoid levels in flamingos. Comp. Biochem. Physiol. 1970, 34, 707–713.
- Fox, D.; Smith, V.E.; Wolfson, A.A. Carotenoid selectivity in blood and feathers of lesser (African), Chilean and greater (European) flamingos. Comp. Biochem. Physiol. 1967, 23, 225–232.
- Lewis, D.; Metallinos-Katsaras, E.; Grivetti, L. Coturnism: Human Poisoning By European Migratory Quail. J. Cult. Geogr. 1987, 7, 51–65.
- Koenig, R. The pink death: Die-offs of the lesser flamingo raise concern. Science 2006, 313, 1724–1725.
- Ballot, A.; Pflugmacher, S.; Wiegand, C.; Kotut, K.; Krause, E.; Metcalf, J.S.; Morrison, L.F.; Codd, G.A.; Krienitz, L. Cyanobacterial toxins, a further contributory cause of mass deaths of flamingos at Kenyan Rift Valley lakes. In Abstracts of the Xth International Conference on Harmful Algae; Tradewinds Conference Center: St. Pete Beach, FL, USA, 2002; p. 20.
- Nonga, H.; Sandvik, M.; Miles, C.; Lie, E.; Mdegela, R.; Mwamengele, G.; Semuguruka, W.; Skaare, J. Possible involvement of microcystins in the unexplained mass mortalities of Lesser Flamingo (Phoeniconaias minor Geoffroy) at Lake Manyara in Tanzania. Hydrobiologia 2011, 678, 167–178.
- Fyumagwa, R.D.; Bugwesa, Z.; Mwita, M.; Kihwele, E.S.; Nyaki, A.; Mdegela, R.H.; Mpanduji, D.G. Cyanobacterial toxins and bacterial infections are the possible causes of mass mortality of lesser flamingos in Soda lakes in northern Tanzania. Res. Opin. Anim. Vet. Sci. 2013, 3, 1–6.
- Kock, N.D.; Kock, R.A.; Wambua, J.; Kamau, G.J.; Mohan, K. Mycobacterium avium related epizootic in free ranging Lesser Flamingos. Kenya J. Wildl. Dis. 1999, 35, 297–300.
- Ndetei, R.; Muhandiki, V.S. Mortalities of Lesser Flamingos in Kenyan Rift Valley saline lakes and the implications for sustainable management of the lakes. Lakes Reserv. Res. Manag. 2005, 10, 51–58.
- Nelson, Y.M.; Thampy, R.J.; Motellin, G.K.; Raini, J.A.; Disante, C.J.; Lion, L.W. Model for trace metal exposure in filter feeding flamingo at an alkaline rift valley lake, Kenya. Environ. Toxicol. Chem. 1998, 17, 2302–2309.
- Foss, A.J.; Miles, C.O.; Samdal, I.A.; Løvberg, K.E.; Wilkins, A.L.; Rise, F.; Jaabæk, J.A.H.; McGowan, P.C.; Aubel, M.T. Analysis of free and metabolized microcystins in samples following a bird mortality event. Harmful Algae 2018, 80, 117–129.
- Sileo, L.; Grootenhuise, J.G.; Tuite, G.H.; Hopcraft, H.D. Microbacteriosis in the Lesser Flamingo of Lake Nakuru, Kenya. J. Wildl. Dis. 1979, 15, 387–390.
- Miller, E.; Brunner, E.; Driscoll, C.; McGowan, P. Botulism.Or Is It? Wildl. Rehabil. Bull. 2013, 31, 1–12.
- WWF–LNCDP. Annual Report. World Wide Fund for Nature–Lake Nakuru Conservation and Development Project (WWF–LNCDP); WWF–LNCDP: Nakuru, Kenya, 1994.
- IUCN. Flamingo. Bulletin of the IUCN-SSC/Wetlands International. 2015. Available online: https://www.wetlands.org/wp-content/uploads/2015/11/Flamingo-Newsletter-14-2006.pdf (accessed on 1 December 2022).
- Matsunaga, H.; Harada, K.I.; Senma, M.; Ito, Y.; Yasuda, N.; Ushida, S.; Kimura, Y. Possible cause of unnatural mass death of wild birds in a pond in Nishinomiya, Japan: Sudden appearance of toxic cyanobacteria. Nat. Toxins 1999, 7, 81–84.
- Henriksen, P.; Carmichael, W.W.; An, J.; Moestrup, Ø. Detection of an anatoxin-a (s)-like anticholinesterase in natural blooms and cultures of cyanobacteria/blue-green algae from Danish lakes and in the stomach contents of poisoned birds. Toxicon 1997, 35, 901–913.
- Papadimitriou, T.; Katsiapi, M.; Vlachopoulos, K.; Christopoulos, A.; Laspidou, C.; Moustaka-Gouni, M.; Kormas, K. Cyanotoxins as the “common suspects” for the Dalmatian pelican (Pelecanus crispus) deaths in a Mediterranean reconstructed reservoir. Envirn. Pollut. 2018, 234, 779–787.
- Driscoll, C.; McGowan, P.; Miller, E.; Carmichael, W. Case Report: Great blue heron (Ardea herodias) morbidity and mortality investigation in Maryland’s Chesapeake Bay. In Proceedings of the Southeast Fish and Wildlife Conference, Baltimore, MD, USA, 24 October 2002.
- Botana, L.; James, K.; Crowley, J.; Duphard, J.; Lehane, M.; Furey, A. Anatoxin-a and Analogues: Discovery, Distribution, and Toxicology. In Phycotoxins: Chemistry and Biochemistry; Blackwell Publishing: Hoboken, NJ, USA, 2007; pp. 141–158.
- Aráoz, R.; Molgó, J.; Tandeau de Marsac, N. Neurotoxic cyanobacterial toxins. Toxicon 2010, 56, 813–828.
- Osswald, J.; Rellán, S.; Gago, A.; Vasconcelos, V. Toxicology and detection methods of the alkaloid neurotoxin produced by cyanobacteria, anatoxin-a. Environ. Int. 2007, 33, 1070–1089.
- Aas, P.; Eriksen, S.; Kolderup, J.; Lundy, P.; Haugen, J.E.; Skulberg, O.M.; Fonnum, F. Enhancement of acetylcholine release by homoanatoxin-a from Oscillatoria formosa. Environ. Toxicol. Pharmacol. 1996, 2, 223–232.
- Wood, S.A.; Selwood, A.I.; Rueckert, A.; Holland, P.T.; Milne, J.R.; Smith, K.F.; Smits, B.; Watts, L.F.; Cary, C.S. First report of homoanatoxin-a and associated dog neurotoxicosis in New Zealand. Toxicon 2007, 50, 292–301.
- Griffiths, D.J.; Saker, M.L. The Palm Island mystery disease 20 years on: A review of research on the cyanotoxin cylindrospermopsin. Environ. Toxicol. Chem. 2003, 18, 78–93.
- Chichova, M.; Tasinov, O.; Shkodrova, M.; Mishonova, M.; Sazdova, I.; Ilieva, B.; Doncheva-Stoimenova, D.; Kiselova-Kaneva, Y.; Raikova, N.; Uzunov, B.; et al. New Data on Cylindrospermopsin Toxicity. Toxins 2021, 13, 41.
- Froscio, S.; Humpage, A.; Wickramasinghe, W.; Shaw, G.; Falconer, I. Interaction of the cyanobacterial toxin cylindrospermopsin with the eukaryotic protein synthesis system. Toxicon 2008, 51, 191–198.
- Humpage, A.R.; Fenech, M.; Thomas, P.; Falconer, I.R. Micronucleus induction and chromosome loss in transformed human white cells indicate clastogenic and aneugenic action of the cyanobacterial toxin, cylindrospermopsin. Mutat. Res. 2000, 472, 155–161.
- Thomas, A.D.; Saker, M.L.; Norton, J.H.; Olsen, R.D. Cyanobacterium Cylindrospermopsis raciborskii as a probable cause of death in cattle in northern Queensland. Aust. Vet. J 1998, 76, 592–594.
- Đorđević, N.; Simić, S.; Ćirić, A. First identification of the cylindrospermopsin (cyn)-producing cyanobacterium Cylindrospermopsis raciborskii (Woloszyńska) Seenayya & Subba Raju in Serbia. Fresenius Env. Bull. 2015, 24, 3736–3742.
- Beasley, V.R. Harmful Algal Blooms (Phycotoxins). In Reference Module in Earth Systems and Environmental Sciences; Elsevier: Amsterdam, The Netherlands, 2020.
- Chen, Y.; Shen, D.; Fang, D. Nodularins in poisoning. Clin. Chim. Acta 2013, 425, 18–29.
- Zegura, B.; Straser, A.; Filipič, M. Genotoxicity and potential carcinogenicity of cyanobacterial toxins—A review. Mutat. Res. 2011, 727, 16–41.
- Flores, M.; Goodrich, D.W. Retinoblastoma Protein Paralogs and Tumor Suppression. Front. Genet. 2022, 13, 818719.
- Štern, A.; Rotter, A.; Novak, M.; Filipič, M.; Žegura, B. Genotoxic effects of the cyanobacterial pentapeptide nodularin in HepG2 cells. Food Chem. Toxicol. 2019, 124, 349–358.
- Sivonen, K. Cyanobacterial toxins and toxin production. Phycologia 1996, 190, 267–275.
- Harding, W.R.; Rowe, N.; Wessels, J.C.; Beattie, K.A.; Codd, G.A. Death of a dog attributed to the cyanobacterial (blue-green algal) hepatotoxin nodularin in South Africa. J. S. Afr. Vet. Assoc. 1995, 66, 256–259.





