
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Daryoush Shahbazi-Gahrouei | -- | 2864 | 2022-11-28 13:22:09 | | | |
| 2 | Lindsay Dong | + 6 word(s) | 2870 | 2022-11-29 11:21:46 | | |
Video Upload Options
Breast cancer is the most prevalent cancer among women and the leading cause of death. Diffusion-weighted imaging (DWI) and diffusion tensor imaging (DTI) are advanced magnetic resonance imaging (MRI) procedures that are widely used in the diagnostic and treatment evaluation of breast cancer. Because of the propensity of malignancies to limit diffusion, DWI can improve MRI diagnostic specificity. Diffusion tensor imaging gives additional information about diffusion directionality and anisotropy over traditional DWI. Recent findings showed that DWI and DTI and their characteristics may facilitate earlier and more accurate diagnosis, followed by better treatment. Overall, with the development of instruments and novel MRI modalities, it may be possible to diagnose breast cancer more effectively in the early stages.
1. Introduction
2. DWI in the Field of Diagnostic Breast Cancer
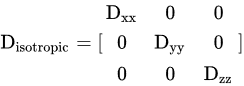
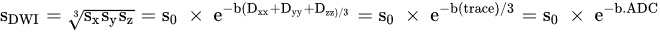
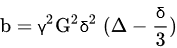
3. Comparison of DWI with Other Modalities
4. Different Models in DWI
5. DWI in Treatment Evaluation of Breast Cancer
6. DTI in the Diagnosis of Breast Cancer
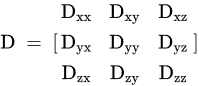
7. DTI in Treatment Evaluation of Breast Cancer
8. Amide Proton Transfer-Weighted Imaging in Breast Cancer Diagnosis
9. Diffusion Kurtosis Imaging in Breast Cancer Diagnosis
10. Conclusions
References
- Khaniabadi, P.M.; Shahbazi-Gahrouei, D.; Aziz, A.A.; Dheyab, M.A.; Khaniabadi, B.M.; Mehrdel, B.; Jameel, M.S. Trastuzumab conjugated porphyrin-superparamagnetic iron oxide nanoparticle: A potential PTT-MRI bimodal agent for herceptin positive breast cancer. Photodiagnosis Photodyn. Ther. 2020, 31, 101896.
- Shahbazi-Gahrouei, D.; Khaniabadi, P.M.; Khaniabadi, B.M.; Shahbazi-Gahrouei, S. Medical imaging modalities using nanoprobes for cancer diagnosis: A literature review on recent findings. J. Res. Med. Sci. Off. J. Isfahan Univ. Med. Sci. 2019, 24, 38.
- Pereira, F.P.A.; Martins, G.; de Oliveira, R.d.V.C. Diffusion magnetic resonance imaging of the breast. Magn. Reson. Imaging Clin. 2011, 19, 95–110.
- Tsougos, I.; Bakosis, M.; Tsivaka, D.; Athanassiou, E.; Fezoulidis, I.; Arvanitis, D.; Vassiou, K. Diagnostic performance of quantitative diffusion tensor imaging for the differentiation of breast lesions at 3 T MRI. Clin. Imaging 2019, 53, 25–31.
- Yabuuchi, H.; Matsuo, Y.; Sunami, S.; Kamitani, T.; Kawanami, S.; Setoguchi, T.; Sakai, S.; Hatakenaka, M.; Kubo, M.; Tokunaga, E. Detection of non-palpable breast cancer in asymptomatic women by using unenhanced diffusion-weighted and T2-weighted MR imaging: Comparison with mammography and dynamic contrast-enhanced MR imaging. Eur. Radiol. 2011, 21, 11–17.
- Shahbazi-Gahrouei, D.; Khaniabadi, P.M.; Shahbazi-Gahrouei, S.; Khorasani, A.; Mahmoudi, F. A literature review on multimodality molecular imaging nanoprobes for cancer detection. Pol. J. Med. Phys. Eng. 2019, 25, 57–68.
- Partridge, S.C.; Nissan, N.; Rahbar, H.; Kitsch, A.E.; Sigmund, E.E. Diffusion-weighted breast MRI: Clinical applications and emerging techniques. J. Magn. Reson. Imaging 2017, 45, 337–355.
- Baliyan, V.; Das, C.J.; Sharma, R.; Gupta, A.K. Diffusion weighted imaging: Technique and applications. World J. Radiol. 2016, 8, 785.
- Malayeri, A.A.; El Khouli, R.H.; Zaheer, A.; Jacobs, M.A.; Corona-Villalobos, C.P.; Kamel, I.R.; Macura, K.J. Principles and applications of diffusion-weighted imaging in cancer detection, staging, and treatment follow-up. Radiographics 2011, 31, 1773–1791.
- Fohlin, H.; Bekkhus, T.; Sandström, J.; Fornander, T.; Nordenskjöld, B.; Carstensen, J.; Stål, O. RAB6C is an independent prognostic factor of estrogen receptor-positive/progesterone receptor-negative breast cancer. Oncol. Lett. 2020, 19, 52–60.
- Rajagopalan, V.; Jiang, Z.; Yue, G.H.; Radic, J.S.; Pioro, E.P.; Wylie, G.R.; Das, A. A Basic Introduction to Diffusion Tensor Imaging Mathematics and Image Processing Steps. Brain Disord 2017, 6, 2.
- Wang, Y.; Zhang, X.-P.; Li, Y.-L.; Li, X.-T.; Hu, Y.; Cui, Y.; Sun, Y.-S.; Zhang, X.-Y. Optimization of the parameters for diffusion tensor magnetic resonance imaging data acquisition for breast fiber tractography at 1.5 T. Clin. Breast Cancer 2014, 14, 61–67.
- Koay, C.G.; Chang, L.-C.; Carew, J.D.; Pierpaoli, C.; Basser, P.J. A unifying theoretical and algorithmic framework for least squares methods of estimation in diffusion tensor imaging. J. Magn. Reson. 2006, 182, 115–125.
- Le Bihan, D.; Poupon, C.; Amadon, A.; Lethimonnier, F. Artifacts and pitfalls in diffusion MRI. J. Magn. Reson. Imaging Off. J. Int. Soc. Magn. Reson. Med. 2006, 24, 478–488.
- Sinha, S.; Lucas-Quesada, F.A.; Sinha, U.; DeBruhl, N.; Bassett, L.W. In vivo diffusion-weighted MRI of the breast: Potential for lesion characterization. J. Magn. Reson. Imaging: Off. J. Int. Soc. Magn. Reson. Med. 2002, 15, 693–704.
- Iima, M.; Kataoka, M.; Kanao, S.; Onishi, N.; Kawai, M.; Ohashi, A.; Sakaguchi, R.; Toi, M.; Togashi, K. Intravoxel incoherent motion and quantitative non-Gaussian diffusion MR imaging: Evaluation of the diagnostic and prognostic value of several markers of malignant and benign breast lesions. Radiology 2018, 287, 432–441.
- Geijer, B.; Sundgren, P.; Lindgren, A.; Brockstedt, S.; Ståhlberg, F.; Holtås, S. The value of b required to avoid T2 shine-through from old lacunar infarcts in diffusion-weighted imaging. Neuroradiology 2001, 43, 511–517.
- Dorrius, M.D.; Dijkstra, H.; Oudkerk, M.; Sijens, P.E. Effect of b value and pre-admission of contrast on diagnostic accuracy of 1.5-T breast DWI: A systematic review and meta-analysis. Eur. Radiol. 2014, 24, 2835–2847.
- Ohlmeyer, S.; Laun, F.B.; Bickelhaupt, S.; Palm, T.; Janka, R.; Weiland, E.; Uder, M.; Wenkel, E. Ultra-High b-Value Diffusion-Weighted Imaging-Based Abbreviated Protocols for Breast Cancer Detection. Investig. Radiol. 2021, 56, 629–636.
- Amornsiripanitch, N.; Bickelhaupt, S.; Shin, H.J.; Dang, M.; Rahbar, H.; Pinker, K.; Partridge, S.C. Diffusion-weighted MRI for unenhanced breast cancer screening. Radiology 2019, 293, 504–520.
- Baltzer, P.; Mann, R.M.; Iima, M.; Sigmund, E.E.; Clauser, P.; Gilbert, F.J.; Martincich, L.; Partridge, S.C.; Patterson, A.; Pinker, K. Diffusion-weighted imaging of the breast—a consensus and mission statement from the EUSOBI International Breast Diffusion-Weighted Imaging working group. Eur. Radiol. 2020, 30, 1436–1450.
- Iima, M.; Partridge, S.C.; Le Bihan, D. Six DWI questions you always wanted to know but were afraid to ask: Clinical relevance for breast diffusion MRI. Eur. Radiol. 2020, 30, 2561–2570.
- Huang, S.Y.; Seethamraju, R.T.; Patel, P.; Hahn, P.F.; Kirsch, J.E.; Guimaraes, A.R. Body MR imaging: Artifacts, k-Space, and solutions. Radiographics 2015, 35, 1439–1460.
- Baxter, G.C.; Patterson, A.J.; Woitek, R.; Allajbeu, I.; Graves, M.J.; Gilbert, F. Improving the image quality of DWI in breast cancer: Comparison of multi-shot DWI using multiplexed sensitivity encoding to conventional single-shot echo-planar imaging DWI. Br. J. Radiol. 2020, 93, 20200427.
- Park, J.Y.; Shin, H.J.; Shin, K.C.; Sung, Y.S.; Choi, W.J.; Chae, E.Y.; Cha, J.H.; Kim, H.H. Comparison of readout segmented echo planar imaging (EPI) and EPI with reduced field-of-view diffusion-weighted imaging at 3T in patients with breast cancer. J. Magn. Reson. Imaging 2015, 42, 1679–1688.
- Kuhl, C.K.; Schrading, S.; Leutner, C.C.; Morakkabati-Spitz, N.; Wardelmann, E.; Fimmers, R.; Kuhn, W.; Schild, H.H. Mammography, breast ultrasound, and magnetic resonance imaging for surveillance of women at high familial risk for breast cancer. J. Clin. Oncol. 2005, 23, 8469–8476.
- Lord, S.; Lei, W.; Craft, P.; Cawson, J.; Morris, I.; Walleser, S.; Griffiths, A.; Parker, S.; Houssami, N. A systematic review of the effectiveness of magnetic resonance imaging (MRI) as an addition to mammography and ultrasound in screening young women at high risk of breast cancer. Eur. J. Cancer 2007, 43, 1905–1917.
- Bonelli, L.A.; Calabrese, M.; Belli, P.; Corcione, S.; Losio, C.; Montemezzi, S.; Pediconi, F.; Petrillo, A.; Zuiani, C.; Camera, L.; et al. MRI versus Mammography plus Ultrasound in Women at Intermediate Breast Cancer Risk: Study Design and Protocol of the MRIB Multicenter, Randomized, Controlled Trial. Diagnostics 2021, 11, 1635.
- Gelardi, F.; Ragaini, E.M.; Sollini, M.; Bernardi, D.; Chiti, A. Contrast-Enhanced Mammography versus Breast Magnetic Resonance Imaging: A Systematic Review and Meta-Analysis. Diagnostics 2022, 12, 1890.
- Zhang, H.; Zhang, X.Y.; Wang, Y. Value of magnetic resonance diffusion combined with perfusion imaging techniques for diagnosing potentially malignant breast lesions. World J. Clin. Cases 2022, 10, 6021–6031.
- Lehman, C.D.; Isaacs, C.; Schnall, M.D.; Pisano, E.D.; Ascher, S.M.; Weatherall, P.T.; Bluemke, D.A.; Bowen, D.J.; Marcom, P.K.; Armstrong, D.K. Cancer yield of mammography, MR, and US in high-risk women: Prospective multi-institution breast cancer screening study. Radiology 2007, 244, 381–388.
- Woodhams, R.; Matsunaga, K.; Iwabuchi, K.; Kan, S.; Hata, H.; Kuranami, M.; Watanabe, M.; Hayakawa, K. Diffusion-weighted imaging of malignant breast tumors: The usefulness of apparent diffusion coefficient (ADC) value and ADC map for the detection of malignant breast tumors and evaluation of cancer extension. J. Comput. Assist. Tomogr. 2005, 29, 644–649.
- Yabuuchi, H.; Matsuo, Y.; Okafuji, T.; Kamitani, T.; Soeda, H.; Setoguchi, T.; Sakai, S.; Hatakenaka, M.; Kubo, M.; Sadanaga, N. Enhanced mass on contrast-enhanced breast MR imaging: Lesion characterization using combination of dynamic contrast-enhanced and diffusion-weighted MR images. J. Magn. Reson. Imaging Off. J. Int. Soc. Magn. Reson. Med. 2008, 28, 1157–1165.
- Zhang, M.; Horvat, J.V.; Bernard-Davila, B.; Marino, M.A.; Leithner, D.; Ochoa-Albiztegui, R.E.; Helbich, T.H.; Morris, E.A.; Thakur, S.; Pinker, K. Multiparametric MRI model with dynamic contrast-enhanced and diffusion-weighted imaging enables breast cancer diagnosis with high accuracy. J. Magn. Reson. Imaging 2019, 49, 864–874.
- Vidić, I.; Egnell, L.; Jerome, N.P.; White, N.S.; Karunamuni, R.; Rakow-Penner, R.; Dale, A.M.; Bathen, T.F.; Goa, P.E. Modeling the diffusion-weighted imaging signal for breast lesions in the b = 200 to 3000 s/mm2 range: Quality of fit and classification accuracy for different representations. Magn. Reson. Med. 2020, 84, 1011–1023.
- Camps-Herrero, J. Diffusion-weighted imaging of the breast: Current status as an imaging biomarker and future role. BJR| Open 2019, 1, 20180049.
- Patterson, D.M.; Padhani, A.R.; Collins, D.J. Technology insight: Water diffusion MRI—A potential new biomarker of response to cancer therapy. Nat. Clin. Pract. Oncol. 2008, 5, 220–233.
- Noij, D.P.; Pouwels, P.J.; Ljumanovic, R.; Knol, D.L.; Doornaert, P.; de Bree, R.; Castelijns, J.A.; de Graaf, P. Predictive value of diffusion-weighted imaging without and with including contrast-enhanced magnetic resonance imaging in image analysis of head and neck squamous cell carcinoma. Eur. J. Radiol. 2015, 84, 108–116.
- Liu, H.; Zhan, H.; Zhang, Y.; He, G.; Wang, H.; Zhang, Q.; Zheng, L. Comparison of BSGI and MRI as Approaches to Evaluating Residual Tumor Status after Neoadjuvant Chemotherapy in Chinese Women with Breast Cancer. Diagnostics 2021, 11, 1846.
- Lee, J.; Kang, B.J.; Park, G.E.; Kim, S.H. The Usefulness of Magnetic Resonance Imaging (MRI) for the Detection of Local Recurrence after Mastectomy with Reconstructive Surgery in Breast Cancer Patients. Diagnostics 2022, 12, 2203.
- Turan, U.; Aygun, M.; Duman, B.B.; Kelle, A.P.; Cavus, Y.; Tas, Z.A.; Dirim, A.B.; Irkorucu, O. Efficacy of US, MRI, and F-18 FDG-PET/CT for Detecting Axillary Lymph Node Metastasis after Neoadjuvant Chemotherapy in Breast Cancer Patients. Diagnostics 2021, 11, 2361.
- Choi, J.H.; Lim, H.I.; Lee, S.K.; Kim, W.W.; Kim, S.M.; Cho, E.; Ko, E.Y.; Han, B.K.; Park, Y.H.; Ahn, J.S. The role of PET CT to evaluate the response to neoadjuvant chemotherapy in advanced breast cancer: Comparison with ultrasonography and magnetic resonance imaging. J. Surg. Oncol. 2010, 102, 392–397.
- Lai, H.-W.; Chen, C.-J.; Lin, Y.-J.; Chen, S.-L.; Wu, H.-K.; Wu, Y.-T.; Kuo, S.-J.; Chen, S.-T.; Chen, D.-R. Does Breast Magnetic Resonance Imaging Combined with Conventional Imaging Modalities Decrease the Rates of Surgical Margin Involvement and Reoperation?: A Case–Control Comparative Analysis. Medicine 2016, 95, e3810.
- Furman-Haran, E.; Nissan, N.; Ricart-Selma, V.; Martinez-Rubio, C.; Degani, H.; Camps-Herrero, J. Quantitative evaluation of breast cancer response to neoadjuvant chemotherapy by diffusion tensor imaging: Initial results. J. Magn. Reson. Imaging 2018, 47, 1080–1090.
- Mao, C.; Jiang, W.; Huang, J.; Wang, M.; Yan, X.; Yang, Z.; Wang, D.; Zhang, X.; Shen, J. Quantitative Parameters of Diffusion Spectrum Imaging: HER2 Status Prediction in Patients with Breast Cancer. Front. Oncol. 2022, 12, 817070.
- Cho, E.; Baek, H.J.; Szczepankiewicz, F.; An, H.J.; Jung, E.J.; Lee, H.J.; Lee, J.; Gho, S.M. Clinical experience of tensor-valued diffusion encoding for microstructure imaging by diffusional variance decomposition in patients with breast cancer. Quant. Imaging Med. Surg. 2022, 12, 2002–2017.
- Zhou, J.; Payen, J.-F.; Wilson, D.A.; Traystman, R.J.; van Zijl, P. Using the amide proton signals of intracellular proteins and peptides to detect pH effects in MRI. Nat. Med. 2003, 9, 1085–1090.
- Zaric, O.; Farr, A.; Rodriguez, E.P.; Mlynarik, V.; Bogner, W.; Gruber, S.; Asseryanis, E.; Singer, C.F.; Trattnig, S. 7T CEST MRI: A potential imaging tool for the assessment of tumor grade and cell proliferation in breast cancer. Magn. Reson. Imaging 2019, 59, 77–87.
- Crescenzi, R.; Donahue, P.M.; Mahany, H.; Lants, S.K.; Donahue, M.J. CEST MRI quantification procedures for breast cancer treatment-related lymphedema therapy evaluation. Magn. Reson. Med. 2020, 83, 1760–1773.
- Someya, Y.; Iima, M.; Imai, H.; Yoshizawa, A.; Kataoka, M.; Isoda, H.; Le Bihan, D.; Nakamoto, Y. Investigation of breast cancer microstructure and microvasculature from time-dependent DWI and CEST in correlation with histological biomarkers. Sci. Rep. 2022, 12, 6523.
- Meng, N.; Wang, X.; Sun, J.; Han, D.; Bai, Y.; Wei, W.; Wang, Z.; Jia, F.; Wang, K.; Wang, M. A comparative study of the value of amide proton transfer-weighted imaging and diffusion kurtosis imaging in the diagnosis and evaluation of breast cancer. Eur. Radiol. 2021, 31, 1707–1717.





