Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Agnieszka Potęga | -- | 4487 | 2022-08-25 13:02:27 | | | |
| 2 | Vivi Li | -1 word(s) | 4486 | 2022-08-26 04:44:22 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Potęga, A. Glutathione-Mediated Conjugation of Anticancer Drugs. Encyclopedia. Available online: https://encyclopedia.pub/entry/26499 (accessed on 23 May 2026).
Potęga A. Glutathione-Mediated Conjugation of Anticancer Drugs. Encyclopedia. Available at: https://encyclopedia.pub/entry/26499. Accessed May 23, 2026.
Potęga, Agnieszka. "Glutathione-Mediated Conjugation of Anticancer Drugs" Encyclopedia, https://encyclopedia.pub/entry/26499 (accessed May 23, 2026).
Potęga, A. (2022, August 25). Glutathione-Mediated Conjugation of Anticancer Drugs. In Encyclopedia. https://encyclopedia.pub/entry/26499
Potęga, Agnieszka. "Glutathione-Mediated Conjugation of Anticancer Drugs." Encyclopedia. Web. 25 August, 2022.
Copy Citation
The effectiveness of many anticancer drugs depends on the creation of specific metabolites that may alter their therapeutic or toxic properties. One significant route of biotransformation is a conjugation of electrophilic compounds with reduced glutathione, which can be non-enzymatic and/or catalyzed by glutathione-dependent enzymes. Glutathione usually combines with anticancer drugs and/or their metabolites to form more polar and water-soluble glutathione S-conjugates, readily excreted outside the body. In this regard, glutathione plays a role in detoxification, decreasing the likelihood that a xenobiotic will react with cellular targets.
anticancer drugs
glutathione
mechanisms of glutathione conjugation reaction
detoxification
bioactivation
1. Introduction: The Place of Glutathione in Drug Metabolism
Living organisms are continuously and unavoidably exposed to xenobiotics, including anticancer drugs. Many of these compounds are lipophilic in nature, and the body can only expel them through chemical modifications, known as biotransformations. Generally, these occur by mechanisms conveniently categorized as phase I and phase II metabolic transformations, which act in a tightly integrated manner [1][2]. Phase I metabolism alters the chemical structure of the drug by ‘functionalization reactions’ which introduce a polar functional group onto the molecule. Thus, phase I reactions are usually oxidation, reduction, dealkylation, epoxidation, oxidative deamination reactions, etc., which are primarily catalyzed by enzymes of the cytochrome P450 (P450; E.C. 1.14.-.-.) superfamily [1][3]. These reactions typically result in metabolites that are more water-soluble than the parent compounds and still retain some of their pharmacological activity [4]. Phase II metabolism generally serves as a detoxifying step in the metabolism of drugs and other xenobiotics and involves ‘conjugation reactions’ that couple the drug and/or its phase I metabolite molecule with an activated conjugating molecule. Conjugation usually renders the compound pharmacologically inert, more polar, and water soluble, so it can easily be excreted out of the body. The most prevalent phase II metabolic pathways include glucuronidation, sulfation, and glutathione conjugation [1][5]. The concept of two phases can also be extended to phase III, which is the excretion of the resulting phase II metabolites (conjugates) [6].
Although metabolic transformations aim to inactivate drugs, some chemical modifications, both in phase I and II metabolism, may also lead to the formation of highly reactive species, i.e., reactive drug metabolites, which are more pharmacologically active and/or toxic in comparison to the original compound [4][7]. Therefore, early knowledge about pharmacological and/or toxicological effects of metabolites of drug candidates is extremely desired for assessing their bioavailability, activity, and safety profiles in humans. In this regard, much attention is also paid to understanding the mechanisms of reactive metabolite generation.
This work is focused on the conjugation reaction of xenobiotics with the reduced tripeptide glutathione (GSH = L-γ-glutamyl-L-cysteinyl-glycine), which is a significant route of drug elimination in phase II metabolism in many species [8]. GSH conjugation reaction may be non-enzymatic (i.e., chemical), but it is significantly accelerated by various GSH-dependent enzymes [9][10], mainly a family of GSH S-transferase (GST; E.C. 2.5.1.18) enzymes [11]. A number of anticancer therapeutics are known to undergo GSH conjugation reaction [12][13][14][15][16][17]. GSH can combine with electrophilic anticancer drugs and/or their phase I metabolites to form less toxic compounds than parent compounds and more polar, water-soluble GSH S-conjugates (thioethers). Further, GSH S-conjugates are substrates for transporters (phase III) involved in the biliary and renal excretion, which facilitates their clearance from the body [18]. Thus, GSH plays a role in detoxification, decreasing the likelihood that a xenobiotic will react with molecular and cellular targets. This may affect the efficacy and interactions of a variety of anticancer interventions. Moreover, numerous studies support the view that, in some cases, GSH conjugation may also play an important role in bioactivation reactions as it is able to generate GSH S-conjugates that are more active than the original parent xenobiotic [18][19][20]. If these reactive metabolites interact with critical cellular macromolecules (i.e., proteins, nucleic acids), toxicity can be ensued. By participating in the formation of toxic metabolites of some anticancer drugs, GSH may also affect the cellular uptake of other agents. For this reason, knowledge of these interactions may be useful in designing combination therapy for neoplastic diseases.
Given this background, the study on GSH conjugation is a crucial factor for the determination of drug therapeutic efficacy and potential toxicity. Depending on the structure and physico-chemical properties of the substrate, the conjugation reaction may proceed according to various mechanisms. In this entry, I discuss some of them in the context of anticancer drugs. In addition, I also indicate the potential significance of GSH conjugation in the detoxification and bioactivation of clinically important anticancer drugs.
To continue the search for potential anticancer drugs, a completely novel class of acridine derivatives of extraordinary structures, unsymmetrical bisacridines (UAs), has been synthesized and developed by researchers' group [21][22]. They were characterized with respect to their physico-chemical properties [23] and the role of phase I and phase II metabolic transformations in their action [24][25]. UAs exhibited high cytotoxic activity against a lot of cancer cell lines and high anticancer efficacy against several types of human cancer xenografts in nude mice [26]. These are mainly cancers that are extremely resistant to chemotherapy and are usually characterized by increased activity of various GST isoenzymes relative to normal tissues. Preliminary studies with rat liver microsomal and cytosolic subfractions showed the ability of C-2028 (9-{N-[(imidazo [4,5,1-de]-acridin-6-on-5-yl)aminopropyl]-N-methylaminopropylamino}-1′-nitroacridine, a representative UA) to GST-mediated and/or direct GSH conjugation [25]. This finding may suggest an important role of detoxifying transformations in UA metabolism, the consequences of which for the anticancer and/or toxic activities of the compounds are not yet well understood. Herein, I would like to present some considerations on this issue and indicate the likely mechanism of UA-GSH S-conjugate formation.
2. Glutathione
2.1. Glutathione Structure as a Determinant of Its Biological Functions
Glutathione is the predominant intracellular thiol-containing tripeptide found in all animal tissues, plants, fungi, and some microorganisms [27][28]. Under physiological conditions, it is mainly present in the cytoplasm in the reduced form (GSH), which is also the biologically active form. GSH is less easily oxidized than its precursors, cysteine and γ-glutamylcysteine; the fully oxidized form with a disulfide between two identical GSH molecules (GSSG) represents less than 1% of the total GSH pool in the cell [29]. GSH concentration in human cells typically ranges from 0.1 to 10 mmol/L, being most focused in the liver (up to 10 mmol/L), spleen, kidney, lens, erythrocytes, and leukocytes [30][31], wherein its depletion and/or altered level are associated with various diseases, including cancer, cardiovascular, inflammatory, immune, metabolic, and neurodegenerative diseases [32]. Maintaining optimal GSH:GSSG ratios in the cell is critical to survival; hence, tight regulation of this system is imperative [33].
The characteristic structural features of the GSH molecule (Figure 1) determine its many diverse biological functions. In contrast to an α-peptide linkage usually found in a number of endogenous peptides, the L-glutamic acid (Glu) and L-cysteine (Cys) of GSH are joined by an unusual γ-peptide linkage [28]. Such a bond promotes GSH resistance to hydrolysis by most intracellular aminopeptidases as well as provides for specificity in other GSH-enzyme interactions [34]. In turn, the activity of the high electron-donating sulfhydryl (thiol, -SH) group of Cys residue supports the reducing properties of GSH by way of a thiol-exchange system (-SH to -S-S-), enabling the participation of GSH in intracellular antioxidative and detoxifying reactions [35]. The reactivity of -SH is due to the thiolate anion (S−), the relative concentration of which is regulated by the acidity of thiol (pKa = 9.2). At physiological pH, for every 100 GSH molecules in the -SH state, approximately 3.7 are in a thiolate form [36]. Due to the polarizability of the sulfur atom, GSH is a strong ‘soft’ nucleophile, and unlike other phase II enzyme cofactors such as uridine 5′-diphosphoglucuronic acid (UDPGA) and 3′-phosphoadenosine-5′-phosphosulfate (PAPS), it easily reacts with various ‘soft’ electrophiles [10], which may also be anticancer drugs. Net-negative charge of cysteinyl residue and overall GSH hydrophilicity greatly increase the aqueous solubility of the lipophilic moieties with which it becomes conjugated. GSH S-conjugates usually achieve a molecular weight higher than 300–500 g/mol (average molecular weight of GSH = 307.3235 g/mol) and are thus preferentially secreted via the biliary system. Then, the final cysteinyl conjugates are reabsorbed into the liver, from where they travel to the kidney for acetylation and excretion as a mercapturic acid [37].
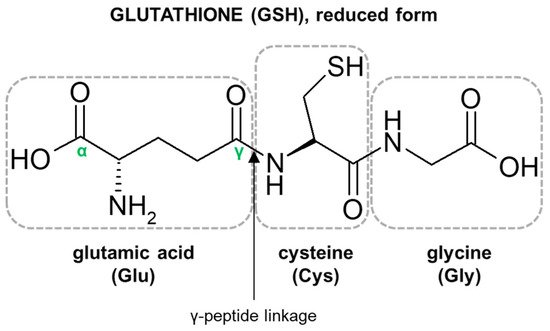
Figure 1. Structure of the tripeptide glutathione (GSH). Glutamic acid (Glu) is linked in a γ-peptide linkage (via its γ-carboxyl group) to cysteine (Cys), which in turn forms an α-peptide linkage with glycine (Gly).
2.2. Biological Functions of Glutathione
Reduced GSH has been adopted through evolution to perform multiple significant cellular functions in living organisms. It is responsible for the correct thiol–disulfide balance and the associated oxidation-reduction potential of cells [38]. The biologically important role of GSH is related to the possibility of regeneration of the -SH moieties of proteins, which counteracts the effects of oxidative reactions, inactivating cell proteins [39]. This compound is also involved in the reduction of ribonucleotides to deoxyribonucleotides, i.e., it has a direct impact on DNA biosynthesis and the related proliferation process [8]. Moreover, it mediates in the synthesis of proteins and in amino acid transport [40]. Further, GSH serves as a reservoir and transporter of cysteine [41], a regulator of calcium ion homeostasis [42], a versatile cofactor for many cytoplasmic enzymes [8], and it is a link in the mitochondrial mechanism to cell death [43].
In addition to the functions mentioned, GSH is an important component of the system that detoxifies both electrophilic xenobiotics and metabolically produced free radicals, i.e., reactive oxygen species (ROS), by the formation of GSH S-conjugates. Thus, it plays a central role in the protection of cells against a variety of exogenous and endogenous potentially harmful compounds [28][44]. The reactions to form GSH S-conjugates may be non-enzymatic, although they are greatly accelerated by GSH-dependent enzymes such as GSTs [11]. Hence, the effectiveness of the detoxification pathway depends upon the intracellular concentration of GSH, the presence of GSTs of appropriate specificity, and/or the capacity of the cell for rapid resynthesis of GSH [45]. Conjugation reactions of GSH with electrophilic compounds to GSH S-conjugates occur mainly in the liver, which exports GSH and has the highest GST activity [28]. Although by conjugation with GSH many compounds are rendered less toxic than the original parent xenobiotic, it has also been reported that some drugs, including those with anticancer activity, become more reactive following this reaction. Thus, GSH conjugation may also play an important role in drug bioactivation processes [18][19][20].
3. Overview of Mechanisms of Glutathione Conjugation of Anticancer Drugs
As previously stated, the important part of the GSH molecule in terms of its chemical activity is the nucleophilic sulfhydryl (thiol, -SH) group of the cysteine residue. Generally, as illustrated in Figure 2, conjugation with GSH involves attachment of the xenobiotic molecule or its phase I metabolite (assigned as R-X) with this group to form the GSH S-conjugate (assigned as R-SG). In fact, the reactive nucleophilic species is the thiolate anion of GSH (GS−), whose concentrations at physiological pH are approximately 1% of GSH concentration (pKa of GSH = 9.2) [38]. Due to the low electronegativity and high polarizability of the sulfur atom, GSH acts as a ‘soft’ nucleophile and, as such, can be used to sense the reactivity of ‘soft’ electrophiles (i.e., compounds that contain an acceptor atom with high polarizability, low electronegativity, and often unshared p or d valence electrons) [46]. Xenobiotics that are conjugated with GSH are either very electrophilic right away or are metabolized to electrophilic compounds. Some reactions of the tripeptide GSH with cellular electrophiles have spontaneous rates which vary considerably depending on the reactivity of the electrophile, and frequently, but not always, are catalyzed by various GST isoenzymes [34]. However, non-enzymatic reactions are usually much slower than those catalyzed by GSTs.
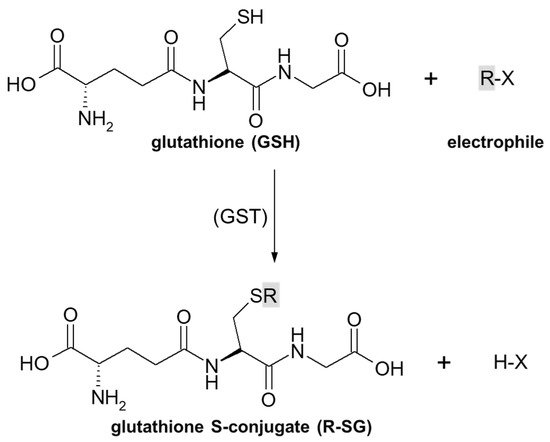
Figure 2. General scheme of glutathione (GSH) conjugation to a generic electrophile (R-X) to form the glutathione S-conjugate (R-SG). The reaction may be catalyzed by the enzyme glutathione S-transferase (GST).
3.1. Nucleophilic Substitution
Substitution reactions, which involve the reaction of a nucleophile with an electrophile, are prevalent in physiological and metabolic processes, in the action of some drugs, and in the chemical synthesis of nearly all drugs [47]. Thus, nucleophilic substitution is also the basic and the most widespread mechanism of GSH conjugation of electrophilic compounds. It is observed in several families of anticancer drugs, including alkyl, allylic, benzylic, and aryl halides, nitrogen mustard derivatives, or platinum complexes [33]. Electrophiles are positively charged or have a polarized bond with a partial positive character. Electrophiles capable of undergoing substitution reactions have a leaving group—a species that can accept and stabilize the pair of electrons that make up the bond being broken [47].
A common type of nucleophilic substitution reaction is the bimolecular nucleophilic substitution reaction, or SN2 reaction for short, where one atom or functional group is replaced with an electronegative GSH molecule (the frame in Figure 3). In this case, bond formation and bond breaking occur simultaneously and the leaving group tends to be a weaker base than the nucleophile. Halide ions, such as I−, Br−, and Cl−, are very good leaving groups and thus give fast reactions [48].
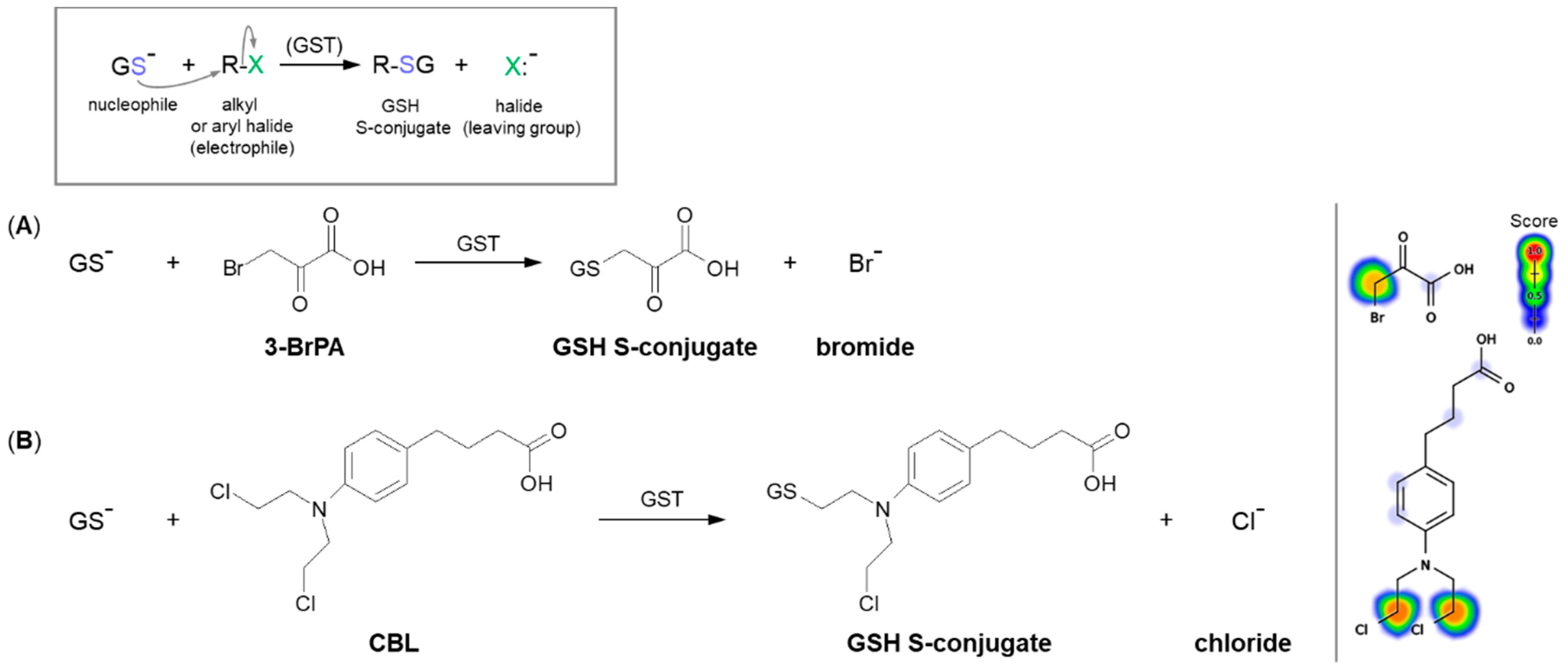
Figure 3. Reaction schemes of nucleophilic substitution of the thiolate anion of GSH (GS–) to the halogen atom (leaving group) in (A) 3-bromopyruvic acid (3-BrPA) [17] and (B) chlorambucil (CBL) [49]. The frame shows the general scheme of the GSH conjugation reaction. Predicted bioactive sites of GSH conjugation for anticancer drugs were obtained by XenoSite Reactivity Predictor available at https://swami.wustl.edu/xenosite/p/reactivity (accessed on 18 May 2022).
3.1.1. Halogen Atom as a Leaving Group
The SN2 reaction mechanism of alkyl halide with GSH can be illustrated by the conjugation of 3-bromopyruvic acid (3-BrPA) with GSH (Figure 3A). 3-BrPA is the brominated derivative of pyruvic acid with proven anticancer activity against many different cancers in children and adults [50][51]. Like other α-bromoketones, it is widely known as a strong alkylating agent with a high affinity for protein binding as well as an antimetabolite [17]. Moreover, being an inhibitor of key glycolysis enzymes, including hexokinase II [52] and glyceraldehyde 3-phosphate dehydrogenase [53], 3-BrPA inhibits the growth of neoplastic cells that perform aerobic glycolysis known as the ‘Warburg effect’ [54]. Additionally, it also selectively blocks mitochondrial oxidative phosphorylation, angiogenesis, and energy production in cancer cells [54][55].
Based on a chemical view, the thiolate anion of GSH (GS−) easily attacks the carbon atom to which the bromine atom is attached. This reaction occurs because of the imbalance of the electron density between the carbon and halogen since it is a polar covalent bond. The more electronegative bromine atom pulls the electron density, thus making the carbon partially positively charged (an electrophilic center) and susceptible to a nucleophilic GS− attack [56]. A bromine atom built into the structure of the 3-BrPA molecule is a good leaving group because the negatively charged bromine atom (bromide) is stable enough to exist on its own when it leaves the molecule. Hence, the conjugation to GSH does not require any prior metabolic activation of the parent compound. 3-BrPA was reported to form GSH S-conjugate both under GST catalysis and also in an enzyme-free system. Further, it is eliminated through the mercapturic acid synthesis pathway where it is excreted from the cell by ATP-binding cassette transporter proteins [17][57].
Another example of a similar SN2 reaction is the GSH conjugation of chlorambucil (CBL; the brand name: Leukeran) (Figure 3B). CBL is an alkylating agent approved for use in various malignant and non-malignant neoplasms, such as chronic lymphocytic leukemia [58], lymphosarcoma [59], and giant follicular lymphoma [60]. In the presence of GSH, CBL behaves as an efficient substrate for GSTA1-1 and GSTP1-1 isoenzymes [49][61]. As with the 3-BrPA, the formation of the corresponding GSH S-conjugate undergoes without prior CBL activation as the negatively charged chlorine atom (chloride) is a good enough leaving group. Moreover, kinetic data suggest that the rate-limiting of the catalytic reaction between CBL and GSH is the reaction product release [49].
3.1.2. Tensioned Ring-Opening Reaction
The next type of GSH conjugation mechanism is the attachment of the thiolate anion (GS–) to the epoxide, four-membered lactone, or three-membered aziridine resulting in ring-opening (Figure 4). Such reactions take place quite easily because the rings are composed of three or four carbon atoms which are highly tensed and their energy is quite high. Additionally, such a process will be easier, if the tensed system includes a heteroatom, because it will inductively decrease the electron density on adjacent carbon atoms [62][63]. Under basic conditions, ring-opening occurs by an SN2 mechanism, and the less substituted carbon is the site of GS– nucleophilic attack. The described transformation can take place stereoselectively, depending on the GST isoenzyme that catalyzes a reaction.
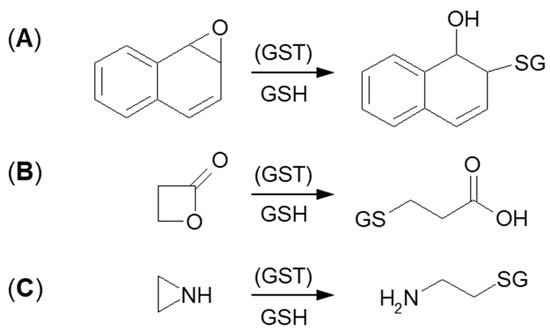
Figure 4. GSH conjugation via opening the tensioned ring of (A) epoxy, (B) lactone, and (C) aziridine.
Formation of GSH S-conjugate, accompanied by the aziridine ring-opening, takes place, e.g., in the case of cyclophosphamide and thiotepa. Cyclophosphamide (CP; the brand name: Cytoxan) belongs to the alkylating agent and nitrogen mustard family of medications [64][65]. Its mechanism of action, quite similar to that of chlorambucil, relies on interfering with the duplication of DNA and RNA synthesis [66]. It is a chemotherapeutic approved to treat malignant lymphomas, neuroblastoma, multiple myeloma, leukemias, ovarian, breast, and many other cancers [67]. Such a wide spectrum of clinical uses makes it an essential component of numerous combination chemotherapeutic regimens. Moreover, CP is also used to suppress the immune system [68].
A prerequisite for conjugation with GSH is the presence of a reactive electrophilic metabolite(s). Unlike 3-BrPA and CBL, CP must undergo previous metabolic activation by hepatic microsomal mixed-function oxidases (i.e., P450s, mainly 2B6, 2C8, and 2C9) (Figure 5) [69][70]. The generally accepted mechanism for the generation of active metabolites of CP involves oxidation to the primary 4-hydroxycyclophosphamide (4-OH-CP) metabolite, which stays in tautomeric equilibrium with the ring-opened aldophosphamide (APA). Then, non-enzymatic cleavage of APA results in the formation of two toxic species—phosphoramide mustard (PAM) and acrolein. PAM is believed to unfold cell toxicity by DNA alkylation [71] while acrolein is held to be responsible for some aspects of host toxicity, such as hemorrhagic cystitis [72]. Therefore, GSH conjugation can result in the formation of three types of GSH S-conjugates, i.e., 4-monoglutathionyl CP (4-GSCP) (1), mono- (2) and diglutathionyl PAMs (3) [13][73]. The formation of 4-GSCP was found to be reversible, and by APA hydrolysis, PAM was formed. Thus, 4-GSCP can be considered a stable reservoir for the generation of PAM that subsequently undergoes two consecutive GSH conjugations. The reaction with nucleophilic GSH was established to proceed through the positively charged and highly polarized aziridinium ion (aziridinium intermediate) that can be opened in both the enzyme-catalyzed and the chemical reactions. On the other hand, the second GSH conjugation reaction was shown to occur through the direct displacement of chloride [13]. The 4-GSCP formation reaction can be catalyzed by various GSTs isoenzymes, whereas GST1A-1 has the greatest effect on the rate of monoglutathionyl PAM formation. Melphalan, mechlorethamine, ifosfamide, carmustine, lomustine, and nimustine are examples of other anticancer-active nitrogen mustard derivatives that react with GSH in a manner similar to that described for CP [13][70].
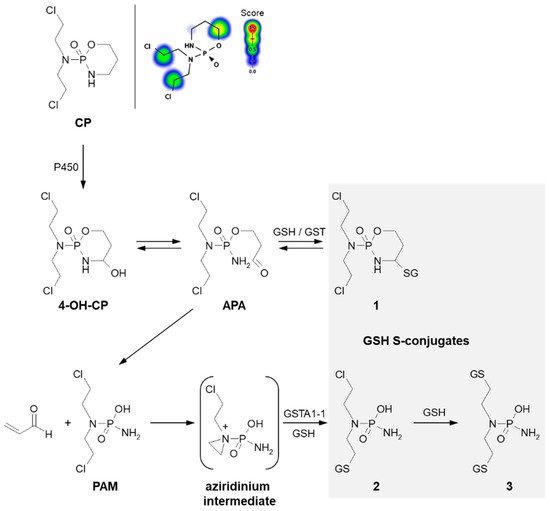
Figure 5. Scheme of cyclophosphamide (CP) activation and drug reaction with GSH [64]. Possible GSH S-conjugates: 1: 4-monoglutathionyl cyclophosphamide, 2: monoglutathionyl phosphoramide mustard, 3: diglutathionyl phosphoramide mustard. 4-OH-CP = 4-hydroxycyclophosphamide; APA = aldophosphamide; PAM = phosphoramide mustard; P450 = cytochrome P450. Predicted bioactive sites of GSH conjugation for anticancer drug were obtained by XenoSite Reactivity Predictor available at https://swami.wustl.edu/xenosite/p/reactivity (accessed on 18 May 2022).
Thiotepa (N,N′,N″-triethylenethiophosphoramide; the brand name: Tepadina) also acts as an alkylating agent [74]. Being active against a wide variety of cancers, it is commonly used in combination with other chemotherapeutic agents to treat ovarian cancer, bladder cancer, and breast cancer [75][76]. Chemically, it is an organophosphorus compound containing a four-coordinated phosphorus atom and three aziridine moieties (Figure 6) through which the drug probably induces crosslinks with DNA, interfering with DNA replication and cell division [77]. The major metabolite formed from thiotepa during P450-catalyzed transformation (i.e., desulfuration reaction) is N,N′,N″-triethylenephosphoramide (tepa) [78]. In turn, when reacting with GSH, the tensioned ring of thiotepa containing a nitrogen atom as a heteroatom is opened, leading to the formation of mono- (1) and diglutathionyl thiotepa (2). In the metabolic pathway of thiotepa, 2-aminoethyl GSH (3), which is the product of the direct GSH conjugation of aziridine, was also characterized. The results confirmed that only thiotepa but not its monoglutathionyl S-conjugate is a substrate for GSTs (mainly A1-1 and P1-1 isoenzymes). Moreover, the non-enzymatic reaction of the aziridinium moieties of thiotepa with GSH is strongly dependent on the pH, and the yield of the reaction is greatest at low pH [79].
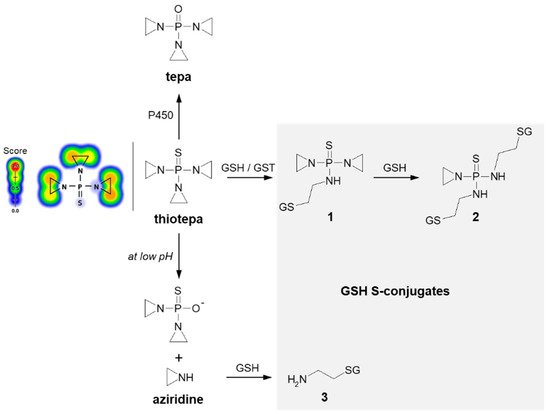
Figure 6. Scheme of thiotepa activation and drug reaction with GSH [79]. Possible GSH S-conjugates: 1: monoglutathionyl thiotepa, 2: diglutathionyl thiotepa, 3: 2-aminoethyl glutathione. P450 = cytochrome P450. Predicted bioactive sites of GSH conjugation for anticancer drug were obtained by XenoSite Reactivity Predictor available at https://swami.wustl.edu/xenosite/p/reactivity (accessed on 18 May 2022).
3.2. Nucleophilic Addition (Michael Addition)
Some reactions leading to the formation of GSH S-conjugates occur through Michael addition (or conjugate addition). Such a mechanism applies to α,β-unsaturated compounds (the so-called Michael acceptors) characterized by having carbon–carbon double (C=C) or carbon–carbon triple (C≡C) bonds with a strongly electron-withdrawing substituent(s) (e.g., a carbonyl, carboxyl, or nitro group) (Figure 7) [80]. This results in a polarizable electron density at the π bond, where the β-carbon atom (β-C) is positively polarized and becomes the preferred site of an attack of a soft nucleophile (Michael donor), e.g., the thiol group of cysteine in GSH. Although many of them can form thioethers non-enzymatically, GST-catalyzed Michael addition is much faster [10][81]. Compounds possessing Michael acceptor units feature a broad spectrum of bioactivity. They are considered to be particularly reactive and are thus capable of bonding with biological macromolecules [82].
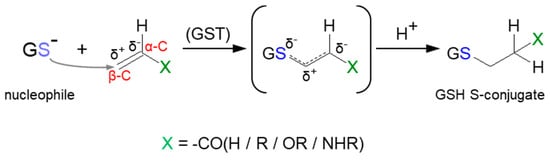
Figure 7. General scheme of Michael addition (or conjugate addition) to α,β-unsaturated compound with carbon−carbon double (C=C) bond.
Mitoxantrone (MTX; the brand name: Novantrone), a synthetic anthraquinone antineoplastic agent, is an example of a compound that undergoes GSH conjugation by Michael addition. MTX is a potent type II topoisomerase inhibitor that disrupts DNA synthesis and DNA repair in both healthy cells and cancer cells by intercalation between DNA bases [83]. It is commonly applied in the treatment of breast [84] and prostate [85] cancers, lymphomas [86], and leukemias, primarily acute myeloid leukemia [87], with excellent efficacy.
In the case of MTX, at least two GSH conjugation pathways were identified (Figure 8). MTX is known to resist reductive enzymatic activation but is subject to facile oxidative enzymatic action [88]. The development of GSH-dependent resistance provides further evidence that the oxidative activation may be a relevant mode of drug action. The in vivo and in vitro studies of Mewes et al. [88] performed using minipigs, cultured rat hepatocytes, and human HepG2 hepatoma cells, respectively, revealed the formation of a major monoglutathionyl MTX (1) and its various degradation products. The ability of MTX to react with GSH enables the formation of an MTX quinone derivative by two-electron oxidation of the parent drug, which is in the form of a hydroquinone. Additionally, there are also data confirming the possibility of the formation of another MTX-GSH S-conjugate (2) [83]. Following the MTX enzymatic oxidation process within the aromatic ring containing polyamine side chains, the intramolecular Michael addition occurs. Presumably, the mechanism of this GSH conjugation reaction takes place via a labile iminium ion intermediate which activates the aromatic ring towards the attack of the external cellular nucleophile (e.g., GSH, DNA).
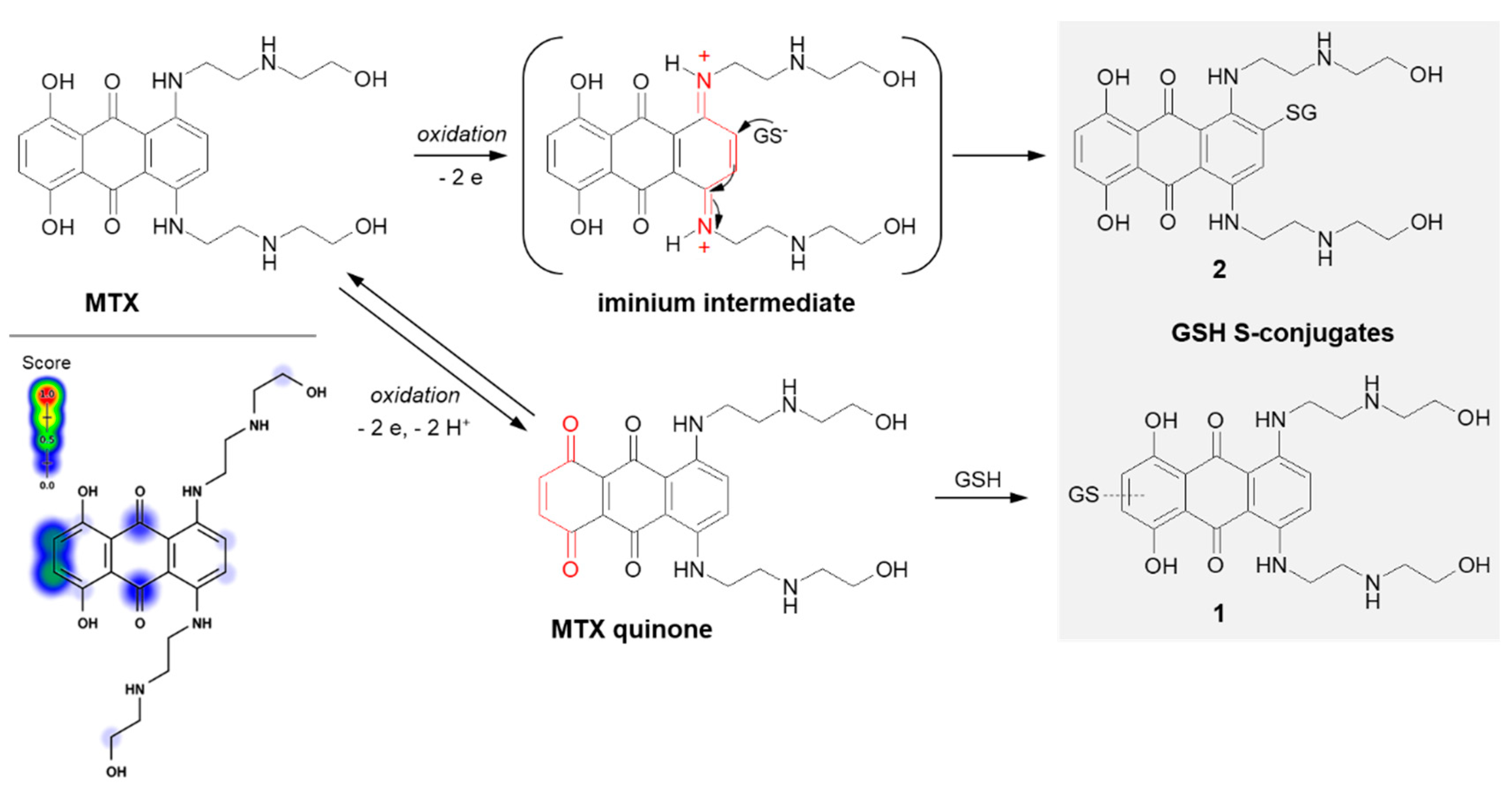
Figure 8. Scheme of mitoxantrone (MTX) activation and drug reaction with GSH [16][83]. The Michael acceptor moieties are marked in red. Predicted bioactive sites of GSH conjugation for anticancer drug were obtained by XenoSite Reactivity Predictor available at https://swami.wustl.edu/xenosite/p/reactivity (accessed on 18 May 2022).
Another example of a Michael addition-type reaction is infigratinib (INF, NVP-BGJ398). It is a novel small-molecule chemotherapeutic drug used for first-line treatment of advanced or metastatic cholangiocarcinoma (bile duct cancer). It was discovered that INF inhibits human fibroblast growth factor receptors (FGFRs), which are a family of receptor tyrosine kinases that may be upregulated in different cancer cell types [89]. For this reason, it is an investigational drug under development for the treatment of patients with various FGFR-driven diseases [90][91]. Al-Shakliah et al. [92] detected at least three GSH S-conjugate metabolites of INF using liquid chromatography ion trap mass spectrometry (LC-ITMS). As shown in Figure 9, the halogenated benzene ring of the INF structure undergoes metabolic bioactivation sequentially by dechlorination, O-demethylation, and oxidation to form the reactive 1,4-benzoquinone intermediate that is attacked by GSH [92][93].
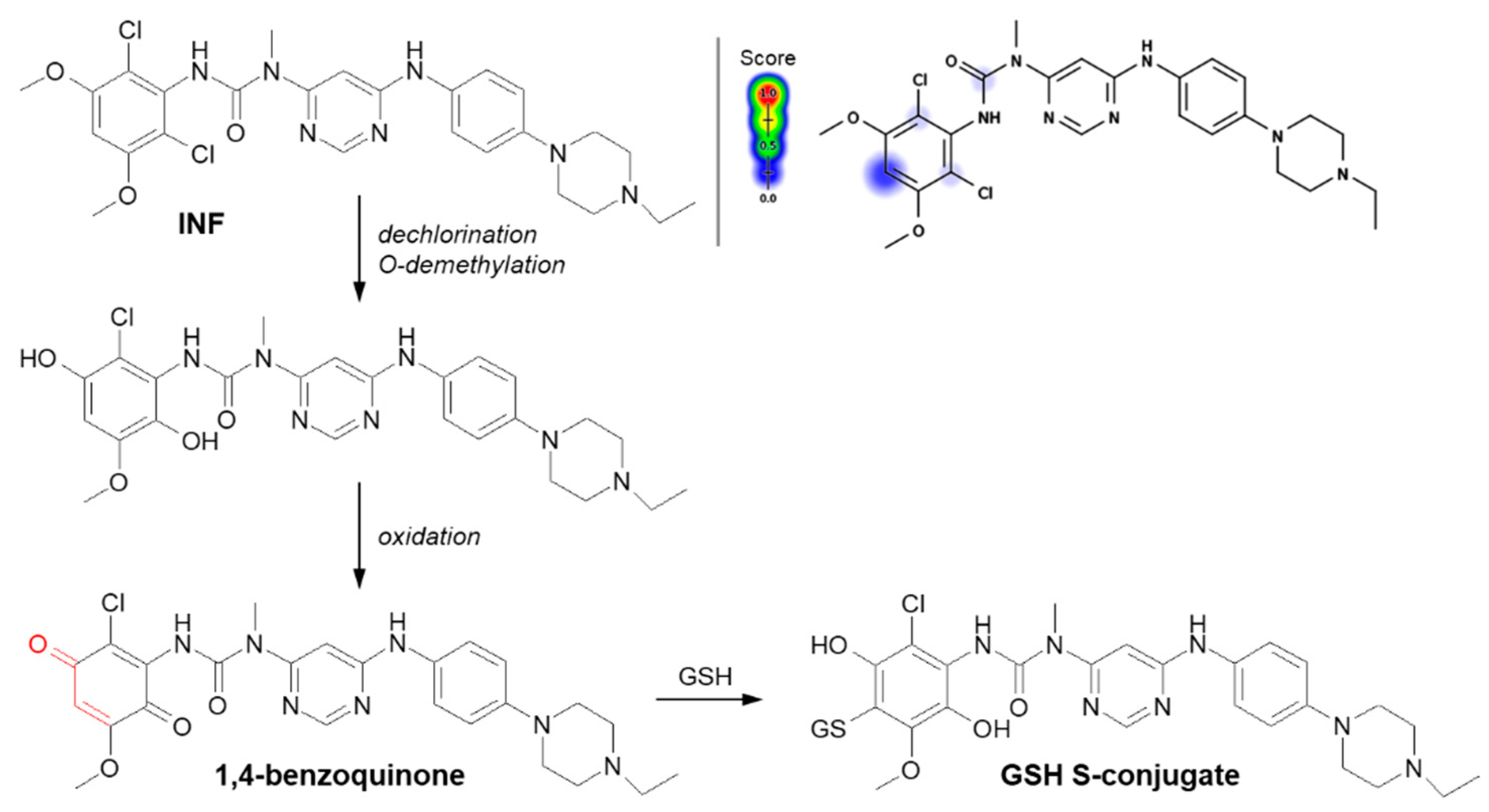
Figure 9. Reaction scheme of infigratinib (INF) conjugation with GSH [92]. A Michael acceptor moiety is marked in red. Predicted bioactive sites of GSH conjugation for anticancer drug were obtained by XenoSite Reactivity Predictor available at https://swami.wustl.edu/xenosite/p/reactivity (accessed on 18 May 2022).
The next example is helenalin, a natural sesquiterpene lactone present in a large number of species mostly from the Asteraceae family, which has a variety of observed effects in vitro, including anti-inflammatory and anticancer activities [94][95][96]. As the compound structure contains reactive Michael acceptor systems (i.e., α,β-unsaturated ketone moiety and α,β-unsaturated lactone moiety), it is able to easily react with GSH [97]. The 2β-mono- (1) and 2,13β-diglutathionyl (2) conjugates of helenalin were shown to be formed by spontaneous Michael addition at physiological pH (Figure 10A). Interestingly, these were found to inhibit GST from horse liver, while free helenalin showed no inhibitory activity [98].
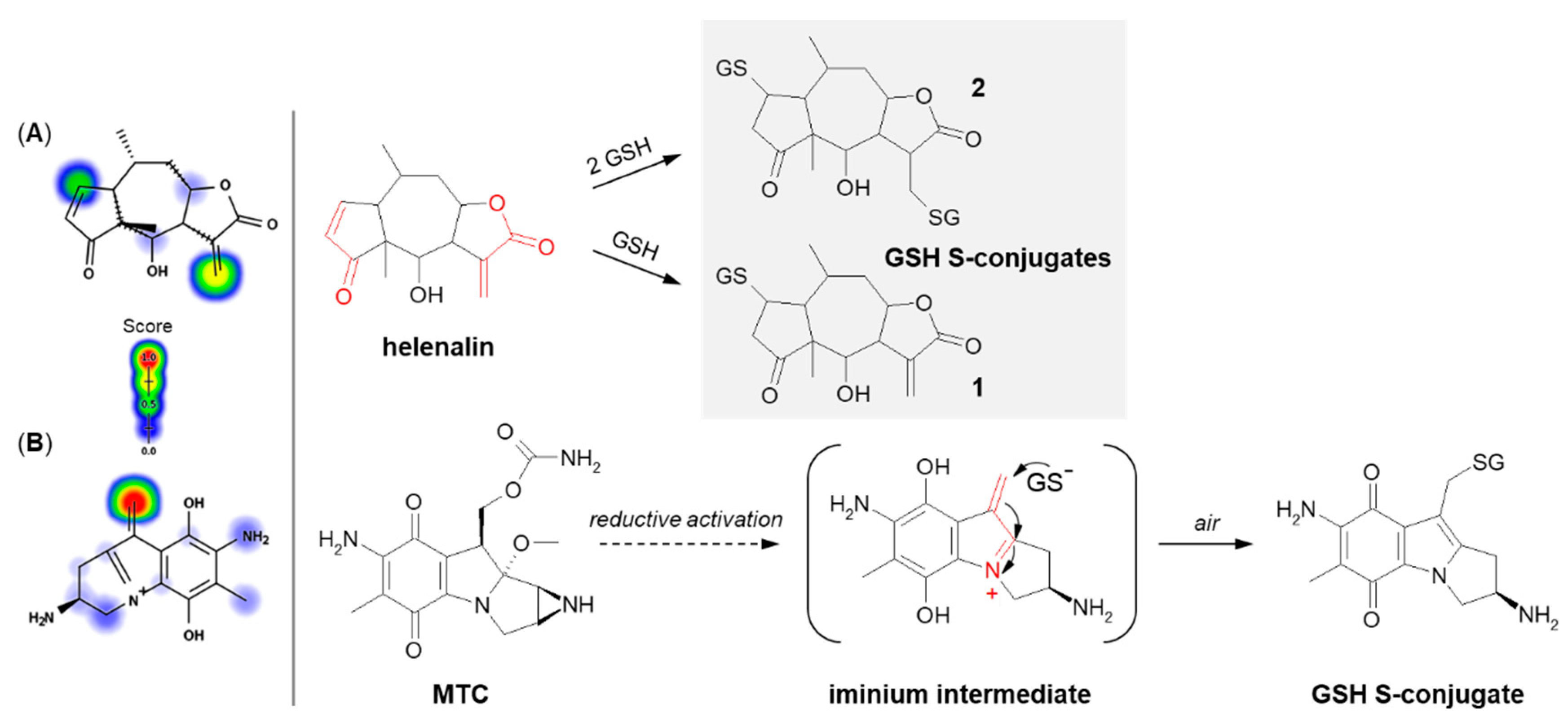
Figure 10. Reaction schemes of (A) helenalin [97] and (B) mitomycin C (MTC) [99] conjugation with GSH. The Michael acceptor moieties are marked in red. Predicted bioactive sites of GSH conjugation for anticancer drugs were obtained by XenoSite Reactivity Predictor available at https://swami.wustl.edu/xenosite/p/reactivity (accessed on 18 May 2022).
Mitomycin C (MTC) is a natural cytostatic antibiotic used as a chemotherapeutic agent by virtue of its anticancer activity. The mechanism of drug action is typical for that based on DNA alkylation [100][101]. Importantly, MTC requires previous activation via enzymatic reduction (bioreductive activation) to exert its biological effects. One-electron reduction steps to the corresponding semiquinone and then to hydroquinone initiates a cascade of consecutive reactions (i.e., spontaneous elimination of methanol from hydroquinone, elimination of the carbamate group, opening of the aziridine ring) that gives an unstable iminium intermediate which reacts with GSH through a Michael-type reaction [99][102] (Figure 10B). MTC was shown to form both mono- and diglutathionyl conjugates. It was also found that GSH itself did not reduce MTC, and unreduced drug did not form conjugates with GSH [103].
A few more examples of Michael acceptor-containing anticancer therapeutics metabolizing through GSH conjugation are afatinib, ibrutinib, and neratinib (Figure 11) [104][105]. They are all inhibitors of various tyrosine kinases and use their own Michael acceptor moiety for irreversible binding to a free cysteine residue of the targeted protein. In recent years, such a targeted covalent modification of regulatory proteins by Michael acceptors became recognized as a promising approach to drug discovery [106]. It can be expected that GSH plays an integral role in the clearance of these electrophilic drugs. Shibata and Chiba [104] showed that both afatinib and neratinib undergo extensive conjugation with GSH in buffer and cytosolic subfraction deriving from liver and kidney tissues, whereas ibrutinib has exhibited much lower degree of GSH/GST-dependent conjugation [104]. These findings may be helpful in optimizing pharmacokinetics in humans during the development stage of other targeted covalent inhibitors.
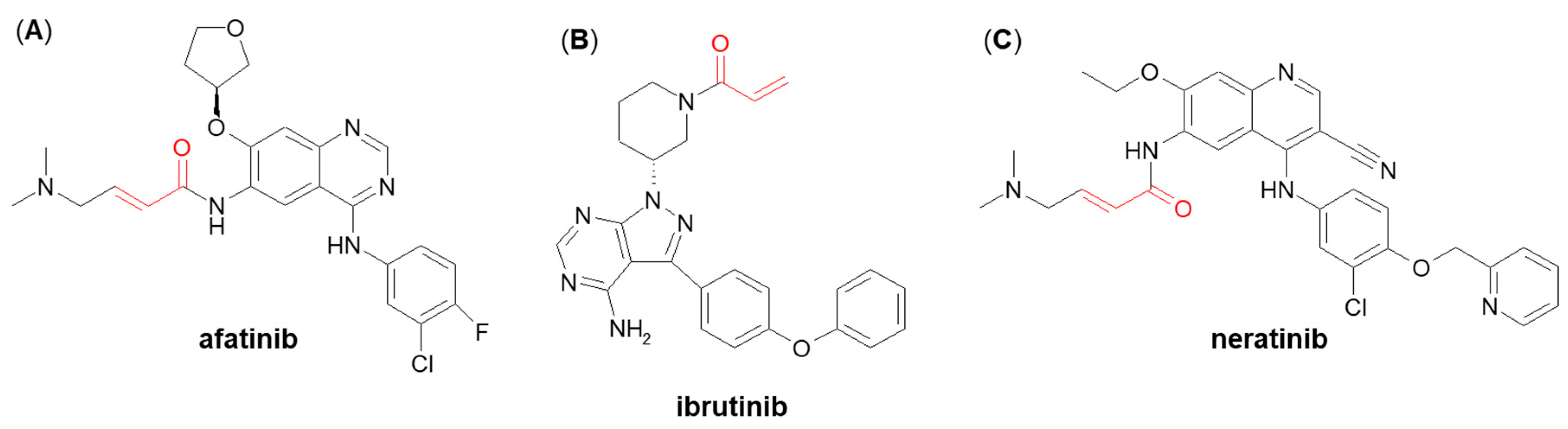
Figure 11. Chemical structures of (A) afatinib, (B) ibrutinib, and (C) neratinib. The Michael acceptor moieties are marked in red.
References
- Kumar, G.N.; Surapaneni, S. Role of drug metabolism in drug discovery and development. Med. Res. Rev. 2001, 21, 397–411.
- Bachmann, K. Chapter 8—Drug Metabolism. In Pharmacology: Principles and Practise, 1st ed.; Hacker, M., Messer, W., Bachmann, K., Eds.; Academic Press: San Diego, CA, USA, 2009; pp. 131–173.
- Penner, N.; Woodward, C.; Prakash, C. Drug Metabolizing Enzymes and Biotransformation Reactions. In ADME-Enabling Technologies in Drug Design and Development, 1st ed.; Zhang, D., Surapaneni, S., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2012; pp. 545–565.
- Baillie, T.A. Cytochrome P450 and Other Enzymes in Drug Discovery and Development. In Drug Metabolizing Enzymes, 1st ed.; Lee, J., Obach, R.S., Fisher, M.B., Eds.; CRC Press, LLC: Boca Raton, FL, USA, 2003; pp. 147–154.
- Jančová, P.; Šiller, M. Phase II Drug Metabolism. In Topics on Drug Metabolism; Paxton, J., Ed.; IntechOpen: London, UK, 2012; pp. 35–60.
- Döring, B.; Petzinger, E. Phase 0 and phase III transport in various organs: Combined concept of phases in xenobiotic transport and metabolism. Drug Metab. Rev. 2014, 46, 261–282.
- Attia, S.M. Deleterious effects of reactive metabolites. Oxidative Med. Cell. Longev. 2010, 3, 238–253.
- Lushchak, V.I. Glutathione homeostasis and functions: Potential targets for medical interventions. J. Amino Acids 2012, 2012, 736837.
- Satoh, K. The high non-enzymatic conjugation rates of some glutathione S-transferase (GST) substrates at high glutathione concentrations. Carcinogenesis 1995, 16, 869–874.
- Deponte, M. Glutathione catalysis and the reaction mechanisms of glutathione-dependent enzymes. Biochim. Biophys. Acta Gen. Sub. 2013, 1830, 3217–3266.
- Armstrong, R.N. Structure, catalytic mechanism, and evolution of the glutathione transferases. Chem. Res. Toxicol. 1997, 10, 2–18.
- Dirven, H.A.A.M.; Megens, L.; Oudshoorn, M.J.; Dingemanse, M.A.; Van Ommen, B.; Van Bladeren, P.J. Glutathione conjugation of the cytostatic drug ifosfamide and the role of human glutathione S-transferases. Chem. Res. Toxicol. 1995, 8, 979–986.
- Dirven, H.A.A.M.; Van Ommen, B.; Van Bladeren, P.J. Glutathione conjugation of alkylating cytostatic drugs with a nitrogen mustard group and the role of glutathione S-transferases. Chem. Res. Toxicol. 1996, 9, 351–360.
- Cnubben, N.H.; Rommens, A.J.; Oudshoorn, M.J.; Van Bladeren, P.J. Glutathione-dependent biotransformation of the alkylating drug thiotepa and transport of its metabolite monoglutathionylthiotepa in human MCF-7 breast cancer cells. Cancer Res. 1998, 58, 4616–4623.
- Zhang, K.; Wong, K.P.; Chow, P. Conjugation of chlorambucil with GSH by GST purified from human colon adenocarcinoma cells and its inhibition by plant polyphenols. Life Sci. 2003, 72, 2629–2640.
- Rossato, L.G.; Costa, V.M.; De Pinho, P.G.; Arbo, M.D.; De Freitas, V.; Vilain, L.; De Lourdes Bastos, M.; Palmeira, C.; Remião, F. The metabolic profile of mitoxantrone and its relation with mitoxantrone-induced cardiotoxicity. Arch. Toxicol. 2013, 87, 1809–1820.
- El Sayed, S.M.; Baghdadi, H.; Zolaly, M.; Almaramhy, H.H.; Ayat, M.; Donki, J.G. The promising anticancer drug 3-bromopyruvate is metabolized through glutathione conjugation which affects chemoresistance and clinical practice: An evidence-based view. Med. Hypotheses 2017, 100, 67–77.
- Cooper, A.J.L.; Hanigan, M.H. 10.17—Metabolism of Glutathione S-Conjugates: Multiple Pathways. In Comprehensive Toxicology, 3rd ed.; McQueen, C.A., Ed.; Elsevier Ltd.: Oxford, UK, 2018; pp. 363–406.
- Van Bladeren, P.J. Glutathione conjugation as a bioactivation reaction. Chem. Biol. Interact. 2000, 129, 61–76.
- Dekant, W. The Role of Biotransformation and Bioactivation in Toxicity. In Molecular, Clinical and Environmental Toxicology; Luch, A., Ed.; Experientia Supplementum: Basel, Switzerland, 2009; pp. 57–86.
- Konopa, J.K.; Horowska, B.; Paluszkiewicz, E.M.; Borowa-Mazgaj, B.; Augustin, E.A.; Skwarska, A.; Mazerska, Z. Asymmetric Bis-Acridines with Antitumour Activity and Use Thereof. European Patent EP3070078A1, 4 October 2017.
- Konopa, J.K.; Horowska, B.; Paluszkiewicz, E.M.; Borowa-Mazgaj, B.; Augustin, E.A.; Skwarska, A.; Mazerska, Z. Asymmetric Bis-Acridines with Antitumour Activity and Their Uses. United. States Patent US10202349B2, 12 February 2019.
- Kosno, M.; Laskowski, T.; Frackowiak, J.E.; Potęga, A.; Kurdyn, A.; Andrałojć, W.; Borzyszkowska-Bukowska, J.; Szwarc-Karabyka, K.; Mazerska, Z. Acid–base equilibrium and self-association in relation to high antitumor activity of selected unsymmetrical bisacridines established by extensive chemometric analysis. Molecules 2022, 27, 3995.
- Mieszkowska, A.; Nowicka, A.M.; Kowalczyk, A.; Potęga, A.; Pawłowska, M.; Kosno, M.; Augustin, E.; Mazerska, Z. Metabolic Profiles of new unsymmetrical bisacridine antitumor agents in electrochemical and enzymatic noncellular systems and in tumor cells. Pharmaceuticals 2021, 14, 317.
- Potęga, A.; Kosno, M.; Mazerska, Z. Novel insights into conjugation of antitumor-active unsymmetrical bisacridine C-2028 with glutathione: Characteristics of non-enzymatic and glutathione S-transferase-mediated reactions. J. Pharm. Anal. 2021, 11, 791–798.
- Paluszkiewicz, E.; Horowska, B.; Borowa-Mazgaj, B.; Peszyńska-Sularz, G.; Paradziej-Łukowicz, J.; Augustin, E.; Konopa, J.; Mazerska, Z. Design, synthesis and high antitumor potential of new unsymmetrical bisacridine derivatives towards human solid tumors, specifically pancreatic cancers and their unique ability to stabilize DNA G-quadruplexes. Eur. J. Med. Chem. 2020, 204, 11259.
- Chasseaud, L.F. The Role of Glutathione and Glutathione S-Transferases in the Metabolism of Chemical Carcinogens and Other Electrophilic Agents. In Advances in Cancer Research; Klein, G., Weinhouse, S., Eds.; Academic Press: San Diego, CA, USA, 1979; pp. 175–274.
- Anderson, M.E. Glutathione: An overview of biosynthesis and modulation. Chem. Biol. Interact. 1998, 111–112, 1–14.
- Lenton, K.J.; Therriault, H.; Wagner, J.R. Analysis of glutathione and glutathione disulfide in whole cells and mitochondria by postcolumn derivatization high-performance liquid chromatography with ortho-phthalaldehyde. Anal. Biochem. 1999, 274, 125–130.
- Meister, A. Glutathione metabolism and its selective modification. J. Biol. Chem. 1988, 263, 17205–17208.
- Josephy, P.D.; Mannervik, B. Biochemistry of Glutathione. In Molecular Toxicology, 2nd ed.; Oxford University Press, Inc.: New York, NY, USA, 2006; pp. 333–364.
- Ballatori, N.; Krance, S.M.; Notenboom, S.; Shi, S.; Tieu, K.; Hammond, C.L. Glutathione dysregulation and the etiology and progression of human diseases. Biol. Chem. 2009, 390, 191–214.
- Townsend, D.M.; Tew, K.D.; Tapiero, H. The importance of glutathione in human disease. Biomed. Pharmacother. 2003, 57, 145–155.
- Ketterer, B.; Coles, B.; Meyer, D.J. The role of glutathione in detoxication. Environ. Health Perspect. 1983, 49, 59–69.
- Park, H.-A.; Khanna, S.; Rink, C.; Gnyawali, S.; Roy, S.; Sen, C.K. Glutathione disulfide induces neural cell death via a 12-lipoxygenase pathway. Cell Death Differ. 2009, 16, 1167–1179.
- Aldini, G.; Altomare, A.; Baron, G.; Vistoli, G.; Carini, M.; Borsani, L.; Sergio, F. N-Acetylcysteine as an antioxidant and disulphide breaking agent: The reasons why. Free Radic. Res. 2018, 52, 751–762.
- Jeffery, E.H. 2—Biochemical Basis of Toxicity. In Handbook of Toxicologic Pathology, 2nd ed.; Haschek, W.M., Rousseaux, C.G., Wallig, M.A., Eds.; Academic Press: San Diego, CA, USA, 2002; pp. 15–37.
- Ulrich, K.; Jakob, U. The role of thiols in antioxidant systems. Free Radic. Biol. Med. 2019, 140, 14–27.
- Xiong, Y.; Uys, J.D.; Tew, K.D.; Townsend, D.M. S-Glutathionylation: From molecular mechanisms to health outcomes. Antioxid. Redox Signal. 2011, 15, 233–270.
- Sies, H. Glutathione and its role in cellular functions. Free Radic. Biol. Med. 1999, 27, 916–921.
- Lu, S.C. Regulation of glutathione synthesis. Mol. Aspects. Med. 2009, 30, 42–59.
- Gomez, L.D.; Noctor, G.; Knight, M.R.; Foyer, C.H. Regulation of calcium signalling and gene expression by glutathione. J. Exp. Bot. 2004, 55, 1851–1859.
- Circu, M.L.; Aw, T.J. Glutathione and apoptosis. Free Radic. Res. 2009, 42, 689–706.
- Forman, H.J.; Zhang, H.; Rinna, A. Glutathione: Overview of its protective roles, measurement, and biosynthesis. Mol. Asp. Med. 2009, 30, 1–12.
- Arrick, B.A.; Nathan, C.F. Glutathione metabolism as a determinant of therapeutic efficacy: A review. Cancer Res. 1984, 44, 4224–4232.
- Cooper, A.J.L.; Hanigan, M.H. 4.17—Enzymes Involved in Processing Glutathione Conjugates. In Comprehensive Toxicology, 2nd ed.; McQueen, C.A., Ed.; Elsevier Ltd.: Oxford, UK, 2010; pp. 323–366.
- Li, J.C.; Renslo, A. Chapter 6: Nucleophilic Substitution, Addition, and Elimination Reactions. In The Organic Chemistry of Medicinal Agents; Renslo, A., Ed.; McGraw Hill: New York, NY, USA, 2016.
- Ouellette, R.J.; Rawn, J.D. Nucleophilic Substitutions and Elimination Reactions. In Organic Chemistry: Structure, Mechanism, Synthesis, 2nd ed.; Ouellette, R.J., Rawn, J.D., Eds.; Academic Press: San Diego, CA, USA, 2018; pp. 299–319.
- Karpusas, M.; Axarli, I.; Chiniadis, L.; Papakyriakou, A.; Bethanis, K.; Scopelitou, K.; Clonis, Y.D.; Labrou, N.E. The interaction of the chemotherapeutic drug chlorambucil with human glutathione transferase A1-1: Kinetic and structural analysis. PLoS ONE 2013, 8, e56337.
- Azevedo-Silva, J.; Queiros, O.; Baltazar, F.; Ułaszewski, S.; Goffeau, A.; Ko, Y.H.; Pedersen, P.L.; Preto, A.; Casal, M. The anticancer agent 3-bromopyruvate: A simple but powerful molecule taken from the lab to the bedside. J. Bioenerg. Biomembr. 2016, 48, 349–362.
- El Sayed, S.M.; Mohamed, W.G.; Seddik, M.-A.H.; Ahmed, A.-S.A.; Mahmoud, A.G.; Amer, W.H.; Helmy Nabo, M.M.; Hamed, A.R.; Ahmed, N.S.; Abd-Allah, A.A.-R. Safety and outcome of treatment of metastatic melanoma using 3-bromopyruvate: A concise literature review and case study. Chin. J. Cancer. 2014, 33, 356–364.
- Rai, Y.; Yadav, P.; Kumari, N.; Kalra, N.; Bhatt, A.N. Hexokinase II inhibition by 3-bromopyruvate sensitizes myeloid leukemic cells K-562 to anti-leukemic drug, daunorubicin. Biosci. Rep. 2019, 39, 1–18.
- Ganapathy-Kanniappan, S.; Geschwind, J.-F.H.; Kunjithapatham, R.; Buijs, M.; Vossen, J.A.; Tchernyshyov, I.; Cole, R.N.; Syed, L.H.; Rao, P.P.; Ota, S.; et al. Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) is pyruvylated during 3-bromopyruvate mediated cancer cell death. Anticancer Res. 2009, 29, 4909–4918.
- Fan, T.; Sun, G.; Sun, X.; Zhao, L.; Zhong, R.; Peng, Y. Tumor energy metabolism and potential of 3-bromopyruvate as an inhibitor of aerobic glycolysis: Implications in tumor treatment. Cancers 2019, 11, 317.
- Attia, Y.M.; El-Abhar, H.S.; Al Marzabani, M.M.; Shouman, S.A. Targeting glycolysis by 3-bromopyruvate improves tamoxifen cytotoxicity of breast cancer cell lines. BMC Cancer 2015, 15, 838.
- Fischer, G.; Sieber, M.; Schellenberger, A. The carbonyl reactivity of 3-bromopyruvate and related compounds. Bioorg. Chem. 1982, 11, 478–484.
- Sadowska-Bartosz, I.; Szewczyk, R.; Jaremko, L.; Jaremko, M.; Bartosz, G. Anticancer agent 3-bromopyruvic acid forms a conjugate with glutathione. Pharmacol. Rep. 2016, 68, 502–505.
- Goede, V.; Eichhorst, B.; Fischer, K.; Wendtner, C.-M.; Hallek, M. Past, present and future role of chlorambucil in the treatment of chronic lymphocytic leukemia. Leuk. Lymphoma 2015, 56, 1585–1592.
- Leblond, V.; Johnson, S.; Chevret, S.; Copplestone, A.; Rule, S.; Tournilhac, O.; Seymour, J.F.; Patmore, R.D.; Wright, D.; Morel, P.; et al. Results of a randomized trial of chlorambucil versus fludarabine for patients with untreated Waldenström macroglobulinemia, marginal zone lymphoma, or lymphoplasmacytic lymphoma. J. Clin. Oncol. 2013, 31, 301–307.
- Ardeshna, K.M.; Smith, P.; Norton, A.; Hancock, B.W.; Hoskin, P.J.; MacLennan, K.A.; Marcus, R.E.; Jelliffe, A.; Vaughan Hudson, G.; Linch, D.C. Long-term effect of a watch and wait policy versus immediate systemic treatment for asymptomatic advanced-stage non-Hodgkin lymphoma:a randomised controlled trial. Lancet 2003, 362, 516–522.
- Parker, L.J.; Ciccone, S.; Italiano, L.C.; Primavera, A.; Oakley, A.J.; Morton, C.J.; Hancock, N.C.; Lo Bello, M.; Parker, M.W. The anti-cancer drug chlorambucil as a substrate for the human polymorphic enzyme glutathione transferase P1-1: Kinetic properties and crystallographic characterisation of allelic variants. J. Mol. Biol. 2008, 380, 131–144.
- Cobb, D.; Boehlert, C.; Lewis, D.; Armstrong, R.N. Stereoselectivity of isozyme C of glutathione S-transferase toward arene and azaarene oxides. Biochemistry 1983, 22, 805–812.
- Andrew, A.W.; Hayes, W.; Kruger, C.L. Hayes’ Principles and Methods of Toxicology; CRC Press, LLC: Boca Raton, FL, USA, 2014; p. 71.
- Hassan, F.; Preiss, R. Cyclophosphamide and related anticancer drugs. J. Chromatogr. B Biomed. Appl. 2001, 764, 173–192.
- Singh, R.K.; Kumar, S.; Prasad, D.N.; Bhardwaj, T.R. Therapeutic journery of nitrogen mustard as alkylating anticancer agents: Historic to future perspectives. Eur. J. Med. Chem. 2018, 151, 401–433.
- Colvin, O.M. An overview of cyclophosphamide development and clinical applications. Curr. Pharm. Des. 1999, 5, 555–560.
- Hubbard, R.D.; Fidanze, S. 7.06—Alkylating and Platinum Antitumor Compounds. In Comprehensive Medicinal Chemistry II.; Taylor, J.B., Triggle, D.J., Eds.; Elsevier Ltd.: Oxford, UK, 2007; pp. 129–148.
- Ahlmann, M.; Hempel, G. The effect of cyclophosphamide on the immune system: Implications for clinical cancer therapy. Cancer Chemother. Pharmacol. 2016, 78, 661–671.
- Bagley, C.M.; Bostick, F.W.; DeVita, V.T. Clinical pharmacology of cyclophosphamide. Cancer Res. 1973, 33, 226–233.
- Li, F.; Patterson, A.D.; Höfer, C.C.; Krausz, K.W.; Gonzalez, F.J.; Idle, J.R. Comparative metabolism of cyclophosphamide and ifosfamide in the mouse using UPLC-ESI-QTOFMS-based metabolomics. Biochem. Pharmacol. 2010, 80, 1063–1074.
- Groehler, A.; Villalta, P.W.; Campbell, C.; Tretyakova, N. Covalent DNA-protein cross-linking by phosphoramide mustard and nornitrogen mustard in human cells. Chem. Res. Toxicol. 2016, 29, 190–202.
- Mills, K.A.; Chess-Williams, R.; McDermott, C. Novel insights into the mechanism of cyclophosphamide-induced bladder toxicity: Chloroacetaldehyde′s contribution to urothelial dysfunction in vitro. Arch. Toxicol. 2019, 93, 3291–3303.
- Yuan, Z.M.; Smith, P.B.; Brundrett, R.B.; Colvin, M.; Fenselau, C. Glutathione conjugation with phosphoramide mustard and cyclophosphamide. A mechanistic study using tandem mass spectrometry. Drug Metab. Dispos. 1991, 19, 625–629.
- Cohen, N.A.; Egorin, M.J.; Snyder, S.W.; Ashar, B.; Wietharn, B.E.; Pan, S.S.; Ross, D.D.; Hilton, J. Interaction of N,N′,N″-triethylenethiophosphoramide and N,N’,N″-triethylenephosphoramide with cellular DNA. Cancer Res. 1991, 51, 4360–4366.
- Van der Wall, E.; Beijnen, J.H.; Rodenhuis, S. High-dose chemotherapy regimens for solid tumors. Cancer Treat. Rev. 1995, 21, 105–132.
- Van Maanen, M.J.; Smeets, C.J.M.; Beijnen, J.H. Chemistry, pharmacology and pharmacokinetics of N,N′,N″-triethylenethiophosphoramide (ThioTEPA). Cancer Treat. Rev. 2000, 26, 257–268.
- Torabifard, H.; Fattahi, A. DFT study on Thiotepa and Tepa interactions with their DNA receptor. Struct. Chem. 2013, 24, 1–11.
- Jacobson, P.A.; Green, K.; Birnbaum, A.; Remmel, R.P. Cytochrome P450 isozymes 3A4 and 2B6 are involved in the in vitro human metabolism of thiotepa to TEPA. Cancer Chemother. Pharmacol. 2002, 49, 461–467.
- Dirven, H.A.A.M.; Dictus, E.L.J.T.; Broeders, N.L.H.L.; Van Ommen, B.; Van Bladeren, P.J. The role of human glutathione S-transferase isoenzymes in the formation of glutathione conjugates of the alkylating cytostatic drug thiotepa. Cancer Res. 1995, 55, 1701–1706.
- Schwöbel, J.A.H.; Wondrousch, D.; Koleva, Y.K.; Madden, J.C.; Cronin, M.T.D.; Schüürmann, G. Prediction of Michael-type acceptor reactivity toward glutathione. Chem. Res. Toxicol. 2010, 23, 1576–1585.
- Ruzza, P.; Calderan, A. Glutathione transferase (GST)-activated prodrugs. Pharmaceutics 2013, 5, 220–231.
- Schultz, T.W.; Yarbrough, J.W.; Hunter, R.S.; Aptula, A.O. Verification of the structural alerts for Michael acceptors. Chem. Res. Toxicol. 2007, 20, 1359–1363.
- Evison, B.J.; Sleebs, B.E.; Watson, K.G.; Phillips, D.R.; Cutts, S.M. Mitoxantrone, more than just another topoisomerase II poison. Med. Res. Rev. 2016, 36, 248–299.
- Hainswort, J.D.; Andrews, M.B.; Johnson, D.H.; Greco, F.A. Mitoxantrone, fluorouracil, and high-dose leucovorin: An effective, well-tolerated regimen for metastatic breast cancer. J. Clin. Oncol. 1991, 9, 1731–1735.
- Basch, E.M.; Scholz, M.; De Bono, J.S.; Vogelzang, N.; De Souza, P.; Marx, G.; Vaishampayan, U.; George, S.; Schwarz, J.K.; Antonarakis, E.S.; et al. Cabozantinib versus mitoxantrone-prednisone in symptomatic metastatic castration-resistant prostate cancer: A randomized Phase 3 trial with a primary pain endpoint. Eur. Urol. 2019, 75, 929–937.
- Nastoupil, L.J.; McLaughlin, P.; Feng, L.; Neelapu, S.S.; Samaniego, F.; Hagemeister, F.B.; Ayala, A.; Romaguera, J.E.; Goy, A.H.; Neal, E.; et al. High ten-year remission rates following rituximab, fludarabine, mitoxantrone and dexamethasone (R-FND) with interferon maintenance in indolent lymphoma: Results of a randomized study. Br. J. Haematol. 2017, 177, 263–270.
- Advani, A.S.; Cooper, B.; Visconte, V.; Elson, P.; Chan, R.; Carew, J.; Wei, W.; Mukherjee, S.; Gerds, A.; Carraway, H.; et al. A Phase I/II trial of MEC (Mitoxantrone, Etoposide, Cytarabine) in combination with ixazomib for relapsed refractory acute myeloid leukemia. Clin. Cancer. Res. 2019, 25, 4231–4237.
- Mewes, K.; Blanz, J.; Ehninger, G.; Gebhardt, R.; Zeller, K.-P. Cytochrome P-450-induced cytotoxicity of mitoxantrone by formation of electrophilic intermediates. Cancer Res. 1993, 53, 5135–5142.
- Guagnano, V.; Furet, P.; Spanka, C.; Bordas, V.; Le Douget, M.; Stamm, C.; Brueggen, J.; Jensen, M.R.; Schnell, C.; Schmid, H.; et al. Discovery of 3-(2,6-dichloro-3,5-dimethoxy-phenyl)-1--1-methyl-urea (NVP-BGJ398), a potent and selective inhibitor of the fibroblast growth factor receptor family of receptor tyrosine kinase. J. Med. Chem. 2011, 27, 7066–7083.
- Komla-Ebri, D.; Dambroise, E.; Kramer, I.; Benoist-Lasselin, C.; Kaci, N.; Le Gall, C.; Martin, L.; Busca, P.; Barbault, F.; Graus-Porta, D.; et al. Tyrosine kinase inhibitor NVP-BGJ398 functionally improves FGFR3-related dwarfism in mouse model. J. Clin. Investig. 2016, 126, 1871–1884.
- Javle, M.; Lowery, M.; Shroff, R.T.; Weiss, K.H.; Springfeld, C.; Borad, M.J.; Ramanathan, R.K.; Goyal, L.; Sadeghi, S.; Macarulla, T.; et al. Phase II Study of BGJ398 in patients with FGFR-altered advanced cholangiocarcinoma. J. Clin. Oncol. 2018, 36, 276–282.
- Al-Shakliah, N.S.; Attwa, M.W.; Kadi, A.A.; AlRabiah, H. Identification and characterization of in silico, in vivo, in vitro, and reactive metabolites of infigratinib using LC-ITMS: Bioactivation pathway elucidation and in silico toxicity studies of its metabolites. RSC Adv. 2020, 10, 16231–16244.
- Zhao, Z.; Koeplinger, K.A.; Padbury, G.E.; Hauer, M.J.; Bundy, G.L.; Banitt, L.S.; Schwartz, T.M.; Zimmermann, D.C.; Harbach, P.R.; Mayo, J.K. Bioactivation of 6,7-dimethyl-2,4-di-1-pyrrolidinyl-7h-pyrrolopyrimidine (U-89843) to reactive intermediates that bind covalently to macromolecules and produce genotoxicity. Chem. Res. Toxicol. 1996, 9, 1230–1239.
- Lyss, G.; Schmidt, T.J.; Merfort, I.; Pahl, H.L. Helenalin, an anti-inflammatory sesquiterpene lactone from Arnica, selectively inhibits transcription factor NF-κB. Biol. Chem. 1997, 378, 951–962.
- Lim, C.B.; Fu, P.Y.; Ky, N.; Zhu, H.S.; Feng, X.L.; Li, J.; Srinivasan, K.G.; Hamza, M.S.; Zhao, Y. NF-κB p65 repression by the sesquiterpene lactone, helenalin, contributes to the induction of autophagy cell death. BMC Complement. Altern. Med. 2012, 12, 93.
- Jakobs, A.; Steinmann, S.; Henrich, S.M.; Schmidt, T.J.; Klempnaue, K.H. Helenalin acetate, a natural sesquiterpene lactone with anti-inflammatory and anti-cancer activity, disrupts the cooperation of CCAAT box/enhancer-binding protein β (C/EBPβ) and co-activator p300. J. Biol. Chem. 2016, 9, 26098–26108.
- Jürgens, F.M.; Behrens, M.; Humpf, H.-U.; Robledo, S.M.; Schmidt, T.J. In vitro metabolism of helenalin acetate and 11α,13-dihydrohelenalin acetate: Natural sesquiterpene lactones from Arnica. Metabolites 2022, 12, 88.
- Schmidt, T.J. Glutathione adducts of helenalin and 11α,13-dihydrohelenalin acetate inhibit glutathione S-transferase from horse liver. Planta Med. 2000, 66, 106–109.
- Lang, W.; Caldwell, G.W.; Masucci, J.A. Evaluation of the effect of oxygen exposure on human liver microsomal metabolism of mitomycin C in the presence of glutathioneusing liquid chromatography–quadrupole time of flight mass spectrometry. Anal. Biochem. 2005, 343, 268–276.
- Verweij, J.; Pinedo, H.M. Mitomycin C: Mechanism of action, usefulness and limitations. Anticancer Drugs 1990, 1, 5–13.
- Tomasz, M. Mitomycin C: Small, fast and deadly (but very selective). Chem. Biol. 1995, 2, 575–579.
- Avendaño, C.; Menéndez, J.C. Chapter 6Anticancer Drugs That Interact with the DNA Minor Groove. In Medicinal Chemistry of Anticancer Drugs, 2nd ed.; Avendaño, C., Menéndez, J.C., Eds.; Elsevier Ltd.: Oxford, UK, 2015; pp. 243–271.
- Sharma, M.; Tomasz, M. Conjugation of glutathione and other thiols with bioreductively activated mitomycin C. Effect of thiols on the reductive activation. Chem. Res. Toxicol. 1994, 7, 390–400.
- Shibata, Y.; Chiba, M. The role of extrahepatic metabolism in the pharmacokinetics of the targeted covalent inhibitors afatinib, ibrutinib, and neratinib. Drug Metab. Dispos. 2015, 43, 375–384.
- Rood, J.J.M.; Dormans, P.J.A.; Van Haren, M.J.; Schellens, J.H.M.; Beijnen, J.H.; Sparidans, R.W. Bioanalysis of ibrutinib, and its dihydrodiol- and glutathione cycle metabolites by liquid chromatography-tandem mass spectrometry. J. Chromatogr. B 2018, 1090, 14–21.
- Piesche, M.; Roos, J.; Kühn, B.; Fettel, J.; Hellmuth, N.; Brat, C.; Maucher, I.V.; Awad, O.; Matrone, C.; Comerma Steffensen, S.G.; et al. The emerging therapeutic potential of nitro fatty acids and other Michael acceptor-containing drugs for the treatment of inflammation and cancer. Front. Pharmacol. 2020, 11, 1297.
More
Information
Subjects:
Biochemistry & Molecular Biology
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
2.8K
Revisions:
2 times
(View History)
Update Date:
26 Aug 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





