Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Hasi Rani Barai | -- | 3364 | 2022-07-25 23:27:19 | | | |
| 2 | Camila Xu | Meta information modification | 3364 | 2022-07-26 07:24:53 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Atia, G.A.N.; Shalaby, H.K.; Zehravi, M.; Ghobashy, M.M.; Ahmad, Z.; Khan, F.S.; Dey, A.; Rahman, M.H.; Joo, S.W.; Barai, H.R.; et al. Hormones for Oral Bone and Periodontal Tissue Engineering. Encyclopedia. Available online: https://encyclopedia.pub/entry/25500 (accessed on 09 June 2026).
Atia GAN, Shalaby HK, Zehravi M, Ghobashy MM, Ahmad Z, Khan FS, et al. Hormones for Oral Bone and Periodontal Tissue Engineering. Encyclopedia. Available at: https://encyclopedia.pub/entry/25500. Accessed June 09, 2026.
Atia, Gamal Abdel Nasser, Hany K. Shalaby, Mehrukh Zehravi, Mohamed Mohamady Ghobashy, Zubair Ahmad, Farhat S. Khan, Abhijit Dey, Md. Habibur Rahman, Sang Woo Joo, Hasi Rani Barai, et al. "Hormones for Oral Bone and Periodontal Tissue Engineering" Encyclopedia, https://encyclopedia.pub/entry/25500 (accessed June 09, 2026).
Atia, G.A.N., Shalaby, H.K., Zehravi, M., Ghobashy, M.M., Ahmad, Z., Khan, F.S., Dey, A., Rahman, M.H., Joo, S.W., Barai, H.R., & Cavalu, S. (2022, July 25). Hormones for Oral Bone and Periodontal Tissue Engineering. In Encyclopedia. https://encyclopedia.pub/entry/25500
Atia, Gamal Abdel Nasser, et al. "Hormones for Oral Bone and Periodontal Tissue Engineering." Encyclopedia. Web. 25 July, 2022.
Copy Citation
Hormones are unique regulatory chemicals that govern fertility, growth, maturation, and microenvironmental maintenance, in addition to power generation, usage, and storage. Hormones play a fundamental function in the maintenance of the integrity of both periodontium and bone. Bone tissue is made up of many cell types and an extracellular matrix that is mostly made up of collagen proteins. Bone’s primary roles are structural support, mechanical motions, blood cell formation, and tissue preservation; it also serves as a depot of calcium and phosphate ions in the body.
tissue engineering
periodontium
bone
hormones
1. Introduction
Hormones are unique regulatory chemicals that govern fertility, growth, maturation, and microenvironmental maintenance, in addition to power generation, usage, and storage [1]. Hormones play a fundamental function in the maintenance of the integrity of both periodontium and bone. Many hormones are involved in the maturation, growth, and maintenance of both periodontium and bone, including IGF-1 and thyroid hormone, as well as sexual hormones, calcium-regulating hormones, parathyroid hormone, and vitamin D. Overall, the hormonal effect on periodontal health, bone development, and maximal bone mass is considerable [2][3][4][5][6]. The periodontium is a functionally organized system of several tissues that encircle and reinforce the tooth, in addition to other periodontal tissues, such as periodontal ligament (PDL) and alveolar bone (AB) [7]. Periodontitis is a chronic irritating illness that affects the periodontium. Periodontal disease is described as the deterioration of periodontal tissue, which includes gums, alveolar bone, periodontal ligament, and cementum.
Periodontal diseases have a wide range of pathophysiology.
The interaction between etiological factors and the host’s immune responses results in the creation of many enzymes, chemokines, and mediators, which induces periodontal disease [8].
Bone tissue is made up of many cell types and an extracellular matrix that is mostly made up of collagen proteins. Bone’s primary roles are structural support, mechanical motions, blood cell formation, and tissue preservation; it also serves as a depot of calcium and phosphate ions in the body [9][10]. To maintain skeletal structure, bone resorption and production are closely controlled and managed by bone equilibrium. Osteoblasts, osteoclasts, and osteocytes are all kinds of cells found in bone tissue. Mesenchymal stem cells (MSCs) are responsible for the formation of osteoblasts and osteocytes, while hematopoietic stem cells give rise to osteoclasts.
Osteocytes make about 90% of the bone cell population and serve as the major cells for bone production, mineralization, and cell signaling regulation.
During remodeling, osteoclasts decompose naturally damaged bone and osteoblasts produce new bone, which is then replenished [11]. The rhythm between bone creation driven by osteoblasts and bone degeneration facilitated by osteoclasts is essential for bone homeostasis. Abnormal bone loss occurs when this equilibrium is disrupted, promoting osteoclastic activity, as observed in pathological conditions including periodontitis [12].
Numerous substances have already been discovered as being significant in bone morphology and performance maintenance. Current treatment modalities of both of periodontal and bone diseases, such as, but not confined to, guided tissue regeneration, guided bone regeneration, and surgery, have limited results and can only repair damaged tissues, rather than their regeneration [13].
An innovative alternative is provided by tissue engineering, which is capable of the regeneration of tissues and restoration of their complete function. Tissue engineering is an interdisciplinary approach along with chemistry, pharmaceutics, genetics, and biomedical engineering [14]. Tissue engineering has received attention as a viable strategy in the discipline of tissue regeneration in recent decades, providing a new option for the rehabilitation of teeth, periodontium, bone [15], as well as blood vessels [16][17]. The scaffold, cells, and signaling molecules are three key components of biomedical engineering, as shown in Figure 1 [18]. Several studies have described distinct scaffolds for various types of tissue regeneration; for instance, oral bone and periodontal tissues [19][20]. Stem cells are categorized into totipotent, pluripotent, or multipotent based on their ability to develop into various cell types. [21][22][23][24]. Totipotent cells may give rise to the entire organism, whereas pluripotent cells (iPSC, such as embryonic stem (ES) cells), can actually lead to all cell types in an organism excluding extra-embryonic organs such as the placenta.
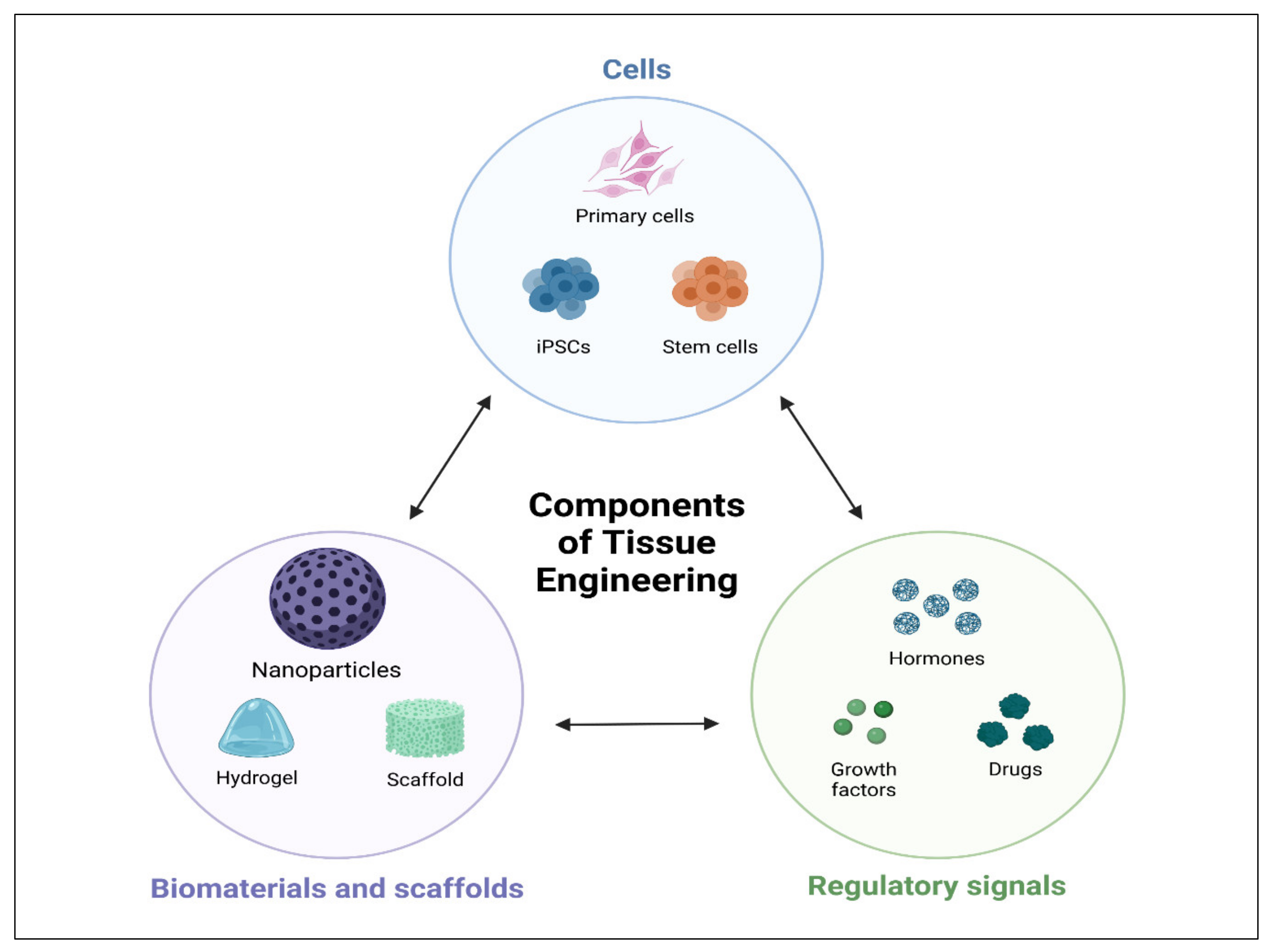 Figure 1. Schematic illustrations of tissue engineering triad. Cell, biomaterials, scaffolds, and regulatory signals.
Figure 1. Schematic illustrations of tissue engineering triad. Cell, biomaterials, scaffolds, and regulatory signals.Mature stem cells that can develop into a particular cell lineage are known as multipotent stem cells (MSC) [25]. Biologically active substances, such as growth factors [26], medicines [26], and hormones [27], can be delivered locally [28], and were reported to induce oral bone and periodontal regeneration.
2. Hormones
Hormones are essentially characterized as a stimulants, inhibitors, or chemical messengers that, after being released into the systemic circulation, cause a specific alteration in the cellular activity of target sites. Figure 2 shows main glands in the human body.
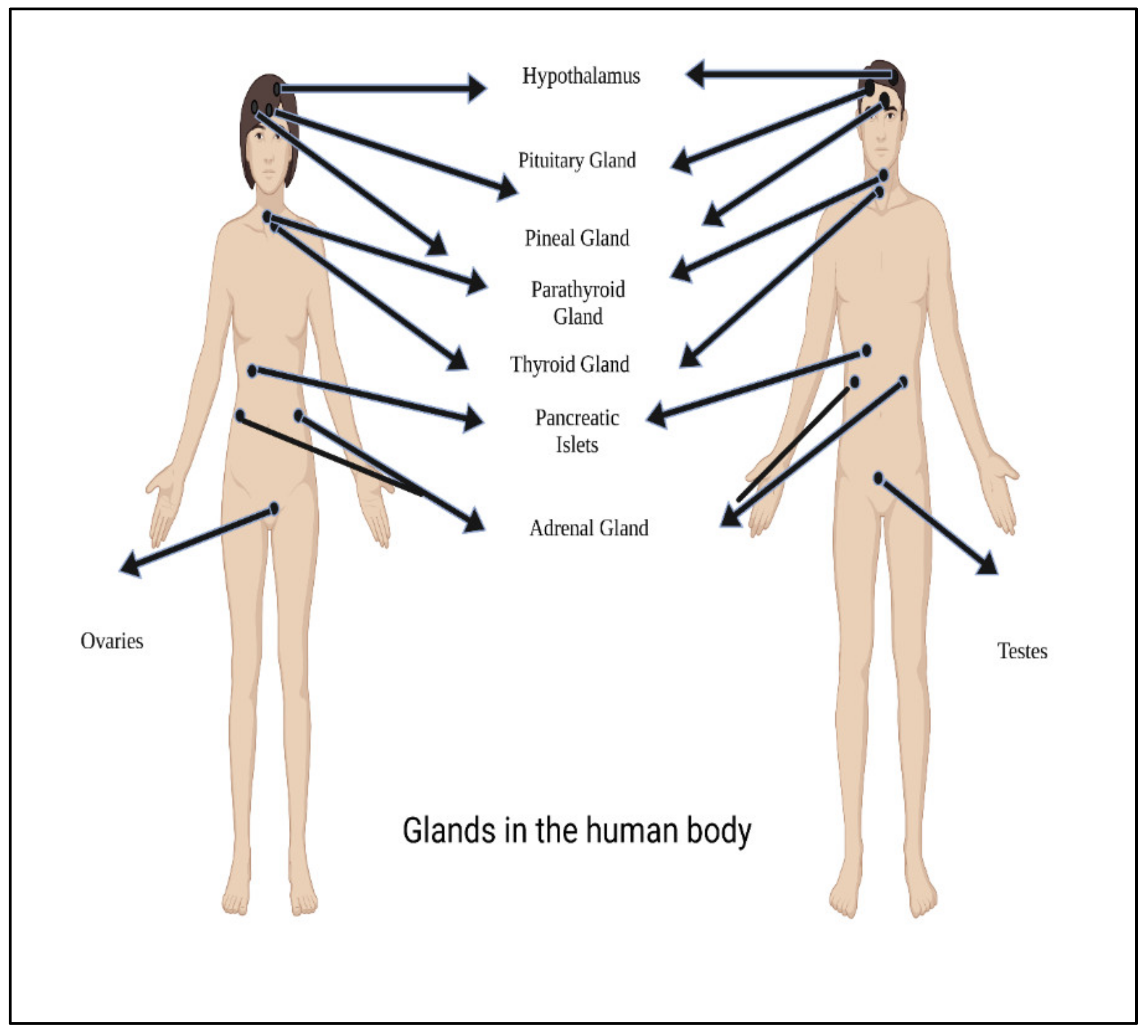 Figure 2. Main glands in the human body.
Figure 2. Main glands in the human body.Hormones are classified according to their composition, such as amino acids, tyrosine (catechol amines and thyroid hormones), tryptophan (serotonin), etc., as shown in Table 1. Hormone action could be endocrine (site of their actions distant from the site of release), and may also be paracrine (functioning on nearby cells by diffusion), autocrine (acting on the secreting cells by diffusion), or intracrine (working in secreting cells without release). Agents that work in this manner are frequently referred to as factors instead of hormones, as shown in Figure 3. Indeed, these substances (for example, hormones) may be generated in the majority of cells throughout the body instead of defined endocrine glands [29].
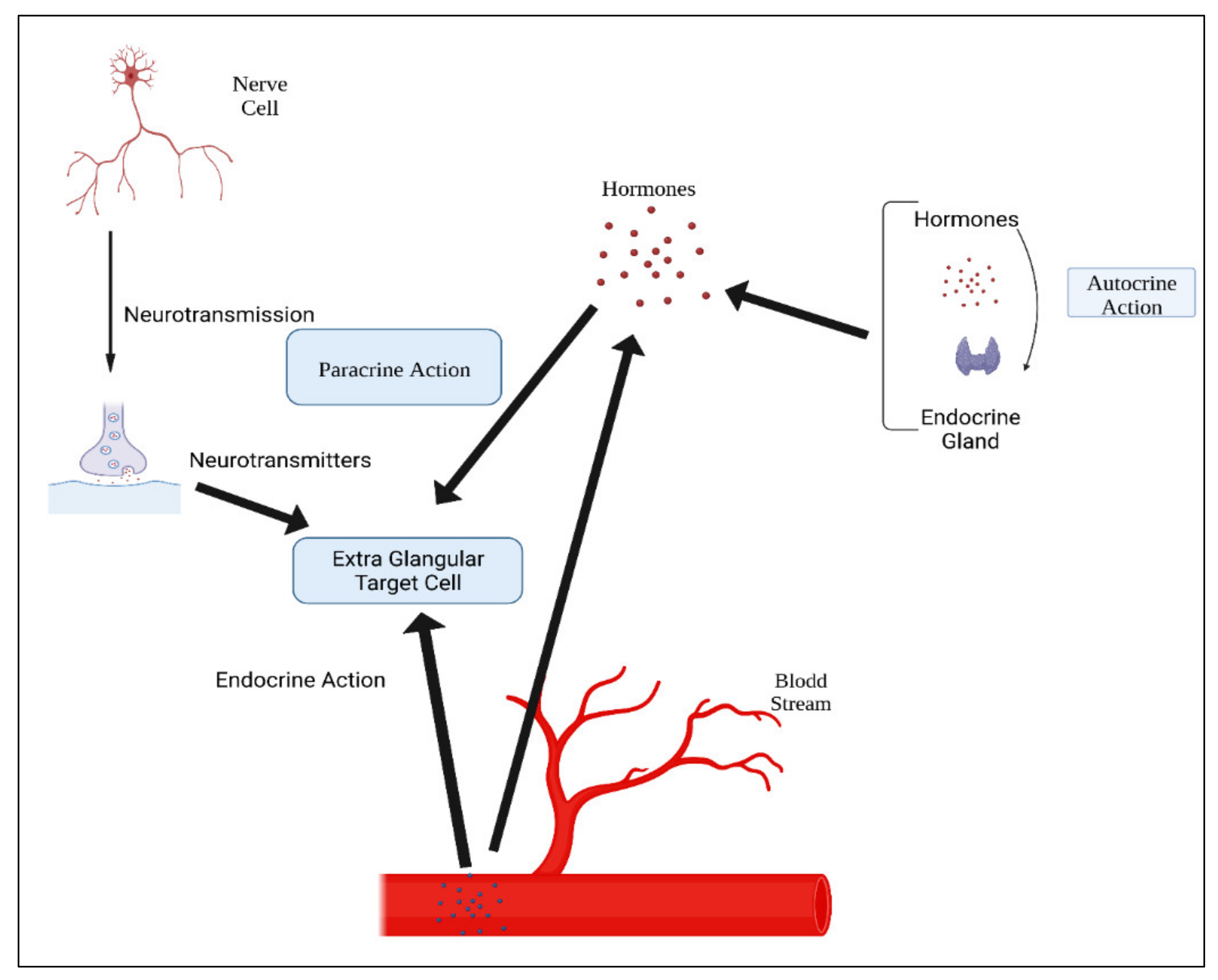 Figure 3. Mechanisms of hormonal actions.
Figure 3. Mechanisms of hormonal actions.Table 1. Classification of hormones.
| Peptides | Glycoproteins | Amines | Eicosanoids | Steroid Hormones |
|---|---|---|---|---|
| Source: made up of amino acid residues | Source: conjugated protein bound to carbohydrate | Source: modification of amino acids | Source: small fatty acid derivatives with a variety of arachidonic acid | Source: derived from cholesterol |
|
e.g., Thyrotropin (TSH) | e.g., thyroid hormones and catecholamines | e.g., Prostaglandins | Examples:
|
| Short peptides | e.g.,
|
|||
| Intermediate peptides | e.g.,
|
|||
| Glycoproteins |
|
|||
| Peptide-based hormones | ||||
| Amino acid derivatives | ||||
| Iodothyronines |
|
|||
| Amines | i.e., • Melatonin |
|||
| Steroidal hormones | ||||
|
||||
3. Examples of Repositioned Hormones for Bone and Periodontal Tissue Engineering
As previously stated, GFs-based therapies are costly and may cause side effects and immunological reactions in certain individuals. To counteract these disadvantages, various hormones have been designed and tested as viable replacements to growth factors. Hormones are inexpensive to produce, can be readily designed and manufactured, and have little immunogenicity due to their flexibility [30]. Figure 4 shows examples of the action of some hormones on osteoblasts and osteoclasts.
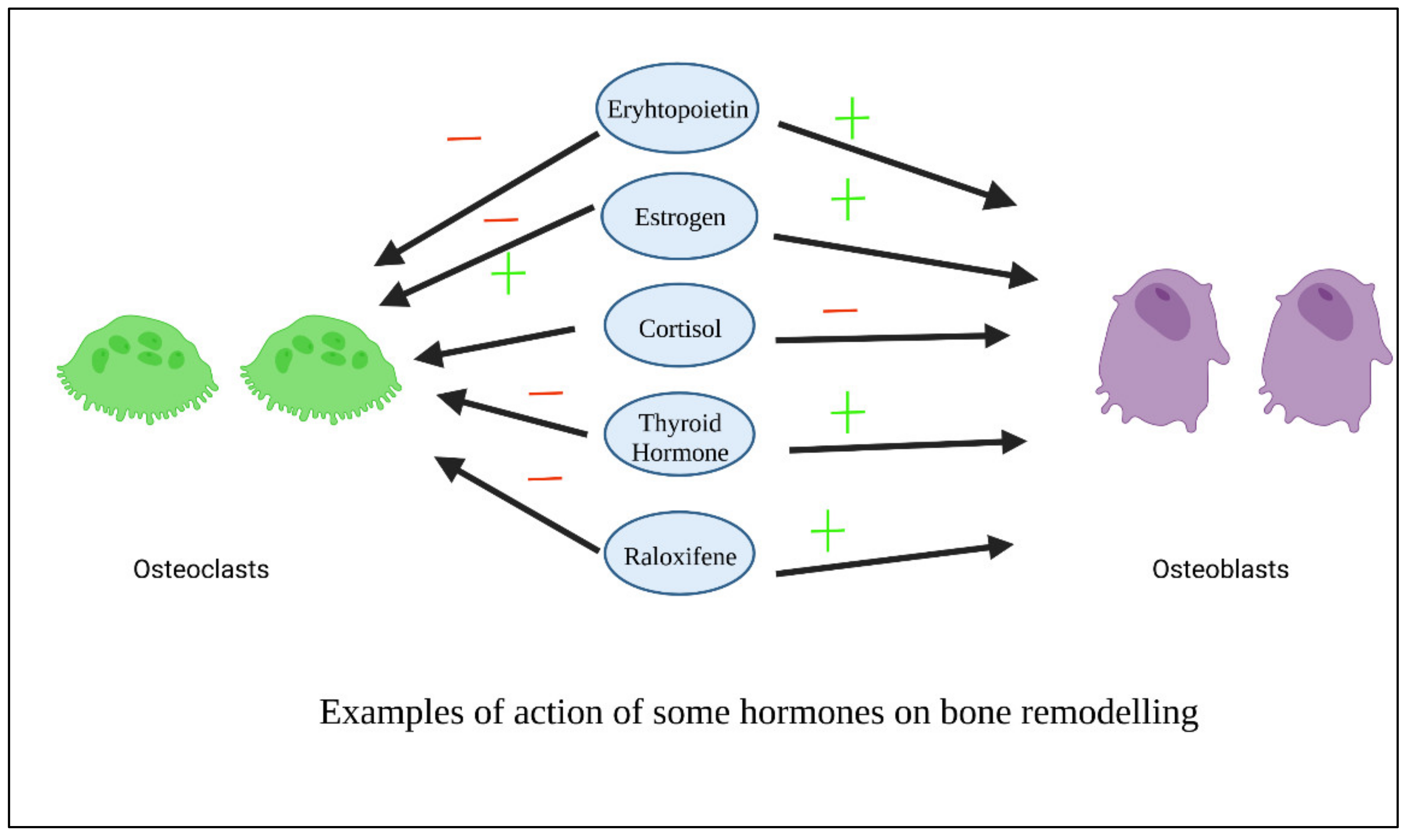 Figure 4. Examples of action of some hormones on osteoblasts.
Figure 4. Examples of action of some hormones on osteoblasts.3.1. Thyroxin
Thyroxin is an essential hormone that performs a range of physiological tasks in the human body. One of them is its capacity to stimulate angiogenesis through a variety of methods [31]. By stimulating integrin v3, thyroxin promotes the production of mediators of angiogenesis [32]. Thyroid hormones also influenced cellular metabolic reactions and cell growth [33]. Chitosan/collagen-based thyroxin-loaded hydrogels have a neovascularization capability, which suggests that they might be useful materials for prospective tissue engineering applications [31]. Chitosan composite enclosed with varying doses of thyroxin were demonstrated to be biocompatible, and these hydrogels with pro-angiogenic activities have a high promising applications in periodontal regeneration [34]. In comparison to chitosan, thyroxin-containing membranes demonstrated significant revascularization and rapid wound healing in rats [35].
3.2. Oxytocin
Oxytocin (OT) is a fundamental anabolic hormone found in animals during breastfeeding that also has local impacts on bone turnover in addition to the systemic endocrine route [36]. This hormone improves bone production by favorable control of osteoblast development, osteoclast activities, and overexpression of bone morphogenic protein 2 (BMP2) [37][38]. Despite oxytocin being researched in a variety of medicinal applications, its influence on in situ osteogenesis has not been explored, most likely because of its limited half-life and instability versus hydrolysis [39]. The impact of this hormone is only temporary in the absence of an adequate carrier and encapsulation technique, and the physicochemical stabilization cannot be preserved over the bone healing period. Thanks to their unique features, poly (D, Llactide coglycolide) PLGA copolymers have been used as local drug carrier for different types of biomolecules [40]. Sustained release micro spherical oxytocin hormone in a polymeric hydrogel scaffold mixed with biphasic calcium phosphates combination promotes bone repair in the rat calvarias [41]. Furthermore, OT-loaded b-TCP increases osteogenesis in rats with calvarias bone defects via an osteoinductive mechanism of action [42]. In vitro, OT increased PDLSC proliferation, aggregation, and osteogenic differentiation. Additionally, OT’s influence on osteogenic development was driven by the ERK and AKT pathways. As a result, OT has the potential to be used in periodontal regeneration [43].
3.3. Dexamethasone
Dexamethasone (DEX) has been demonstrated to enhance osteoblast development in vitro and bone tissue creation in vivo by enhancing osteoblast-related gene transcription [44][45]. DEX has long been employed as an osteoinductive factor due to its excellent integrity as well as osteogenesis [46][47]. High DEX concentrations, on the other hand, would inhibit osteoblast growth and create hazardous adverse effects [46][48], As a result, its additional functional applicability in bone tissue engineering is limited. Thus, prolonged release of DEX is essential to maximize effectiveness while minimizing negative effects on bone regeneration. Porous bio composite matrices comprise the chitosan-alginate-gelatin scaffold in addition to the accumulation of calcium phosphate and DEX-loaded nano silica. Doping was manufactured and demonstrated increased growth and osteogenesis in rats, suggesting that they might be extremely good as potential local insertable frameworks for possible uses in bone tissue engineering [49]. Dexamethasone (DEX) has been demonstrated to initiate bone marrow differentiation as well as guide cells toward maturation [50][51]. Injectable hydrogels loaded with dexamethasone have a promising potential as an injectable drug-depot for bone repair therapy in cases of chronic inflammation [52].
3.4. Androgens
In males, testosterone is the major sexual hormone and anabolic factor. In humans, testosterone is crucial in the male sexual organs, for example the testes, as well as in the promotion of secondary sexual traits such as increased muscular and bone mass [53]. PLGA-coated pericardial inserts or membranes combining topical gradual administration of supplementary quantities of testosterone and alendronate may be a viable approach for stimulating in situ osteogenesis, leading to enhanced implant osseo-integration and repair of bone defects and fractures [54]. In mice, testosterone delivered with a scaffold has similar effects to the Bone Morphologic Protein-2 in enhancing bone regeneration [55].
3.5. Parathyroid Hormone (PTH)
The endogenous parathyroid hormone is a critical mediator of bone remodeling as well as a crucial regulator of calcium-phosphate equilibrium. This hormone promotes bone formation by activating numerous mechanisms involved in stem/preosteoblast cell osteo differentiation. Inhibiting osteoblast apoptosis can also increase the quantity of osteoblasts. PTH causes osteoblasts to release a number of growth factors, and it causes osteocytes to produce less sclerostin and DKK, two anti-osteoclastic and Wnt signaling inhibitors. Furthermore, PTH may indirectly trigger osteoclasts to accomplish bone resorption. PTH stimulates osteoblast RANKL synthesis and increases RANKL binding to osteoclast surface receptors, resulting in osteoclast activation [56]. The amount and duration of PTH exposure influence bone production (anabolism) and bone resorption (catabolism). Constant and high hormone dosages promote bone breakdown, whereas minimal and inconsistent levels promote osteogenesis and increased mineral density [57].
3.6. Insulin
Insulin is a hormone which affects energy production and balance, as well as being an important part in bone formation metabolism. Skeletal anomalies linked to Diabetes type I can be cured with insulin treatment [58][59]. Clinically, it is frequently noted that insulin shortage increases the possibility of fracture. The use of insulin therapy dramatically boosted bone formation in patients with type 2 diabetes, which can minimize the risk of fracture [60][61]. Insulin/IGF-1 has been proven in vivo to induce angiogenesis and give nourishment for bone growth. [62][63][64]. Insulin can successfully enhance local skull bone growth in the mouse skull by raising the quantity of bone forming cells and the surface area of the osteoid [65], and has the ability to control osteoclastic activity [66]. In recent years, research has discovered that IGF-1 can also influence the formation and maturation of osteoblasts, hence increasing bone repair [67]. Given the success of nanoparticles in drug loading, a variety of insulin carriers have been innovated, which could be breakthroughs in bioengineering technology [68]. In another study, insulin-loaded poly lactic-co-glycolic-acid (PLGA) Nano spheres were incorporated into nano hydroxyapatite/collagen (nHAC) scaffolds, where insulin was successfully distributed from the nano spheres and aided bone regeneration in significant size impairments in the rabbit mandible [69]. Furthermore, insulin-encapsulated PLGA microspheres greatly enhanced the insert’s stability in rabbits at Week 4, indicating that it is possible to lower the implant’s early failure rate without affecting serum biochemical markers [70]. New bioactive injectable composites loaded with insulin have been developed and might be used to treat bone defects, notably as an economic promotion/substitute to BMP-2 approaches [71]. Local insulin infiltration at the implant–bone contact has the potential to have significant therapeutic ramifications by spontaneously increasing the effectiveness of oral implantation in diabetic rats [72].
3.7. Estrogen
Estrogen is a natural steroidal hormone that regulates bone mass and maintains bone tissue balance. The estrogen’s activity is directly connected to the regulation of osteoblast proliferation and differentiation. In addition, estrogen reduces apoptosis in osteocytes and osteoblasts while inducing apoptosis in osteoclasts. By decreasing the synthesis osteoclastic mediators, estrogen reduces the creation of active osteoclasts. Moreover, it increases the creation of osteoprotegerin by osteoblasts and osteocytes (OPG) [73][74]. 17-estradiol (E2) is the most powerful hormone in the body system, and it adheres to estrogen receptors (ERs) in both bone cells and MSCs. Estradiol can encourage MSCs to differentiate into osteoblasts and improve osteogenesis by boosting the expression of BMP-2, TGF-1, and IGF-1 [75]. Estrogen activity causes bone remodeling to be balanced and bone metabolism to be modulated. As a result, estrogen deprivation reduces osseous density, raises the possibility of osteoporotic fractures, and causes bone loss [76]. Systemic estrogen treatment can help reduce osteoporotic fractures in postmenopausal women. Accumulation in organs, on the other hand, generates negative consequences, for example, cardiovascular disease and breast cancer [77]. A controlled release to administer the lowest therapeutic dosage while avoiding systemic adverse effects may be a desired method for extending estrogen clinical uses. Various tissue-engineering technologies have been investigated in order to create local delivery for an osteoporotic bone fracture. Nano materials have recently been identified as an excellent choice for the transport of biomolecules. 17-estradiol (E2) was put into a nano fibrous matrix, which demonstrated improved cell growth and osteoblast development mediators [78].
3.8. Selective Estrogen Receptor Modulators (SERMs)
Selective estrogen receptor modulators (SERMs) are non-steroidal compounds that have estrogenic actions on the bone, vascular system, and lipid profile, while also having anti-estrogenic effects on the breast and uterine [79][80]. Through an estrogenic action on the skeletal structure, they promote endochondral ossification, bone production, and callus remodeling [81]. By reducing osteoblast and osteoclast bone turnover, selective estrogen receptor modulators decrease bone degradation and lessen the fracture probability [82][83]. Several SERMs are now being used in clinical settings, including Raloxifene, Tamoxifene, bazedoxifene, Lasofoxifene, Ospemifene, Arzoxifene, Droloxifene, Idoxifene, and Fulvestrant [84][85]. Tamoxifen is a therapy for breast cancer that reduces osteoclast-mediated bone resorption [86][87]. Both raloxifene and bazedoxifene are SERMs that have been demonstrated to reduce bone resorption activity in postmenopausal osteoporosis patients [83][85][88][89] and have been utilized to keep bone fragility fractures at bay. SERM binding to estrogen receptors (ERs) modifies the receptor’s structure or capacity to form a combination with co-regulators, altering their expression levels [90][91][92][93][94].
3.9. 1, 25(OH) 2 Vitamin D3
Vitamin D is a fat-soluble hormone that governs bone development and strength and helps to maintain calcium-phosphorus proportions. Scientific proof suggests that vitamin D plays an autocrine function in bone production, mineralization, and degeneration. 1, 25(OH) 2 D3 influences osteoblastic protein production via the (MAPK) ERK1/2 system [95][96]. Many studies have demonstrated that vitamin D has a high capability in both osteoinduction and odontiinduction. At modest doses of this chemical, the expression of OCN, OPN, DSPP, DMP-1, and bone mineralization has enhanced [97]. Bordini et al. created a scaffold loaded with 1 nM 1, 25-dihydroxy vitamin D3. They discovered that vitamin D3 can boost odontoblastic marker expression [98].
A cellulose/hydroxyapatite/mesoporous silica scaffold was created and supplemented with vitamin D3 in a similar work. In vitro research revealed that vitamin D3 might improve cell adhesion and proliferation (MG63). Furthermore, the ALP activity and calcium accumulation assays validated the synergistic effects of hydroxyapatite and vitamin D [99].
3.10. Melatonin
Melatonin’s (ML) involvement in hard tissues has gotten a lot of attention [100][101]. The indoleamine ML (N-acetyl-5-metoxy-tryptamine) is produced and released by the pineal gland in a circadian rhythm [102]. Melatonin is also produced in possibly all organs in numbers of orders of magnitude greater than in the pineal gland and bloodstream [103]. ML may be implicated in the formation of hard tissues such as bone and teeth [104]. ML stimulates alkaline phosphatase activity and tissue mineralization [105]. As previously indicated, ML has been employed for its anti-inflammatory, antioxidant, and free-radical-scavenging qualities [106][107] and cytoprotective properties [108][109]. When there is a large quantity of ML, the generation of inflammatory mediators decreases via modulating the NFkB activity, which contributes to the signaling route.
While the favorable benefits of ML on periodontal regeneration have been proven in gingival fibroblasts as well as in experimental animals, more research is needed. [108]. ML has a circulation half-life of around 23 min [110]. As a result, a few writers have advocated for the use of vehicles in ML to slowly release it and enhance the duration of action in tissues. Steady ML release using poly-lactic-co-glycolic acid micro particles has been demonstrated to convert human mesenchymal stem cells into osteoblasts. Melatonin-loaded chitosan (ML-CS) micro particles (MPs) can modulate Mel release over time, accelerating osteogenic differentiation of preosteoblast cells in vitro [111]. Local administration of 2 mg melatonin gel is a viable treatment method for effective bone and PDL regeneration in diabetic rats [112]. Melatonin has the potential to be a promising implant coating. When powdered melatonin was applied to implant sites, it caused considerably increased bone growth and bone mineralization in canines in comparison with control groups [113]. Melatonin improves the osteogenic properties of bone grafts around dental implants in canines [86]. The findings of a 3-month clinical investigation demonstrate that melatonin may be therapeutically useful in improving the Osseo integration of dental implants [114] Novel ML delivery methods, such as ML microspheres, have demonstrated tremendous potential for application in regenerative medicine and dentistry, particularly in bone-grafting techniques, to stimulate new bone growth [115].
3.11. Erythropoietin
Erythropoietin (EPO), a glycoprotein that is generally known as an important stimulant of erythropoiesis, is released by kidneys in adult animals and in the liver during intrauterine life [116]. Erythropoietin (EPO) is a glycoprotein hormone with a low molecular weight (30–36 kDa) that stimulates erythropoiesis. RhEPO received FDA approval in 1989, and it is now used to treat anemia caused by renal insufficiency, chemotherapy, bone marrow transplant, and AIDS [117][118][119]. EPO has non hematopoietic cellular receptors in skin, and the presence of EPO receptors on endothelial cells [92][93] and macrophages has been documented [120][121] in macrophages [122], fibroblasts, and mast cells [123][124]. Erythropoietin and its ligands are found in both the central and peripheral nervous systems [125][126]. Erythropoietin boosts anti oxidative enzyme synthesis, antagonizes glutamate cytotoxicity, influences neurotransmitter release, and induces neo angiogenesis [127]. Unlike previously held beliefs that EPO was exclusively beneficial in the formation of erythropoiesis, Epo has been shown to have multiple effects, such as tissue modulation in a variety of cell types [128][129][130][131]. There is growing evidence that EPO plays biological roles in tissues outside than the hematopoietic system, which has sparked major experimental interest. EPO is a tissue-protective hormone that promotes wound healing in a variety of damage scenarios such as tissue/organ inflammation [132]. The healing of skin lesions in rats with intentionally induced diabetes is expedited by the local administration of recombinant human EPO to the wounds, which stimulates angiogenesis, reepithelialization, and collagen deposition, while inhibiting inflammatory process and apoptosis [133]. Fibronectin supplements EPO’s positive effects on wound healing in diabetics (FN). FN promotes the establishment of the preliminary wound matrix and keeps it from dissolving [134].
3.12. Calcitonin (CTN)
Calcitonin (CTN), a hormone secreted by par follicular cells (C cells) in the thyroid gland, is crucial in bone maintenance and calcium metabolic control [135]. CTN binds to osteoclasts only in bone tissues, demonstrating the greatest expression of calcitonin receptor (CTR), and triggers osteoclast activity to cease [135][136]. CTN, according to Granholm et al., suppresses osteoclast development in mouse hematopoietic cells through modulating RANK signaling [137]. CTN has also been used to treat hypercalcemia from cancer and postmenopausal osteoporosis [138]. In rats with periodontitis, local injection of CTN reduced alveolar bone resorption through controlling osteoclast activation [139].
References
- Mariotti, A. Sex steroid hormones and cell dynamics in the periodontium. Crit. Rev. Oral. Biol. Med. 1994, 5, 27–53.
- McCauley, L.K.; Tozum, T.; Rosol, T. Estrogen Receptors in Skeletal Metabolism: Lessons from Genetically Modified Models of Receptor Function. Crit. Rev. Eukaryot. Gene Expr. 2002, 12, 89–100.
- Riggs, B.L.; Khosla, S.; Melton, L.J. Sex steroids and the construction and conservation of the adult skeleton. Endocr. Rev. 2002, 23, 279–302.
- Lorenzo, J. A new hypothesis for how sex steroid hormones regulate bone mass. J. Clin. Investig. 2003, 111, 1641–1643.
- Wang, Q.; Alén, M.; Nicholson, P.H.F.; Halleen, J.M.; Alatalo, S.L.; Ohlsson, C.; Suominen, H.; Cheng, S. Differential Effects of Sex Hormones on Peri- and Endocortical Bone Surfaces in Pubertal Girls. J. Clin. Endocrinol. Metab. 2006, 91, 277–282.
- Mascarenhas, P.; Gapski, R.; Al-Shammari, K.; Wang, H.-L. Influence of sex hormones on the periodontium. J. Clin. Periodontol. 2003, 30, 671–681.
- Nagai, N.; Yunoki, S.; Suzuki, T.; Sakata, M.; Tajima, K.; Munekata, M. Application of cross-linked salmon atelocollagen to the scaffold of human periodontal ligament cells. J. Biosci. Bioeng. 2004, 97, 389–394.
- AlRowis, R.; AlMoharib, H.S.; AlMubarak, A.; Bhaskardoss, J.; Preethanath, R.S.; Anil, S. Oral fluid-based biomarkers in periodontal disease–Part 2. Gingival crevicular fluid. J. Int. Oral Health 2014, 6, 126.
- Yang, J.; Sun, X.; Zhang, Y.; Chen, Y. The application of natural polymer–based hydrogels in tissue engineering. In Hydrogels Based on Natural Polymers; Elsevier: Amsterdam, The Netherlands, 2020; pp. 273–307.
- Kini, U.; Nandeesh, B. Physiology of bone formation, remodeling, and metabolism. In Radionuclide and Hybrid Bone Imaging; Springer: Berlin/Heidelberg, Germany, 2012; pp. 29–57.
- Zhang, D.; Wu, X.; Chen, J.; Lin, K. The development of collagen based composite scaffolds for bone regeneration. Bioact. Mater. 2018, 3, 129–138.
- Cavalu, S.; Fritea, L.; Brocks, M.; Barbaro, K.; Murvai, G.; Costea, T.O.; Antoniac, I.; Verona, C.; Romani, M.; Latini, A.; et al. Novel Hybrid Composites Based on PVA/SeTiO2 Nanoparticles and Natural Hydroxyapatite for Orthopedic Applications: Correlations between Structural, Morphological and Biocompatibility Properties. Materials 2020, 13, 2077.
- Huang, Q.; Huang, X.; Gu, L. Periodontal Bifunctional Biomaterials: Progress and Perspectives. Materials 2021, 14, 7588.
- Zang, S.; Mu, R.; Chen, F.; Wei, X.; Zhu, L.; Han, B.; Yu, H.; Bi, B.; Chen, B.; Wang, Q.; et al. Injectable chitosan/β-glycerophosphate hydrogels with sustained release of BMP-7 and ornidazole in periodontal wound healing of class III furcation defects. Mater. Sci. Eng. C 2019, 99, 919–928.
- Radulescu, D.-E.; Neacsu, I.A.; Grumezescu, A.-M.; Andronescu, E. Novel Trends into the Development of Natural Hydroxyapatite-Based Polymeric Composites for Bone Tissue Engineering. Polymers 2022, 14, 899.
- Iqbal, N.; Khan, A.S.; Asif, A.; Yar, M.; Haycock, J.W.; Rehman, I.U. Recent concepts in biodegradable polymers for tissue engineering paradigms: A critical review. Int. Mater. Rev. 2018, 64, 91–126.
- Lu, H.-T.; Lu, T.-W.; Chen, C.-H.; Mi, F.-L. Development of genipin-crosslinked and fucoidan-adsorbed nano-hydroxyapatite/hydroxypropyl chitosan composite scaffolds for bone tissue engineering. Int. J. Biol. Macromol. 2019, 128, 973–984.
- Bottino, M.C.; Thomas, V.; Schmidt, G.; Vohra, Y.K.; Chu, T.-M.G.; Kowolik, M.J.; Janowski, G.M. Recent advances in the development of GTR/GBR membranes for periodontal regeneration—A materials perspective. Dent. Mater. 2012, 28, 703–721.
- Moioli, E.K.; Clark, P.A.; Xin, X.; Lal, S.; Mao, J.J. Matrices and scaffolds for drug delivery in dental, oral and craniofacial tissue engineering. Adv. Drug Deliv. Rev. 2007, 59, 308–324.
- Yadegari, A.; Fahimipour, F.; Rasoulianboroujeni, M.; Dashtimoghadarm, E.; Omidi, M.; Golzar, H.; Tahriri, M.; Tayebi, L. 10—Specific considerations in scaffold design for oral tissue engineering. In Biomaterials for Oral and Dental Tissue Engineering; Tayebi, L., Moharamzadeh, K., Eds.; Woodhead Publishing: Sawston, UK, 2017; pp. 157–183. ISBN 9780081009611.
- Zhao, H.; Chai, Y. Stem cells in teeth and craniofacial bones. J. Dent. Res. 2015, 94, 1495–1501.
- Hughes, D.; Song, B. Dental and Nondental Stem Cell Based Regeneration of the Craniofacial Region: A Tissue Based Approach. Stem Cells Int. 2016, 2016, 1–20.
- El-Sayed, K.M.F.; Dörfer, C.E. Gingival Mesenchymal Stem/Progenitor Cells: A Unique Tissue Engineering Gem. Stem Cells Int. 2016, 2016, 7154327.
- Najeeb, Z.S.; Zafar, M.S.; Khurshid, Z.; Zohaib, S.; Almas, K. The role of nutrition in periodontal health: An update. Nutrients 2016, 8, 530.
- Péault, B.; Asatrian, G.; Pham, D.; Hardy, W.R.; James, A.W. Stem cell technology for bone regeneration: Current status and potential applications. Stem Cells Cloning Adv. Appl. 2015, 8, 39–48.
- Szulc, M.; Zakrzewska, A.; Zborowski, J. Local drug delivery in periodontitis treatment: A review of contemporary literature. Dent. Med Probl. 2018, 55, 333–342.
- Woo, H.N.; Cho, Y.J.; Tarafder, S.; Lee, C.H. The recent advances in scaffolds for integrated periodontal regeneration. Bioact. Mater. 2021, 6, 3328–3342.
- Greenstein, G.; Polson, A. The Role of Local Drug Delivery in the Management of Periodontal Diseases: A Comprehensive Review. J. Periodontol. 1998, 69, 507–520.
- Styne, D.M. Introduction to Pediatric Endocrinology: The Endocrine System; Springer: Berlin/Heidelberg, Germany, 2016; pp. 1–10.
- Visser, R.; Rico-Llanos, G.A.; Pulkkinen, H.; Becerra, J. Peptides for bone tissue engineering. J. Control. Release 2016, 244, 122–135.
- Aleem, A.R.; Shahzadi, L.; Alvi, F.; Khan, A.F.; Chaudhry, A.A.; ur Rehman, I.; Yar, M. Thyroxin releasing chitosan/collagen based smart hydrogels to stimulate neovascularization. Mater. Des. 2017, 133, 416–425.
- Luidens, M.K.; Mousa, S.A.; Davis, F.B.; Lin, H.-Y.; Davis, P.J. Thyroid hormone and angiogenesis. Vasc. Pharmacol. 2010, 52, 142–145.
- Sirakov, M.; Skah, S.; Nadjar, J.; Plateroti, M. Thyroid hormone’s action on progenitor/stem cell biology: New challenge for a classic hormone? Biochim. Biophys. Acta 2013, 1830, 3917–3927.
- Malik, M.H.; Shahzadi, L.; Batool, R.; Safi, S.Z.; Khan, A.S.; Khan, A.F.; Chaudhry, A.A.; Rehman, I.U.; Yar, M. Thyroxine-loaded chitosan/carboxymethyl cellulose/hydroxyapatite hydrogels enhance angiogenesis in in-ovo experiments. Int. J. Biol. Macromol. 2020, 145, 1162–1170.
- Shahzadi, L.; Bashir, M.; Tehseen, S.; Zehra, M.; Mehmood, A.; Chaudhry, A.A.; Rehman, I.U.; Yar, M. Thyroxine impregnated chitosan-based dressings stimulate angiogenesis and support fast wounds healing in rats: Potential clinical candidates. Int. J. Biol. Macromol. 2020, 160, 296–306.
- Colaianni, G.; Sun, L.; Di Benedetto, A.; Tamma, R.; Zhu, L.-L.; Cao, J.; Grano, M.; Yuen, T.; Colucci, S.; Cuscito, C.; et al. Bone Marrow Oxytocin Mediates the Anabolic Action of Estrogen on the Skeleton. J. Biol. Chem. 2012, 287, 29159–29167.
- Colaianni, G.; Di Benedetto, A.; Zhu, L.-L.; Tamma, R.; Li, J.; Greco, G.; Peng, Y.; Dell’Endice, S.; Zhu, G.; Cuscito, C.; et al. Regulated production of the pituitary hormone oxytocin from murine and human osteoblasts. Biochem. Biophys. Res. Commun. 2011, 411, 512–515.
- Elnagar, A.; El-Dawy, K.; El-Belbasi, H.I.; Rehan, I.F.; Embark, H.; Al-Amgad, Z.; Shanab, O.; Mickdam, E.; Batiha, G.E.; Alamery, S.; et al. Ameliorative Effect of Oxytocin on FBN1 and PEPCK Gene Expression, and Behavioral Patterns in Rats’ Obesity-Induced Diabetes. Front. Public Health 2022, 10, 777129.
- Dawood, M.Y. Novel approach to oxytocin induction-augmentation of labor. Application of oxytocin physiology during pregnancy. Single Mol. Single Cell Seq. 1995, 395, 585–594.
- Makadia, H.K.; Siegel, S.J. Poly lactic-co-glycolic acid (PLGA) As biodegradable controlled drug delivery carrier. Polymers 2011, 3, 1377–1397.
- Akay, A.S.; Arısan, V.; Cevher, E.; Sessevmez, M.; Cam, B. Oxytocin-loaded sustained-release hydrogel graft provides accelerated bone formation: An experimental rat study. J. Orthop. Res. 2020, 38, 1676–1687.
- Park, J.-W.; Kim, J.-M.; Lee, H.-J.; Jeong, S.-H.; Suh, J.-Y.; Hanawa, T. Bone healing with oxytocin-loaded microporous β-TCP bone substitute in ectopic bone formation model and critical-sized osseous defect of rat. J. Clin. Periodontol. 2014, 41, 181–190.
- Ge, B.; Liu, H.; Liang, Q.; Shang, L.; Wang, T.; Ge, S. Oxytocin facilitates the proliferation, migration and osteogenic differentiation of human periodontal stem cells in vitro. Arch. Oral. Biol. 2019, 99, 126–133.
- Chen, Y.; Kawazoe, N.; Chen, G. Preparation of dexamethasone-loaded biphasic calcium phosphate nanoparticles/collagen porous composite scaffolds for bone tissue engineering. Acta Biomater. 2018, 67, 341–353.
- Jørgensen, N.; Henriksen, Z.; Sørensen, O.; Civitelli, R. Dexamethasone, BMP-2, and 1,25-dihydroxyvitamin D enhance a more differentiated osteoblast phenotype: Validation of an in vitro model for human bone marrow-derived primary osteoblasts. Steroids 2004, 69, 219–226.
- Martins, A.; Duarte, A.R.C.; Faria, S.; Marques, A.P.; Reis, R.L.; Neves, N.M. Osteogenic induction of hBMSCs by electrospun scaffolds with dexamethasone release functionality. Biomaterials 2010, 31, 5875–5885.
- Li, L.; Zhou, G.; Wang, Y.; Yang, G.; Ding, S.; Zhou, S. Controlled dual delivery of BMP-2 and dexamethasone by nanoparticle-embedded electrospun nanofibers for the efficient repair of critical-sized rat calvarial defect. Biomaterials 2015, 37, 218–229.
- Ren, H.; Chen, S.; Jin, Y.; Zhang, C.; Yang, X.; Ge, K.; Liang, X.-J.; Li, Z.; Zhang, J. A traceable and bone-targeted nanoassembly based on defect-related luminescent mesoporous silica for enhanced osteogenic differentiation. J. Mater. Chem. B 2017, 5, 1585–1593.
- Zhou, X.; Liu, P.; Nie, W.; Peng, C.; Li, T.; Qiang, L.; He, C.; Wang, J. Incorporation of dexamethasone-loaded mesoporous silica nanoparticles into mineralized porous biocomposite scaffolds for improving osteogenic activity. Int. J. Biol. Macromol. 2020, 149, 116–126.
- Porter, R.M.; Huckle, W.R.; Goldstein, A.S. Effect of dexamethasone withdrawal on osteoblastic differentiation of bone marrow stromal cells. J. Cell. Biochem. 2003, 90, 13–22.
- Kim, H.; Suh, H.; Jo, S.A.; Kim, H.W.; Lee, J.M.; Kim, E.H.; Reinwald, Y.; Park, S.-H.; Min, B.-H.; Jo, I. In vivo bone formation by human marrow stromal cells in biodegradable scaffolds that release dexamethasone and ascorbate-2-phosphate. Biochem. Biophys. Res. Commun. 2005, 332, 1053–1060.
- Chauhan, N.; Gupta, P.; Arora, L.; Pal, D.; Singh, Y. Dexamethasone-loaded, injectable pullulan-poly(ethylene glycol) hydrogels for bone tissue regeneration in chronic inflammatory conditions. Mater. Sci. Eng. C 2021, 130, 112463.
- Koons, G.L.; Diba, M.; Mikos, A.G. Materials design for bone-tissue engineering. Nat. Rev. Mater. 2020, 5, 584–603.
- Van de Ven, C.J.J.M.; Bakker, N.E.C.; Link, D.P.; Geven, E.J.W.; Gossen, J.A. Sustained release of ancillary amounts of testosterone and alendronate from PLGA coated pericard membranes and implants to improve bone healing. PLoS ONE 2021, 16, e0251864.
- Cheng, B.-H.; Chu, T.-M.G.; Chang, C.; Kang, H.-Y.; Huang, K.-E. Testosterone Delivered with a Scaffold Is as Effective as Bone Morphologic Protein-2 in Promoting the Repair of Critical-Size Segmental Defect of Femoral Bone in Mice. PLoS ONE 2013, 8, e70234.
- Teitelbaum, S. Bone resorption by osteoclasts. Science 2000, 289, 1504–1508.
- Capriani, C.; Irani, D.; Bilezikian, J.P.; Research, M. Safety of osteoanabolic therapy: A decade of experience. JBMR 2012, 27, 2419–2428.
- Vanea, E.; Moraru, C.; Vulpoi, A.; Cavalu, S.; Simon, V. Freeze-dried and spray-dried zinc-containing silica microparticles entrapping insulin. J. Biomater. Appl. 2014, 28, 1190–1199.
- Maratova, K.; Soucek, O.; Matyskova, J.; Hlavka, Z.; Petruzelkova, L.; Obermannova, B.; Pruhova, S.; Kolouskova, S.; Sumnik, Z. Muscle functions and bone strength are impaired in adolescents with type 1 diabetes. Bone 2018, 106, 22–27.
- Nordklint, A.K.; Almdal, T.P.; Vestergaard, P.; Lundby-Christensen, L.; Jørgensen, N.R.; Boesgaard, T.W.; Breum, L.; Gade-Rasmussen, B.; Sneppen, S.B.; Gluud, C.; et al. Effect of Metformin vs. Placebo in Combination with Insulin Analogues on Bone Markers P1NP and CTX in Patients with Type 2 Diabetes Mellitus. Calcif. Tissue Res. 2020, 107, 160–169.
- Vestergaard, P.; Rejnmark, L.; Mosekilde, L. Relative fracture risk in patients with diabetes mellitus, and the impact of insulin and oral antidiabetic medication on relative fracture risk. Diabetologia 2005, 48, 1292–1299.
- Hynes, B.; Kumar, A.; O’Sullivan, J.; Buneker, C.K.; Leblond, A.-L.; Weiss, S.; Schmeckpeper, J.; Martin, K.; Caplice, N.M. Potent endothelial progenitor cell-conditioned media-related anti-apoptotic, cardiotrophic, and pro-angiogenic effects post-myocardial infarction are mediated by insulin-like growth factor-1. Eur. Hear. J. 2011, 34, 782–789.
- Paglia, D.N.; Wey, A.; Breitbart, E.A.; Faiwiszewski, J.; Mehta, S.K.; Al-Zube, L.; Vaidya, S.; Cottrell, J.A.; Graves, D.; Benevenia, J.; et al. Effects of local insulin delivery on subperiosteal angiogenesis and mineralized tissue formation during fracture healing. J. Orthop. Res. 2012, 31, 783–791.
- Rabinovsky, E.D.; Draghia-Akli, R. Insulin-like growth factor I plasmid therapy promotes in vivo angiogenesis. Mol. Ther. 2004, 9, 46–55.
- Cornish, J.; Callon, K.E.; Reid, I.R. Insulin increases histomorphometric indices of bone formation in vivo. Calcif. Tissue Int. 1996, 59, 492–495.
- Thomas, D.; Udagawa, N.; Hards, D.; Quinn, J.; Moseley, J.; Findlay, D.; Best, J.D. Insulin receptor expression in primary and cultured osteoclast-like cells. Bone 1998, 23, 181–186.
- Xian, L.; Wu, X.; Pang, L.; Lou, M.; Rosen, C.J.; Qiu, T.; Crane, J.; Frassica, F.; Zhang, L.; Rodriguez, J.P.; et al. Matrix IGF-1 maintains bone mass by activation of mTOR in mesenchymal stem cells. Nat. Mat. 2012, 18, 1095–1101.
- Zhang, X.; Xing, H.; Qi, F.; Liu, H.; Gao, L.; Wang, X. Local delivery of insulin/IGF-1 for bone regeneration: Carriers, strategies, and effects. Nanotheranostics 2020, 4, 242–255.
- Wang, X.; Zhang, G.; Qi, F.; Cheng, Y.; Lu, X.; Wang, L.; Zhao, J.; Zhao, B. Enhanced bone regeneration using an insulin-loaded nano-hydroxyapatite/collagen/PLGA composite scaffold. Int. J. Nanomed. 2017, 13, 117–127.
- Wang, X.; Qi, F.; Xing, H.; Zhang, X.; Lu, C.; Zheng, J.; Ren, X. Uniform-sized insulin-loaded PLGA microspheres for improved early-stage peri-implant bone regeneration. Drug Deliv. 2019, 26, 1178–1190.
- Krajcer, A.; Klara, J.; Horak, W.; Lewandowska-Łańcucka, J. Bioactive injectable composites based on insulin-functionalized silica particles reinforced polymeric hydrogels for potential applications in bone tissue engineering. J. Mater. Sci. Technol. 2022, 105, 153–163.
- Wang, B.; Song, Y.; Wang, F.; Li, D.; Zhang, H.; Ma, A.; Huang, N. Effects of local infiltration of insulin around titanium implants in diabetic rats. Br. J. Oral Maxillofac. Surg. 2011, 49, 225–229.
- Hofbauer, L.C.; Heufelder, A.E. Role of receptor activator of nuclear factor-κB ligand and osteoprotegerin in bone cell biology. J. Mol. Med. 2001, 79, 243–253.
- Khosla, S.; Oursler, M.J.; Monroe, D.G. Estrogen and the skeleton. Trends Endocrinol. Metab. 2012, 23, 576–581.
- Irmak, G.; Demirtaş, T.T.; Altındal, D.Ç.; Çalış, M.; Gümüşderelioğlu, M. Sustained release of 17β-estradiol stimulates osteogenic differentiation of adipose tissue-derived mesenchymal stem cells on chitosan-hydroxyapatite scaffolds. Cells Tissues Organs 2014, 199, 37–50.
- Ettinger, B.; Pressman, A.; Sklarin, P.; Bauer, D.C.; Cauley, J.A.; Cummings, S.R. Associations between Low Levels of Serum Estradiol, Bone Density, and Fractures among Elderly Women: The Study of Osteoporotic Fractures1. J. Clin. Endocrinol. Metab. 1998, 83, 2239–2243.
- Bowring, C.; Francis, R.M. National Osteoporosis Society’s Position statement on hormone replacement therapy in the prevention and treatment of osteoporosis. Menopause Int. 2011, 17, 63–65.
- Wang, D.; Steffi, C.; Wang, Z.; Kong, C.H.; Lim, P.N.; Shi, Z.; Thian, E.S.; Wang, W. Beta-cyclodextrin modified mesoporous bioactive glass nanoparticles/silk fibroin hybrid nanofibers as an implantable estradiol delivery system for the potential treatment of osteoporosis. Nanoscale 2018, 10, 18341–18353.
- Ott, S.M.; Oleksik, A.; Lu, Y.; Harper, K.; Lips, P. Bone Histomorphometric and Biochemical Marker Results of a 2-Year Placebo-Controlled Trial of Raloxifene in Postmenopausal Women. J. Bone Miner. Res. 2002, 17, 341–348.
- Urano, T.; Shiraki, M.; Kuroda, T.; Tanaka, S.; Uenishi, K.; Inoue, S. Preventive effects of raloxifene treatment on agerelated weight loss in postmenopausal women. J. Bone Miner. Metab. 2017, 35, 108–113.
- Spiro, A.S.; Khadem, S.; Jeschke, A.; Marshall, R.P.; Pogoda, P.; Ignatius, A.; Amling, M.; Beil, F.T. The SERM raloxifene improves diaphyseal fracture healing in mice. J. Bone Miner. Metab. 2013, 31, 629–636.
- Stringhetta-Garcia, C.T.; Singulani, M.P.; Santos, L.F.; Louzada, M.J.Q.; Nakamune, A.C.S.; Chaves-Neto, A.H.; Rossi, A.C.; Ervolino, E.; Dornelles, R.C.M. The effects of strength training and raloxifene on bone health in aging ovariectomized rats. Bone 2016, 85, 45–54.
- Ettinger, B.; Black, D.M.; Mitlak, B.H.; Knickerbocker, R.K.; Nickelsen, T.; Genant, H.K.; Christiansen, C.; Delmas, P.D.; Zanchetta, J.R.; Stakkestad, J.; et al. Reduction of vertebral fracture risk in postmenopausal women with osteoporosis treated with raloxifene: Results from a 3-year randomized clinical trial. JAMA 1999, 282, 637–645.
- Mirkin, S.; Pickar, J.H. Selective estrogen receptor modulators (SERMs): A review of clinical data. Maturitas 2015, 80, 52–57.
- Pinkerton, J.V.; Thomas, S. Use of SERMs for treatment in postmenopausal women. J. Steroid Biochem. Mol. Biol. 2014, 142, 142–154.
- Powles, T.J.; Hickish, T.; Kanis, J.A.; Tidy, A.; Ashley, S. Effect of tamoxifen on bone mineral density measured by dual-energy x-ray absorptiometry in healthy premenopausal and postmenopausal women. J. Clin. Oncol. 1996, 14, 78–84.
- Love, R.; Mazess, R.; Barden, H.; Epstein, S.; Newcomb, P.; Jordan, V.; Carbone, P.; DeMets, D.L. Effects of tamoxifen on bone material density and metabolism in postmenopausal women with breast cancer. N. Engl. J. Med. 1992, 326, 852–856.
- Blair, J.M.; Hanson, D.L.; Jones, J.L.; Dworkin, M.S. Trends in Pregnancy Rates Among Women With Human Immunodeficiency Virus. Obstet. Gynecol. 2004, 103, 663–668.
- Silverman, S.L.; Christiansen, C.; Genant, H.K.; Vukicevic, S.; Zanchetta, J.R.; de Villiers, T.J.; Constantine, G.D.; Chines, A.A. Efficacy of bazedoxifene in reducing new vertebral fracture risk in postmenopausal women with osteoporosis: Results from a 3-year, randomized, placebo-, and active-controlled clinical trial. J. Bone Miner. Res. 2008, 23, 1923–1934.
- Zhang, X.; Jeyakumar, M.; Petukhov, S.; Bagchi, M.K. A nuclear receptor corepressor modulates transcriptional activity of antagonist-occupied steroid hormone receptor. Mol. Endocrinol. 1998, 12, 513–524.
- Couse, J.F.; Korach, K.S. Estrogen receptor null mice: What have we learned and where will they lead us? Endocr. Rev. 1999, 20, 358–417.
- Aranda, A.; Pascual, A. Nuclear hormone receptors and gene expression. Physiol. Rev. 2001, 81, 1269–1304.
- Riggs, B.L.; Hartmann, L.C. Selective estrogen-receptor modulators—Mechanisms of action and application to clinical practice. N. Engl. J. Med. 2003, 348, 618–629.
- Arnott, J.; Martinkovich, S.; Planey, S.L.; Shah, D. Selective estrogen receptor modulators: Tissue specificity and clinical utility. Clin. Interv. Aging 2014, 9, 1437–1452.
- Yang, D.; Anderson, P.H.; Wijenayaka, A.R.; Barratt, K.R.; Triliana, R.; Stapledon, C.J.; Zhou, H.; Findlay, D.M.; Morris, H.A.; Atkins, G.J.; et al. Both ligand and VDR expression levels critically determine the effect of 1α, 25-dihydroxyvitamin-D3 on osteoblast differentiation. J. Steroid. Biochem. Mol. Biol. 2018, 177, 83–90.
- Griffin, A.C.; Kern, M.J.; Kirkwood, K. MKP-1 is essential for canonical vitamin D-induced signaling through nuclear import and regulates RANKL expression and function. Mol. Endocrinol. 2012, 26, 1682–1693.
- Mucuk, G.; Sepet, E.; Erguven, M.; Ekmekcı, O.; Bılır, A. 1,25-Dihydroxyvitamin D3 stimulates odontoblastic differentiation of human dental pulp-stem cells in vitro. Connect. Tissue Res. 2017, 58, 531–541.
- Bordini, E.A.F.; Cassiano, F.B.; Silva, I.S.P.; Usberti, F.R.; Anovazzi, G.; Pacheco, L.E.; Pansani, T.N.; Leite, M.L.; Hebling, J.; de Souza Costa, C.A. Synergistic potential of 1α, 25-dihydroxyvitamin D3 and calcium–aluminate–chitosan scaffolds with dental pulp cells. Clin. Oral. Investig. 2020, 24, 663–674.
- Sumathra, M.; Munusamy, M.A.; Alarfaj, A.A.; Rajan, M. Osteoblast response to Vitamin D3 loaded cellulose enriched hydroxyapatite Mesoporous silica nanoparticles composite. Biomed. Pharmacother. 2018, 103, 858–868.
- Leonida, A.; Favero, G.; Caccianiga, P.; Ceraulo, S.; Rodella, L.F.; Rezzani, R.; Caccianiga, G. Concentrated Growth Factors (CGF) Combined with Melatonin in Guided Bone Regeneration (GBR): A Case Report. Diagnostics 2022, 12, 1257.
- Shino, H.; Hasuike, A.; Arai, Y.; Honda, M.; Isokawa, K.; Sato, S. Melatonin enhances vertical bone augmentation in rat calvaria secluded spaces. Oral Surg. 2016, 21, e122–e126.
- Meenakshi, S.S.; Malaiappan, S. Role of melatonin in periodontal disease—A systematic review. Indian J. Dent. Res. 2020, 31, 593.
- Acuña-Castroviejo, D.; Rahim, I.; Acuña-Fernández, C.; Fernández-Ortiz, M.; Solera-Marín, J.; Sayed, R.K.A.; Díaz-Casado, M.E.; Rusanova, I.; Lopez, L.C.; Escames, G. Melatonin, clock genes and mitochondria in sepsis. Cell. Mol. Life Sci. 2017, 74, 3965–3987.
- Liu, J.; Huang, F.; He, H.-W. Melatonin Effects on Hard Tissues: Bone and Tooth. Int. J. Mol. Sci. 2013, 14, 10063–10074.
- Liu, J.; Zhou, H.; Fan, W.; Dong, W.; Fu, S.; He, H.; Huang, F. Melatonin influences proliferation and differentiation of rat dental papilla cells in vitro and dentine formation in vivo by altering mitochondrial activity. J. Pineal Res. 2012, 54, 170–178.
- Köse, O.; Arabaci, T.; Kizildag, A.; Erdemci, B.; Eminoğlu, D.; Gedikli, S.; Özkanlar, S.; Zihni, M.; Albayrak, M.; Kara, A.; et al. Melatonin prevents radiation-induced oxidative stress and periodontal tissue breakdown in irradiated rats with experimental periodontitis. J. Periodontal Res. 2016, 52, 438–446.
- Fernández-Ortiz, M.; Sayed, R.K.; Fernández-Martínez, J.; Cionfrini, A.; Aranda-Martínez, P.; Escames, G.; de Haro, T.; Acuña-Castroviejo, D. Melatonin/Nrf2/NLRP3 connection in mouse heart mitochondria during aging. Antioxidants 2020, 9, 1187.
- Dos Santos, R.M.; Marani, F.; Chiba, F.Y.; Mattera, M.S.D.L.C.; Tsosura, T.V.S.; Tessarin, G.W.L.; Pereira, R.F.; Belardi, B.E.; Pinheiro, B.C.E.S.; Sumida, D.H. Melatonin promotes reduction in TNF levels and improves the lipid profile and insulin sensitivity in pinealectomized rats with periodontal disease. Life Sci. 2018, 213, 32–39.
- Fernández-Gil, B.; Moneim, A.E.A.; Ortiz, F.; Shen, Y.-Q.; Soto-Mercado, V.; Mendivil-Perez, M.; Guerra-Librero, A.; Acuna-Castroviejo, D.; Molina-Navarro, M.M.; García-Verdugo, J.; et al. Melatonin protects rats from radiotherapy-induced small intestine toxicity. PLoS ONE 2017, 12, e0174474.
- Gibbs, F.P.; Vriend, J. The half-life of melatonin elimination from rat plasma. Endocrinology 1981, 109, 1796–1798.
- Huang, R.-Y.; Hsiao, P.-Y.; Mau, L.-P.; Tsai, Y.-W.C.; Cochran, D.L.; Weng, P.-W.; Cheng, W.-C.; Chung, C.-H.; Huang, Y.-C. Synthesis and Characterization of Melatonin-Loaded Chitosan Microparticles Promote Differentiation and Mineralization in Preosteoblastic Cells. J. Oral Implant. 2020, 46, 562–570.
- Yousuf, D.; Afify, O.; El Soudany, K.; Ghoniem, S. The effect of local application of melatonin gel on the healing of periodontal osseous defects in experimentally induced diabetes in rabbits. Tanta Dent. J. 2013, 10, 48–57.
- Cutando, A.; Gómez-Moreno, G.; Arana, C.; Muñoz, F.; Lopez-Peña, M.; Stephenson, J.; Reiter, R.J. Melatonin stimulates osteointegration of dental implants. J. Pineal. Res. 2008, 45, 174–179.
- Elgammal, M.Y.A.; Salem, A.S.; Anees, M.M.; Tawfik, M.A.-M. Clinical and Radiographic Evaluation of Immediate Loaded Dental Implants With Local Application of Melatonin: A Preliminary Randomized Controlled Clinical Trial. J. Oral Implant. 2016, 42, 119–125.
- Boga, J.A.; Caballero, B.; Potes, Y.; Perez-Martinez, Z.; Reiter, R.J.; Vega-Naredo, I.; Coto-Montes, A. Therapeutic potential of melatonin related to its role as an autophagy regulator: A review. J. Pineal Res. 2018, 66, e12534.
- Rangarajan, V.; Juul, S.E. Erythropoietin: Emerging role of erythropoietin in neonatal neuroprotection. Pediatr. Neurol. 2014, 51, 481–488.
- Bulmer, C.; Margaritis, A.; Xenocostas, A. Production and characterization of novel chitosan nanoparticles for controlled release of rHu-Erythropoietin. Biochem. Eng. J. 2012, 68, 61–69.
- Fayed, B.E.; Tawfik, A.F.; Yassin, A.E.B. Novel erythropoietin-loaded nanoparticles with prolonged in vivo response. J. Microencapsul. 2012, 29, 650–656.
- Jelkmann, W. Erythropoietin: Structure, control of production, and function. Physiol. Rev. 1992, 72, 449–489.
- Anagnostou, A.; Lee, E.S.; Kessimian, N.; Levinson, R.; Steiner, M. Erythropoietin has a mitogenic and positive chemotactic effect on endothelial cells. Proc. Natl. Acad. Sci. USA 1990, 87, 5978–5982.
- Anagnostou, A.; Liu, Z.; Steiner, M.; Chin, K.; Lee, E.S.; Kessimian, N.; Noguchi, C.T. Erythropoietin receptor mRNA expression in human endothelial cells. Proc. Natl. Acad. Sci. USA 1994, 91, 3974–3978.
- Haroon, Z.A.; Amin, K.; Jiang, X.; Arcasoy, M.O. A Novel Role for Erythropoietin During Fibrin-Induced Wound-Healing Response. Am. J. Pathol. 2003, 163, 993–1000.
- Isogai, R.; Takahashi, M.; Aisu, K.; Horiuti, Y.; Aragane, Y.; Kawada, A.; Tezuka, T. The receptor for erythropoietin is present on cutaneous mast cells. Arch. Derm. Res. 2006, 297, 389–394.
- Bodó, E.; Kromminga, A.; Funk, W.; Laugsch, M.; Duske, U.; Jelkmann, W.; Paus, R. Human hair follicles are an extrarenal source and a nonhematopoietic target of erythropoietin. FASEB J. 2007, 21, 3346–3354.
- Campana, W.M.; Myers, R.R. Erythropoietin and erythropoietin receptors in the peripheral nervous system: Changes after nerve injury. FASEB J. 2001, 15, 1804–1806.
- Bianchi, R.; Buyukakilli, B.; Brines, M.; Savino, C.; Cavaletti, G.; Oggioni, N.; Lauria, G.; Borgna, M.; Lombardi, R.; Cimen, B.; et al. Erythropoietin both protects from and reverses experimental diabetic neuropathy. Proc. Natl. Acad. Sci. USA 2004, 101, 823–828.
- Lykissas, M.G.; Korompilias, A.V.; Vekris, M.D.; Mitsionis, G.I.; Sakellariou, E.; Beris, A.E. The role of erythropoietin in central and peripheral nerve injury. Clin. Neurol. Neurosurg. 2007, 109, 639–644.
- Broxmeyer, H.E. Erythropoietin: Multiple targets, actions, and modifying influences for biological and clinical consideration. J. Exp. Med. 2013, 210, 205–208.
- Jelkmann, W. Erythropoietin: Back to basics. Blood 2010, 115, 4151–4152.
- Jelkmann, W.; Elliott, S. Erythropoietin and the vascular wall: The controversy continues. Nutr. Metab. Cardiovasc. Dis. 2013, 23, S37–S43.
- Nairz, M.; Sonnweber, T.; Schroll, A.; Theurl, I.; Weiss, G. The pleiotropic effects of erythropoietin in infection and inflammation. Microbes Infect. 2012, 14, 238–246.
- Brines, M.; Cerami, A. Erythropoietin-mediated tissue protection: Reducing collateral damage from the primary injury response. J. Intern. Med. 2008, 264, 405–432.
- Hamed, S.; Ullmann, Y.; Masoud, M.; Hellou, E.; Khamaysi, Z.; Teot, L. Topical Erythropoietin Promotes Wound Repair in Diabetic Rats. J. Investig. Dermatol. 2010, 130, 287–294.
- Hamed, S.; Ullmann, Y.; Egozi, D.; Daod, E.; Hellou, E.; Ashkar, M.; Gilhar, A.; Teot, L. Fibronectin Potentiates Topical Erythropoietin-Induced Wound Repair in Diabetic Mice. J. Investig. Dermatol. 2011, 131, 1365–1374.
- De Paula, F.J.A.; Rosen, C.J. Back to the Future: Revisiting Parathyroid Hormone and Calcitonin Control of Bone Remodeling. Horm. Metab. Res. 2010, 42, 299–306.
- Wallach, S.; Carstens, J.; Avioli, L.V. Calcitonin, osteoclasts, and bone turnover. Calcif. Tissue Int. 1990, 47, 388–391.
- Granholm, S.; Lundberg, P.; Lerner, U.H. Calcitonin inhibits osteoclast formation in mouse haematopoetic cells independently of transcriptional regulation by receptor activator of NF-kappa B and c-Fms. J. Endocrinol 2007, 195, 415–428.
- Zaidi, M.; Moonga, B.S.; Abe, E. Calcitonin and bone formation: A knockout full of surprises. J. Clin. Investig. 2002, 110, 1769–1771.
- Wada-Mihara, C.; Seto, H.; Ohba, H.; Tokunaga, K.; Kido, J.-I.; Nagata, T.; Naruishi, K. Local administration of calcitonin inhibits alveolar bone loss in an experimental periodontitis in rats. Biomed. Pharmacother. 2018, 97, 765–770.
More
Information
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
2.7K
Entry Collection:
Peptides for Health Benefits
Revisions:
2 times
(View History)
Update Date:
26 Jul 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





