Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Hasnae Boughaleb | -- | 1824 | 2022-11-30 10:03:54 | | | |
| 2 | Camila Xu | Meta information modification | 1824 | 2022-12-01 00:48:54 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Boughaleb, H.; Lobysheva, I.; Zotti, F.D.; Balligand, J.; Montiel, V. Vascular Sources of Nitric Oxide. Encyclopedia. Available online: https://encyclopedia.pub/entry/37324 (accessed on 10 June 2026).
Boughaleb H, Lobysheva I, Zotti FD, Balligand J, Montiel V. Vascular Sources of Nitric Oxide. Encyclopedia. Available at: https://encyclopedia.pub/entry/37324. Accessed June 10, 2026.
Boughaleb, Hasnae, Irina Lobysheva, Flavia Dei Zotti, Jean-Luc Balligand, Virginie Montiel. "Vascular Sources of Nitric Oxide" Encyclopedia, https://encyclopedia.pub/entry/37324 (accessed June 10, 2026).
Boughaleb, H., Lobysheva, I., Zotti, F.D., Balligand, J., & Montiel, V. (2022, November 30). Vascular Sources of Nitric Oxide. In Encyclopedia. https://encyclopedia.pub/entry/37324
Boughaleb, Hasnae, et al. "Vascular Sources of Nitric Oxide." Encyclopedia. Web. 30 November, 2022.
Copy Citation
Nitric oxide (NO) is implicated in numerous physiological processes, including vascular homeostasis. Reduced NO bioavailability is a hallmark of endothelial dysfunction, a prequel to many cardiovascular diseases. Biomarkers of an early NO-dependent endothelial dysfunction obtained from routine venous blood sampling would be of great interest but are currently lacking.
nitric oxide
NO-dependent endothelial function
NO bioavailability
1. Introduction
The vascular endothelium is one of the largest organs in the body and consists of an active cell monolayer producing a wide range of vascular homeostatic mediators. Nitric Oxide (NO) is the main endothelium-derived substance that maintains the vasculature in a quiescent state by inhibition of contractile tone, cellular proliferation, and thrombosis.
NO was firstly discovered by Joseph Priestley in 1772 [1] as a gas denominated “nitrous air”, but it was not until 1979 that Gruetter highlighted the vasodilator function of NO by demonstrating that precontracted bovine coronary arteries relaxed after exposure to a gas mixture of NO [2]. Few years later, Furchgott found that the ability of rabbit thoracic aorta to relax after exposure to acetylcholine was dependent on the presence of an intact vessel intima, concluding that relaxation involved endothelial cells [3] releasing an endothelium-derived relaxation factor (EDRF). Two independents studies [4][5] in 1987 finally identified NO as this EDRF involved in vascular smooth muscle relaxation through the activation of soluble guanylyl cyclase (sGC). The significance of NO-cGMP was recognized by the 1998 Nobel Prize in Physiology and Medicine awarded to Professors Furchgott, Ignarro and Murad for their discoveries on NO as a signaling molecule in the cardiovascular system [6].
Today, the most commonly accepted functional feature of endothelial dysfunction refers to abnormalities in the regulation of the vessel lumen with impaired NO bioavailability [7]. Such endothelial dysfunction is the initiating step towards the development of atherosclerosis, a major cause of mortality and morbidity accounting for up to 31% of deaths worldwide in 2015 [8]. Impairment of the endothelial function is therefore recognized as the prequel to any cardiovascular and metabolic disease associated with hypertension, atherosclerosis, diabetes, hypercholesterolemia [9].
Since NO plays a major role in the maintenance of endothelial function, early detection of its reduced bioavailability would be of great interest to prevent cardiovascular events. Nevertheless, an easy quantification of NO in circulating blood is precluded by its very short half-life (approximately 2 milliseconds) related to its chemical reactivity, itself influenced by the redox status in the vasculature. A reliable assay of the bioavailability of NO in human circulation in vivo would be of high interest both for the screening of high-risk, yet asymptomatic cardiovascular patients and for the monitoring of existing endothelial dysfunction. Such an assessment would also be of interest as a surrogate biomarker to quantitatively measure the effects of new drugs developed to treat endothelial dysfunction.
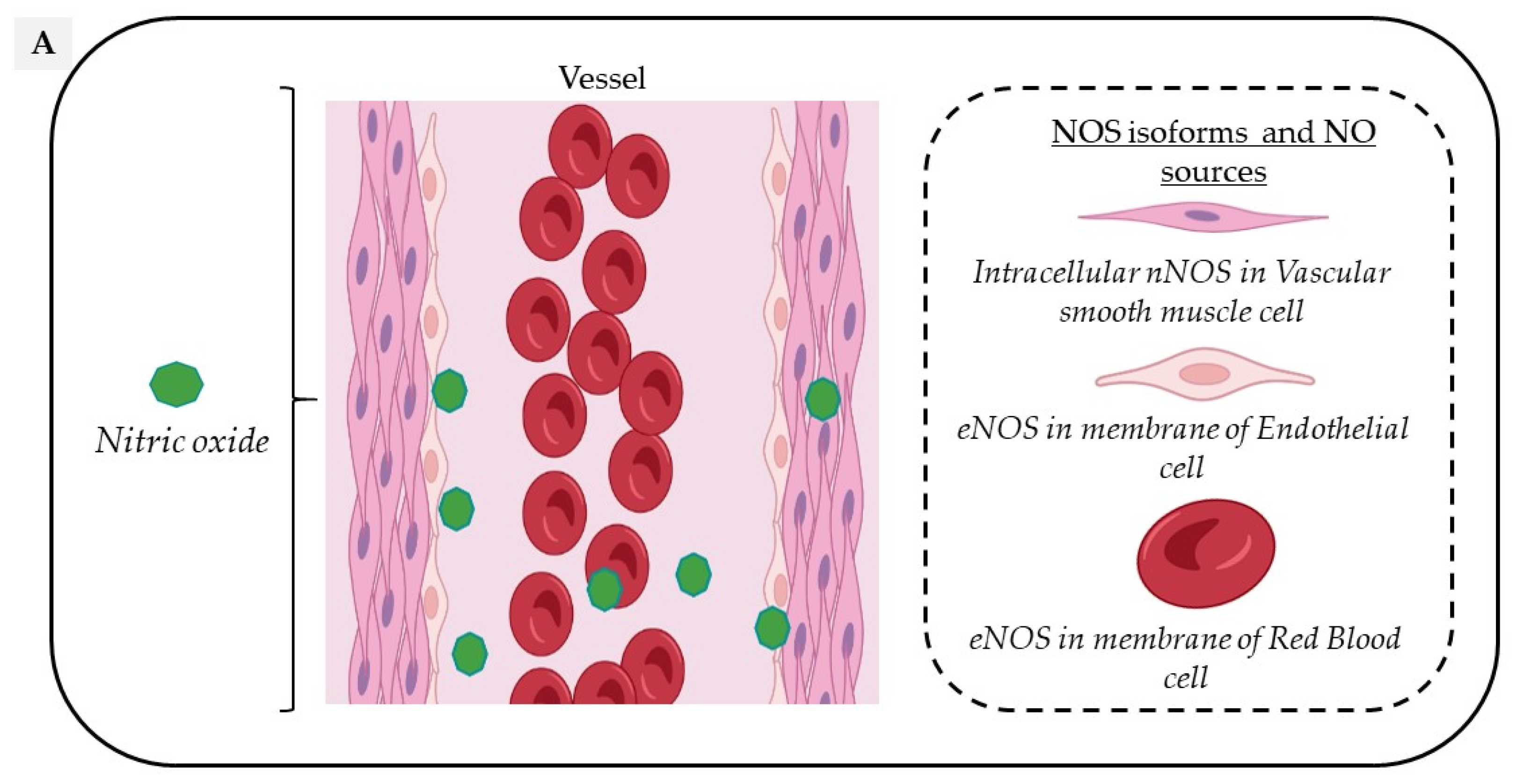
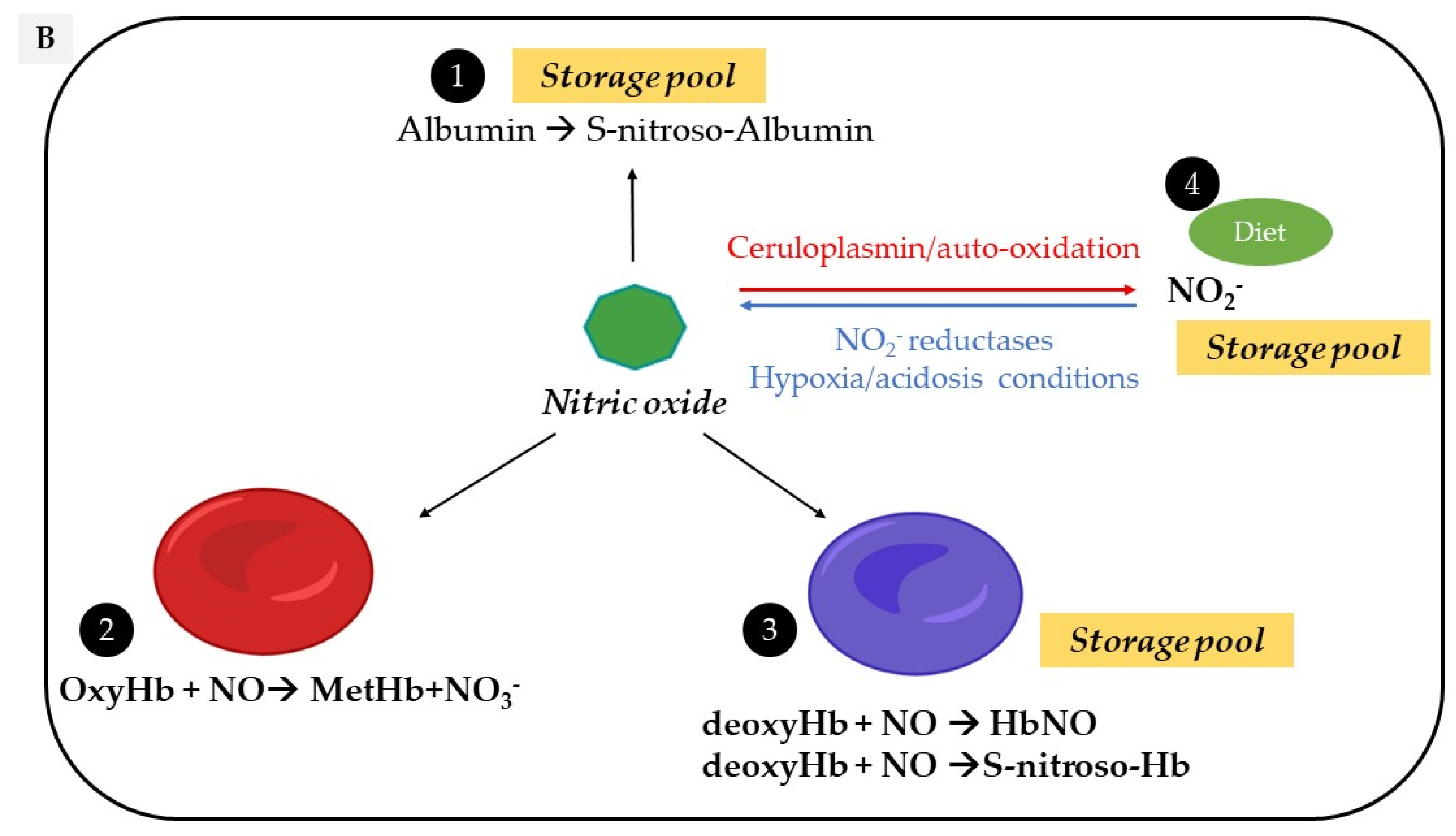
2. Vascular Sources of NO
Circulating NO is mainly regulated through the activity of endothelial nitric oxide synthase (eNOS) but also from the nitrite-dependent NO synthesis, while nitroso/nitrosyl species in biological compounds can also act as a reservoir in the bloodstream (Figure 1).


Figure 1. Vascular sources and “reservoirs” of NO. (A) NO, produced by eNOS, diffuses to VSMC and the lumen of vessels. (B) In the lumen, NO reacts with albumin to form S-nitroso-albumin (1), with OxyHb to form MetHb and nitrate in normoxic conditions (2) and with deoxy-Hb to form HbNO (3) and S-nitroso-Hb in hypoxic conditions (3). S-nitroso-albumin (1) and S-nitroso-Hb (3) serve as storage pools of NO. NO can also be oxidized into nitrite under normoxic condition (4). Combined with the dietary intake, nitrite, in turn, it serves as the storage pool and can be reduced to NO under hypoxic and acidic conditions (4). In VSMC, NO is also produced by nNOS. deoxy-Hb: deoxygenated hemoglobin; EC: endothelial cells, eNOS: endothelial Nitric Oxide synthase; HbNO: nitrosylated hemoglobin; MetHb: methemoglobin; nNOS: neuronal Nitric Oxide synthase; NO: nitric oxide; NO2−: nitrite; NO3−: nitrate; OxyHb: oxygenated hemoglobin; RBC: red blood cells; S-nitroso-Hb: S-nitroso-hemoglobin; VSMC: vascular smooth muscular cells.
2.1. Oxygen-Dependent Nitric Oxide Synthesis
In the vascular system, NO is mainly produced from the conversion of L-Arginine to L-Citrulline by the constitutive vascular endothelial NOS isoform (eNOS). However, all three NOS isoforms are potentially expressed in various cell types composing the vascular tissue and act in a coordinated manner through their appropriate cellular–subcellular localization. In blood vessels, eNOS is mainly localized in the caveolae of endothelial cells, while the neuronal NOS (nNOS) is expressed in the sarcoplasmic reticulum from vascular smooth muscle cells (VSMC); in addition, the inducible NOS (iNOS) is expressed in the cytosol of all cell types upon exposure to inflammatory cytokines such as Interleukin 1 beta (IL1-beta) and Interferon-gamma. Both eNOS and nNOS are constitutively expressed but are strongly regulated by transcriptional, post-transcriptional and post-translational mechanisms, including S-glutathionylation, S-nitrosation (which both inhibit its function) or protein–protein interactions, such as the inhibitory interaction of eNOS with caveolin-1 at the plasma membrane [10].
Both these enzymes constantly produce small amounts of NO in the vessel wall and in the lumen—particularly eNOS, which is activated by shear stress and stretch [11]. In contrast, the inducible iNOS, once expressed, produces much higher amounts of NO that is mainly involved in nitrosative reactions associated with pathological vascular remodeling [12].
The functional eNOS is organized as a homodimer and needs oxygen and cofactors such as reduced nicotinamide-adenine-dinucleotide phosphate (NADPH), Flavin adenine dinucleotide (FAD), Flavin mononucleotide (FMN) and tetrahydro-L-biopterin (BH4) to be catalytically active [13]. Full eNOS activation requires the binding of Ca2+, Calmodulin and a number of kinases in order to phosphorylate the enzyme on Ser1177, its main activation site (e.g., by protein kinase A, B, C, G, AMP-activated protein kinase and Calmodulin-Dependent Protein Kinase II) and phosphatases to dephosphorylate the negative regulatory site on Thr495 [14]. Under physiological circumstances, and as long as eNOS remains dimeric, NO signaling participates to the vascular homeostasis through a cyclic guanosine-5-monophosphate (cGMP)-dependent or via a cGMP-independent pathway [15][16].
eNOS is mainly expressed in endothelial cells, but is also found in platelets [17] and erythrocytes [18]. Endothelial eNOS-derived NO appears as the central regulator of vascular homeostasis by playing crucial roles both in the vascular wall by relaxing and inhibiting VSMC hypertrophy, promoting angiogenesis, but also by diffusion into the vessel lumen where it inhibits leukocyte adhesion, platelet aggregation and thrombosis. An additional involvement in vascular relaxation is provided by nNOS through an Angiotensin II- dependent stimulation mainly in hypertension [19].
Through a functional, calcium-dependent, eNOS-like enzyme, erythrocytes also produce, store and distribute NO in peripheral tissues with a potential role in hypoxia-induced vasodilation [20], platelet inhibition [21] and regulation of red blood cells (RBC) deformability and aggregation [22]. While some contradictory results have questioned the specific role of erythrocyte NOS [23], its involvement is supported by decreased erythrocyte-released NO metabolites (nitrite and nitrate) levels in response to NOS inhibition [24]. A specific contribution of erythrocyte eNOS to blood pressure regulation and nitrite homeostasis has gained recognition from experiments using bone marrow transplantation in chimeric mice competent or deficient for eNOS expression in circulating blood cells [25]. Erythrocyte eNOS expression and function have also emerged as independent risk factors of cardiovascular disease [26].
2.2. Nitrite-Dependent Nitric Oxide Synthesis
An alternative pathway can contribute additional NO from the inorganic anions, nitrate (NO3−) and nitrite (NO2−). Diet, particularly from vegetables, is the main provider of exogenous nitrate [27] and from cured meats (∼30%), of nitrite, to a lesser extent [28]. Nitrate is reduced into nitrite through the nitrate reductase activity of the bacteria in the oral cavity and enters the stomach where it undergoes an acid-dependent reduction into nitrous acid (HNO2) [27]. Nitrous acid then decomposes into nitrogen species, which are partially absorbed into the circulation and recycled to form NO or other bioactive nitrogen species. The reduction of nitrite to NO is enhanced by ascorbic acid and polyphenols, both reducing compounds commonly found in vegetables and fruits. After dietary intake, under normoxic conditions, the L-Arginine-NOS pathway is the main supplier of NO oxidation products from the rapid oxidation of NO by heme proteins (e.g., hemoglobin, to form nitrate with production of methemoglobin) and by cytochrome c oxidase and ceruloplasmin (to form nitrite) [29]. In addition, NO auto-oxidation can also occur [30][31]. Nitrite is considered as a reservoir of NO that can be reduced to bioactive NO through the nitrite reductase activity of a number of proteins, particularly under hypoxic and acidic conditions [28], while hemoglobin and myoglobin would act as oxygen sensors. This mechanism may therefore serve as a backup system mainly when oxygen-dependent NOS function is limited [32].
2.3. Nitroso/Nitrosyl Species
As a free radical, the NO “nitrosyl” moiety can easily react either with carbon, nitrogen, oxygen or sulfur atoms, resulting in a nitrosation reaction (where the incorporated NO is referred to as a nitroso group) [33]. NO reaction with a metal-centered protein (e.g., the catalytic site of a metalloenzyme) will induce a nitrosylation reaction (where the incorporated NO is referred to as nitrosyl group) [34]. Protein S-nitrosation (R-SNO) with the thiol group of a cysteine residue has emerged as a main posttranslational modification involved in the regulation of cell signaling proteins (including eNOS as reported above) while repeated transnitrosation reactions enable the incorporation of NO groups into other organic molecules [35]. These compounds naturally occur in vivo; stabilize NO; potentiate its biological effects and mostly act as a storage pool for NO (particularly S-nitroso-Albumin, S-nitroso-Hemoglobin and S-nitrosated proteins) [36].
2.4. Red Blood Cells
Erythrocytes have been considered as “NO sinks” due to different interactions be-tween NO and hemoglobin depending on its state. Hemoglobin exists in two distinct conformational states with less affinity for oxygen in the T-state (tense) than the R-state (relaxed). The T-state corresponds to the deoxy-form of hemoglobin and is also known as deoxyhemoglobin (deoxy-Hb) [37]. A dioxygenation reaction in which NO reacts with oxygenated hemoglobin (oxy-Hb) induces the formation of inactive methemoglobin and nitrate, which cannot be converted back to nitrite in the circulation. An interaction of NO with deoxy-Hb produces both iron-nitrosyl hemoglobin (NO bound to the heme group; HbNO) and S-nitroso hemoglobin (NO bound to the thiol group of cysteine 93 in the β-globin chain) [38][39]. These two reactions have both high association and slow dissociation rates supporting the theory of a “NO sink” in erythrocytes and hemoglobin as scavenger of NO [38][40]. However, in vivo, pressures gradients push erythrocytes into the center of the blood vessel, creating a cell-free zone that attenuates the interaction between endothelial cells, NO and hemoglobin, thereby limiting scavenging [41]. Moreover, NO consumption by erythrocytes is 1000 times slower than free hemoglobin, suggesting intrinsic factors limiting NO consumption, including extracellular diffusion limitations [42]. In addition, two pathways have been suggested to mediate the export of NO from the erythrocytes via band 3, either in a metabolon complex with deoxyhemoglobin promoting its nitrite reductase activity [43] or by band 3-specific transnitrosation from S-nitroso-hemoglobin to other NO acceptors outside the cell [44].
These propositions combined with the production of NO from the erythrocyte NOS and of a significant amount of NO derivatives (including S-nitrosothiols and S-nitroso-hemoglobin) preserving NO from binding to oxyhemoglobin argue against the NO “scavenging” in erythrocytes.
Thus, erythrocytes should rather be considered as NO transporters reflecting vascular NO contents and, in a limited fashion, as NO generators through their eNOS activity, albeit with marginal influence on erythrocyte NO content.
References
- West, J.B. Joseph Priestley, Oxygen, and the Enlightenment. Am. J. Physiol. Lung Cell. Mol. Physiol. 2014, 306, L111–L119.
- Gruetter, C.A.; Barry, B.K.; McNamara, D.B.; Gruetter, D.Y.; Kadowitz, P.J.; Ignarro, L. Relaxation of Bovine Coronary Artery and Activation of Coronary Arterial Guanylate Cyclase by Nitric Oxide, Nitroprusside and a Carcinogenic Nitrosoamine. J. Cyclic Nucleotide Res. 1979, 5, 211–224.
- Furchgott, R.F.; Zawadzki, J.V. The Obligatory Role of Endothelial Cells in the Relaxation of Arterial Smooth Muscle by Acetylcholine. Nature 1980, 288, 373–376.
- Palmer, R.M.; Ferrige, A.G.; Moncada, S. Nitric Oxide Release Accounts for the Biological Activity of Endothelium-Derived Relaxing Factor. Nature 1987, 327, 524–526.
- Ignarro, L.J.; Buga, G.M.; Wood, K.S.; Byrns, R.E.; Chaudhuri, G. Endothelium-Derived Relaxing Factor Produced and Released from Artery and Vein Is Nitric Oxide. Proc. Natl. Acad. Sci. USA 1987, 84, 9265–9269.
- SoRelle, R. Nobel Prize Awarded to Scientists for Nitric Oxide Discoveries. Circulation 1998, 98, 2365–2366.
- Hadi, H.A.R.; Carr, C.S.; Al Suwaidi, J. Endothelial Dysfunction: Cardiovascular Risk Factors, Therapy, and Outcome. Vasc. Health Risk Manag. 2005, 1, 183–198.
- Kim, H.; Kim, S.; Han, S.; Rane, P.P.; Fox, K.M.; Qian, Y.; Suh, H.S. Prevalence and Incidence of Atherosclerotic Cardiovascular Disease and Its Risk Factors in Korea: A Nationwide Population-Based Study. BMC Public Health 2019, 19, 1112.
- Vanhoutte, P.M.; Shimokawa, H.; Tang, E.H.C.; Feletou, M. Endothelial Dysfunction and Vascular Disease. Acta Physiol. 2009, 196, 193–222.
- Fleming, I.; Busse, R. Signal Transduction of ENOS Activation. Cardiovasc. Res. 1999, 43, 532–541.
- Humphrey, J.D.; Schwartz, M.A. Vascular Mechanobiology: Homeostasis, Adaptation, and Disease. Annu. Rev. Biomed. Eng. 2021, 23, 1–27.
- Farah, C.; Michel, L.Y.M.; Balligand, J.-L. Nitric Oxide Signalling in Cardiovascular Health and Disease. Nat. Rev. Cardiol. 2018, 15, 292–316.
- Forstermann, U.; Sessa, W.C. Nitric Oxide Synthases: Regulation and Function. Eur. Heart J. 2012, 33, 829–837.
- Mount, P.F.; Kemp, B.E.; Power, D.A. Regulation of Endothelial and Myocardial NO Synthesis by Multi-Site ENOS Phosphorylation. J. Mol. Cell Cardiol. 2007, 42, 271–279.
- Kraehling, J.R.; Sessa, W.C. Contemporary Approaches to Modulating the Nitric Oxide–CGMP Pathway in Cardiovascular Disease. Circ. Res. 2017, 120, 1174–1182.
- Lima, B.; Forrester, M.T.; Hess, D.T.; Stamler, J.S. S-Nitrosylation in Cardiovascular Signaling. Circ. Res. 2010, 106, 633–646.
- Wallerath, T.; Gath, I.; Aulitzky, W.E.; Pollock, J.S.; Kleinert, H.; Förstermann, U. Identification of the NO Synthase Isoforms Expressed in Human Neutrophil Granulocytes, Megakaryocytes and Platelets. Thromb. Haemost. 1997, 77, 163–167.
- Kleinbongard, P.; Schulz, R.; Rassaf, T.; Lauer, T.; Dejam, A.; Jax, T.; Kumara, I.; Gharini, P.; Kabanova, S.; Ozüyaman, B.; et al. Red Blood Cells Express a Functional Endothelial Nitric Oxide Synthase. Blood 2006, 107, 2943–2951.
- Boulanger, C.M.; Heymes, C.; Benessiano, J.; Geske, R.S.; Lévy, B.I.; Vanhoutte, P.M. Neuronal Nitric Oxide Synthase Is Expressed in Rat Vascular Smooth Muscle Cells: Activation by Angiotensin II in Hypertension. Circ. Res. 1998, 83, 1271–1278.
- Singel, D.J.; Stamler, J.S. Chemical Physiology of Blood Flow Regulation by Red Blood Cells: The Role of Nitric Oxide and S-Nitrosohemoglobin. Annu. Rev. Physiol. 2005, 67, 99–145.
- Chen, L.Y.; Mehta, J.L. Evidence for the Presence of L-Arginine-Nitric Oxide Pathway in Human Red Blood Cells: Relevance in the Effects of Red Blood Cells on Platelet Function. J. Cardiovasc. Pharmacol. 1998, 32, 57–61.
- Cortese-Krott, M.M.; Kelm, M. Endothelial Nitric Oxide Synthase in Red Blood Cells: Key to a New Erythrocrine Function? Redox Biol. 2014, 2, 251–258.
- Dei Zotti, F.; Lobysheva, I.I.; Balligand, J.-L. Nitrosyl-Hemoglobin Formation in Rodent and Human Venous Erythrocytes Reflects NO Formation from the Vasculature in Vivo. PLoS ONE 2018, 13, e0200352.
- Ulker, P.; Sati, L.; Celik-Ozenci, C.; Meiselman, H.J.; Baskurt, O.K. Mechanical Stimulation of Nitric Oxide Synthesizing Mechanisms in Erythrocytes. Biorheology 2009, 46, 121–132.
- Wood, K.C.; Cortese-Krott, M.M.; Kovacic, J.C.; Noguchi, A.; Liu, V.B.; Wang, X.; Raghavachari, N.; Boehm, M.; Kato, G.J.; Kelm, M.; et al. Circulating Blood Endothelial Nitric Oxide Synthase Contributes to the Regulation of Systemic Blood Pressure and Nitrite Homeostasis. ATVB 2013, 33, 1861–1871.
- Cortese-Krott, M.M.; Rodriguez-Mateos, A.; Sansone, R.; Kuhnle, G.G.C.; Thasian-Sivarajah, S.; Krenz, T.; Horn, P.; Krisp, C.; Wolters, D.; Heiß, C.; et al. Human Red Blood Cells at Work: Identification and Visualization of Erythrocytic ENOS Activity in Health and Disease. Blood 2012, 120, 4229–4237.
- Lundberg, J.O.; Weitzberg, E.; Gladwin, M.T. The Nitrate–Nitrite–Nitric Oxide Pathway in Physiology and Therapeutics. Nat. Rev. Drug Discov. 2008, 7, 156–167.
- Shiva, S. Nitrite: A Physiological Store of Nitric Oxide and Modulator of Mitochondrial Function. Redox Biol. 2013, 1, 40–44.
- Moncada, S.; Higgs, A. The L-Arginine-Nitric Oxide Pathway. N. Engl. J. Med. 1993, 329, 2002–2012.
- Ford, P.C.; Wink, D.A.; Stanbury, D.M. Autoxidation Kinetics of Aqueous Nitric Oxide. FEBS Lett. 1993, 326, 1–3.
- Shiva, S.; Wang, X.; Ringwood, L.A.; Xu, X.; Yuditskaya, S.; Annavajjhala, V.; Miyajima, H.; Hogg, N.; Harris, Z.L.; Gladwin, M.T. Ceruloplasmin Is a NO Oxidase and Nitrite Synthase That Determines Endocrine NO Homeostasis. Nat. Chem. Biol. 2006, 2, 486–493.
- Lundberg, J.O.; Weitzberg, E. Nitric Oxide Signaling in Health and Disease. Cell 2022, 185, 2853–2878.
- Heinrich, T.A.; da Silva, R.S.; Miranda, K.M.; Switzer, C.H.; Wink, D.A.; Fukuto, J.M. Biological Nitric Oxide Signalling: Chemistry and Terminology: NO Chemical Biology and Terminology. Br. J. Pharmacol. 2013, 169, 1417–1429.
- Maron, B.A.; Tang, S.-S.; Loscalzo, J. S-Nitrosothiols and the S-Nitrosoproteome of the Cardiovascular System. Antioxid. Redox Signal. 2013, 18, 270–287.
- Hess, D.T.; Matsumoto, A.; Kim, S.-O.; Marshall, H.E.; Stamler, J.S. Protein S-Nitrosylation: Purview and Parameters. Nat. Rev. Mol. Cell Biol. 2005, 6, 150–166.
- Belcastro, E.; Wu, W.; Fries-Raeth, I.; Corti, A.; Pompella, A.; Leroy, P.; Lartaud, I.; Gaucher, C. Oxidative Stress Enhances and Modulates Protein S-Nitrosation in Smooth Muscle Cells Exposed to S-Nitrosoglutathione. Nitric Oxide 2017, 69, 10–21.
- Ahmed, M.H.; Ghatge, M.S.; Safo, M.K. Hemoglobin: Structure, Function and Allostery. In Vertebrate and Invertebrate Respiratory Proteins, Lipoproteins and other Body Fluid Proteins; Hoeger, U., Harris, J.R., Eds.; Subcellular Biochemistry; Springer International Publishing: Cham, Switzerland, 2020; Volume 94, pp. 345–382. ISBN 978-3-030-41768-0.
- Kim-Shapiro, D.B.; Schechter, A.N.; Gladwin, M.T. Unraveling the Reactions of Nitric Oxide, Nitrite, and Hemoglobin in Physiology and Therapeutics. Arter. Thromb. Vasc. Biol. 2006, 26, 697–705.
- Gladwin, M.T.; Schechter, A.N. NO Contest: Nitrite Versus S -Nitroso-Hemoglobin. Circ. Res. 2004, 94, 851–855.
- Helms, C.C.; Liu, X.; Kim-Shapiro, D.B. Recent Insights into Nitrite Signaling Processes in Blood. Biol. Chem. 2017, 398, 319–329.
- Helms, C.C.; Gladwin, M.T.; Kim-Shapiro, D.B. Erythrocytes and Vascular Function: Oxygen and Nitric Oxide. Front. Physiol. 2018, 9, 125.
- Vaughn, M.W.; Huang, K.T.; Kuo, L.; Liao, J.C. Erythrocytes Possess an Intrinsic Barrier to Nitric Oxide Consumption. J. Biol. Chem. 2000, 275, 2342–2348.
- Gladwin, M.T.; Crawford, J.H.; Patel, R.P. The Biochemistry of Nitric Oxide, Nitrite, and Hemoglobin: Role in Blood Flow Regulation. Free Radic. Biol. Med. 2004, 36, 707–717.
- Stamler, J.S.; Jia, L.; Eu, J.P.; McMahon, T.J.; Demchenko, I.T.; Bonaventura, J.; Gernert, K.; Piantadosi, C.A. Blood Flow Regulation by S-Nitrosohemoglobin in the Physiological Oxygen Gradient. Science 1997, 276, 2034–2037.
More
Information
Subjects:
Biology
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.8K
Entry Collection:
Hypertension and Cardiovascular Diseases
Revisions:
2 times
(View History)
Update Date:
01 Dec 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





