Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Milena Magalhães Aleluia | -- | 1221 | 2022-06-22 15:32:11 | | | |
| 2 | Milena Magalhães Aleluia | Meta information modification | 1221 | 2022-06-27 17:13:48 | | | | |
| 3 | Amina Yu | -173 word(s) | 1048 | 2022-06-28 03:32:09 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Santos, E.D.C.; Melo, G.I.V.; Santana, P.V.B.; Quadros, I.G.S.; Yahouédéhou, S.C.M.A.; Guarda, C.C.D.; Santiago, R.P.; Fiuza, L.M.; Carvalho, S.P.; Adorno, E.V.; et al. Hemolytic Component in Sickle Leg Ulcer. Encyclopedia. Available online: https://encyclopedia.pub/entry/24354 (accessed on 09 May 2026).
Santos EDC, Melo GIV, Santana PVB, Quadros IGS, Yahouédéhou SCMA, Guarda CCD, et al. Hemolytic Component in Sickle Leg Ulcer. Encyclopedia. Available at: https://encyclopedia.pub/entry/24354. Accessed May 09, 2026.
Santos, Edvan Do Carmo, Gabriela Imbassahy Valentim Melo, Paulo Vinícius Bispo Santana, Idaiara Graziele Silva Quadros, Sètondji Cocou Modeste Alexandre Yahouédéhou, Caroline Conceição Da Guarda, Rayra Pereira Santiago, Luciana Magalhães Fiuza, Suéllen Pinheiro Carvalho, Elisângela Vitória Adorno, et al. "Hemolytic Component in Sickle Leg Ulcer" Encyclopedia, https://encyclopedia.pub/entry/24354 (accessed May 09, 2026).
Santos, E.D.C., Melo, G.I.V., Santana, P.V.B., Quadros, I.G.S., Yahouédéhou, S.C.M.A., Guarda, C.C.D., Santiago, R.P., Fiuza, L.M., Carvalho, S.P., Adorno, E.V., Kaneto, C.M., Fonseca, T.C.C., Goncalves, M.S., & Aleluia, M.M. (2022, June 22). Hemolytic Component in Sickle Leg Ulcer. In Encyclopedia. https://encyclopedia.pub/entry/24354
Santos, Edvan Do Carmo, et al. "Hemolytic Component in Sickle Leg Ulcer." Encyclopedia. Web. 22 June, 2022.
Copy Citation
Sickle leg ulcers (SLU) are malleoli lesions with exuberant hemolytic pathophysiology. The microRNAs are potential genetic biomarkers for several pathologies. Thereby, the expression of circulating miR-199a-5p, miR-144, and miR-126 in association with hemolytic biomarkers in SLU was assessed.
sickle cell disease
sickle leg ulcer
microRNA
hemolysis
biomarkers
1. Introduction
Sickle cell disease (SCD) is a common genetic disorder in Brazil, with 3500 new cases annually, and characterized by intravascular hemolysis, sterile inflammation, reduced nitric oxide (NO) bioavailability, and endothelial dysfunction [1]. The chronic hemolysis leads to vaso-occlusive episodes and clinical manifestations, such as painful crises, acute chest syndrome, stroke, and sickle leg ulcers (SLU) [2].
The SLU are cutaneous lesions with raised edges that frequently affect the malleolus of SCD patients [3]. SLU wound bed may present unviable tissues, such as necrotic tissue due to accumulation of dead cells or sloughy tissue without vascularization and constituted by cells fragments. In addition, the SLU wound bed may present viable granulation tissue with the proliferation of blood capillaries and epithelized tissue consisting of regenerated and dry epidermis [3][4].
The SLU occurrence has been associated with morbidity, work disability, social restriction, frequent recurrence, and recalcitration [2][5]. Thus, the evaluation of these factors is insufficient to understand the SLU clinical profile. Molecular biology techniques revealed the existence of microRNAs (miRNAs) which may modulate pathophysiology mechanisms [6]. miRNAs are small non-coding RNA molecules which act as positive or negative regulators in several pathologies, including SCD [6][7]. In this sense, miR-199a-5p has been associated with NO metabolism in vitro [8] and miR-144 may regulate fetal hemoglobin (HbF) production through gene silencing [6]. miR-126 has been associated with protective mechanisms against vascular damage in vitro and in vivo [9]. However, the role of these miRNAs in the intravascular hemolysis related to SLU occurrence is unknown.
2. Circulating miRNAs Expression in SCD Patients and between HbSS and HbSC Genotype with and without SLU
Regarding SLU, there was higher miR-199a-5p (Figure 1a) and miR-144 (Figure 1b) expression, as well as lower miR-126 (Figure 1c) expression in SLU+ patients (p < 0.05).
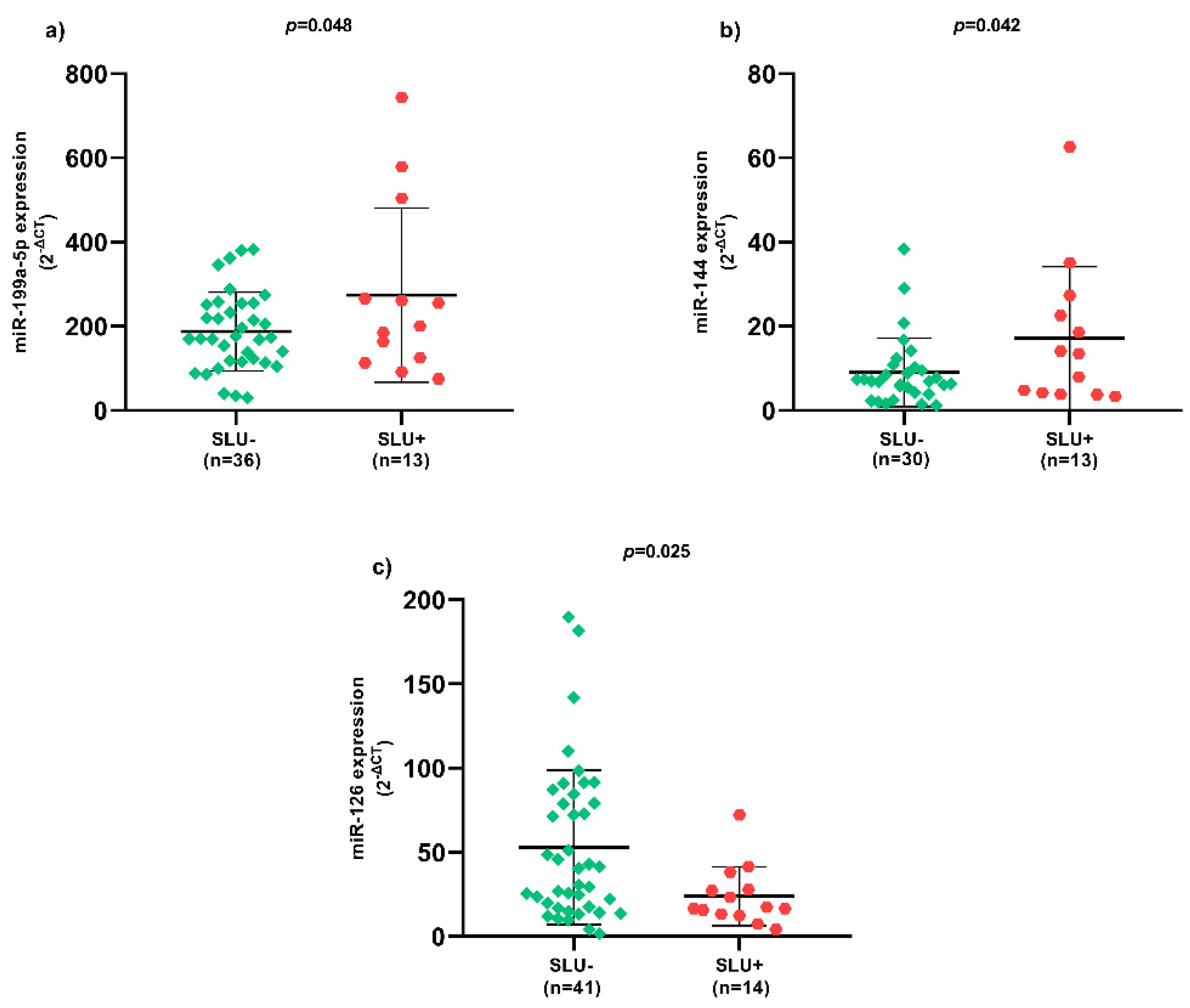
Figure 1. Expression of circulating miRNAs between SLU- and SLU+ patients. (a) Expression of circulating miR-199a-5p between SLU- and SLU+ patients; (b) Expression of circulating miR-144 between SLU- and SLU+ patients; (c) Expression of circulating miR-126 between SLU- and SLU+ patients. SLU-, patients without sickle leg ulcer; SLU+, patients with active sickle leg ulcer or previous history; miR, microRNA; p-value obtained using independent t-test.
In SLU- patients with HbSC genotype, there was higher expression of circulating miR-199a-5p, miR-144, and miR-126. In addition, circulating miR-126 expression was higher in SLU+ patients with HbSC genotype.
3. Correlation Coefficients between Hemolytic Biomarkers and Circulating miRNAs Expression in SLU+ Patients
miR-199a-5p expression was positively correlated with NOm, LDH, AST, GGT, iron, and ferritin levels, and negatively correlated with red blood cell (RBC) count and hemoglobin levels (p < 0.05) (Figure 2a). Moreover, there were positive correlations between miR-144 expression and NOm, LDH, indirect bilirubin, AST, GGT, iron, and ferritin levels, as well as negative correlations with RBC count and hemoglobin levels (p < 0.05) (Figure 2b). Regarding miR-126 expression, there were negative correlations with indirect bilirubin, iron and ferritin levels (p < 0.05) (Figure 2c).
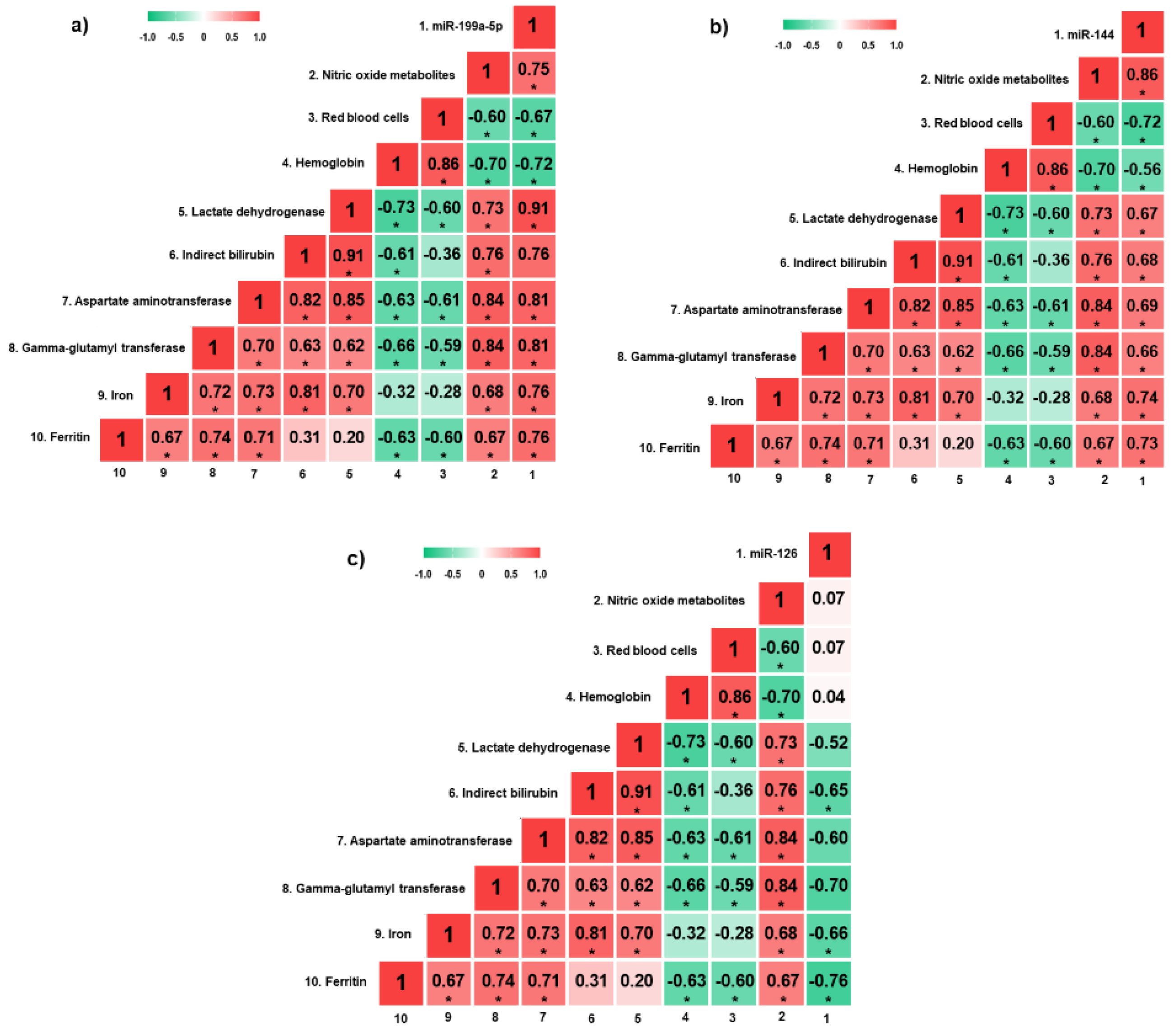
Figure 2. Heatmap of correlation coefficients between hemolytic biomarkers and circulating miRNAs in SLU+ patients. (a) Correlation analyses between hemolytic biomarkers and circulating miR-199a-5p; (b) correlation analyses between hemolytic biomarkers and circulating miR-144; (c) correlation analyses between hemolytic biomarkers and circulating miR-126. miRNAs, microRNAs; SLU+, patients with active sickle leg ulcer or previous history. * p < 0.05. p-value obtained by Pearson correlation.
4. Circulating miRNAs Expression in Active SLU
Patients whose SLU wound bed was constituted by unviable tissues, such as necrotic and sloughy tissues, presented higher miR-144 expression (Figure 3a) and lower miR-126 expression (Figure 3b) (p < 0.05). Patients with more than one SLU simultaneously presented higher miR-144 (Figure 3c) and miR-199a-5p (Figure 3d) expression (p < 0.05). In addition, SLU recurrence between 4 to 7 episodes presented higher miR-199a-5p expression (Figure 3e) and lower miR-126 expression (Figure 3f) (p < 0.05). SLU with sick edges were associated with higher miR-199a-5p (Figure 3g) and miR-144 (Figure 3h) expression (p < 0.05).
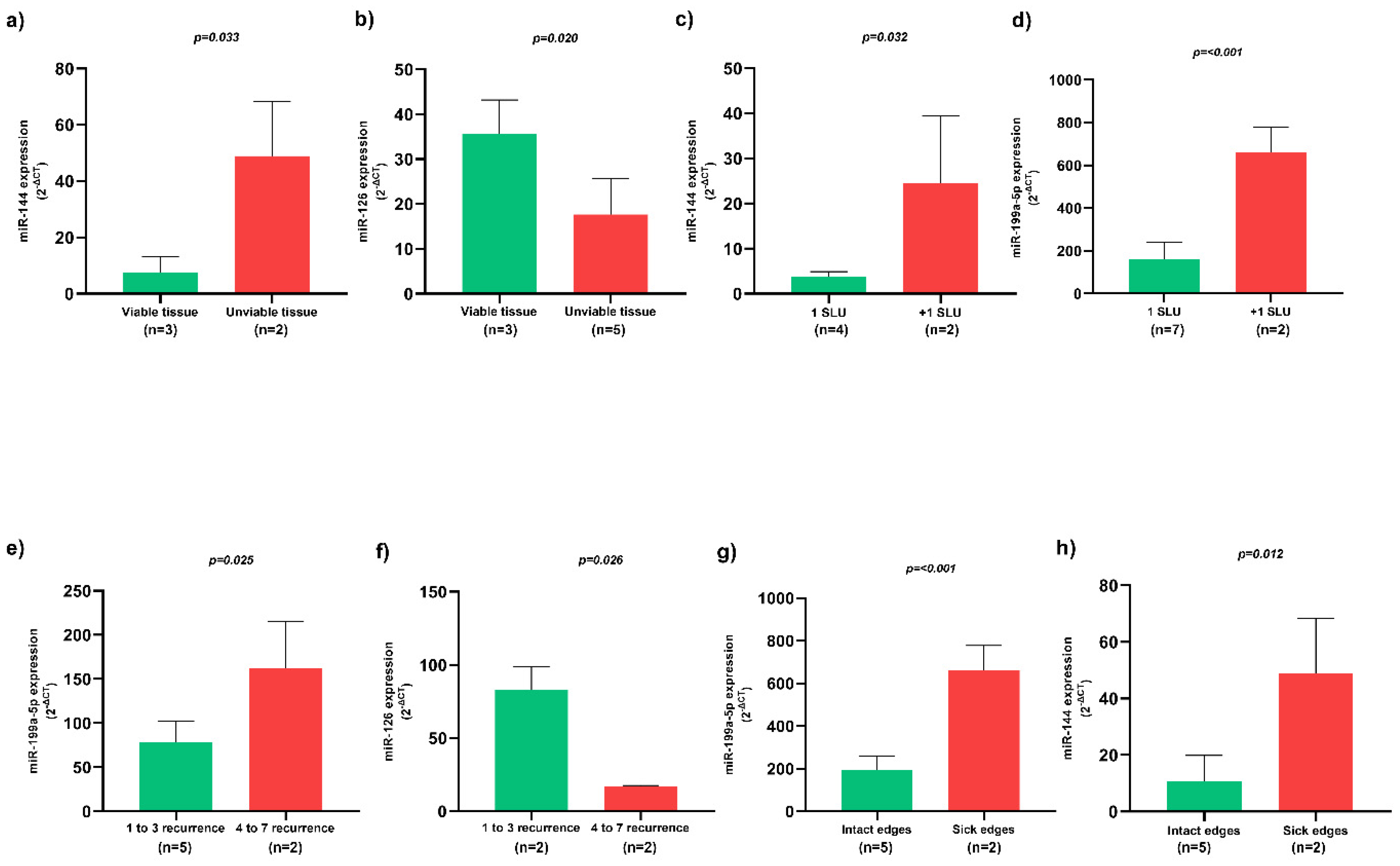
Figure 3. Association of circulating miRNAs and clinical characteristics of active SLU. (a) Expression of circulating miR-144 in SLU with viable and unviable tissues; (b) Expression of circulating miR-126 in SLU with viable and unviable tissues; (c) Expression of circulating miR-144 in patients with one and more than one SLU; (d) Expression of circulating miR-199a-5p in patients with one and more than one SLU; (e) Expression of circulating miR-199a-5p in patients with multiple SLU recurrence; (f) Expression of circulating miR-126 in patients with multiple SLU recurrence; (g) Expression of circulating miR-199a-5p in SLU with intact and sick edges; (h) Expression of circulating miR-144 in SLU with intact and sick edges. SLU, sickle leg ulcers; miR, miRNA. 1 SLU, one sickle leg ulcer; +1 SLU, more than one sickle leg ulcer; p-value obtained using independent t-test.
5. Association of HU with Circulating miRNAs Expression in Patients with and without SLU
Two MLR models were performed with HU as dependent variable. In SLU- patients, miR-199a-5p and miR-126 expression were independently associated with HU (R2 = 0.486; p < 0.05), while in SLU+ patients only miR-126 expression presented independent association (R2 = 0.779; p < 0.05) (Table 1).
Table 1. Multivariate linear regression models of hydroxyurea in association with confounding variables.
| Independent Variables | Dependent Variable | p-Value | β | R2 | p-Value of the Model | |
|---|---|---|---|---|---|---|
| SLU- patients N = 43 |
Circulating miR-199a-5p | Hydroxyurea | 0.007 | 0.695 | 0.486 | 0.022 |
| Circulating miR-144 | 0.096 | 0.468 | ||||
| Circulating miR-126 | 0.009 | −0.834 | ||||
| SLU+ patients N = 15 |
Circulating miR-199a-5p | 0.485 | 0.191 | 0.779 | 0.043 | |
| Circulating miR-144 | 0.997 | −0.001 | ||||
| Circulating miR-126 | 0.011 | −0.885 |
SLU-, patients without sickle leg ulcers; SLU+, patients with active sickle leg ulcers or previous history; miR, microRNA; R2: coefficient of determination; β: coefficient of regression; Bold p-values indicate significance at p < 0.05.
6. Target Gene Prediction with Biological Processes of miR-199a-5p to SCD Patients
The results demonstrate interaction networks of miR-199a-5p and target genes using miRWalk analysis (Figure 4a). Correlation between level significance by the score for these genes is presented (Figure 4b), as well as biological processes (Figure 4c).
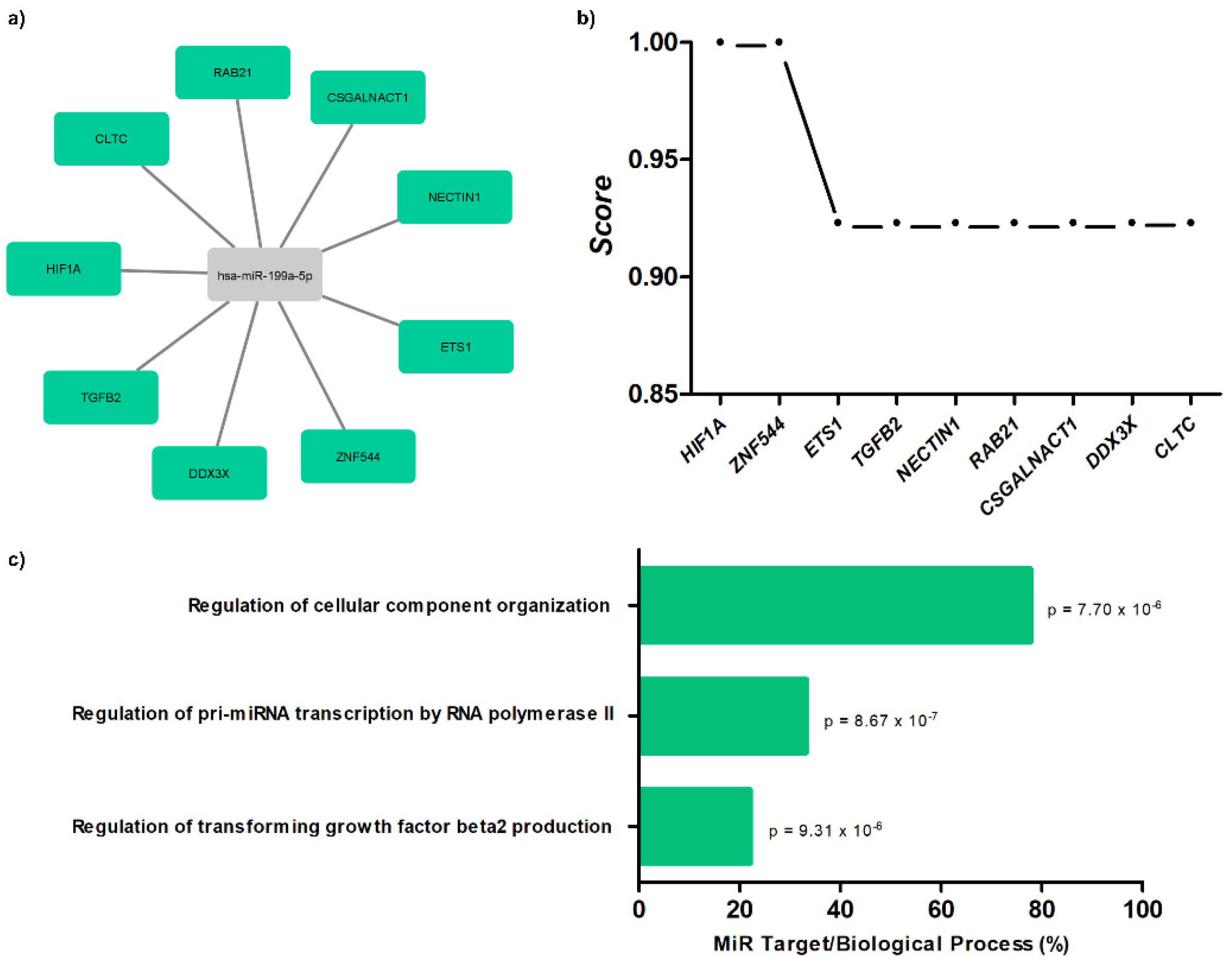
Figure 4. Target gene prediction with biological processes of miR-199a-5p. (a) Interaction networks of miR-199a-5p and target genes using miRWalk analysis; (b) correlation between level significance by score for genes; (c) biological processes for miR-199a-5p with p-values.
References
- Njoku, F.; Zhang, X.; Shah, B.N.; Machado, R.F.; Han, J.; Saraf, S.L.; Gordeuk, V.R. Biomarkers of clinical severity in treated and untreated sickle cell disease: A comparison by genotypes of a single center cohort and African Americans in the NHANES study. Br. J. Haematol. 2021, 194, 767–778.
- Antwi-Boasiako, C.; Andemariam, B.; Colombatti, R.; Asare, E.V.; Strunk, C.; Piccone, C.M.; Manwani, D.; Boruchov, D.; Farooq, F.; Urbonya, R.; et al. A study of the geographic distribution and associated risk factors of leg ulcers within an international cohort of sickle cell disease patients: The CASiRe group analysis. Ann. Hematol. 2020, 99, 2073–2079.
- Sickle Cell Disease: Ulcers: Prevention and Treatment, 1st ed.; Ministério da Saúde. Secretaria de Atenção à Saúde. Departamento de Atenção Especializada: Brasília-DF, Brasil, 2012; ISBN 978-85-334-1957-5.
- Senet, P.; Blas-Chatelain, C.; Levy, P.; Manea, E.M.; Peschanski, M.; Mirault, T.; Stankovic-Stojanovic, K.; Debure, C.; Debbache, K.; Girot, R.; et al. Factors predictive of leg-ulcer healing in sickle cell disease: A multicentre, prospective cohort study. Br. J. Dermatol. 2017, 177, 206–211.
- Umeh, N.I.; Ajegba, B.; Buscetta, A.J.; Abdallah, K.E.; Minniti, C.P.; Bonham, V.L. The psychosocial impact of leg ulcers in patients with sickle cell disease: I don’t want them to know my little secret. PLoS ONE 2017, 12, e0186270.
- Li, B.; Zhu, X.; Ward, C.M.; Starlard-Davenport, A.; Takezaki, M.; Berry, A.; Ward, A.; Wilder, C.; Neunert, C.; Kutlar, A.; et al. MIR-144-mediated NRF2 gene silencing inhibits fetal hemoglobin expression in sickle cell disease. Exp. Hematol. 2019, 70, 85–96.
- Yang, X.; Zheng, Y.; Tan, J.; Tian, R.; Shen, P.; Cai, W.; Liao, H. miR-199a-5p–HIF-1α-STAT3 positive feedback loop contributes to the progression of non-small cell lung cancer. Front. Cell Dev. Biol. 2021, 8, 1–13.
- Le, N.-T.; Abe, J. MicroRNA 199a and the eNOS (Endothelial NO Synthase)/NO Pathway. Arterioscler. Thromb. Vasc. Biol. 2018, 38, 2278–2280.
- Pan, Q.; Zheng, J.; Du, D.; Liao, X.; Ma, C.; Yang, Y.; Chen, Y.; Zhong, W.; Ma, X. microRNA-126 priming enhances functions of endothelial progenitor cells under physiological and hypoxic conditions and their therapeutic efficacy in cerebral ischemic damage. Stem Cells Int. 2018, 2018, 1–13.
More
Information
Subjects:
Hematology
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
601
Revisions:
3 times
(View History)
Update Date:
28 Jun 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





