
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Stuart Russell Maudsley | -- | 3484 | 2022-09-26 11:30:43 | | | |
| 2 | Beatrix Zheng | + 7 word(s) | 3491 | 2022-09-27 04:00:52 | | |
Video Upload Options
During the aging process human's body becomes less well equipped to deal with cellular stress, resulting in an increase in unrepaired damage. This causes varying degrees of impaired functionality and an increased risk of mortality. One of the most effective anti-aging strategies involves interventions that combine simultaneous glucometabolic support with augmented DNA damage protection/repair. Thus, it seems prudent to develop therapeutic strategies that target this combinatorial approach. Studies have shown that the ADP-ribosylation factor (ARF) GTPase activating protein GIT2 (GIT2) acts as a keystone protein in the aging process. GIT2 can control both DNA repair and glucose metabolism. Through in vivo co-regulation analyses it was found that GIT2 forms a close co-expression based relationship with the relaxin-3 receptor (RXFP3). Cellular RXFP3 expression is directly affected by DNA damage and oxidative stress. Overexpression or stimulation of this receptor, by its endogenous ligand relaxin 3 (RLN3), can regulate the DNA damage response and repair processes. Interestingly, RLN3 is an insulin-like peptide and has been shown to control multiple disease processes linked to aging mechanisms, e.g., anxiety, depression, memory dysfunction, appetite, and anti-apoptotic mechanisms.
1. Introduction
1.1. Aging and Aging-Related Disorders
1.2. Relaxin-Family Peptide Receptor 3
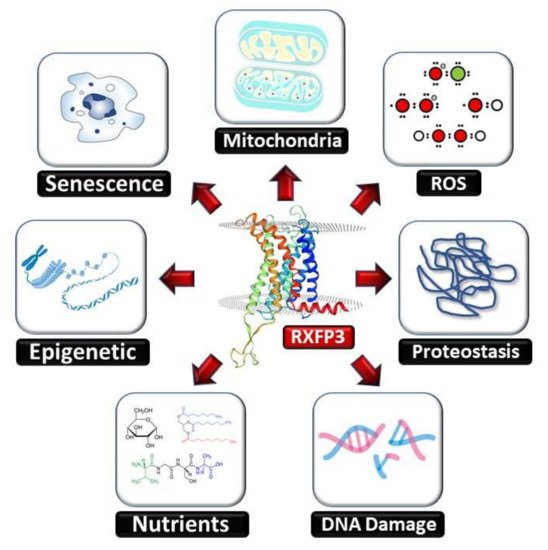
2. RXFP3 in Aging-Related Disorders
2.1. Alzheimer’s Disease
2.2. Anxiety and Post-Traumatic Stress Disorder
2.3. Schizophrenia
2.4. Obesity and Metabolic Dysfunction
2.5. Ischemic Stroke
2.6. Reproductive Aging
2.7. Alcohol Abuse
References
- Liguori, I.; Russo, G.; Curcio, F.; Bulli, G.; Aran, L.; Della-Morte, D.; Gargiulo, G.; Testa, G.; Cacciatore, F.; Bonaduce, D.; et al. Oxidative stress, aging, and diseases. Clin. Interv. Aging. 2018, 13, 757–772.
- Chadwick, W.; Martin, B.; Chapter, M.C.; Park, S.; Wang, L.; Daimon, C.M.; Brenneman, R.; Maudsley, S. GIT2 acts as a potential keystone protein in functional hypothalamic networks associated with age-related phenotypic changes in rats. PLoS ONE 2012, 7, e36975.
- Cai, H.; Cong, W.N.; Ji, S.; Rothman, S.; Maudsley, S.; Martin, B. Metabolic dysfunction in Alzheimer’s disease and related neurodegenerative disorders. Curr. Alzheimer Res. 2012, 9, 5–17.
- Li, H.; Mitchell, J.R.; Hasty, P. DNA double-strand breaks: A potential causative factor for mammalian aging? Mech. Ageing Dev. 2008, 129, 416–424.
- Martin, B.; Chadwick, W.; Janssens, J.; Premont, R.T.; Schmalzigaug, R.; Becker, K.G.; Lehrmann, E.; Wood, W.H.; Zhang, Y.; Siddiqui, S.; et al. GIT2 acts as a systems-level coordinator of neurometabolic activity and pathophysiological aging. Front. Endocrinol. 2016, 6, 191.
- Chadwick, W.; Zhou, Y.; Park, S.; Wang, L.; Mitchell, N.; Stone, M.D.; Becker, K.G.; Martin, B.; Maudsley, S. Minimal peroxide exposure of neuronal cells induces multifaceted adaptive responses. PLoS ONE 2010, 5, e14352.
- Lu, D.; Cai, H.; Park, S.S.; Siddiqui, S.; Premont, R.T.; Schmalzigaug, R.; Paramasivam, M.; Seidman, M.; Bodogai, I.; Biragyn, A.; et al. Nuclear GIT2 is an ATM substrate and promotes DNA repair. Mol. Cell. Biol. 2015, 35, 1081–1096.
- Premont, R.T.; Claing, A.; Vitale, N.; Perry, S.J.; Lefkowitz, R.J. The GIT family of ADP-ribosylation factor GTPase-activating proteins. Functional diversity of GIT2 through alternative splicing. J. Biol. Chem. 2000, 275, 22373–22380.
- Premont, R.T.; Perry, S.J.; Schmalzigaug, R.; Roseman, J.T.; Xing, Y.; Claing, A. The GIT/PIX complex: An oligomeric assembly of GIT family ARF GTPase-activating proteins and PIX family Rac1/Cdc42 guanine nucleotide exchange factors. Cell. Signal. 2004, 16, 1001–1011.
- Van Gastel, J.; Etienne, H.; Azmi, A.; Maudsley, S. The synergistic GIT2-RXFP3 system in the brain and its importance in age-related disorders. Front. Aging Neurosci. 2016, 3, 8.
- Roux, B.T.; Cottrell, G.S. G protein-coupled receptors: What a difference a ‘partner’ makes. Int. J. Mol. Sci. 2014, 15, 1112–1142.
- Bathgate, R.A.; Oh, M.H.; Ling, W.J.; Kaas, Q.; Hossain, M.A.; Gooley, P.R.; Rosengren, J. Elucidation of relaxin-3 binding interactions in the extracellular loops of RXFP3. Front. Endocrinol. 2013, 4, 13.
- Matsumoto, M.; Kamohara, M.; Sugimoto, T.; Hidaka, K.; Takasaki, J.; Saito, T.; Okada, M.; Yamaguchi, T.; Furuichi, K. The novel G-protein coupled receptor SALPR shares sequence similarity with somatostatin and angiotensin receptors. Gene 2000, 248, 183–189.
- Olucha-Bordonau, F.E.; Albert-Gascó, H.; Ros-Bernal, F.; Rytova, V.; Ong-Pålsson, E.K.E.; Ma, S.; Sánchez-Pérez, A.M.; Gundlach, A.L. Modulation of forebrain function by nucleus incertus and relaxin-3/RXFP3 signaling. CNS Neurosci. Ther. 2018, 24, 694–702.
- Smith, C.M.; Shen, P.J.; Banerjee, A.; Bonaventure, P.; Ma, S.; Bathgate, R.A.D.; Sutton, S.W.; Gundlach, A.L. Distribution of relaxin-3 and RXFP3 within arousal, stress, affective, and cognitive circuits of mouse brain. J. Comp. Neurol. 2010, 518, 4016–4045.
- Ganella, D.E.; Ma, S.; Gundlach, A.L. Relaxin-3/RXFP3 Signaling and Neuroendocrine Function—A Perspective on Extrinsic Hypothalamic Control. Front. Endocrinol. 2013, 4, 128.
- Halls, M.L.; Bathgate, R.A.; Sutton, S.W.; Dschietzig, T.B.; Summers, R.J. International Union of Basic and Clinical Pharmacology. XCV. Recent advances in the understanding of the pharmacology and biological roles of relaxin family peptide receptors 1–4, the receptors for relaxin family peptides. Pharmacol. Rev. 2015, 67, 389–440.
- Aaboud, M.; Aad, G.; Abbott, B.; Abdallah, J.; Abdinov, O.; Abeloos, B.; Aben, R.; AbouZeid, O.S.; Abraham, N.L.; Abramowicz, H.; et al. Measurement of the Inelastic Proton-Proton Cross Section at sqrt = 13 TeV with the ATLAS Detector at the LHC. Phys. Rev. Lett. 2016, 117, 182002.
- Van der Westhuizen, E.T.; Werry, T.D.; Sexton, P.M.; Summers, R.J. The relaxin family peptide receptor 3 activates extracellular signal-regulated kinase 1/2 through a protein kinase C-dependent mechanism. Mol. Pharmacol. 2007, 71, 1618–1629.
- Wilkinson, T.N.; Speed, T.P.; Tregear, G.W.; Bathgate, R.A. Evolution of the relaxin-like peptide family: From neuropeptide to reproduction. Ann. N. Y. Acad. Sci. 2005, 1041, 530–533.
- Yegorov, S.; Good-Avila, S.V.; Parry, L.; Wilson, B.C. Relaxin family genes in humans and teleosts. Ann. N. Y. Acad. Sci. 2009, 1160, 42–44.
- Bathgate, R.A.; Samuel, C.S.; Burazin, T.C.; Layfield, S.; Claasz, A.A.; Grace, I.; Reytomas, T.; Dawson, N.F.; Zhao, C.; Bond, C.; et al. Human relaxin gene 3 (H3) and the equivalent mouse relaxin (M3) gene. Novel members of the relaxin peptide family. J. Biol. Chem. 2002, 277, 1148–1157.
- Good, S.; Yegorov, S.; Martijn, J.; Franck, J.; Bogerd, J. New insights into ligand-receptor pairing and coevolution of relaxin family peptides and their receptors in teleosts. Int. J. Evol. Biol. 2012, 2012, 310278.
- Van Gastel, J.; Leysen, H.; Santos-Otte, P.; Hendrickx, J.O.; Azmi, A.; Martin, B.; Maudsley, S. The RXFP3 receptor is functionally associated with cellular responses to oxidative stress and DNA damage. Aging 2019, 11, 11268–11313.
- Tanaka, M.; Iijima, N.; Miyamoto, Y.; Fukusumi, S.; Itoh, Y.; Ozawa, H.; Ibata, Y. Neurons expressing relaxin 3/INSL 7 in the nucleus incertus respond to stress. Eur. J. Neurosci. 2005, 21, 1659–1670.
- Ryan, P.J.; Buchler, E.; Shabanpoor, F.; Hossain, M.A.; Wade, J.D.; Lawrence, A.J.; Gundlach, A.L. Central relaxin-3 receptor (RXFP3) activation decreases anxiety- and depressive-like behaviours in the rat. Behav. Brain Res. 2013, 244, 142–151.
- Lee, J.H.; Koh, S.Q.; Guadagna, S.; Francis, P.T.; Esiri, M.M.; Chen, C.P.; Wong, P.T.H.; Dawe, G.S.; Lai, M.K.P. Altered relaxin family receptors RXFP1 and RXFP3 in the neocortex of depressed Alzheimer’s disease patients. Psychopharmacology 2016, 233, 591–598.
- Zhang, C.; Baimoukhametova, D.V.; Smith, C.M.; Bains, J.S.; Gundlach, A.L. Relaxin-3/RXFP3 signalling in mouse hypothalamus: No effect of RXFP3 activation on corticosterone, despite reduced presynaptic excitatory input onto paraventricular CRH neurons in vitro. Psychopharmacology 2017, 234, 1725–1739.
- McGowan, B.M.; Stanley, S.A.; Smith, K.L.; Minnion, J.S.; Donovan, J.; Thompson, E.L.; Patterson, M.; Connolly, M.M.; Abbott, C.R.; Small, C.J.; et al. Effects of acute and chronic relaxin-3 on food intake and energy expenditure in rats. Regul. Pept. 2006, 136, 72–77.
- Smith, C.M.; Ryan, P.J.; Hosken, I.T.; Ma, S.; Gundlach, A.L. Relaxin-3 systems in the brain—The first 10 years. J. Chem. Neuroanat. 2011, 42, 262–275.
- Ryan, P.J.; Kastman, H.E.; Krstew, E.V.; Rosengren, K.J.; Hossain, M.A.; Churilov, L.; Wade, J.D.; Gundlach, A.L.; Lawrence, A.J. Relaxin-3/RXFP3 system regulates alcohol-seeking. Proc. Natl. Acad. Sci. USA 2013, 110, 20789–20794.
- Opdebeeck, B.; Maudsley, S.; Azmi, A.; De Maré, A.; De Leger, W.; Meijers, B.; Verhulst, A.; Evenepoel, P.; D’Haese, P.C.; Neven, E.J. Indoxyl Sulfate and p-Cresyl Sulfate Promote Vascular Calcification and Associate with Glucose Intolerance. Am. Soc. Nephrol. 2019, 30, 751–766.
- Moons, R.; Konijnenberg, A.; Mensch, C.; Van Elzen, R.; Johannessen, C.; Maudsley, S.; Lambeir, A.M.; Sobott, F. Metal ions shape α-synuclein. Sci. Rep. 2020, 10, 16293.
- Zhang, X.; Pan, L.; Yang, K.; Fu, Y.; Liu, Y.; Chen, W.; Ma, X.; Yin, X. Alterations of relaxin and its receptor system components in experimental diabetic cardiomyopathy rats. Cell Tissue Res. 2017, 370, 297–304.
- Kim, W.; Doyle, M.E.; Liu, Z.; Lao, Q.; Shin, Y.K.; Carlson, O.D.; Kim, H.S.; Thomas, S.; Napora, J.K.; Lee, E.K.; et al. Cannabinoids inhibit insulin receptor signaling in pancreatic beta-cells. Diabetes 2011, 60, 1198–1209.
- Martin, B.; Shin, Y.K.; White, C.M.; Ji, S.; Kim, W.; Carlson, O.D.; Napora, J.K.; Chadwick, W.; Chapter, M.; Waschek, J.A.; et al. Vasoactive intestinal peptide-null mice demonstrate enhanced sweet taste preference, dysglycemia, and reduced taste bud leptin receptor expression. Diabetes 2010, 59, 1143–1152.
- Chapter, M.C.; White, C.M.; DeRidder, A.; Chadwick, W.; Martin, B.; Maudsley, S. Chemical modification of class II G protein-coupled receptor ligands: Frontiers in the development of peptide analogs as neuroendocrine pharmacological therapies. Pharmacol. Ther. 2010, 125, 39–54.
- Zhang, X.; Fu, Y.; Li, H.; Shen, L.; Chang, Q.; Pan, L.; Hong, S.; Yin, X. H3 relaxin inhibits the collagen synthesis via ROS- and P2X7R-mediated NLRP3 inflammasome activation in cardiac fibroblasts under high glucose. J. Cell. Mol. Med. 2018, 22, 1816–1825.
- Zhou, J.; Livak, M.F.A.; Bernier, M.; Muller, D.C.; Carlson, O.D.; Elahi, D.; Maudsley, S.; Egan, J.M. Ubiquitination is involved in glucose-mediated downregulation of GIP receptors in islets. Am. J. Physiol. Endocrinol. Metab. 2007, 293, E538–E547.
- Camell, C.D. Adipose tissue microenvironments during aging: Effects on stimulated lipolysis. Biochim. Biophys. Acta. Mol. Cell Biol. Lipids 2022, 1867, 159118.
- Lu, B.; Huang, L.; Cao, J.; Li, L.; Wu, W.; Chen, X.; Ding, C.J. Adipose tissue macrophages in aging-associated adipose tissue function. Physiol. Sci. 2021, 71, 38.
- Fonseca, G.W.P.D.; von Haehling, S. The fatter, the better in old age: The current understanding of a difficult relationship. Curr. Opin. Clin. Nutr. Metab. Care 2022, 25, 1–6.
- Yamamoto, H.; Shimokawa, H.; Haga, T.; Fukui, Y.; Iguchi, K.; Unno, K.; Hoshino, M.; Takeda, A. The expression of relaxin-3 in adipose tissue and its effects on adipogenesis. Protein Pept. Lett. 2014, 21, 517–522.
- Gallander, G.E.; Bathgate, R.A.D. Relaxin family peptide systems and the central nervous system. Cell. Mol. Life Sci. 2010, 67, 2327–2341.
- Munro, J.; Skrobot, O.; Sanyoura, M.; Kay, V.; Susce, M.T.; Glaser, P.E.; de Leon, J.; Blakemore, A.I.; Arranz, M.J. Relaxin polymorphisms associated with metabolic disturbance in patients treated with antipsychotics. J. Psychopharmacol. 2012, 26, 374–379.
- Klaips, C.L.; Jayaraj, G.G.; Hartl, F.U. Pathways of cellular proteostasis in aging and disease. J. Cell Biol. 2018, 217, 51–63.
- De Avila, C.; Chometton, S.; Lenglos, C.; Calvez, J.; Gundlach, A.L.; Timofeeva, E. Differential effects of relaxin-3 and a selective relaxin-3 receptor agonist on food and water intake and hypothalamic neuronal activity in rats. Behav. Brain Res. 2018, 336, 135–144.
- Miquel, S.; Champ, C.; Day, J.; Aarts, E.; Bahr, B.A.; Bakker, M.; Bánáti, D.; Calabrese, V.; Cederholm, T.; Cryan, J.; et al. Poor cognitive ageing: Vulnerabilities, mechanisms and the impact of nutritional interventions. Ageing Res. Rev. 2018, 42, 40–55.
- Maudsley, S.; Chadwick, W. Progressive and unconventional pharmacotherapeutic approaches to Alzheimer’s disease therapy. Curr. Alzheimer Res. 2012, 9, 1–4.
- Haidar, M.; Tin, K.; Zhang, C.; Nategh, M.; Covita, J.; Wykes, A.D.; Rogers, J.; Gundlach, A.L. Septal GABA and Glutamate Neurons Express RXFP3 mRNA and Depletion of Septal RXFP3 Impaired Spatial Search Strategy and Long-Term Reference Memory in Adult Mice. Front. Neuroanat. 2019, 13, 30.
- Haidar, M.; Guèvremont, G.; Zhang, C.; Bathgate, R.A.D.; Timofeeva, E.; Smith, C.M.; Gundlach, A.L. Relaxin-3 inputs target hippocampal interneurons and deletion of hilar relaxin-3 receptors in "floxed-RXFP3" mice impairs spatial memory. Hippocampus 2017, 27, 529–546.
- García-Díaz, C.; Gil-Miravet, I.; Albert-Gasco, H.; Mañas-Ojeda, A.; Ros-Bernal, F.; Castillo-Gómez, E.; Gundlach, A.L.; Olucha-Bordonau, F.E. Relaxin-3 Innervation from the Nucleus Incertus to the Parahippocampal Cortex of the Rat. Front. Neuroanat. 2021, 15, 674649.
- Vida, C.; González, E.M.; De la Fuente, M. Increase of oxidation and inflammation in nervous and immune systems with aging and anxiety. Curr. Pharm. Des. 2014, 20, 4656–4678.
- Green, E.; Fairchild, J.K.; Kinoshita, L.M.; Noda, A.; Yesavage, J. Effects of Posttraumatic Stress Disorder and Metabolic Syndrome on Cognitive Aging in Veterans. Gerontologist 2016, 56, 72–81.
- Rutter, L.A.; Vahia, I.V.; Passell, E.; Forester, B.P.; Germine, L. The role of intraindividual cognitive variability in posttraumatic stress syndromes and cognitive aging: A literature search and proposed research agenda. Int. Psychogeriatr. 2021, 33, 677–687.
- Mañas-Ojeda, A.; Ros-Bernal, F.; Olucha-Bordonau, F.E.; Castillo-Gómez, E. Becoming Stressed: Does the Age Matter? Reviewing the Neurobiological and Socio-Affective Effects of Stress throughout the Lifespan. Int. J. Mol. Sci. 2020, 21, 5819.
- Rytova, V.; Ganella, D.E.; Hawkes, D.; Bathgate, R.A.D.; Ma, S.; Gundlach, A.L. Chronic activation of the relaxin-3 receptor on GABA neurons in rat ventral hippocampus promotes anxiety and social avoidance. Hippocampus 2019, 29, 905–920.
- Zhang, C.; Chua, B.E.; Yang, A.; Shabanpoor, F.; Hossain, M.A.; Wade, J.D.; Rosengren, K.J.; Smith, C.M.; Gundlach, A.L. Central relaxin-3 receptor (RXFP3) activation reduces elevated, but not basal, anxiety-like behaviour in C57BL/6J mice. Behav. Brain Res. 2015, 292, 125–132.
- Palmer, B.W.; Shir, C.; Chang, H.; Mulvaney, M.; Hall, J.M.H.; Shu, I.W.; Jin, H.; Lohr, J.B. Increased Prevalence of Metabolic Syndrome in Veterans with PTSD Untreated with Antipsychotic Medications. Int. J. Ment. Health 2021, 5, 10.
- Anckaerts, C.; van Gastel, J.; Leysen, V.; Hinz, R.; Azmi, A.; Simoens, P.; Shah, D.; Kara, F.; Langbeen, A.; Bols, P.; et al. Image-guided phenotyping of ovariectomized mice: Altered functional connectivity, cognition, myelination, and dopaminergic functionality. Neurobiol. Aging 2019, 74, 77–89.
- Mayer, A.; Mizdrak, M.; Babić, M.; Mastelić, T.; Glavina, T.; Božić, J.; Kurir, T.T. Knowledge, Attitudes, and Screening for Obstructive Sleep Apnea and Diabetes Mellitus among War Veterans Seeking Treatment of Posttraumatic Stress Disorder. Healthcare 2021, 9, 1698.
- Schmalzigaug, R.; Rodriguiz, R.M.; Phillips, L.E.; Davidson, C.E.; Wetsel, W.C.; Premont, R.T. Anxiety-like behaviors in mice lacking GIT2. Neurosci. Lett. 2009, 451, 156–161.
- Albert-Gasco, H.; Sanchez-Sarasua, S.; Ma, S.; García-Díaz, C.; Gundlach, A.L.; Sanchez-Perez, A.M.; Olucha-Bordonau, F.E. Central relaxin-3 receptor (RXFP3) activation impairs social recognition and modulates ERK-phosphorylation in specific GABAergic amygdala neurons. Brain Struct. Funct. 2019, 224, 453–469.
- Ganella, D.E.; Ryan, P.J.; Bathgate, R.A.D.; Gundlach, A.L. Increased feeding and body weight gain in rats after acute and chronic activation of RXFP3 by relaxin-3 and receptor-selective peptides: Functional and therapeutic implications. Behav. Pharmacol. 2012, 23, 516–525.
- Kania, A.; Szlaga, A.; Sambak, P.; Gugula, A.; Blasiak, E.; Di Bonaventura, M.V.M.; Hossain, M.A.; Cifani, C.; Hess, G.; Gundlach, A.L.; et al. RLN3/RXFP3 Signaling in the PVN Inhibits Magnocellular Neurons via M-like Current Activation and Contributes to Binge Eating Behavior. J. Neurosci. 2020, 40, 5362–5375.
- DeChristopher, B.; Park, S.; Vong, L.; Bamford, D.; Cho, H.; Duvadie, R.; Fedolak, A.; Hogan, C.; Honda, T.; Pandey, P.; et al. Discovery of a small molecule RXFP3/4 agonist that increases food intake in rats upon acute central administration. Bioorg. Med. Chem. Lett. 2019, 29, 991–994.
- Walker, A.W.; Smith, C.M.; Chua, B.E.; Krstew, E.V.; Zhang, C.; Gundlach, A.L.; Lawrence, A.J. Relaxin-3 receptor (RXFP3) signalling mediates stress-related alcohol preference in mice. PLoS ONE 2015, 10, e0122504.
- Van Gastel, J.; Hendrickx, J.O.; Leysen, H.; Martin, B.; Veenker, L.; Beuning, S.; Coppens, V.; Morrens, M.; Maudsley, S. Enhanced Molecular Appreciation of Psychiatric Disorders Through High-Dimensionality Data Acquisition and Analytics. Methods Mol. Biol. 2019, 2011, 671–723.
- Solomon, H.V.; Sinopoli, M.; DeLisi, L.E. Ageing with schizophrenia: An update. Curr. Opin. Psychiatry 2021, 34, 266–274.
- Jones, R.; MacCabe, J.H.; Price, M.J.; Liu, X.; Upthegrove, R. Effect of age on the relative efficacy of clozapine in schizophrenia. Acta Psychiatr. Scand. 2020, 142, 109–120.
- Kumar, J.R.; Rajkumar, R.; Jayakody, T.; Marwari, S.; Mei Hong, J.; Ma, S. Relaxin’ the brain: A case for targeting the nucleus incertus network and relaxin-3/RXFP3 system in neuropsychiatric disorders. Br. J. Pharmacol. 2017, 174, 1061–1076.
- Smith, C.M.; Walker, A.W.; Hosken, I.T.; Chua, B.E.; Zhang, C.; Haidar, M.; Gundlach, A.L. Relaxin-3/RXFP3 networks: An emerging target for the treatment of depression and other neuropsychiatric diseases? Front. Pharmacol. 2014, 5, 46.
- Hosken, I.T.; Sutton, S.W.; Smith, C.M.; Gundlach, A.L. Relaxin-3 receptor (Rxfp3) gene knockout mice display reduced running wheel activity: Implications for role of relaxin-3/RXFP3 signalling in sustained arousal. Behav. Brain Res. 2015, 278, 167–175.
- Corremans, R.; Neven, E.; Maudsley, S.; Leysen, H.; De Broe, M.E.; D’Haese, P.C.; Vervaet, B.A.; Verhulst, A. Progression of established non-diabetic chronic kidney disease is halted by metformin treatment in rats. Kidney Int. 2022, in press.
- Martin, B.; Pearson, M.; Kebejian, L.; Golden, E.; Keselman, A.; Bender, M.; Carlson, O.; Egan, J.; Ladenheim, B.; Cadet, J.L.; et al. Sex-dependent metabolic, neuroendocrine, and cognitive responses to dietary energy restriction and excess. Endocrinology 2007, 148, 4318–4333.
- Lee, S.; Huang, K. Epigenetic profiling of human brain differential DNA methylation networks in schizophrenia. BMC Med. Genomics 2016, 9, 68.
- Mattison, J.A.; Wang, M.; Bernier, M.; Zhang, J.; Park, S.; Maudsley, S.; An, S.S.; Santhanam, L.; Martin, B.; Faulkner, S.; et al. Resveratrol prevents high fat/sucrose diet-induced central arterial wall inflammation and stiffening in nonhuman primates. Cell Metab. 2014, 20, 183–190.
- Colleluori, G.; Villareal, D.T. Aging, obesity, sarcopenia and the effect of diet and exercise intervention. Exp. Gerontol. 2021, 155, 111561.
- Gutiérrez-Cuevas, J.; Galicia-Moreno, M.; Monroy-Ramírez, H.C.; Sandoval-Rodriguez, A.; García-Bañuelos, J.; Santos, A.; Armendariz-Borunda, J. The Role of NRF2 in Obesity-Associated Cardiovascular Risk Factors. Antioxidants 2022, 11, 235.
- Yamamoto, T.; Sano, M. Deranged Myocardial Fatty Acid Metabolism in Heart Failure. Int. J. Mol. Sci. 2022, 23, 996.
- Mezhnina, V.; Ebeigbe, O.P.; Poe, A.; Kondratov, R.V. Circadian Control of Mitochondria in Reactive Oxygen Species Homeostasis. Antioxid. Redox Signal. 2022, in press.
- Smith, C.M.; Chua, B.E.; Zhang, C.; Walker, A.W.; Haidar, M.; Hawkes, D.; Shabanpoor, F.; Hossain, M.A.; Wade, J.D.; Rosengren, K.J.; et al. Central injection of relaxin-3 receptor (RXFP3) antagonist peptides reduces motivated food seeking and consumption in C57BL/6J mice. Behav. Brain Res. 2014, 268, 117–126.
- Calvez, J.; de Ávila, C.; Timofeeva, E. Sex-specific effects of relaxin-3 on food intake and body weight gain. Br. J. Pharmacol. 2017, 174, 1049–1060.
- Lenglos, C.; Mitra, A.; Guèvremont, G.; Timofeeva, E. Regulation of expression of relaxin-3 and its receptor RXFP3 in the brain of diet-induced obese rats. Neuropeptides 2014, 48, 119–132.
- Hida, T.; Takahashi, E.; Shikata, K.; Hirohashi, T.; Sawai, T.; Seiki, T.; Tanaka, H.; Kawai, T.; Ito, O.; Arai, T.; et al. Chronic intracerebroventricular administration of relaxin-3 increases body weight in rats. J. Recept. Signal Transduct. Res. 2006, 26, 147–158.
- Hossain, M.A.; Smith, C.M.; Ryan, P.J.; Büchler, E.; Bathgate, R.A.; Gundlach, A.L.; Wade, J.D. Chemical synthesis and orexigenic activity of rat/mouse relaxin-3. Amino Acids. 2013, 44, 1529–1536.
- Benjamin, E.J.; Virani, S.S.; Callaway, C.W.; Chamberlain, A.M.; Chang, A.R.; Cheng, S.; Chiuve, S.E.; Cushman, M.; Delling, F.N.; Deo, R.; et al. Heart Disease and Stroke Statistics-2018 Update: A Report From the American Heart Association. Circulation 2018, 137, e67–e492.
- Bergeron, L.H.; Willcox, J.M.; Alibhai, F.J.; Connell, B.J.; Saleh, T.M.; Wilson, B.C.; Summerlee, A.J.S. Relaxin peptide hormones are protective during the early stages of ischemic stroke in male rats. Endocrinology 2015, 156, 638–646.
- Chhunchha, B.; Singh, P.; Stamer, W.D.; Singh, D.P. Prdx6 retards senescence and restores trabecular meshwork cell health by regulating reactive oxygen species. Cell Death Discov. 2017, 3, 17060.
- Martin, B.; Pearson, M.; Brenneman, R.; Golden, E.; Wood, W.H., 3rd; Prabhu, V.; Becker, K.G.; Mattson, M.P.; Maudsley, S. Gonadal transcriptome alterations in response to dietary energy intake: Sensing the reproductive environment. PLoS ONE 2009, 4, e4146.
- Gao, Z.; Lei, W.I.; Lee, L.T.O. The Role of Neuropeptide-Stimulated cAMP-EPACs Signalling in Cancer Cells. Molecules 2022, 27, 311.
- Yousufuddin, M.; Young, N. Aging and ischemic stroke. Aging 2019, 11, 2542–2544.
- Podolak, A.; Woclawek-Potocka, I.; Lukaszuk, K. The Role of Mitochondria in Human Fertility and Early Embryo Development: What Can We Learn for Clinical Application of Assessing and Improving Mitochondrial DNA? Cells 2022, 11, 797.
- DePina, A.S.; Iser, W.B.; Park, S.S.; Maudsley, S.; Wilson, M.A.; Wolkow, C.A. Regulation of Caenorhabditis elegans vitellogenesis by DAF-2/IIS through separable transcriptional and posttranscriptional mechanisms. BMC Physiol. 2011, 11, 11.
- Martin, B.; Golden, E.; Carlson, O.D.; Egan, J.M.; Mattson, M.P.; Maudsley, S. Caloric restriction: Impact upon pituitary function and reproduction. Ageing Res. Rev. 2008, 7, 209–224.
- Ivell, R.; Kotula-Balak, M.; Glynn, D.; Heng, K.; Anand-Ivell, R. Relaxin family peptides in the male reproductive system--a critical appraisal. Mol. Hum. Reprod. 2011, 17, 71–84.
- Wilson, B.C.; Burnett, D.; Rappaport, R.; Parry, L.J.; Fletcher, E.K. Relaxin-3 and RXFP3 expression, and steroidogenic actions in the ovary of teleost fish. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2009, 153, 69–74.
- McGowan, B.M.; Stanley, S.A.; Donovan, J.; Thompson, E.L.; Patterson, M.; Semjonous, N.M.; Gardiner, J.V.; Murphy, K.G.; Ghatei, M.A.; Bloom, S.R. Relaxin-3 stimulates the hypothalamic-pituitary-gonadal axis. Am. J. Physiol. Endocrinol. Metab. 2008, 295, E278–E286.
- Pomrenze, M.B.; Millan, E.Z.; Hopf, F.W.; Keiflin, R.; Maiya, R.; Blasio, A.; Dadgar, J.; Khazaria, V.; De Guglielmo, G.; Crawford, E.; et al. A transgenic rat for investigating the anatomy and function of corticotrophin releasing factor circuits. Front. Neurosci. 2015, 9, 487.
- Kilpatrick, D.G.; McAlhany, D.A.; McCurdy, R.L.; Shaw, D.L.; Roitzsch, J.C. Aging, alcoholism, anxiety, and sensation seeking: An exploratory investigation. Addict. Behav. 1982, 7, 97–100.
- Matthews, D.B.; Imhoff, B.M. Age modifies the effect of ethanol on behavior: Investigations in adolescent, adult and aged rats. Int. Rev. Neurobiol. 2022, 161, 251–275.
- Leber, W.R.; Parsons, O.A. Premature aging and alcoholism. Int. J. Addict. 1982, 17, 61–88.
- Topiwala, A.; Ebmeier, K.P. Effects of drinking on late-life brain and cognition. Evid. Based Ment. Health 2018, 21, 12–15.
- Sullivan, E.V.; Pfefferbaum, A. Brain-behavior relations and effects of aging and common comorbidities in alcohol use disorder: A review. Neuropsychology 2019, 33, 760–780.
- León, B.E.; Kang, S.; Franca-Solomon, G.; Shang, P.; Choi, D.S. Alcohol-Induced Neuroinflammatory Response and Mitochondrial Dysfunction on Aging and Alzheimer’s Disease. Front. Behav. Neurosci. 2022, 15, 778456.
- Nunes, P.T.; Kipp, B.T.; Reitz, N.L.; Savage, L.M. Aging with alcohol-related brain damage: Critical brain circuits associated with cognitive dysfunction. Int. Rev. Neurobiol. 2019, 148, 101–168.





