Cyanobacteria, algal blooms and cyanotoxins have become common environmental enigmas in marine, freshwater and estuarine ecosystems. IThere is also compelling scientific consensus that cyanotoxins can bioaccumulate in edible aquatic organisms and affects water quality, which impacts ecosystem integrity, public health and aggravates water insecurity. Herein, this entry highlights the current knowledge on the taxonomy, bloom dynamics, toxic effects, human and ecological health implications of cyanobacteria, harmful algal blooms and cyanotoxins in the East African lakes, mainlyCommunity (EAC) lakes. Available literature shows that the major toxigenic microalgae in East African lakes belong to the Microcystis, Arthrospira, Dolichospermum, Planktolyngbya and Anabaenopsis genera. There haspeci been increased incidences of cyanobacteria have been responsible for the production of analgal blooms in eutrophic EAC lakes but there are few reports on the same in oligotrophic lakes, which indicates a gap in the current understanding of algal blooms in the region. Anatoxin-a, homoanatoxin-a, microcystins (MCs), cylindrospermopsin and nodularin have been quantified in water from below method detection limits to 81 µg L-1, with peak concentrations characteristically reported for the wet season. In edible Mifish tissues, MCs have been quantified from 2.4 to 1,479.24 μg kg-1, which can pose human health rocystinisks to a daily consumer. No episodes of human poisoning due to cyanotoxins has been reported in the EAC but MCs and anatoxin-a (up to 51.4 μg g-1) have been implicated as the proximal cause of indiscriminate fish deaths and epornitic mortality of lesser flamingos. With the unequivocal increase in climate change and variability, algal blooms and cyanotoxins will increase in frequency and severity, and this will necessitate swift action towards the mitigation of nutrient-rich pollutants loading into lakes and other water resources in the region. As some cyanobacteria encountered in EAC lakes produce other cyanotoxins (notably β-N-methylamino-L-alanine and saxitoxins), studies targeting such cyanobacterial metabolites should be initiated. Research examining the risk of hepatocellular cancer steming from ingestion of cyanobacteria and mycotoxin-contaminated water and foods, and hepatitis virus should be launched. Another potential relationship with microplastics should be assessed because they are known to accumulate toxins and amplify their toxicity
- Arthrospira fusiformis
- cylindrospermopsin
- hepatotoxicity
- lesser flamingos
- Lake Victoria
- crater lakes
1. Introduction
2. Overview of cyanobacteria, harmful algal blooms and cyanotoxins in East African lacustrine ecosystems
The East African Community (EAC), is a regional cooperation constituted by seven sovereign states as of April 2022 namely: Burundi, South Sudan, Rwanda, Democratic Republic of Congo, Tanzania, Uganda and Kenya. The region is blessed with water resources, but the water demands are largely met by eutrophic water bodies [8][9][10][13,14,15]. In thise regionEAC, MCs and anatoxin-a (ATX) are the main cyanotoxins that garnered early have been extensively recieved toxicological interest (Figure 1). For example, iIn the DRC, the oligotrophic Lake Tanganyika haswas reported to have CYB (Anabaenopsis species, Dolichospermum flosaquae, and Limnococcus limneticus) [11][12][28,29]. Another lake in DRC (Lake Kivu) is a deep oligotrophic and meromictic water resource with large volumes of exploitable methane in DRC (Lake Kivu) also has been cited to harbour . Nevertheless, CYB (Synechococcus species and Planktolyngbya limnetica) [13][14] have been indicated to be prevalent in this lake, followed by pennate diatoms (Nitzschia bacata and Fragilaria danica) [30,31].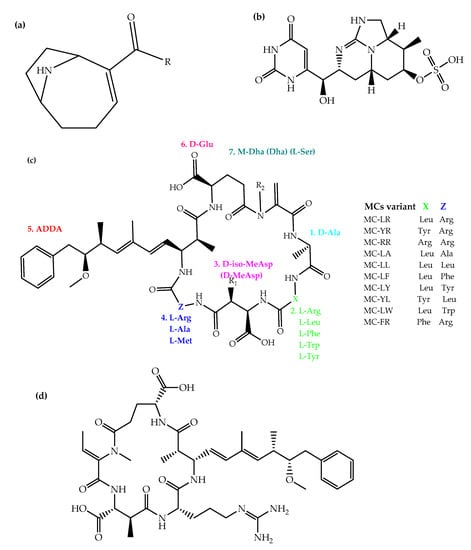
2.3. Tanzania
Pr study in Lake Naivasha confirmed the dominance of Microcystis viouspecies in a 2010 CYAHAB in the lake [20]. From a recereational perspective, a 2023 study [21] substantudiated that cyanobacterial (Microcystis as cond Dolichospermum species) cell counts in water from Winam Gulf of L. Victoria exceeded WHO standards for recreational risk in 84% of water samples. The same samples had MCs (0.02 to 23.31 ), whcih surpasses the provisional WHO drinking water guideline for MCs. In the United Republic of Tanzania, studies done on laed about lakes: Big Momela, Embagai and Manyara recorded indicated that CYB (>50%) namley:, mostly Anabaenopsis elenkenii, A. fusiformis (Lake Big Momela), A. fusiformis, Oscillatoria, Hantzschia (Lake Embagai), Oscillatoria jenensis and Pseudoanabaena terebriformis (Lake Manyara), [22][23]were dominant in the phytoplankton [50,51]. Similarly, CYB (specifically A. fusiformis ) have equally been prevalent in Momela Lakes and Lake Natron with MCs detected in water at concentrations of 0.1–4.5 μg mL−1 of scum in the latter [24][52]. In the lentic wate Fors of L. Victoria, the occurrence of CYB (upto 82%) report was quantified in samples from seeveral parts of the southern portion. A later investigation by art. Miles et al. [25][53] ifoundicated the presence of p putative MCs analogues in extracts of a bloom sampled from Tanznaia's cyanobacterial bloom from Mwanza Gulf, but no cyanotoxin but did not quantifications were doney them. On 27 islands of Ukerewe district, MCs (0.0028 to 0.0102 μg L−1) were reported [26][54]. Other studies in L. Victoria (several bays, open water and Gulfs) have found MCs (up to 13 µg MC-LR eq L−1). An incidence of multiple cyanotoxins: CYN (0.004 to 0.01 μg L−1), NODs (0.010 μg L−1) and MCs (0.0028 to 0.0118 μg L−1) in water from L. Victoria has been communicated [26][54]. The report emphasized that multiple and repeated exposure to phycotoxins could amplify their toxicity and/or adverse effects.2.4. Uganda
From the available literature, Uganda has the highest number of reports about CYB in thseve regionral lakes. Of these, Western Uganda crater lakes (Kyaninga, Saaka, Nyabikere, Nyinambuga, Munyayange, Kikorongo, Maseche, Murumuli, Bunyampaka, Katwe, Bagusa, Nyamunuka, Mwamba, Katanda, Karolero, Kerere, Kacuba, Mwengenyi, Kyerbwato, Katanda, Kanyamukali, Nkugute, Kyanga, Mirambi, Nyanswiga, Kitere, Chibwera, Lugembe, Nyanswiga, Kamweru, Nyahirya, Nyabikere, Kyasanduka, Kifuruka, Wandakara, Nyamusingire, Nyungu and Katinda) were found to contain CYB (35% to 100%), primarily of the genera Planktolyngbya, Microcystis, Anabaena and Cylindrospermopsis. No determination of cyanotoxins was performed [27][28][55,56]. Other interesting studies are available on the Albertine lakes (Edward and George) [29][57]. Species from Raphidiopsis and Anabaenopsis genera are the primary community in Lake Edward, though Aphanocapsa, Merismopedia, Microcystis, Aphanothece and Anathece genera are also present. According to several authors, shallow Ugandan lakes near Mount Rwenzori (Lake George, Lake Edward and Lake Mburo) are eutrophic, with Microcystis species being the most abundant CYB [29][30][31][32][33][57,58,59,60,61]. Lake Mburo was earlier reported to have more than 90% of its phytoplanktonic community as CYB [34][35][62,63]. In the Ugandan part of L. Victoria, Microcystis, Dolichospermum and Cylindrospermopsis species are the prevalent CYB (>80%) (Table S1). Cyanotoxin analyses have reported concentrations of NT to 93 µg L−1 of MCs in water from Murchison Bay, Napoleon gulf and open lake water. Worth citing are pioneering studies in Murchison Bay where MCs were quantified in Oreochromis niloticus (Nile tilapia fish), unveiling that the concentrations in biota and aqueous phase were correlated. The study highlighted that there has been an increase in MCs-producing CYB in the lake which are plausibly ingested by fish, agreeing with previous research findings [33][36][37][61,65,66]. The maximum concentration of total MCs reported for guts, liver and muscles of phytoplanktivorous Oreochromis niloticus (Nile tilapia) and Lates niloticus (Nile perch) from Murchison Bay of L. Victoria (1.86 to 1479.24 μg kg−1) is slightly higher than those from other Ugandan lakes such as Lake Mburo (73.10 to 1312 μg kg−1) [36][65].2.5. Rwanda
The remaining report on cyantoxins in the EAC is a single only report on CYB fromin a Rwandese Lake (Lake Muhazi). showed that it contains mainly Microcystis aeruginosa , folloccurwed along with thby the dinoflagellate Cerutium hirundinellu [38][68]. and tThis was confirmese are ingested by a follow up study which Nile tilapia present in the lake [69], subggestantitated that the CYB were being ingested Nile tilapiaing the need to establish the concentrations of cyanotoxins in water and fish from the same lake [39]is lake. Overall, volcanic and tectonic lakes in the East African Great Rift Valley possess distinguished extents of hydrological connections. Volcanicity in the region resulted in endorheic basins whose bedrock, groundwater connection and climate have favored schizohaline water formation [40][70]. These, in turn, have contributed to the dominance of CYB, and occurrence of CYBHAB and cyanotoxins. The present literature reveals that toxigenic microalgae recorded from EAC lakes are Dolichospermum, Microcystis, Arthrospira, Planktolyngbya and Anabaenopsis species. The prevalence of CYBHAB and cyanotoxins in EAC lakes is of concern due to potential bioaccumulation and trophic transfer in zooplanktivorous and carnivorous fish species [33][37][61,66]. Moreover, the observed levels of MCs in whole fish, gut, liver and muscles (2.4 to 1479.24 μg kg−1) could pose human health risks to a daily consumer, as the WHO daily intake limit of MCs in fish is 0.04 µg kg−1 [36][65].3. Toxicity, Human and Ecological Health Implications of Cyanotoxins in EAC Lakes
3.1. MCs
MCs are hepatotoxins, majorly produced as secondary metabolites of planktonic cyanobacterial species from genera such as Microcystis, Cylindrospermopsis, Anabaena, Oscillatoria (Planktothrix), Anabaenopsis, Nostoc, Arthrospira, Hapalosiphon, Limnothrix, Lyngbya, Phormidium, Rivularia, Synechocystis and Synechococcus [41][75]. Acute effects such as nausea, diarrhea, dermal, eye and throat irritations have been associated with their ingestion. Chronic exposure to MCs culminates in hepatic necrosis, retarded growth, reduced reproduction potential and, ultimately, death in fish and humans. The neurotoxicity of MCs is also known, but this applies specifically to invertebrates without livers [42][76]. In addition, exposure to MCs is associated with colorectal and primary liver cancer, with MC-LR receiving classification as a possible human carcinogen (group 2B) [43][77]. For humans, exposure to MCs occurs principally through the ingestion of contaminated aquatic organisms (e.g., fish) or water, as well as through the recreational use of water. Upon ingestion and absorption into the liver by organic anion transport proteins, MCs inhibit protein phosphatases, thereby selectively distorting cytoskeleton formation, degrading hepatic ultrastructure in eukaryotic cells, resulting in hepatic failure, intrahepatic hemorrhage and shock [44][45][78,79]. In EAC, MCs and ATX were implicated in the death of Phoeniconaias minor Geoffroy Saint-Hilaire 1798 (lesser flamingos) [46][80]. The pink birds feed on A. fusiformis [47][81], which confers upon them the pink plumage following the accumulation of ingested cyanobacterial pigments [48][82]. While this phenomenon is not new (e.g., in the Greater flamingos and Western Tanager [49][50][83,84]), it should be anticipated that other nutrition-based compounds may become bioaccumulated in lesser flamingos, e.g., potentially toxic metals. Event-driven reports of lesser flamingo die-offs are available for soda lakes such as Bogoria and Nakuru of Kenya [51][52][85,86], Momela, Natron, Rishateni, Manyara and Empakai Crater of Tanzania [8][23][53][54][13,51,87,88]. While it is still debated that MCs may be a potential initiator of avian botulism, other probable causes of the unnatural mass death of wild birds include avian tuberculosis [55][96], cholera, botulism, heavy metals [56][57][95,106], pesticide residues, or combinations of these [51][58][59][60][61][85,98,103,107,108]. Indeed, mycobacteriosis was reported in lesser flamingos from Lake Nakuru, Kenya [62][109]. Nevertheless, anatoxins and MCs (at concentrations higher than reported in EAC flamingos) have been associated with avian mortalities [58][63][64][65][66][98,101,102,110,111].3.2. Anatoxin-a
ATX is toxicologically known as Very Fast Death Factor for its fast lethal effect in animals, which could be related to its high rate of absorption into the gastrointestinal tract [67][117]. ATX is a secondary bicyclic amine alkaloid with peracute neurotoxic effects. Its discovery and identification in the 1960s and 1972 from CYB (Anabaena flos-aquae) followed the mortality of cattle herds that ingested contaminated water from Saskatchewan Lake in Ontario [67][117]. It is known to be biosynthesized by CYB from Arthrospira, Anabaena, Microcystis, Planktothrix, Oscillatoria, Aphanizomenon and Cylindrospermum genus [67][117]. Exposure to ATX (through ingestion of contaminated water or dried algal crusts, accidental swallowing/inhalation) has been associated with burning, tingling, respiratory paralysis and dysrhythmias, which are fatal. ATX antagonizes the activity of neuronal α4β2 and α4 nicotinic acetylcholine receptors (nAchRs) of the central nervous system and (α1)2βγδ muscle-type nAchRs of the neuromuscular junction [68][118]. With an affinity >20 times that of acetylcholine, ATX has the same effect as the former when it binds with nAchRs, i.e., it induces a conformational effect on the receptor, opening the channel pore to permit the passage of ions (Ca2+ and Na+) into the neuron. This culminates into cell depolarization, the generation of action potentials and thus muscle contraction. During ATX-mediated toxicity, the acetylcholine neurotransmitter does not dissociate from the nAchRs, resulting into irreversible inhibition and blockage of neuromuscular transmission [69][119].3.3. Homoanatoxin-a
HATX being structurally a higher homologue of ATX has the same toxic effects as ATX. In addition to its nicotinic agonistic effects, HATX also upregulates acetylcholine release from cholinergic nerves [70][121]. This may explain why the potency of HATX is greater than that of ATX. Mortalities from CYBHAB with HATX are rare, but a report of dog neurotoxicosis from New Zealand (where the animals ingested CYB from Hutt River, lower North Island with 4400 µg kg−1 wet weight of HATX) has been published [71][122].3.4. Cylindrospermopsin
CYN is a hydrophilic potentially hepatotoxic and immunotoxic cyclic guanidinium alkaloid, with characteristic tricyclic hydroxymethyl uracil [42][76]. It has some analogues such as deoxy-CYN (lacking an oxygen atom), demethoxy-CYN and 7-epiCYN (difference in the orientation of hydroxyl group) isolated in CYB Cylindrospermopsis raciborskii. The discovery of CYN toxicity happened when more than 100 children from Palm Island in Queensland, Australia suffered from unprecedented gastroenteritis and hepatomegaly. The ordeal was finally found to be due to the ingestion of CYN in contaminated water with CYBHAB of C. raciborskii [72][123]. However, CYN is also produced by other CYB, including Aphanizomenon flos-aquae, Anabaena species (bergii, and lapponica), Aphanizomenon ovalisporum, Lyngbya wollei, Raphidiopsis curvata Oscillatoria (Planktothrix) species and Umezakia natans [41][75]. With guideline values of 0.5 to 3 μg L−1 in drinking water across continents, CYN is the second-most-studied cyanotoxin known to target the liver, kidneys, heart, spleen, ovary, eye, lung, T lymphocytes, neutrophils and vascular endothelium [73][124]. CYN elicit toxicity through inhibition of protein synthesis, which can also occur at subtoxic concentrations [74][125]. Other toxicologists stated that CYN (with its inherent reactive guanidine) could be largely toxic through the induction of DNA wreckage and disruption of the kinetochore spindle. This could possibly result in chromosome loss, aneugenic and clastogenic effects [75][126]. Chichova et al. [73][124] found that CYN elicited moderate toxicity in human intestinal epithelial cells with suppression of cellular regeneration of the epithelial layer. CYN shows hepatotoxic, nephrotoxic, and cytotoxic effects, suggesting potential carcinogenicity. The neurotoxic potential of CYN has also been cited, though this could be a direct consequence of its cytotoxicity. To this end, the full underlying mechanisms of CYN toxicity needs to be elucidated [42][76]. In the EAC, there are no toxicity reports on CYN, which may be due to the absence of robust data on this cyanotoxin. There are, however, episodes of human and animal CYN-related poisoning from other countries. The most notable human poisoning is the 1979 Solomon dam gastroenteritis and hepatomegaly incidence in children from Palm Island [72][123]. The mortality of a cow and three calves after drinking water from McKinley Shire dam, Northern Queensland (Australia) was also reported. The animals had severe abdominal and thoracic haemorrhagic effusion, hyperaemic mesentery, pale and swollen liver, extremely distended gall bladder with dark yellow bile and epicardial haemorrhages [76][127]. In the subsequent 21 days, another eight animals (two cows and six calves) died, and analyses implicated CYN in C. raciborskii as the cause [76][127]. In Lake Aleksandrovac (Serbia), indiscriminate fish deaths due to the ingestion of CYN (range: 1.91 and 24.28 µg L−1) were reported [77][128]. This report may point to the need to establish CYN levels in EAC lakes where indiscriminate fish deaths have been reported, as CYN may be a contributing factor in addition to MCs.3.5. Nodularins
Nodularins, a class of hepatotoxic non-ribosomal cyclic pentapeptides, possess toxicity mechanisms similar to those of MCs [78][129]. They are structurally analogous to MCs, but differentiable from MCs in their amino acid components. To date, ten naturally occurring variants (isoforms) of NODs have been discovered, but nodularin-R (with Z amino acid = arginine) is the most common, most commercially available and most studied variant. The toxicity of NODs mainly targets the liver, but they also accumulate in the intestines, blood and kidneys [79][130]. Upon ingestion, NODs diffuse from the proximal and distal ileum into the liver [80][131], where they inhibit active sites of serine/threonine protein phosphatases (PP) namely: 1 (PP-1), 2A (PP-2A) and 3 (PP-3). A non-covalent interaction occurs at first with the side chain (ADDA part) and a free D-glutamyl carboxyl group in the cyclic structure of the PP, followed by the inhibition of the phosphatase activities. NODs–phosphatase complexes (NODs-PP-1 and NODs-PP-2A) are formed with exceptionally stable bonds. Thus, the key difference between NODs and MCs in their toxicity via protein phosphatases inhibition is that the former binds non-covalently to phosphatases, while the latter forms a covalent bond [79][130]. Also, NODs also elicit toxicity through formation of superoxide and hydroxyl radicals (reactive oxygen species) according to a yet incompletely elucidated pathway [79][130]. Their tumor-promoting activity is, on the other hand, mediated through the induced gene expression of TNF-alpha and proto-oncogenes, the exact mechanism of which is yet to be unraveled. In addition, the deactivation of the resultant tumor suppressor gene products (retinoblastoma and p53) progresses via phosphorylation, and this inevitably promotes tumorigenesis [81][132]. Overall, the cascade of reactions following NOD ingestion causes cellular disorganizations and damages, apoptosis, necrosis, loss of cell integrity, DNA fragmentation and strand breaks, intrahepatic bleeding and rapid blistering of hepatocytes which results in blood pooling and doubling of the liver weight [82][133]. Thus, mortalities associated with NOD poisoning is mediated through hemorrhagic shocks, which occurs in a few hours when ingested at high concentrations [83][134]. There are no toxicity events involving NODs in the EAC. Nevertheless, animal (cattle, dog, sheep, horse, pig and guinea pig) NOD-poisoning-related mortalities have been reported in other parts of the world. For example, hepatotoxicosis of a South African dog following the ingestion of NODs (0.00000347 µg kg−1 DW) was reported [84][135]. This emphasizes that more studies on this cyanotoxin is warranted in EAC aquatic ecosystems4. Conclusions
ICyan conclusion, CYBobacteria, algal blooms and cyanotoxins have increased in East African lacustrine ecosystems. Dolichospermum, Microcystis, Arthrospira, Planktolyngbya and Anabaenopsis species are the major groups of CYB prevalent in EAC lakes producing ATX, HATX, MCs, CYN and NODs. Shallow EAC lakes exhibit less seasonality in their CYB composition, with Microcystis being the CYB producing MCs under shallow and eutrophic lacustrine conditions. The only direct ecological effects of cyanotoxins in EAC lakes is indiscriminate fish deaths and mass die-offs of lesser flamingos. With the unequivocal increase in climate change and variability, it is inferred that CYBHAB and cyanotoxins will increase in frequency and severity. This calls for urgent action to mitigate nutrient-rich pollutants loading into water resources and the expansion of CYBHAB from eutrophic lakes to the surrounding marine environments. The (eco)toxicological relevance of co-production of phycotoxins should be assessed in the EAC because such exposure may amplify the toxicological outcomes in aquatic biota and humans. As some CYB encountered in EAC lakes produce other cyanotoxins (such as β-N-methylamino-L-alanine and saxitoxins), studies targeting these cyanobacterial metabolites should be initiated. While there are no reports of cyanotoxin poisoning of humans in the EAC, future studies should examine the risk of hepatocellular cancer, the ingestion of CYB and mycotoxin-contaminated water and foods, and hepatitis virus, which were earlier linked to increased primary liver cancer cases in Asia. Another potential relationship with microplastics should be assessed because they are known to accumulate toxins and amplify their toxicity.
