
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Sardar Rehman | -- | 2820 | 2022-11-22 12:51:53 | | | |
| 2 | Lindsay Dong | -3 word(s) | 2817 | 2022-11-23 01:46:53 | | | | |
| 3 | Lindsay Dong | Meta information modification | 2817 | 2022-11-25 09:23:28 | | |
Video Upload Options
Cracking is one of the main ways that concrete ages, allowing pollutants to seep within and potentially lowering the physical and mechanical strength and endurance of concrete structures. One of the healing procedures that merits research is the use of bacterially generated calcium carbonate precipitation in concrete mixtures to mend concrete cracks.
1. Introduction
2. Factor Affecting the MICP
2.1. Nucleation Site
2.2. Bacterial Type
2.3. Bacterial Concentrations and Ureolytic Activity
2.4. PH
2.5. Nutrients
2.6. Temperature
3. Tests for Assessing the Bio-Mineralized Calcium Carbonate Based SELF-Healing Concrete
Several methods for evaluating the crack self-healing effect were put forth in order to evaluate the potential of microbial self-healing concrete (MSHC) to close cracks. For instance, X-ray diffraction, scanning electron microscopy, and energy dispersive spectroscopy (XRD) [68][69] were employed to examine the shape of crystals that precipitated at the break. The ability of the bio-mineralized self-healing precipitate was further evaluated using water permeability, porosity, and chloride ion permeation resistance [70][71] and nondestructive detection methods including ultrasonic and acoustic emission were used to assess the effectiveness of bio-self-healing. concrete [72][73][74][75]. TEM analysis was used to assess the sealing ability or deposition of MICP by [76] on archeological gypsum plasters employed M. xanthus bacteria. However, characterization techniques like SEM, XRD, and water permeability are only suitable for testing in the lab and not real engineering since they cannot concurrently satisfy the demands of operability and nondestructive testing. Acoustic emission and ultrasonic technology are expected to be heavily used in practical engineering due to the advantages of simple detecting equipment, practical operation, and economical detection. The majority of MSHC research is based on previously published experiments.
However, several scientists have chosen to use measurements of the deposited minerals to confirm the encapsulation of bacteria in self-healing concrete [44][77][78][79][80]. In the realm of encapsulating materials in self-healing concrete, techniques of testing based on the restoration of mechanical characteristics by the transmission of ultrasonic waves are still unexamined. Quantification of precipitated crystals in cementitious and biochar materials has been investigated using TGA for concrete or mortar samples.
Microstructure tests are carried out using tools including scanning electron microscopes (SEMs), field-emission scanning electron microscopes (FESEMs), and X-ray diffraction to identify and describe embedded materials after self-healing (XRD) [81] used bio-cement with a variable urea-CaCl2 and bacterial cell density concentration. Based on its 40% greater compressive strength than conventional concrete, improved material finish, aesthetic qualities, and environmental effect, the authors came to the conclusion that bio-cement has potential as a sustainable design material.
ATRIR [82][83] revealed the chemical composition of the deposit, which consisted of a calcite and aragonite combination in addition to two CaCO3 polymorphs. For more confirmation of the characterization results, the XRD is frequently employed, along with SEM-EDX by various researchers. This method was utilized to search for verified healing agents in the precipitates based on macrostructural and SEM inspection. The most prevalent microstructural test used by investigators to trace deposition products in crack specimens was SEM analysis [84]. In addition, for qualitative and quantitative elemental analysis, several researchers have combined EDS with SEM [85][86][87][88]. X-ray tomography, Raman spectroscopy, and Nuclear Magnet Resonance (NMR) can be utilized to monitor and study crack healing qualitatively and quantitatively besides the existing microstructure level research [89][90]. The nanotechnology level study of self-restoration effectiveness by bacteria encased materials has yet to be completed. These tests are performed to ensure that the results of the microstructural testing are as reliable as possible. Nanoscale tests should be performed to determine the bonding strength within the fractures at the interface between the deposited minerals and the cement substrate [91].
4. Sealing Ability and Recovery of Mechanical and Durability Properties
4.1. Recovery of Mechanical Properties
Wang [92] used glass tubes to encapsulate bacteria cells in PU and silica gel. The crack width used to measure mechanical strength recovery was around 0.35 mm. In the case of silica gel, there was more calcium carbonate precipitation, but only around 5% strength recovery. PU-containing specimens recovered between 50 and 80 percent of their potency. Conversely, the bacterial role in regaining mechanical strength was questioned because the strength recovery for live and dead bacteria cells was not significantly different. The precipitate quantity was larger in silica gel than in PU, although PU had a higher strength recovery. It’s reasonable to assume that PU, as an excellent sealing agent, played a major role in the mechanical strength recovery.
4.2. Recovery of Durability Properties
5. Field Application of Bio-Mineralized Self-Healing Concrete
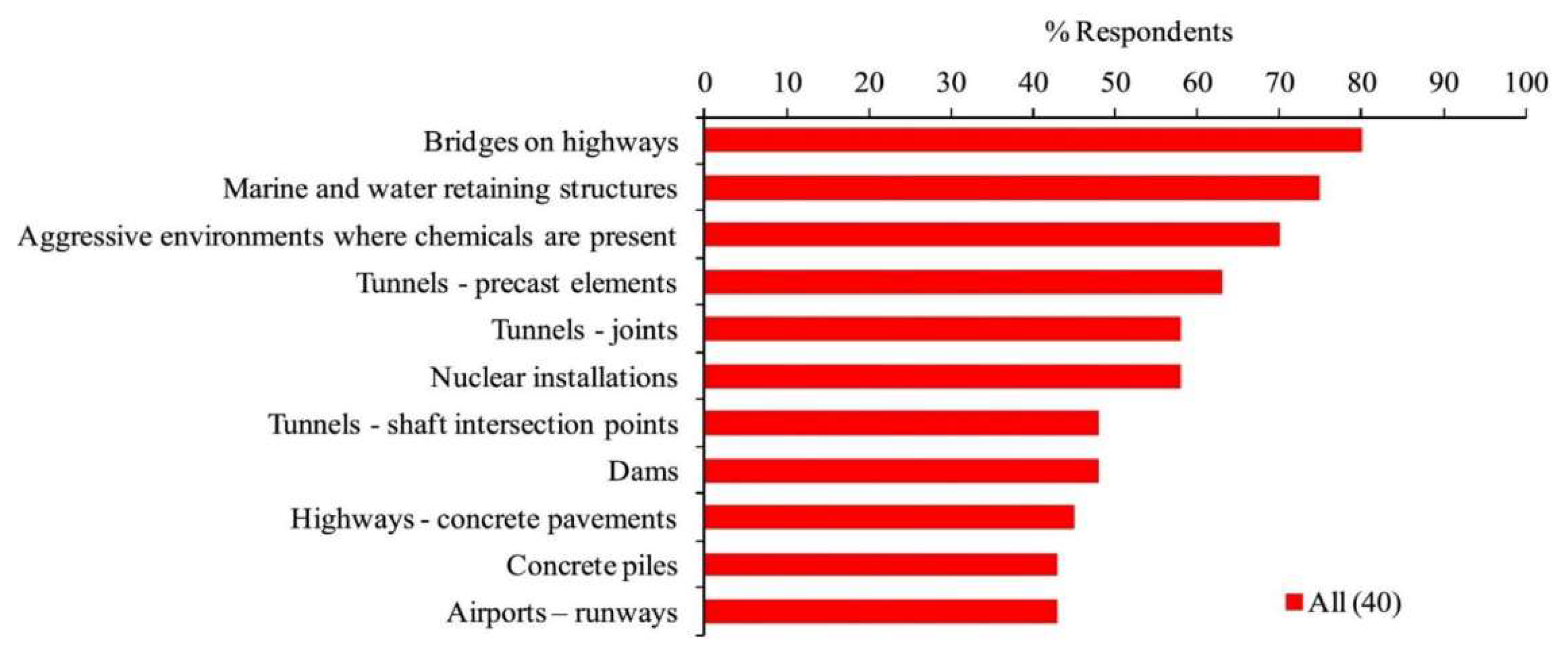
References
- Horszczaruk, E.; Sikora, P.; Cendrowski, K.; Mijowska, E.J.C.; Materials, B. The effect of elevated temperature on the properties of cement mortars containing nanosilica and heavyweight aggregates. Constr. Build. Mater. 2017, 137, 420–431.
- Seifan, M.; Samani, A.K.; Berenjian, A. Induced calcium carbonate precipitation using Bacillus species. Appl. Microbiol. Biotechnol. 2016, 100, 9895–9906.
- Luhar, S.; Luhar, I.; Shaikh, F.U.A. A Review on the Performance Evaluation of Autonomous Self-Healing Bacterial Concrete: Mechanisms, Strength, Durability, and Microstructural Properties. J. Compos. Sci. 2022, 6, 23.
- Chapman, J.; Regan, F.; Sullivan, T. Nanoparticles in Anti-Microbial Materials: Use and Characterisation; Royal Society of Chemistry: London, UK, 2012; Volume 23.
- Biondini, F.; Camnasio, E.; Palermo, A. Lifetime seismic performance of concrete bridges exposed to corrosion. Struct. Infrastruct. Eng. 2014, 10, 880–900.
- Virmani, Y.P.; Clemena, G.G. Corrosion Protection: Concrete Bridges; Federal Highway Administration: Washington, DC, USA, 1998.
- Müller, H.S.; Haist, M.; Vogel, M. Assessment of the sustainability potential of concrete and concrete structures considering their environmental impact, performance and lifetime. Constr. Build. Mater. 2014, 67, 321–337.
- Roels, E.; Terryn, S.; Iida, F.; Bosman, A.W.; Norvez, S.; Clemens, F.; Van Assche, G.; Vanderborght, B.; Brancart, J. Processing of Self-Healing Polymers for Soft Robotics. Adv. Mater. 2022, 34, 2104798.
- Palmer, F. Using Emergent Technologies to Develop Sustainable Architectural Composites. Ph.D. Thesis, Auckland University of Technology, Auckland, New Zealand, 2009.
- Ismail, A.; Adan, N.H. Effect of oxygen concentration on corrosion rate of carbon steel in seawater. Am. J. Eng. Res. 2014, 3, 64–67.
- Andrade, C.; Alonso, C.; Sarria, J. Influence of relative humidity and temperature on-site corrosion rates. Mater. Constr. 1998, 48, 5–17.
- Shevtsov, D.; Cao, N.L.; Nguyen, V.C.; Nong, Q.Q.; Le, H.Q.; Nguyen, D.A.; Zartsyn, I.; Kozaderov, O.J.S. Progress in Sensors for Monitoring Reinforcement Corrosion in Reinforced Concrete Structures—A Review. Sensors 2022, 22, 3421.
- Osterminski, K. Zur Voll-Probabilistischen Modellierung der Korrosion von Stahl in Beton: Ein Beitrag zur Dauerhaftigkeitsbemessung von Stahlbetonbauteilen. Ph.D. Thesis, Technische Universität München, München, Germany, 2013.
- Xian, X.; Zhang, D.; Lin, H.; Shao, Y. Ambient pressure carbonation curing of reinforced concrete for CO2 utilization and corrosion resistance. J. CO2 Util. 2022, 56, 101861.
- Tuutti, K. Corrosion of Steel in Concrete; Cement-Och Betonginst: Stockholm, Schwedisch, 1982.
- Zhang, J.; Shi, X.; Chen, X.; Huo, X.; Yu, Z. Microbial-Induced Carbonate Precipitation: A Review on Influencing Factors and Applications. Adv. Civ. Eng. 2021, 2021, 9974027.
- Jain, S.; Fang, C.; Achal, V. A critical review on microbial carbonate precipitation via denitrification process in building materials. Bioengineered 2021, 12, 7529–7551.
- Ferris, G.R.; Frink, D.D.; Galang, M.C.; Zhou, J.; Kacmar, K.M.; Howard, J.L. Perceptions of organizational politics: Prediction, stress-related implications, and outcomes. Hum. Relat. 1996, 49, 233–266.
- Warren, L.A.; Haack, E.A. Biogeochemical controls on metal behaviour in freshwater environments. Earth-Sci. Rev. 2001, 54, 261–320.
- Chuo, S.C.; Mohamed, S.F.; Mohd Setapar, S.H.; Ahmad, A.; Jawaid, M.; Wani, W.A.; Yaqoob, A.A.; Mohamad Ibrahim, M.N. Insights into the current trends in the utilization of bacteria for microbially induced calcium carbonate precipitation. Materials 2020, 13, 4993.
- Walker, S.; Flemming, C.; Ferris, F.; Beveridge, T.; Bailey, G. Physicochemical interaction of Escherichia coli cell envelopes and Bacillus subtilis cell walls with two clays and ability of the composite to immobilize heavy metals from solution. Appl. Environ. Microbiol. 1989, 55, 2976–2984.
- Hammes, F.; Verstraete, W. Key roles of pH and calcium metabolism in microbial carbonate precipitation. Rev. Environ. Sci. Biotechnol. 2002, 1, 3–7.
- Beveridge, T.; Davies, J.A. Cellular responses of Bacillus subtilis and Escherichia coli to the Gram stain. J. Bacteriol. 1983, 156, 846–858.
- Salton, M.R.J. The relationship between the nature of the cell wall and the Gram stain. Microbiology 1963, 30, 223–235.
- Zhu, J.; Shen, D.; Jin, B.; Wu, S. Theoretical investigation on the formation mechanism of carbonate ion in microbial self-healing concrete: Combined QC calculation and MD simulation. Constr. Build. Mater. 2022, 342, 128000.
- Wang, J.; Snoeck, D.; Van Vlierberghe, S.; Verstraete, W.; De Belie, N. Application of hydrogel encapsulated carbonate precipitating bacteria for approaching a realistic self-healing in concrete. Constr. Build. Mater. 2014, 68, 110–119.
- Wang, J.; Jonkers, H.M.; Boon, N.; De Belie, N. Bacillus sphaericus LMG 22257 is physiologically suitable for self-healing concrete. Appl. Microbiol. Biotechnol. 2017, 101, 5101–5114.
- Shahid, S.; Aslam, M.A.; Ali, S.; Zameer, M.; Faisal, M. Self-healing of cracks in concrete using Bacillus strains encapsulated in sodium alginate beads. ChemistrySelect 2020, 5, 312–323.
- Khaliq, W.; Ehsan, M.B. Crack healing in concrete using various bio influenced self-healing techniques. Constr. Build. Mater. 2016, 102, 349–357.
- Andalib, R.; Abd Majid, M.Z.; Hussin, M.W.; Ponraj, M.; Keyvanfar, A.; Mirza, J.; Lee, H.-S. Optimum concentration of Bacillus megaterium for strengthening structural concrete. Constr. Build. Mater. 2016, 118, 180–193.
- Dhami, N.K.; Reddy, M.S.; Mukherjee, A. Improvement in strength properties of ash bricks by bacterial calcite. Ecol. Eng. 2012, 39, 31–35.
- Siddique, R.; Singh, K.; Singh, M.; Corinaldesi, V.; Rajor, A. Properties of bacterial rice husk ash concrete. Constr. Build. Mater. 2016, 121, 112–119.
- Chahal, N.; Siddique, R.; Rajor, A. Influence of bacteria on the compressive strength, water absorption and rapid chloride permeability of concrete incorporating silica fume. Constr. Build. Mater. 2012, 37, 645–651.
- Raut, S.H.; Sarode, D.; Lele, S.S. Biocalcification using B. pasteurii for strengthening brick masonry civil engineering structures. World J. Microbiol. Biotechnol. 2014, 30, 191–200.
- Castanier, S.; Le Métayer-Levrel, G.; Perthuisot, J.-P. Ca-carbonates precipitation and limestone genesis—The microbiogeologist point of view. Sediment. Geol. 1999, 126, 9–23.
- Jonkers, H.M.; Thijssen, A.; Muyzer, G.; Copuroglu, O.; Schlangen, E. Application of bacteria as self-healing agent for the development of sustainable concrete. Ecol. Eng. 2010, 36, 230–235.
- Zhang, J.; Liu, Y.; Feng, T.; Zhou, M.; Zhao, L.; Zhou, A.; Li, Z. Immobilizing bacteria in expanded perlite for the crack self-healing in concrete. Constr. Build. Mater. 2017, 148, 610–617.
- Seifan, M.; Sarabadani, Z.; Berenjian, A. Development of an innovative urease-aided self-healing dental composite. Catalysts 2020, 10, 84.
- Stocks-Fischer, S.; Galinat, J.K.; Bang, S.S. Microbiological precipitation of CaCO3. Soil Biol. Biochem. 1999, 31, 1563–1571.
- Tomalia, D.A.; Baker, H.; Dewald, J.; Hall, M.; Kallos, G.; Martin, S.; Roeck, J.; Ryder, J.; Smith, P. A new class of polymers: Starburst-dendritic macromolecules. Polym. J. 1985, 17, 117–132.
- Seifan, M.; Samani, A.K.; Berenjian, A. New insights into the role of pH and aeration in the bacterial production of calcium carbonate (CaCO3). Appl. Microbiol. Biotechnol. 2017, 101, 3131–3142.
- Achal, V.; Pan, X.; Zhang, D. Remediation of copper-contaminated soil by Kocuria flava CR1, based on microbially induced calcite precipitation. Ecol. Eng. 2011, 37, 1601–1605.
- Ning, F.; Cong, W.; Qiu, J.; Wei, J.; Wang, S. Additive manufacturing of carbon fiber reinforced thermoplastic composites using fused deposition modeling. Compos. Part B Eng. 2015, 80, 369–378.
- De Belie, N.; Gruyaert, E.; Al-Tabbaa, A.; Antonaci, P.; Baera, C.; Bajare, D.; Darquennes, A.; Davies, R.; Ferrara, L.; Jefferson, T. A review of self-healing concrete for damage management of structures. Adv. Mater. Interfaces 2018, 5, 1800074.
- Krajewska, B. Urease-aided calcium carbonate mineralization for engineering applications: A review. J. Adv. Res. 2018, 13, 59–67.
- Seifan, M.; Berenjian, A. Application of microbially induced calcium carbonate precipitation in designing bio self-healing concrete. World J. Microbiol. Biotechnol. 2018, 34, 168.
- Algaifi, H.A.; Bakar, S.A.; Sam, A.R.M.; Ismail, M.; Abidin, A.R.Z.; Shahir, S.; Altowayti, W.A.H. Insight into the role of microbial calcium carbonate and the factors involved in self-healing concrete. Constr. Build. Mater. 2020, 254, 119258.
- De Muynck, W.; De Belie, N.; Verstraete, W. Microbial carbonate precipitation in construction materials: A review. Ecol. Eng. 2010, 36, 118–136.
- Kempe, S.; Kazmierczak, J. The role of alkalinity in the evolution of ocean chemistry, organization of living systems, and biocalcification processes. Bull. Inst. Océanograph. 1994, 13, 61–117.
- Gowthaman, S.; Iki, T.; Nakashima, K.; Ebina, K.; Kawasaki, S. Feasibility study for slope soil stabilization by microbial induced carbonate precipitation (MICP) using indigenous bacteria isolated from cold subarctic region. SN Appl. Sci. 2019, 1, 1480.
- Seifan, M.; Berenjian, A. Microbially induced calcium carbonate precipitation: A widespread phenomenon in the biological world. Appl. Microbiol. Biotechnol. 2019, 103, 4693–4708.
- Nannipieri, P.; Ascher, J.; Ceccherini, M.; Landi, L.; Pietramellara, G.; Renella, G. Microbial diversity and soil functions. Eur. J. Soil Sci. 2003, 54, 655–670.
- Rusznyák, A.; Akob, D.M.; Nietzsche, S.; Eusterhues, K.; Totsche, K.U.; Neu, T.R.; Frosch, T.; Popp, J.; Keiner, R.; Geletneky, J. Calcite biomineralization by bacterial isolates from the recently discovered pristine karstic Herrenberg cave. Appl. Environ. Microbiol. 2012, 78, 1157–1167.
- Li, Y.H.; Chen, Y.Y.M.; Burne, R.A. Regulation of urease gene expression by Streptococcus salivarius growing in biofilms. Environ. Microbiol. 2000, 2, 169–177.
- Gorospe, C.M.; Han, S.-H.; Kim, S.-G.; Park, J.-Y.; Kang, C.-H.; Jeong, J.-H.; So, J.-S. Effects of different calcium salts on calcium carbonate crystal formation by Sporosarcina pasteurii KCTC 3558. Biotechnol. Bioprocess Eng. 2013, 18, 903–908.
- Nemati, M.; Greene, E.; Voordouw, G. Permeability profile modification using bacterially formed calcium carbonate: Comparison with enzymic option. Process Biochem. 2005, 40, 925–933.
- Rivadeneyra, M.A.; Delgado, G.; Soriano, M.; Ramos-Cormenzana, A.; Delgado, R. Precipitation of carbonates by Nesterenkonia halobia in liquid media. Chemosphere 2000, 41, 617–624.
- Sadeghzadeh, M.; Maddah, H.; Ahmadi, M.H.; Khadang, A.; Ghazvini, M.; Mosavi, A.; Nabipour, N. Prediction of thermo-physical properties of TiO2-Al2O3/water nanoparticles by using artificial neural network. Nanomaterials 2020, 10, 697.
- Shabani, S.; Samadianfard, S.; Sattari, M.T.; Mosavi, A.; Shamshirband, S.; Kmet, T.; Várkonyi-Kóczy, A.R. Modeling pan evaporation using Gaussian process regression K-nearest neighbors random forest and support vector machines; comparative analysis. Atmosphere 2020, 11, 66.
- Nabipour, N.; Mosavi, A.; Hajnal, E.; Nadai, L.; Shamshirband, S.; Chau, K.-W. Modeling climate change impact on wind power resources using adaptive neuro-fuzzy inference system. Eng. Appl. Comput. Fluid Mech. 2020, 14, 491–506.
- Mitchell, A.C.; Ferris, F.G. The coprecipitation of Sr into calcite precipitates induced by bacterial ureolysis in artificial groundwater: Temperature and kinetic dependence. Geochim. Cosmochim. Acta 2005, 69, 4199–4210.
- Okwadha, G.D.; Li, J. Optimum conditions for microbial carbonate precipitation. Chemosphere 2010, 81, 1143–1148.
- Mosavi, A.; Sajedi Hosseini, F.; Choubin, B.; Goodarzi, M.; Dineva, A.A.; Rafiei Sardooi, E. Ensemble boosting and bagging based machine learning models for groundwater potential prediction. Water Resour. Manag. 2021, 35, 23–37.
- Ghalandari, M.; Shamshirband, S.; Mosavi, A.; Chau, K.-W. Flutter speed estimation using presented differential quadrature method formulation. Eng. Appl. Comput. Fluid Mech. 2019, 13, 804–810.
- Ferris, F.G.; Phoenix, V.; Fujita, Y.; Smith, R. Kinetics of calcite precipitation induced by ureolytic bacteria at 10 to 20 C in artificial groundwater. Geochim. Cosmochim. Acta 2004, 68, 1701–1710.
- Nemati, M.; Voordouw, G. Modification of porous media permeability, using calcium carbonate produced enzymatically in situ. Enzym. Microb. Technol. 2003, 33, 635–642.
- Dhami, N.K.; Reddy, M.S.; Mukherjee, A. Synergistic role of bacterial urease and carbonic anhydrase in carbonate mineralization. Appl. Biochem. Biotechnol. 2014, 172, 2552–2561.
- Zamani, M.; Nikafshar, S.; Mousa, A.; Behnia, A. Bacteria encapsulation using synthesized polyurea for self-healing of cement paste. Constr. Build. Mater. 2020, 249, 118556.
- Pang, B.; Zhou, Z.; Hou, P.; Du, P.; Zhang, L.; Xu, H. Autogenous and engineered healing mechanisms of carbonated steel slag aggregate in concrete. Constr. Build. Mater. 2016, 107, 191–202.
- Zhang, X.; Jin, Z.; Li, M.; Qian, C. Effects of carrier on the performance of bacteria-based self-healing concrete. Constr. Build. Mater. 2021, 305, 124771.
- Qian, C.; Zhang, Y.; Xie, Y. Effect of ion concentration in crack zone on healing degree of microbial self-healing concrete. Constr. Build. Mater. 2021, 286, 122969.
- Qian, C.; Zheng, T.; Zhang, X.; Su, Y. Application of microbial self-healing concrete: Case study. Constr. Build. Mater. 2021, 290, 123226.
- Aggelis, D.; Leonidou, E.; Matikas, T.E. Subsurface crack determination by one-sided ultrasonic measurements. Cem. Concr. Compos. 2012, 34, 140–146.
- Shamshirband, S.; Mosavi, A.; Rabczuk, T.; Nabipour, N.; Chau, K.-W. Prediction of significant wave height; comparison between nested grid numerical model, and machine learning models of artificial neural networks, extreme learning and support vector machines. Eng. Appl. Comput. Fluid Mech. 2020, 14, 805–817.
- Mousavi, S.M.; Ghasemi, M.; Dehghan Manshadi, M.; Mosavi, A. Deep learning for wave energy converter modeling using long short-term memory. Mathematics 2021, 9, 871.
- Jroundi, F.; Gonzalez-Muñoz, M.T.; Garcia-Bueno, A.; Rodriguez-Navarro, C. Consolidation of archaeological gypsum plaster by bacterial biomineralization of calcium carbonate. Acta Biomater. 2014, 10, 3844–3854.
- Xu, J.; Yao, W. Multiscale mechanical quantification of self-healing concrete incorporating non-ureolytic bacteria-based healing agent. Cem. Concr. Res. 2014, 64, 1–10.
- Taherei Ghazvinei, P.; Hassanpour Darvishi, H.; Mosavi, A.; Yusof, K.b.W.; Alizamir, M.; Shamshirband, S.; Chau, K.-W. Sugarcane growth prediction based on meteorological parameters using extreme learning machine and artificial neural network. Eng. Appl. Comput. Fluid Mech. 2018, 12, 738–749.
- Asadi, E.; Isazadeh, M.; Samadianfard, S.; Ramli, M.F.; Mosavi, A.; Nabipour, N.; Shamshirband, S.; Hajnal, E.; Chau, K.-W. Groundwater quality assessment for sustainable drinking and irrigation. Sustainability 2019, 12, 177.
- Kalbasi, R.; Jahangiri, M.; Mosavi, A.; Dehshiri, S.J.H.; Dehshiri, S.S.H.; Ebrahimi, S.; Etezadi, Z.A.-S.; Karimipour, A. Finding the best station in Belgium to use residential-scale solar heating, one-year dynamic simulation with considering all system losses: Economic analysis of using ETSW. Sustain. Energy Technol. Assess. 2021, 45, 101097.
- Lee, C.; Lee, H.; Kim, O.B. Biocement fabrication and design application for a sustainable urban area. Sustainability 2018, 10, 4079.
- Cagatay Ersan, Y.; Hernandez Sanabria, E.; Boon, N.; De Belie, N. Enhanced crack closure performance of microbial mortar through nitrate reduction. Cem. Concr. Compos. 2016, 70, 159–170.
- Wiktor, V.; Jonkers, H.M. Quantification of crack-healing in novel bacteria-based self-healing concrete. Cem. Concr. Compos. 2011, 33, 763–770.
- Brandl, E.; Heckenberger, U.; Holzinger, V.; Buchbinder, D. Additive manufactured AlSi10Mg samples using Selective Laser Melting (SLM): Microstructure, high cycle fatigue, and fracture behavior. Mater. Des. 2012, 34, 159–169.
- Wang, J.; Mignon, A.; Trenson, G.; Van Vlierberghe, S.; Boon, N.; De Belie, N. A chitosan based pH-responsive hydrogel for encapsulation of bacteria for self-sealing concrete. Cem. Concr. Compos. 2018, 93, 309–322.
- Palin, D.; Wiktor, V.; Jonkers, H.M. A bacteria-based self-healing cementitious composite for application in low-temperature marine environments. Biomimetics 2017, 2, 13.
- Mohammadzadeh, S.D.; Kazemi, S.-F.; Mosavi, A.; Nasseralshariati, E.; Tah, J.H. Prediction of compression index of fine-grained soils using a gene expression programming model. Infrastructures 2019, 4, 26.
- Ghalandari, M.; Ziamolki, A.; Mosavi, A.; Shamshirband, S.; Chau, K.-W.; Bornassi, S. Aeromechanical optimization of first row compressor test stand blades using a hybrid machine learning model of genetic algorithm, artificial neural networks and design of experiments. Eng. Appl. Comput. Fluid Mech. 2019, 13, 892–904.
- Snoeck, D.; Malm, F.; Cnudde, V.; Grosse, C.U.; Van Tittelboom, K. Validation of Self-Healing Properties of Construction Materials through Nondestructive and Minimal Invasive Testing. Adv. Mater. Interfaces 2018, 5, 1800179.
- Liu, W.; Li, Y.-Q.; Tang, L.-P.; Dong, Z.-J. XRD and 29Si MAS NMR study on carbonated cement paste under accelerated carbonation using different concentration of CO2. Mater. Today Commun. 2019, 19, 464–470.
- Muhammad, N.Z.; Shafaghat, A.; Keyvanfar, A.; Majid, M.Z.A.; Ghoshal, S.; Yasouj, S.E.M.; Ganiyu, A.A.; Kouchaksaraei, M.S.; Kamyab, H.; Taheri, M.M. Tests and methods of evaluating the self-healing efficiency of concrete: A review. Constr. Build. Mater. 2016, 112, 1123–1132.
- Wang, J.; Van Tittelboom, K.; De Belie, N.; Verstraete, W. Use of silica gel or polyurethane immobilized bacteria for self-healing concrete. Constr. Build. Mater. 2012, 26, 532–540.
- Johannesson, B.; Utgenannt, P. Microstructural changes caused by carbonation of cement mortar. Cem. Concr. Res. 2001, 31, 925–931.
- Choubin, B.; Mosavi, A.; Alamdarloo, E.H.; Hosseini, F.S.; Shamshirband, S.; Dashtekian, K.; Ghamisi, P. Earth fissure hazard prediction using machine learning models. Environ. Res. 2019, 179, 108770.
- Mahmoudi, M.R.; Heydari, M.H.; Qasem, S.N.; Mosavi, A.; Band, S.S. Principal component analysis to study the relations between the spread rates of COVID-19 in high risks countries. Alex. Eng. J. 2021, 60, 457–464.
- Imtiaz, L.; Rehman, S.K.; Ali Memon, S.; Khizar Khan, M.; Faisal Javed, M. A review of recent developments and advances in eco-friendly geopolymer concrete. Appl. Sci. 2020, 10, 7838.
- Cuthbert, M.O.; McMillan, L.A.; Handley-Sidhu, S.; Riley, M.S.; Tobler, D.J.; Phoenix, V.R. A field and modeling study of fractured rock permeability reduction using microbially induced calcite precipitation. Environ. Sci. Technol. 2013, 47, 13637–13643.
- De Muynck, W.; Verbeken, K.; De Belie, N.; Verstraete, W. Influence of temperature on the effectiveness of a biogenic carbonate surface treatment for limestone conservation. Appl. Microbiol. Biotechnol. 2013, 97, 1335–1347.
- Amran, M.; Onaizi, A.M.; Fediuk, R.; Vatin, N.I.; Muhammad Rashid, R.S.; Abdelgader, H.; Ozbakkaloglu, T. Self-Healing Concrete as a Prospective Construction Material: A Review. Materials 2022, 15, 3214.
- Gardner, D.; Lark, R.; Jefferson, T.; Davies, R. A survey on problems encountered in current concrete construction and the potential benefits of self-healing cementitious materials. Case Stud. Constr. Mater. 2018, 8, 238–247.





