
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Ruchi Roy | -- | 2705 | 2023-01-17 19:07:43 | | | |
| 2 | Peter Tang | Meta information modification | 2705 | 2023-01-18 11:20:31 | | | | |
| 3 | Ruchi Roy | Meta information modification | 2705 | 2024-02-20 16:23:00 | | |
Video Upload Options
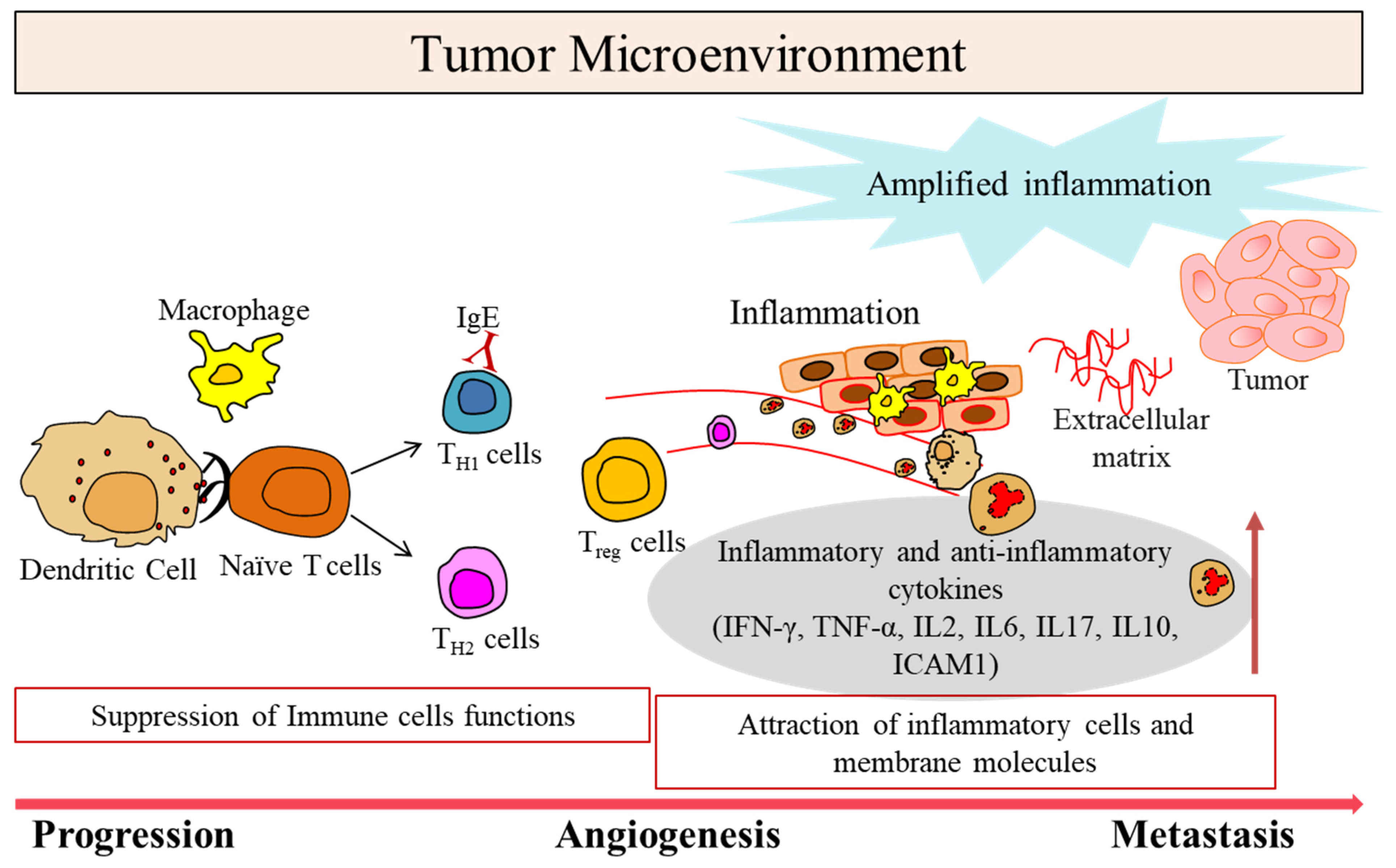
Involvement of the immune system in biological therapies specifically targeting tumor microenvironment has been suggested. Substantial advancement in the treatment of malignant tumors utilizing immune cells, most importantly T cells that play a key role in cell-mediated immunity, have led to success in clinical trials.
1. Tumor Microenvironment in Cancer
2. Immunomodulatory Roles of Lymphangiogenesis in TME
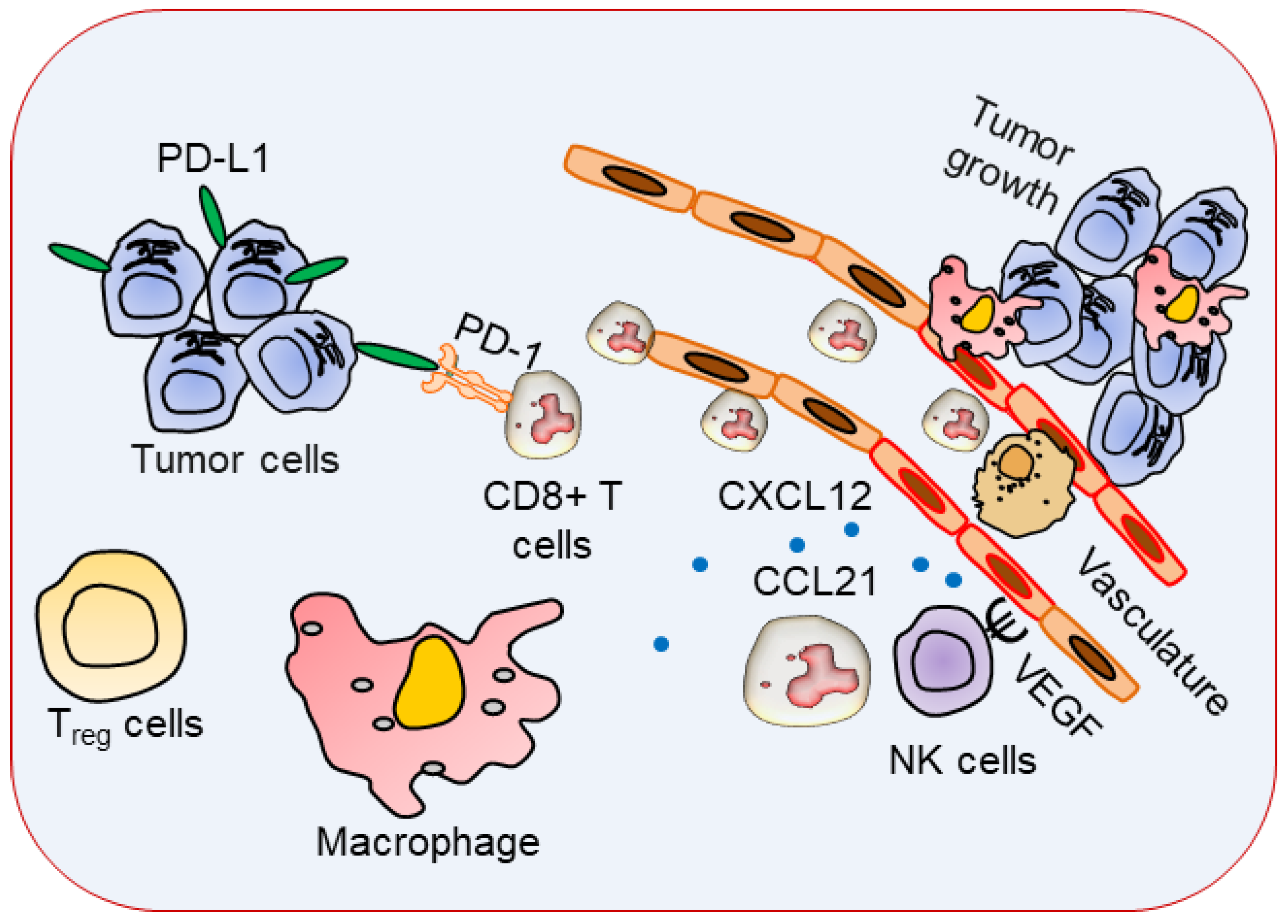
3. Immune Cells with Specific Phenotypes in TME
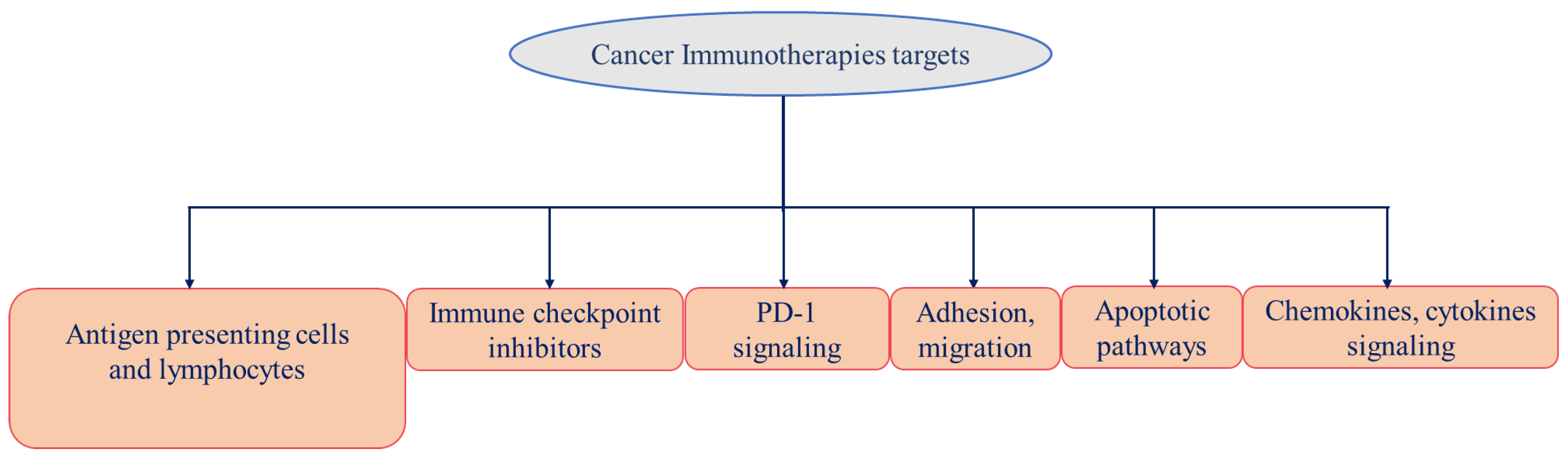
4. Cancer Immunotherapy
|
Target |
Drug INN (Brand Name) |
Cancer Type |
Current Status |
Ref. |
|---|---|---|---|---|
|
Antibodies Based |
||||
|
CTLA-4 |
Ipilimumab (Yervoy) |
Melanoma (2011) and Renal cell carcinoma (2018) |
FDA approved |
|
|
Multiple cancers |
Phase I-III |
[52] |
||
|
Tremelimumab (Imjudo) |
Antineoplastic; liver cancer |
FDA approved |
||
|
PD-1 |
Nivolumab (Opdivo) |
Melanoma (2014), NSCLC (2015), and Renal (2018) cancers, Hodgkin lymphoma |
FDA approved |
|
|
Multiple cancers |
Phase I-III |
[52] |
||
|
Pembrolizumab (Keytruda) |
Melanoma (2014), Various (2015) |
FDA approved |
[56] |
|
|
Multiple cancers |
Phase I-III |
[52] |
||
|
MED10680 |
Multiple cancers |
Phase I |
[52] |
|
|
AMP-224 |
Multiple cancers |
Phase I |
[52] |
|
|
Pidilizumab |
Multiple cancers |
Phase I-II |
[52] |
|
|
Cemiplimab (Libtayo) |
Cutaneous squamous-cell carcinoma (2018) |
FDA approved |
||
|
PD-L1 |
Atezolizumab (Tecentriq) |
Bladder, NSCLC (2016), and TNBC (2019), hepatocellular carcinoma, HCC (2020) |
FDA approved |
[52] |
|
Avelumab (Bavencio) |
Urothelial Carcinoma (2017), Merkel Cell Carcinoma (2017), Renal carcinoma (2019) |
FDA approved |
[57] |
|
|
MED14736 |
Multiple cancers |
Phase III |
[52] |
|
|
Avelumab (Bavencio) |
Merkel cell carcinoma (2017), Rena (2019), Urothelial carcinoma (2020) |
FDA approved |
[57] |
|
|
BMS-936559 |
Multiple cancers |
Phase I |
[52] |
|
|
Durvalumab (IMFINZI) |
Bladder Cancer (2017), NSCLC (2018) |
FDA approved |
||
|
LAG-3 |
IMP321 |
Multiple cancers |
Phase I |
[52] |
|
BMS-986016 |
Multiple cancers |
Phase I |
[52] |
|
|
Relatlimab (Opdualag) |
Melanoma (2022) |
FDA approved |
[53] |
|
|
VEGF |
Bevacizumab |
Colorectal (2004), NSCLC (2006, 2018), Renal (2009), Cervical (2014), Glioblastoma (2009), and Ovarian (2018) Cancers |
FDA approved |
|
|
VEGF-A, Ang-2 |
Faricimab (Vabysmo) |
wAMD, DME |
FDA approved |
[53] |
|
VEGFR2 |
Ramucirumab (Cyramza) |
Gastric cancer (2014), NSCLC (2020), HCC (2019) |
FDA approved |
|
|
EGFR |
Cetuximab |
Colorectal cancer (CRC) (2004, 2012) and Head and neck squamous cell carcinoma (2006, 2011) |
FDA approved |
|
|
Necitumumab (Portrazza) |
NSCLC (2015) |
FDA approved |
||
|
Panitumumab (Vectibix) |
Colorectal Cancer (2006) |
FDA approved |
[59] |
|
|
PDGFRα |
Olaratumab (Lartruvo) |
Soft-tissue sarcoma (2016) |
FDA approved |
[59] |
|
HER2 |
Pertuzumab (Perjeta) |
HER2-positive Breast cancer (2012) |
FDA approved |
|
|
Trastuzumab (Herceptin) |
HER2-positive Breast cancer (1998) |
FDA approved |
||
|
Ado-trastuzumab emtansine (Kadcyla) |
HER2-Breast cancer (2013) |
FDA approved |
[59] |
|
|
Fam-trastuzumab deruxtecan (Enhertu) |
HER2-positive Breast cancer (2019) |
FDA approved |
[59] |
|
|
Trastuzumab tucatinib |
HER2-positive Breast cancer (2020) |
FDA approved |
[57] |
|
|
CCR4 |
Mogamulizumab (Poteligeo) |
Cutaneous T cell lymphoma (2018) |
FDA approved |
[59] |
|
CD20 |
Obinutuzumab (Gazyva) |
Chronic lymphocytic leukemia (2013), follicular lymphoma (2017) |
FDA approved |
|
|
Ofatumumab (Arzerra) |
Chronic lymphocytic leukemia (2014) |
FDA approved |
||
|
Rituximab (MabThera, Rituxan) |
B-Cell Lymphoma (1997) |
FDA approved |
[59] |
|
|
Ibritumomab tiuxetan (Zevalin) |
NHL (2004) |
FDA approved |
||
|
tositumomab Iodine-131 (Bexxar) |
NHL (2003) |
FDA approved |
[59] |
|
|
Ublituximab |
Chronic lymphocytic leukemia, CLL, non-Hodgkin’s lymphoma) and non-cancer (multiple sclerosis) |
Phase III |
||
|
CD33 |
Gemtuzumab ozogamicin (Mylotarg) |
Acute myeloid leukemia (2000) |
FDA approved |
|
|
CD30 |
Brentuximab vedotin (Adcetris) |
Hodgkin’s lymphoma and Anaplastic large-cell lymphoma (2011) |
FDA approved |
|
|
CD79B |
Polatuzumab vedotin (Polivy) |
Diffuse large B-cell lymphoma (2019) |
FDA approved |
|
|
CD22 |
Inotuzumab ozogamicin (BESPONSA) |
Acute lymphoblastic leukemia (2017) |
FDA approved |
|
|
Moxetumomab pasudotox (Lumoxiti) |
Hairy-cell leukemia (2018) |
FDA approved |
||
|
CD19 |
Inebilizumab (Uplizna) |
Neuromyelitis optica and neuromyelitis optica spectrum disorders (2022) |
FDA approved |
[53] |
|
CD19, CD3 |
Blinatumomab (Blincyto) |
Acute lymphoblastic leukemia (2014) |
FDA approved |
|
|
TROP2 |
Sacituzumab govitecan (Trodelvy) |
TNBC (2020) |
FDA approved |
|
|
CD3 |
Muromonab-CD3 (Orthoclone Okt3) |
Reversal of kidney transplant rejection (1986) |
FDA approved |
[60] |
|
CD3, BCMA |
Teclistamab (TECVAYLI) |
Multiple myeloma (2022) |
FDA approved |
[53] |
|
gp100, CD3 |
Tebentafusp (KIMMTRAK) |
Metastatic uveal melanoma (2022) |
FDA approved |
[53] |
|
CD30, CD3 |
Mosunetuzumab (Lunsumio) |
Follicular lymphoma (2022) |
FDA Review |
[53] |
|
CD38 |
Daratumumab (Darzalex) |
Multiple Myeloma (2015) |
FDA approved |
|
|
Isatuximab (Sarclisa) |
Multiple Myeloma (2020) |
FDA approved |
||
|
GD2 |
Dinutuximab (Qarziba; Unituxin) |
Neuroblastoma (2015) |
FDA approved |
|
|
Nectin-4 |
Enfortumab Vedotin (Padcev) |
Bladder cancer (2019), Urothelial cancer (2022) |
FDA approved |
|
|
Small Drugs Based |
||||
|---|---|---|---|---|
|
Target |
Drug INN (Brand Name) |
Cancer Type |
Current Status |
Ref. |
|
EGFR |
Gefitinib |
NSCLC (2015) |
FDA approved |
|
|
Erlotinib HCl (Tarceva) |
NSCLC (2004) |
FDA approved |
[63] |
|
|
Osimertinib mesylate |
NSCLC (2020) |
FDA approved |
||
|
Dacomitinib (Vizimpro) |
EGFR-mutated NSCLC (2018) |
FDA approved |
||
|
Mobocertinib succinate (Exkivity) |
EGFR exon 20-mutated NSCLC (2021) |
FDA approved |
[63] |
|
|
HER2 |
Tucatinib (Tukysa) |
HER2-positive breast cancer (2020) |
FDA approved |
|
|
EGFR, HER2, and HER4 |
Neratinib maleate (Nerlynx) |
HER2-overexpressed breast cancer (2017) |
FDA approved |
[63] |
|
Afatinib dimaleate (Gilotrif) |
Metastatic NSCLC with EGFR exon 19 deletion or exon 21 (L858R) mutation (2013) |
FDA approved |
[63] |
|
|
PARP |
Olaparib (Lynparza) |
Advanced BRCA-mutated ovarian cancer (2020) |
FDA approved |
|
|
Rucaparib camsylate (Rubraca) |
BRCA-positive ovarian cancer (2016) |
FDA approved |
||
|
Niraparib tosylate (Zejula) |
Epithelial ovarian, fallopian tube, or primary peritoneal cancer (2017) |
FDA approved |
[63] |
|
|
PDGFRα |
Avapritinib (Ayvakit) |
metastatic gastrointestinal stromal tumor (GIST) with platelet-derived growth factor receptor alpha (PDGFRA) exon 18 mutations (2020) |
FDA approved |
|
|
Multitarget TKI (VEGFRs, PDGFRα/β, CSF1R, KIT, and FLT3) |
Sunitinib malate (Sutent) |
Imatinib-resistant GIST and advanced RCC (2013) |
FDA approved |
|
|
Multitarget TKI (RET, VEGFRs, KIT, PDGFRα/β, FGFR1/2, RAF1, BRAF, and BRAFV600E) |
Regorafenib (Stivarga) |
Metastatic colorectal cancer (2012) |
FDA approved |
|
|
Multitarget TKI (VEGFR2/3, PDGFRβ, FLT3, KIT, RAF1, and BRAF) |
Sorafenib toylate (Nexavar) |
Advanced RCC (2005) |
FDA approved |
[63] |
|
Multitarget TKI (VEGFRs, PDGFRα/β, FGFR1/2, KIT) |
Pazopanib HCl (Votrient) |
Metastatic RCC (2009) |
FDA approved |
[63] |
|
Multitarget TKI (VEGFRs, FGFRs, PDGFRα, RET, and KIT) |
Lenvatinib mesylate (Lenvima) |
Thyroid cancer (2015) |
FDA approved |
[63] |
|
Multitarget TKI (VEGFR2/3, EGFR, and RET) |
Vandetanib (Caprelsa) |
Unresectable or metastatic medullary thyroid cancer (2011) |
FDA approved |
|
|
Multitarget TKI (VEGFRs, MET, RET, FLT3, KIT, TIE2, and AXL) |
Cabozantinib S-malate (Cometriq) |
Progressive, metastatic medullary thyroid cancer (2012) |
[63] |
|
|
VEGFRs |
Axitinib (Inlyta) |
Advanced RCC (2012) |
FDA approved |
|
|
Tivozanib HCl (Fotivda) |
Advanced RCC (2021) |
FDA approved |
[63] |
|
|
mTOR |
Everolimus (Afinitor) |
Advanced RCC (2009), HER2-negative breast cancer after failure of treatment with letrozole or anastrozole (2012), nonfunctional neuroendocrine tumors of gastrointestinal or lung origin with unresectable, locally advanced, or metastatic disease (2016) |
FDA approved |
|
|
Temsirolimus (Torisel) |
Advanced RCC (2007) |
FDA approved |
[63] |
|
References
- Siegel, R.L.; Miller, K.D.; Fuchs, H.E.; Jemal, A. Cancer statistics, 2022. CA A Cancer J. Clin. 2022, 72, 7–33.
- Zeng, D.; Li, M.; Zhou, R.; Zhang, J.; Sun, H.; Shi, M.; Bin, J.; Liao, Y.; Rao, J.; Liao, W. Tumor Microenvironment Characterization in Gastric Cancer Identifies Prognostic and Immunotherapeutically Relevant Gene Signatures. Cancer Immunol. Res. 2019, 7, 737–750.
- Quail, D.F.; Joyce, J.A. Microenvironmental regulation of tumor progression and metastasis. Nat. Med. 2013, 19, 1423–1437.
- Mantovani, A.; Marchesi, F.; Malesci, A.; Laghi, L.; Allavena, P. Tumour-associated macrophages as treatment targets in oncology. Nat. Rev. Clin. Oncol. 2017, 14, 399–416.
- Kalluri, R. The biology and function of fibroblasts in cancer. Nat. Rev. Cancer 2016, 16, 582–598.
- Fridman, W.H.; Zitvogel, L.; Sautès-Fridman, C.; Kroemer, G. The immune contexture in cancer prognosis and treatment. Nat. Rev. Clin. Oncol. 2017, 14, 717–734.
- Turley, S.J.; Cremasco, V.; Astarita, J.L. Immunological hallmarks of stromal cells in the tumour microenvironment. Nat. Rev. Immunol. 2015, 15, 669–682.
- Christiansen, A.; Detmar, M. Lymphangiogenesis and cancer. Genes Cancer 2011, 2, 1146–1158.
- Fukumura, D.; Xavier, R.; Sugiura, T.; Chen, Y.; Park, E.C.; Lu, N.; Selig, M.; Nielsen, G.; Taksir, T.; Jain, R.K.; et al. Tumor induction of VEGF promoter activity in stromal cells. Cell 1998, 94, 715–725.
- Joukov, V.; Pajusola, K.; Kaipainen, A.; Chilov, D.; Lahtinen, I.; Kukk, E.; Saksela, O.; Kalkkinen, N.; Alitalo, K. A novel vascular endothelial growth factor, VEGF-C, is a ligand for the Flt4 (VEGFR-3) and KDR (VEGFR-2) receptor tyrosine kinases. Embo J. 1996, 15, 290–298.
- Orlandini, M.; Marconcini, L.; Ferruzzi, R.; Oliviero, S. Identification of a c-fos-induced gene that is related to the platelet-derived growth factor/vascular endothelial growth factor family. Proc. Natl. Acad. Sci. USA 1996, 93, 11675–11680.
- Yamada, Y.; Nezu, J.; Shimane, M.; Hirata, Y. Molecular cloning of a novel vascular endothelial growth factor, VEGF-D. Genomics 1997, 42, 483–488.
- Achen, M.G.; Jeltsch, M.; Kukk, E.; Mäkinen, T.; Vitali, A.; Wilks, A.F.; Alitalo, K.; Stacker, S.A. Vascular endothelial growth factor D (VEGF-D) is a ligand for the tyrosine kinases VEGF receptor 2 (Flk1) and VEGF receptor 3 (Flt4). Proc. Natl. Acad. Sci. USA 1998, 95, 548–553.
- Favier, B.; Alam, A.; Barron, P.; Bonnin, J.; Laboudie, P.; Fons, P.; Mandron, M.; Herault, J.P.; Neufeld, G.; Savi, P.; et al. Neuropilin-2 interacts with VEGFR-2 and VEGFR-3 and promotes human endothelial cell survival and migration. Blood 2006, 108, 1243–1250.
- Soker, S.; Miao, H.Q.; Nomi, M.; Takashima, S.; Klagsbrun, M. VEGF165 mediates formation of complexes containing VEGFR-2 and neuropilin-1 that enhance VEGF165-receptor binding. J. Cell Biochem. 2002, 85, 357–368.
- Müller, A.; Homey, B.; Soto, H.; Ge, N.; Catron, D.; Buchanan, M.E.; McClanahan, T.; Murphy, E.; Yuan, W.; Wagner, S.N.; et al. Involvement of chemokine receptors in breast cancer metastasis. Nature 2001, 410, 50–56.
- Hirakawa, S.; Detmar, M.; Kerjaschki, D.; Nagamatsu, S.; Matsuo, K.; Tanemura, A.; Kamata, N.; Higashikawa, K.; Okazaki, H.; Kameda, K.; et al. Nodal lymphangiogenesis and metastasis: Role of tumor-induced lymphatic vessel activation in extramammary Paget’s disease. Am. J. Pathol. 2009, 175, 2235–2248.
- Zhang, J.P.; Lu, W.G.; Ye, F.; Chen, H.Z.; Zhou, C.Y.; Xie, X. Study on CXCR4/SDF-1alpha axis in lymph node metastasis of cervical squamous cell carcinoma. Int. J. Gynecol. Cancer 2007, 17, 478–483.
- Kaifi, J.T.; Yekebas, E.F.; Schurr, P.; Obonyo, D.; Wachowiak, R.; Busch, P.; Heinecke, A.; Pantel, K.; Izbicki, J.R. Tumor-cell homing to lymph nodes and bone marrow and CXCR4 expression in esophageal cancer. J. Natl. Cancer Inst. 2005, 97, 1840–1847.
- Yoshitake, N.; Fukui, H.; Yamagishi, H.; Sekikawa, A.; Fujii, S.; Tomita, S.; Ichikawa, K.; Imura, J.; Hiraishi, H.; Fujimori, T. Expression of SDF-1 alpha and nuclear CXCR4 predicts lymph node metastasis in colorectal cancer. Br. J. Cancer 2008, 98, 1682–1689.
- Llosa, N.J.; Cruise, M.; Tam, A.; Wicks, E.C.; Hechenbleikner, E.M.; Taube, J.M.; Blosser, R.L.; Fan, H.; Wang, H.; Luber, B.S.; et al. The vigorous immune microenvironment of microsatellite instable colon cancer is balanced by multiple counter-inhibitory checkpoints. Cancer Discov. 2015, 5, 43–51.
- Chen, D.S.; Mellman, I. Elements of cancer immunity and the cancer-immune set point. Nature 2017, 541, 321–330.
- Joyce, J.A.; Fearon, D.T. T cell exclusion, immune privilege, and the tumor microenvironment. Science 2015, 348, 74–80.
- Herbst, R.S.; Soria, J.C.; Kowanetz, M.; Fine, G.D.; Hamid, O.; Gordon, M.S.; Sosman, J.A.; McDermott, D.F.; Powderly, J.D.; Gettinger, S.N.; et al. Predictive correlates of response to the anti-PD-L1 antibody MPDL3280A in cancer patients. Nature 2014, 515, 563–567.
- Bai, R.; Lv, Z.; Xu, D.; Cui, J. Predictive biomarkers for cancer immunotherapy with immune checkpoint inhibitors. Biomark Res. 2020, 8, 34.
- Woo, S.R.; Corrales, L.; Gajewski, T.F. Innate immune recognition of cancer. Annu. Rev. Immunol. 2015, 33, 445–474.
- Pagès, F.; Berger, A.; Camus, M.; Sanchez-Cabo, F.; Costes, A.; Molidor, R.; Mlecnik, B.; Kirilovsky, A.; Nilsson, M.; Damotte, D.; et al. Effector memory T cells, early metastasis, and survival in colorectal cancer. N. Engl. J. Med. 2005, 353, 2654–2666.
- Galon, J.; Costes, A.; Sanchez-Cabo, F.; Kirilovsky, A.; Mlecnik, B.; Lagorce-Pagès, C.; Tosolini, M.; Camus, M.; Berger, A.; Wind, P.; et al. Type, density, and location of immune cells within human colorectal tumors predict clinical outcome. Science 2006, 313, 1960–1964.
- Curiel, T.J.; Coukos, G.; Zou, L.; Alvarez, X.; Cheng, P.; Mottram, P.; Evdemon-Hogan, M.; Conejo-Garcia, J.R.; Zhang, L.; Burow, M.; et al. Specific recruitment of regulatory T cells in ovarian carcinoma fosters immune privilege and predicts reduced survival. Nat. Med. 2004, 10, 942–949.
- Gajewski, T.F.; Schreiber, H.; Fu, Y.X. Innate and adaptive immune cells in the tumor microenvironment. Nat. Immunol. 2013, 14, 1014–1022.
- Liu, R.B.; Engels, B.; Arina, A.; Schreiber, K.; Hyjek, E.; Schietinger, A.; Binder, D.C.; Butz, E.; Krausz, T.; Rowley, D.A.; et al. Densely granulated murine NK cells eradicate large solid tumors. Cancer Res. 2012, 72, 1964–1974.
- Delahaye, N.F.; Rusakiewicz, S.; Martins, I.; Ménard, C.; Roux, S.; Lyonnet, L.; Paul, P.; Sarabi, M.; Chaput, N.; Semeraro, M.; et al. Alternatively spliced NKp30 isoforms affect the prognosis of gastrointestinal stromal tumors. Nat. Med. 2011, 17, 700–707.
- Arlauckas, S.P.; Garris, C.S.; Kohler, R.H.; Kitaoka, M.; Cuccarese, M.F.; Yang, K.S.; Miller, M.A.; Carlson, J.C.; Freeman, G.J.; Anthony, R.M.; et al. In vivo imaging reveals a tumor-associated macrophage-mediated resistance pathway in anti-PD-1 therapy. Sci. Transl. Med. 2017, 9, eaal3604.
- Neubert, N.J.; Schmittnaegel, M.; Bordry, N.; Nassiri, S.; Wald, N.; Martignier, C.; Tillé, L.; Homicsko, K.; Damsky, W.; Maby-El Hajjami, H.; et al. T cell-induced CSF1 promotes melanoma resistance to PD1 blockade. Sci. Transl. Med. 2018, 10, eaan3311.
- Riaz, N.; Havel, J.J.; Makarov, V.; Desrichard, A.; Urba, W.J.; Sims, J.S.; Hodi, F.S.; Martín-Algarra, S.; Mandal, R.; Sharfman, W.H.; et al. Tumor and Microenvironment Evolution during Immunotherapy with Nivolumab. Cell 2017, 171, 934–949.e16.
- McDermott, D.F.; Huseni, M.A.; Atkins, M.B.; Motzer, R.J.; Rini, B.I.; Escudier, B.; Fong, L.; Joseph, R.W.; Pal, S.K.; Reeves, J.A.; et al. Clinical activity and molecular correlates of response to atezolizumab alone or in combination with bevacizumab versus sunitinib in renal cell carcinoma. Nat. Med. 2018, 24, 749–757.
- Crist, M.; Yaniv, B.; Palackdharry, S.; Lehn, M.A.; Medvedovic, M.; Stone, T.; Gulati, S.; Karivedu, V.; Borchers, M.; Fuhrman, B.; et al. Metformin increases natural killer cell functions in head and neck squamous cell carcinoma through CXCL1 inhibition. J. Immunother. Cancer 2022, 10, e005632.
- Wang, S.; Lin, Y.; Xiong, X.; Wang, L.; Guo, Y.; Chen, Y.; Chen, S.; Wang, G.; Lin, P.; Chen, H.; et al. Low-Dose Metformin Reprograms the Tumor Immune Microenvironment in Human Esophageal Cancer: Results of a Phase II Clinical Trial. Clin. Cancer Res. 2020, 26, 4921–4932.
- Wei, Z.; Zhang, X.; Yong, T.; Bie, N.; Zhan, G.; Li, X.; Liang, Q.; Li, J.; Yu, J.; Huang, G.; et al. Boosting anti-PD-1 therapy with metformin-loaded macrophage-derived microparticles. Nat. Commun. 2021, 12, 440.
- Liu, Y.; Zhou, Z.; Hou, J.; Xiong, W.; Kim, H.; Chen, J.; Zheng, C.; Jiang, X.; Yoon, J.; Shen, J. Tumor Selective Metabolic Reprogramming as a Prospective PD-L1 Depression Strategy to Reactivate Immunotherapy. Adv. Mater. 2022, 34, e2206121.
- Chen, J.; Zhou, Z.; Zheng, C.; Liu, Y.; Hao, R.; Ji, X.; Xi, Q.; Shen, J.; Li, Z. Chitosan oligosaccharide regulates AMPK and STAT1 pathways synergistically to mediate PD-L1 expression for cancer chemoimmunotherapy. Carbohydr. Polym. 2022, 277, 118869.
- Waldman, A.D.; Fritz, J.M.; Lenardo, M.J. A guide to cancer immunotherapy: From T cell basic science to clinical practice. Nat. Rev. Immunol. 2020, 20, 651–668.
- Fife, B.T.; Bluestone, J.A. Control of peripheral T-cell tolerance and autoimmunity via the CTLA-4 and PD-1 pathways. Immunol. Rev. 2008, 224, 166–182.
- Walker, L.S. Treg and CTLA-4: Two intertwining pathways to immune tolerance. J. Autoimmun. 2013, 45, 49–57.
- Chikuma, S.; Bluestone, J.A. Expression of CTLA-4 and FOXP3 in cis protects from lethal lymphoproliferative disease. Eur. J. Immunol. 2007, 37, 1285–1289.
- Van der Bruggen, P.; Traversari, C.; Chomez, P.; Lurquin, C.; De Plaen, E.; Van den Eynde, B.; Knuth, A.; Boon, T. A gene encoding an antigen recognized by cytolytic T lymphocytes on a human melanoma. Science 1991, 254, 1643–1647.
- Traversari, C.; van der Bruggen, P.; Luescher, I.F.; Lurquin, C.; Chomez, P.; Van Pel, A.; De Plaen, E.; Amar-Costesec, A.; Boon, T. A nonapeptide encoded by human gene MAGE-1 is recognized on HLA-A1 by cytolytic T lymphocytes directed against tumor antigen MZ2-E. J. Exp. Med. 1992, 176, 1453–1457.
- Hanna, M.G., Jr.; Peters, L.C. Immunotherapy of established micrometastases with Bacillus Calmette-Guérin tumor cell vaccine. Cancer Res. 1978, 38, 204–209.
- Guo, C.; Manjili, M.H.; Subjeck, J.R.; Sarkar, D.; Fisher, P.B.; Wang, X.Y. Therapeutic cancer vaccines: Past, present, and future. Adv. Cancer Res. 2013, 119, 421–475.
- Yang, Y. Cancer immunotherapy: Harnessing the immune system to battle cancer. J. Clin. Investig. 2015, 125, 3335–3337.
- Barbee, M.S.; Ogunniyi, A.; Horvat, T.Z.; Dang, T.O. Current status and future directions of the immune checkpoint inhibitors ipilimumab, pembrolizumab, and nivolumab in oncology. Ann. Pharm. 2015, 49, 907–937.
- Zhang, H.; Chen, J. Current status and future directions of cancer immunotherapy. J. Cancer 2018, 9, 1773–1781.
- Mullard, A. FDA approves 100th monoclonal antibody product. Nat. Rev. Drug. Discov. 2021, 20, 491–495.
- Marrone, K.A.; Brahmer, J.R. Using Immune Checkpoint Inhibitors in Lung Cancer. Oncology 2016, 30, 713–721.
- Hatae, R.; Chamoto, K. Immune checkpoint inhibitors targeting programmed cell death-1 (PD-1) in cancer therapy. Rinsho Ketsueki 2016, 57, 2224–2231.
- Swart, M.; Verbrugge, I.; Beltman, J.B. Combination Approaches with Immune-Checkpoint Blockade in Cancer Therapy. Front. Oncol. 2016, 6, 233.
- Olivier, T.; Haslam, A.; Prasad, V. Anticancer Drugs Approved by the US Food and Drug Administration From 2009 to 2020 According to Their Mechanism of Action. JAMA Netw. Open 2021, 4, e2138793.
- Kates, M.; Sopko, N.A.; Matsui, H.; Drake, C.G.; Hahn, N.M.; Bivalacqua, T.J. Immune checkpoint inhibitors: A new frontier in bladder cancer. World J. Urol. 2016, 34, 49–55.
- Zahavi, D.; Weiner, L. Monoclonal Antibodies in Cancer Therapy. Antibodies 2020, 9, 34.
- Lu, R.M.; Hwang, Y.C.; Liu, I.J.; Lee, C.C.; Tsai, H.Z.; Li, H.J.; Wu, H.C. Development of therapeutic antibodies for the treatment of diseases. J. Biomed. Sci. 2020, 27, 1.
- Chen, R.; Chen, B. Brentuximab vedotin for relapsed or refractory Hodgkin’s lymphoma. Drug. Des. Devel. 2015, 9, 1729–1733.
- Chen, R.; Hou, J.; Newman, E.; Kim, Y.; Donohue, C.; Liu, X.; Thomas, S.H.; Forman, S.J.; Kane, S.E. CD30 Downregulation, MMAE Resistance, and MDR1 Upregulation Are All Associated with Resistance to Brentuximab Vedotin. Mol. Cancer 2015, 14, 1376–1384.
- Wu, Q.; Qian, W.; Sun, X.; Jiang, S. Small-molecule inhibitors, immune checkpoint inhibitors, and more: FDA-approved novel therapeutic drugs for solid tumors from 1991 to 2021. J. Hematol. Oncol. 2022, 15, 143.





