
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Carla Giordano | -- | 1860 | 2022-06-22 11:16:57 | | | |
| 2 | Peter Tang | Meta information modification | 1860 | 2022-06-22 11:26:54 | | |
Video Upload Options
A very low-calorie ketogenic diet (VLCKD) is characterized by low daily caloric intake (less than 800 kcal/day), low carbohydrate intake (<50 g/day) and normoproteic (1–1.5 g of protein/kg of ideal body weight) contents. It induces a significant weight loss and an improvement in lipid parameters, blood pressure, glycaemic indices and insulin sensitivity in patients with obesity and type 2 diabetes mellitus. Cushing’s syndrome (CS) is characterized by an endogenous or exogenous excess of glucocorticoids and shows many comorbidities including cardiovascular disease, obesity, type 2 diabetes mellitus and lipid disorders.
1. Introduction
2. Cushing’s Syndrome
2.1. Nutrition and Cushing’s Syndrome
3. Ketogenic Diet
|
KD |
LCKD |
VLCKD |
|
|---|---|---|---|
|
Caloric intake |
Normocaloric |
800–1200 Kcal/day |
<800 Kcal/day |
|
Carbohydrate (%) |
5–10 |
13 |
13 |
|
Protein (%) |
15–20 |
29 |
44 |
|
Fat (%) |
70–80 |
58 |
43 |
|
Foods |
Vegetable oils, fish, eggs, meat, cheese, olives, avocado, coconut |
Natural high biological value proteins (1–2 servings) including meat, fish, eggs, processed meat |
Replacement meals with high biological value proteins composed by 18 g of proteins, 4 g of carbohydrates and 3 g of fats |
|
Recommendations for use [1] |
|
|
|
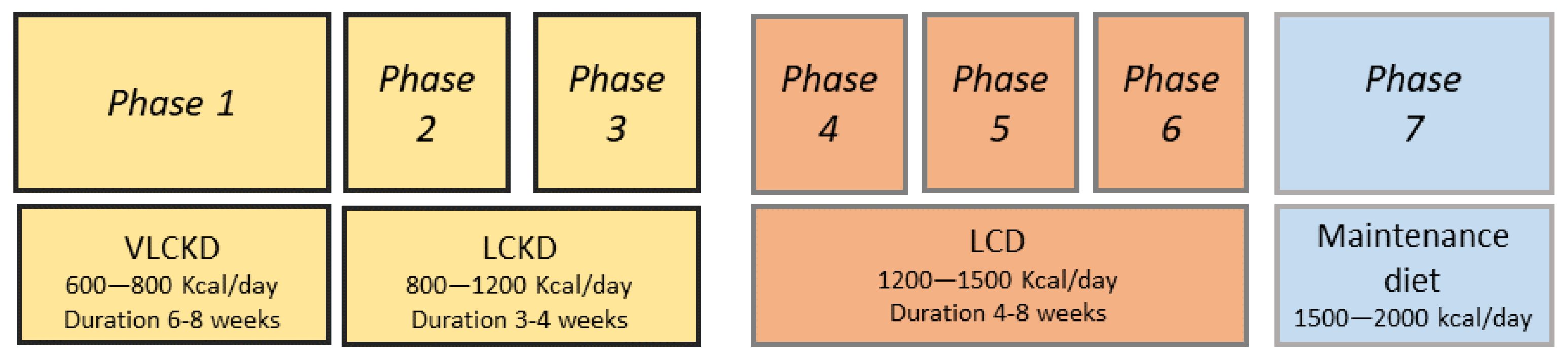
4. VLCKD
References
- Kirkpatrick, C.F.; Bolick, J.P.; Kris-Etherton, P.M.; Sikand, G.; Aspry, K.E.; Soffer, D.E.; Willard, K.E.; Maki, K.C. Review of current evidence and clinical recommendations on the effects of low-carbohydrate and very-low-carbohydrate (including ketogenic) diets for the management of body weight and other cardiometabolic risk factors: A scientific statement from the National Lipid Association Nutrition and Lifestyle Task Force. J. Clin. Lipidol. 2019, 13, 689–711.e1.
- Muscogiuri, G.; El Ghoch, M.; Colao, A.; Hassapidou, M.; Yumuk, V.; Busetto, L.; Obesity Management Task Force (OMTF) of the European Association for the Study of Obesity (EASO). European Guidelines for Obesity Management in Adults with a Very Low-Calorie Ketogenic Diet: A Systematic Review and Meta-Analysis. Obes. Fact. 2021, 14, 222–245.
- Muscogiuri, G.; Barrea, L.; Laudisio, D.; Pugliese, G.; Salzano, C.; Savastano, S.; Colao, A. The management of very low-calorie ketogenic diet in obesity outpatient clinic: A practical guide. J. Transl. Med. 2019, 17, 356.
- Kolb, H.; Kempf, K.; Röhling, M.; Lenzen-Schulte, M.; Schloot, N.C.; Martin, S. Ketone bodies: From enemy to friend and guardian angel. BMC Med. 2021, 19, 313.
- Boden, G.; Sargrad, K.; Homko, C.; Mozzoli, M.; Stein, T.P. Effect of a low-carbohydrate diet on appetite, blood glucose levels, and insulin resistance in obese patients with type 2 diabetes. Ann. Intern. Med. 2005, 142, 403–411.
- Dashti, H.M.; Mathew, T.C.; Khadada, M.; Al-Mousawi, M.; Talib, H.; Asfar, S.K.; Behbahani, A.I.; Al-Zaid, N.S. Beneficial effects of ketogenic diet in obese diabetic subjects. Mol. Cell. Biochem. 2007, 302, 249–256.
- Gupta, L.; Khandelwal, D.; Kalra, S.; Gupta, P.; Dutta, D.; Aggarwal, S. Ketogenic diet in endocrine disorders: Current perspectives. J. Postgrad. Med. 2017, 63, 242–251.
- Barañano, K.W.; Hartman, A.L. The ketogenic diet: Uses in epilepsy and other neurologic illnesses. Curr. Treat. Options Neurol. 2008, 10, 410–419.
- Thammongkol, S.; Vears, D.F.; Bicknell-Royle, J.; Nation, J.; Draffin, K.; Stewart, K.G.; Ingrid, E.; Scheffer, E.; Mackay, M.T. Efficacy of the ketogenic diet: Which epilepsies respond? Epilepsia 2012, 53, e55–e59.
- Rubini, A.; Bosco, G.; Lodi, A.; Cenci, L.; Parmagnani, A.; Grimaldi, K.; Zhongjin, Y.; Paoli, A. Effects of twenty days of the ketogenic diet on metabolic and respiratory parameters in healthy subjects. Lung 2015, 193, 939–945.
- Pivonello, R.; De Martino, M.C.; Iacuaniello, D.; Simeoli, C.; Muscogiuri, G.; Carlomagno, F.; De Leo, M.; Cozzolino, A.; Colao, A. Metabolic Alterations and Cardiovascular Outcomes of Cortisol Excess. Front. Horm. Res. 2016, 46, 54–56.
- Nieman, L.K. Hypertension and Cardiovascular Mortality in Patients with Cushing Syndrome. Endocrinol. Metab. Clin. N. Am. 2019, 48, 717–725.
- Barbot, M.; Zilio, M.; Scaroni, C. Cushing’s syndrome: Overview of clinical presentation, diagnostic tools and complications. Best Pract. Res. Clin. Endocrinol. Metab. 2020, 34, 101380.
- Giordano, C.; Guarnotta, V.; Pivonello, R.; Amato, M.C.; Simeoli, C.; Ciresi, A.; Cozzolino, A.; Colao, A. Is diabetes in Cushing’s syndrome only a consequence of hypercortisolism? Eur. J. Endocrinol. 2013, 170, 311–319.
- Ragnarsson, O. Cushing’s syndrome—Disease monitoring: Recurrence, surveillance with biomarkers or imaging studies. Best Pract. Res. Clin. Endocrinol. Metab. 2020, 34, 101382.
- Ferriere, A.; Tabarin, A. Cushing’s syndrome: Treatment and new therapeutic approaches. Best Pract. Res. Clin. Endocrinol. Metab. 2020, 34, 101381.
- Guarnotta, V.; Prinzi, A.; Pitrone, M.; Pizzolanti, G.; Giordano, C. Circulating Irisin Levels as a Marker of Osteosarcopenic-Obesity in Cushing’s Disease. Diabetes Metab. Syndr. Obes. 2020, 13, 1565–1574.
- Braun, L.T.; Vogel, F.; Reincke, M. Long-term morbidity and mortality in patients with Cushing’s syndrome. J. Neuroendocrinol. 2022, 4, e13113.
- Castinetti, F. Medical management of Cushing’s disease: When and how? J. Neuroendocrinol. 2022, 15, e13120.
- Pivonello, R.; Ferrigno, R.; De Martino, M.C.; Simeoli, C.; Di Paola, N.; Pivonello, C.; Barba, L.; Negri, M.; De Angelis, C.; Colao, A. Medical Treatment of Cushing’s Disease: An Overview of the Current and Recent Clinical Trials. Front. Endocrinol. 2020, 11, 648.
- Tritos, N.A. Adrenally Directed Medical Therapies for Cushing Syndrome. J. Clin. Endocrinol. Metab. 2021, 106, 16–25.
- Brown, D.R.; East, H.E.; Eilerman, B.S.; Gordon, M.B.; King, E.E.; Knecht, L.A.; Salke, B.; Samson, S.L.; Yuen, K.C.J.; Yau, H. Clinical management of patients with Cushing syndrome treated with mifepristone: Consensus recommendations. Clin. Diabetes Endocrinol. 2020, 6, 18.
- Stachowicz, M.; Lebiedzińska, A. The effect of diet components on the level of cortisol. Eur. Food Res. Technol. 2016, 242, 2001–2009.
- Lemmens, S.; Born, J.M.; Martens, E.A.; Martens, M.J.; Westerterp Plantenga, M.S. Influence of consumption of a high-protein vs. high-carbohydrate meal on the physiological cortisol and psychological mood response in men and women. PLoS ONE 2011, 6, e16826.
- Peeters, F.; Nicholson, N.A.; Berkhof, J. Cortisol responses to daily events in major depressive disorder. Psychosom. Med. 2003, 65, 836–841.
- Martens, M.J.; Rutters, F.; Lemmens, S.G.; Born, J.M.; Westerterp-Plantenga, M.S. Effects of single macronutrients on serum cortisol concentrations in normal weight men. Physiol. Behav. 2010, 101, 563–567.
- Bray, G.A.; Most, M.; Rood, J.; Redmann, S.; Smith, S.R. Hormonal responses to a fast-food meal compared with nutritionally comparable meals of different composition. Ann. Nutr. Metab. 2007, 51, 163–171.
- Galvao-Teles, A.; Graves, L.; Burke, C.W.; Fotherby, K.; Fraser, R. Free cortisol in obesity; effect of fasting. Acta Endocrinol. 1976, 81, 321–329.
- Edelstein, C.K.; Roy-Byrne, P.; Fawzy, F.I.; Dornfeld, L. Effects of weight loss on the dexamethasone suppression test. Am. J. Psychiatry 1983, 140, 338–341.
- Berger, M.P.K.; Doerr, P.; Krieg, C.; von Zersseen, D. Influence of weight loss on the dexamethasone suppression test. Arch. Gen. Psychiatry 1983, 19, 585–586.
- Tomiyama, A.J.; Mann, T.; Vinas, D.; Hunger, J.M.; Dejager, J.; Taylor, S.E. Low calorie dieting increases cortisol. Psychosom. Med. 2010, 72, 357–364.
- Dubuc, G.R.; Phinney, S.D.; Stern, J.S.; Havel, P.J. Changes of serum leptin and endocrine and metabolic parameters after 7 days of energy restriction in men and women. Metab. Clin. Exp. 1998, 47, 429–434.
- Pasiakos, S.M.; Caruso, C.M.; Kellogg, M.D.; Kramer, F.M.; Lieberman, H.R. Appetite and endocrine regulators of energy balance after 2 days of energy restriction: Insulin, leptin, ghrelin, and DHEA-S. Obesity 2011, 19, 1124–1130.
- van der Valk, E.S.; Savas, M.; van Rossum, E.F.C. Stress and obesity: Are there more susceptible individuals? Curr. Obes. Rep. 2018, 7, 193–203.
- Valenzano, A.; Polito, R.; Trimigno, V.; Di Palma, A.; Moscatelli, F.; Corso, G.; Sessa, F.; Salerno, M.; Montana, A.; Di Nunno, N.; et al. Effects of very low calorie ketogenic diet on the orexinergic system, visceral adipose tissue, and ROS production. Antioxidants 2019, 8, 643.
- Polito, R.; Messina, G.; Valenzano, A.; Scarinci, A.; Villano, I.; Monda, M.; Cibelli, G.; Porro, C.; Pisanelli, D.; Monda, V.; et al. The Role of Very Low Calorie Ketogenic Diet in Sympathetic Activation through Cortisol Secretion in Male Obese Population. J. Clin. Med. 2021, 10, 4230.
- Stimson, R.H.; Johnstone, A.M.; Homer, N.Z.; Wake, D.J.; Morton, N.M.; Andrew, R.; Lobley, G.E.; Walker, B.R. Dietary macronutrient content alters cortisol metabolism independently of body weight changes in obese men. J. Clin. Endocrinol. Metab. 2007, 92, 4480–4484.
- Stimson, R.H.; Mohd-Shukri, N.A.; Bolton, J.L.; Andrew, R.; Reynolds, R.M.; Walker, B.R. The postprandial rise in plasma cortisol in men is mediated by macronutrient-specific stimulation of adrenal and extra-adrenal cortisol production. J. Clin. Endocrinol. Metab. 2014, 99, 160–168.
- Nakamura, Y.; Walker, B.R.; Ikuta, T. Systematic review and meta-analysis reveals acutely elevated plasma cortisol following fasting but not less severe calorie restriction. Stress 2016, 19, 151–157.
- Capello, A.E.; Marcus, C.R. Effect of sub chronic tryptophan supplementation on stress-induced cortisol and appetite in subjects differing in 5-HTTLPR genotype and trait neuroticism. Psychoneuroendocrinology 2014, 45, 96–107.
- Starks, M.A.; Starks, S.L.; Kingsley, M.; Purpura, M.; Jager, R. The effects of phosphatidylserine on endocrine response to moderate intensity exercise. J. Int. Soc. Sports Nutr. 2008, 5, 11–17.
- Miklos, I.H.; Kovacs, K.J. GABAergic innervation of corticotropin-releasing hormone (CRH)-secreting parvocellular neurons and its plasticity as demonstrated by quantitative immunoelectron micorscopy. Neuroscience 2002, 113, 581–592.
- Song, Z.; Hatton, G.I. Taurine and the control of basal hor mone release from rat neurophysis. Exp. Neurol. 2003, 183, 330–337.
- Wilder, R.M. The effects of ketonemia on the course of epilepsy. Mayo Clin. Proc. 1921, 2, 307–308.
- Sukkar, S.G.; Muscaritoli, M. A Clinical Perspective of Low Carbohydrate Ketogenic Diets: A Narrative Review. Front. Nutr. 2021, 8, 642628.
- Abbasi, J. Interest in the ketogenic diet grows for weight loss and type 2 diabetes. JAMA 2018, 319, 215–217.
- Merra, G.; Gratteri, S.; De Lorenzo, A.; Barrucco, S.; Perrone, M.A.; Avolio, E.; Bernardini, S.; Marchetti, M.; Di Renzo, L. Effects of very-low-calorie diet on body composition, metabolic state, and genes expression: A randomized double-blind placebo-controlled trial. Eur. Rev. Med. Pharmacol. Sci. 2017, 21, 329–345.
- Paoli, A.; Rubini, A.; Volek, J.S.; Grimaldi, K.A. Beyond weight loss: A review of the therapeutic uses of very-low-carbohydrate (ketogenic) diets. Eur. J. Clin. Nutr. 2013, 67, 789–796.
- Cicero, A.F.; Benelli, M.; Brancaleoni, M.; Dainelli, G.; Merlini, D.; Negri, R. Middle and long-term impact of a very low-carbohydrate ketogenic diet on cardiometabolic factors: A multi-center, cross-sectional, clinical study. High Blood Press Cardiovasc. Prev. 2015, 22, 389–394.
- Merra, G.; Miranda, R.; Barrucco, S.; Gualtieri, P.; Mazza, M.; Moriconi, E.; Marchetti, M.; Chang, T.F.M.; De Lorenzo, A.; Di Renzo, L. Very-low-calorie ketogenic diet with aminoacid supplement versus very low restricted-calorie diet for preserving muscle mass during weight loss: A pilot double-blind study. Eur. Rev. Med. Pharmacol. Sci. 2016, 20, 2613–2621.
- Antonio, J.; Ellerbroek, A.; Silver, T.; Vargas, L.; Tamayo, A.; Buehn, R.; Peacock, C.A. A high protein diet has no harmful efects: A oneyear crossover study in resistance-trained males. J. Nutr. Metab. 2016, 2016, 9104792.
- Lafel, L. Ketone bodies: A review of physiology, pathophysiology and application of monitoring to diabetes. Diabetes Metab. Res. Rev. 1999, 15, 412–426.
- McPherson, P.A.; McEneny, J. The biochemistry of ketogenesis and its role in weight management, neurological disease and oxidative stress. J. Physiol. Biochem. 2012, 68, 141–151.
- Dehghan, M.; Mente, A.; Zhang, X.; Swaminathan, S.; Li, W.; Mohan, V.; Iqbal, R.; Kumar, R.; Wentzel-Viljoen, E.; Rosengren, A.; et al. Associations of fats and carbohydrate intake with cardiovascular disease and mortality in 18 countries from five continents (PURE): A prospective cohort study. Lancet 2017, 390, 2050–2062.
- Currenti, W.; Galvano, F. Very low-calorie ketogenic diet (VLCKD): Indicazioni ed efficacia nel trattamento dell’obesità. L’Endocrinologo 2020, 21, 458–463.





