Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Brian Cameron Webb | -- | 2001 | 2022-05-17 00:19:52 | | | |
| 2 | Brian Cameron Webb | Meta information modification | 2001 | 2022-05-17 00:23:38 | | | | |
| 3 | Rita Xu | Meta information modification | 2001 | 2022-05-17 03:29:32 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Webb, B.; Santerre, P.; , . Next-Generation Gingival Graft Substitutes. Encyclopedia. Available online: https://encyclopedia.pub/entry/22988 (accessed on 08 May 2026).
Webb B, Santerre P, . Next-Generation Gingival Graft Substitutes. Encyclopedia. Available at: https://encyclopedia.pub/entry/22988. Accessed May 08, 2026.
Webb, Brian, Paul Santerre, . "Next-Generation Gingival Graft Substitutes" Encyclopedia, https://encyclopedia.pub/entry/22988 (accessed May 08, 2026).
Webb, B., Santerre, P., & , . (2022, May 16). Next-Generation Gingival Graft Substitutes. In Encyclopedia. https://encyclopedia.pub/entry/22988
Webb, Brian, et al. "Next-Generation Gingival Graft Substitutes." Encyclopedia. Web. 16 May, 2022.
Copy Citation
There is a shortage of suitable tissue-engineered solutions for gingival recession, a soft tissue defect of the oral cavity. Autologous tissue grafts lead to an increase in morbidity due to complications at the donor site. Although material substitutes are available on the market, their development is early, and work to produce more functional material substitutes is underway. The latter materials along with newly conceived tissue-engineered substitutes must maintain volumetric form over time and have advantageous mechanical and biological characteristics facilitating the regeneration of functional gingival tissue.
electrospinning
gingival tissue
material substitutes
1. Introduction
Gingival recession with tooth root exposure affects half of the adult U.S. population [1][2]. A more efficient and less painful solution to the current treatment standard could have a widespread impact, improving the lives of millions. Loss of gingival coverage around the tooth at the tooth–tissue margin is referred to as gingival recession and results in the exposure of the tooth’s root surface. This root exposure can lead to tooth sensitivity when eating, increased risk of biofilm accumulation and further tissue loss and aesthetic compromise. Tissue loss is primarily caused by inflammation associated with periodontitis (initiated from agents produced within plaque/biofilm) and mechanical trauma [3]. Not only does gingival recession yield challenges for the patients’ esthetic appearance, but it can also expose the roots surface of the tooth to cariogenic supragingival microbiota leading to an increased risk of dental caries and in the extreme case loss of tooth [3].
The current treatment for the soft tissue defect of gingival recession is primarily autologous soft tissue grafts, usually harvested from the patient’s palate [4]. However, material substitutes can be used in isolation, or with autologous grafts, and are available on the market, such as the Geistlich Fibro-Gide® bovine-collagen-based material [5]. This material still has limitations when compared to the gold standard of care (autologous grafts) [6], while several other more innovative materials that are now being studied and are discussed here. However, the field of tissue material substitutes [7], and tissue-engineered solutions is still in its infancy in this application area. The pain and length of recovery and the time to carry out the procedures could be greatly reduced, when compared to the standard-of-care-associated procedures, if superior scaffold material substitutes and/or pre-vascularized tissue-engineered constructs could be translated into the clinical realm [8][9]. Vascularized tissue-engineered substitutes hold the potential to provide the cells needed for tissue regeneration and anastomosis, and deliver novel scaffolding materials to promote their proliferation and phenotype expression towards successful tissue regeneration outcomes [10].
One promising processing method for fabricating materials for regenerating and/or engineering gingival tissue is electrospinning. The method enables the production of fiber and fibril features that are on the scale of those of host extracellular matrices (ECM). Despite its mention in a recent systematic review looking at engineering vascularized oral tissue (mainly gingiva and alveolar bone), the entry provided no insight into the use of layered electrospun scaffolds, which is gaining interest by many tissue engineering groups attempting to replicate the ECM form and niche residence conditions for related cells to the tissue being grown [11]. It should be noted that while other examples of layered scaffolds for periodontal regeneration have been previously reported, none have addressed the potential use of electrospun elastomeric polymers [7].
2. Physiology and Disease of the Periodontium and Gingival Tissues: Defining Structure Requirements
The periodontium is comprised of four main tissue types: the alveolar bone, periodontal ligaments (connective tissue which allows for the attachment between the alveolar bone and root of the tooth), cementum, which is a mineralized tissue connecting the alveolar bone and the root of the tooth via periodontal ligaments, and gingival tissue which is the mucosal tissue that seals and protects the tooth from bacterial or physical threats as illustrated in Figure 1A [12]. The gingiva has two distinct layers, the epithelial tissue layer and the connective tissue layer (lamina propria) which make up approximately 30% and 70% of the gingiva, respectively [13][14]. The lamina propria can further be described as having two layers, the papillary layer, and the reticular layer [14]. The recession of gingival tissue is primarily caused by prolonged inflammation of periodontal tissue, periodontal treatment, and occlusal trauma [15]. Factors that could predispose an individual to gingival recession include a decrease in the thickness of the alveolar or buccal bone [15].
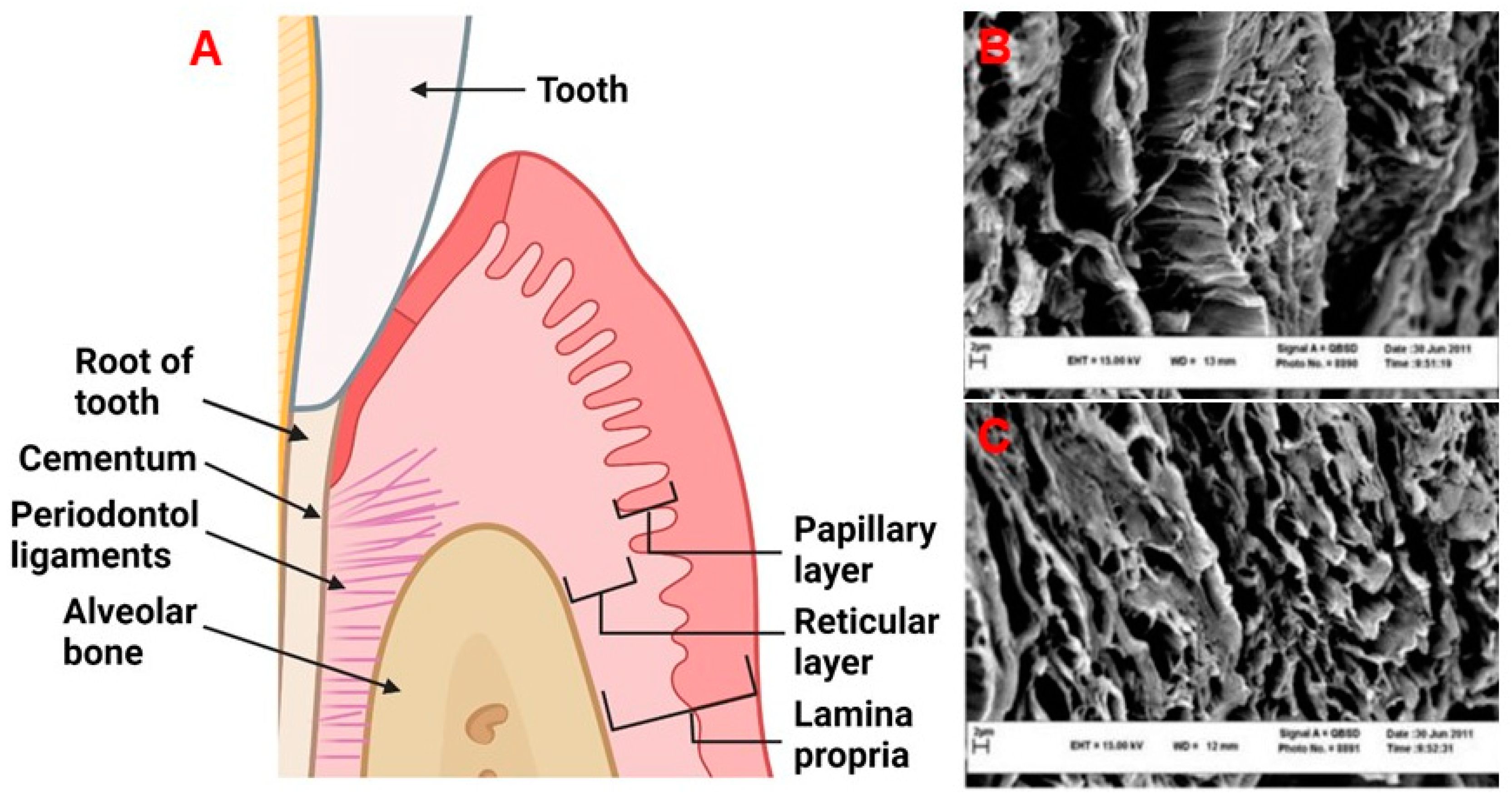
Figure 1. (A) The periodontal tissue anatomy. Created with BioRender.com. (B,C) Decellularized human gingival tissue adapted from previous literature reproduced under terms of the CC-BY license [16]. Copyright 2012, Nasser Mahdavishahri, Maryam Moghatam Matin, Masoud Fereidoni, Zahra Yarjanli, Seyed Ali Banihashem Rad, and Saeedeh Khajeh Ahmadi, published by Iranian Journal of Basic Medical Sciences. Created with BioRender.com, accessed on 8 April 2022.
Healthy gingiva is comprised mostly of collagens [17], elastin [18], laminin [13], and fibronectin [13][19]. Of the collagens found in the gingiva, collagen type I and type III make up 99% of this protein family in human gingival tissue [17]. The remaining 1% is accounted for by collagen type IV, with the presence of collagen type V only increasing during the initial stages of healing. The presence of collagen type V is thought to guide endothelial cells (ECs), facilitating angiogenesis [17]. The major function of the remaining collagen molecules is primarily to provide strength to the lamina propria [17]. The ultra-structure of these collagens and ECM can be found in Figure 1B,C. Within the lamina propria, human gingival fibroblasts (HGFs) are responsible for synthesizing and maintaining the ECM [20]. Gingival fibroblasts are present in the lamina propria at a concentration of 200 million cells/cm3 [14]. The collagen fibrils produced by HGFs are approximately 50–100 nm in diameter [21]. Both the papillary and reticular components contain a dense network of vasculature, consisting of terminal capillary loops in the papillary component and the gingival plexus which is made up of postcapillary venules [14]. The papillary layer contains approximately 50–60 loops/mm2 [14]. The natural gingiva has approximately 10 microvessel lumens/mm2 [22][23], with defined diameters depending on locations and depth as outlined in Table 1.
Table 1. An outline of the vessels found within gingival tissue adapted from previous literature [24].
| Tissue Area | Type of Vessel | Diameter (µm) | Average Depth (µm) |
|---|---|---|---|
| Free gingiva | Capillary loops | ≤30 | 50–200 |
| Connective vessels | 50–100 | 200–700 | |
| Large blood vessels | 200–400 | ≥500 | |
| Attached gingiva | Capillary loops | ≤15 | 50–200 |
| Connective vessels | - | - | |
| Large blood vessels | 200–500 | ≥600 | |
| Alveolar mucosa | Capillary loops | ≤15 | 50–200 |
| Connective vessels | 200–600 | 200–700 | |
| Large blood vessels | ≥600 | ≥700 |
Having the blueprint of the vasculature within gingival tissue is fundamental to its engineering from the standpoint of understanding what is required for the native tissue to function. There are however notable differences in vasculature structure from person to person [25]. How and to what variation blood flow is being supplied to the lamina propria is of relevance when considering how important the anastomosis of pre-vascularized constructs will be. By prohibiting blood flow from certain areas of the papilla, previous literature has demonstrated that some individuals have greater blood flow horizontally or vertically [25]. This is thought to be related to the abundance of blood vessels supplying the gingival area [25]. The differences may also be explained by changes in arteriole-to-arteriole connections (<100 µm in diameter) [24][25][26]. It is also well recognized that males have better blood flow recovery, and quicker anastomosis of coronally advanced flaps than females, which may suggest key factors that can be targeted to enhance the anastomosis of a graft [27]. Characterizing the differences in gingival vasculature is ongoing and will be critical to the application of pre-vascularized tissue engineering to the periodontium. Based on the physiology and anatomy of gingival tissue, it is evident that to prepare a construct resembling the native gingiva, modulating vasculature formation is going to be critical to graft integration and healing.
3. Current Material Options for Gingival Recession Treatment
The current treatment for gingival recession is typically autologous soft tissue grafts [4]. Additionally, material substitutes are available on the market, which have some reports on their efficacy. The two most common types of autologous grafts are connective tissue grafts (CTG) and free gingival grafts (FGG). CTGs involve harvesting connective tissue and grafting it such that root coverage and improved thickness of the gingival tissue are provided, as seen in Figure 2. An FGG entails harvesting connective tissue with surface epithelial tissue and placing it on the defect to cover the exposed root of the tooth and increase keratinized tissue [28]. Some of the major disadvantages of autologous gingival grafting are the increase in morbidity due to the harvest site, interindividual differences in terms of tissue availability, the time associated with the tissue harvesting (FGG takes ~25 min longer than using material substitutes) [29], donor infection, and bleeding from the harvest site [5][30].
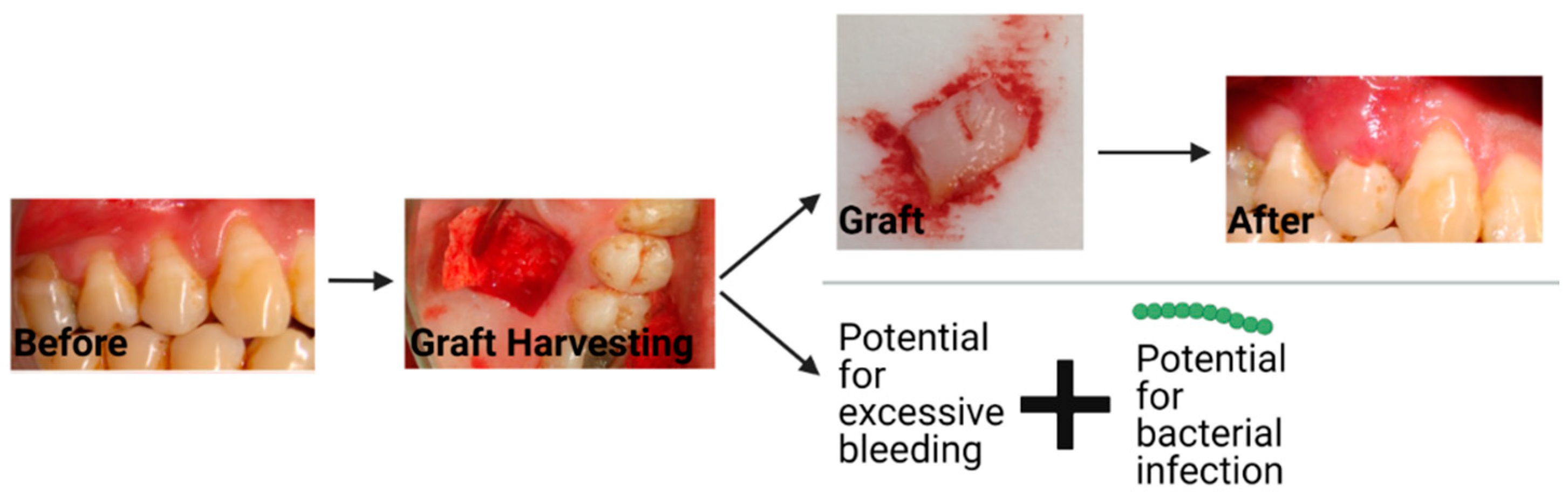
Figure 2. The workflow and potential complications that can occur with a connective tissue graft. The string of green dots represents bacteria. Created with BioRender.com with images from Dr. Michael Glogauer (University of Toronto) and images reproduced with permission under terms of the CC-BY license [31]. Copyright 2014, Sakshee Trivedi, Neeta Bhavsar, Kirti Dulani, and Rahul Trivedi published by the Journal of Clinical and Experimental Dentistry. Created with BioRender.com, accessed on 8 April 2022.
Based on two systematic reviews, the general consensus is that FGGs provide additional efficacy in terms of generating keratinized tissue when compared to material substitutes [6][32]. One of the most popular metrics of efficacy is the width of keratinized tissue [32]. Although the differences between autografts and material substitutes might not be large enough to negate the use of material substitutes given their benefits, autologous grafts remain the “gold standard” due to their ability to provide a greater area of keratinized tissue [6][32]. Taken all together autologous grafts remain the engineering benchmark, in large part due to their superior efficacy quantified by keratinized tissue.
During the differentiation of epithelial cells to keratinocytes, the composition of the underlying connective tissue dictates the subsequent occupancy of the epithelium, due to the keratinocyte interactions with components of the ECM, such as collagen type I [33][34]. Keratinocytes are also involved in the complex processes of healing the underlying connective tissue [35]. Further, changes in epithelial cell integrin binding are associated with changes in the activity of matrix metalloproteinases (MMPs), which participate in tissue remodeling and the migration of keratinocytes [33][36]. The interactions between the underlying connective tissue and the epithelial layer are mediated by integrins such as beta1-integrins [37]. The interactions that epithelial cells, such as keratinocytes, have with such integrins dictate cellular differentiation and survival [37][38]. Additionally, the secretion of paracrine factors such as hepatocyte growth factor from the underlying connective tissue contributes to the formation of keratinocytes [39].
Due to the available supply and lower morbidity associated with using material substitutes, the field is pushing towards their greater adoption. However, their adoption is in part hampered by the lack of efficacy to establish a keratinized structured layer, as discussed above, when compared to the traditional autologous graft. In considering keratinized tissue dependence in relation to their composition and interactions with the underlying connective tissue, it will be important that de novo material substitutes try to facilitate the formation of healthy gingival tissue more rapidly, as in this instance more keratinized tissue can be produced, thus improving the clinical efficacy of the graft.
Currently, the most common types of material substitutes being reported on and used in the clinic appear to be xenogenic scaffolds such as the Geistlich Fibro-Gide® [5], or allogenic grafts such as Alloderm® [40][41]. Their benefits mainly reflect their unlimited supply relative to autografts and lower associated morbidity relative to other material subsitutes [42]. Geistlich Fibro-Gide® by volume is 96% porous and is comprised of 60–96% (w/w) porcine collagen (type I and III) and 4–40% (w/w) elastin [43][44]. A similar product is Mucograft®, which has two layers, one of which is compact, and one which is spongy architecture for supporting cell ingrowth [43]. The compact outer layer can be left exposed to the oral environment and can aid in gaining keratinized tissue, suggesting that having a multilayered structure wherein different layers provide different functions is being used in the clinic [43]. Mucoderm® is a similar product to Mucograft® however is only comprised of a single layer [43]. Alloderm® or decellularized human dermal tissue are used clinically, however, they have been shown to have inferior clinical outcomes when compared to FGGs [45][46][47]. These material substitutes are currently the leading commercial substitutes that are widely available but are clearly still in need of improvement [5].
References
- Kassab, M.M.; Cohen, R.E. The etiology and prevalence of gingival recession. J. Am. Dent. Assoc. 2003, 134, 220–225.
- Eke, P.I.; Dye, B.A.; Wei, L.; Thronton-Evans, G.O.; Genco, R.J. Prevalence of periodontitis in adults in the United States: 2009 and 2010. J. Dent. Res. 2012, 91, 914–920.
- Merijohn, G.K. Management and prevention of gingival recession. Periodontol. 2000 2016, 71, 228–242.
- Moharamzadeh, K.; Colley, H.; Murdoch, C.; Hearnden, V.; Chai, W.L.; Brook, I.M.; Thornhill, M.H.; MacNeil, S. Tissue-engineered oral mucosa. J. Dent. Res. 2012, 91, 642–650.
- Mancini, L.; Fratini, A.; Tarallo, F.; Americo, L.M.; Marchetti, E. 3D analysis at implant sites after soft tissue augmentation with two types of collagen matrices: A pilot study. Plast. Aesthetic Res. 2021, 8, 26.
- Kim, D.M.; Neiva, R. Periodontal Soft Tissue Non–Root Coverage Procedures: A Systematic Review From the AAP Regeneration Workshop. J. Periodontol. 2015, 86, S56–S72.
- Goudouri, O.M.; Kontonasaki, E.; Boccaccini, A.R. Layered Scaffolds for Periodontal Regeneration; Elsevier Ltd.: Amsterdam, The Netherlands, 2017; ISBN 9780081009673.
- Benatti, B.B.; Silvério, K.G.; Casati, M.Z.; Sallum, E.A.; Nociti, F.H., Jr. Physiological features of periodontal regeneration and approaches for periodontal tissue engineering utilizing periodontal ligament cells. J. Biosci. Bioeng. 2007, 103, 1–6.
- Herford, A.S.; Akin, L.; Cicciu, M.; Maiorana, C.; Boyne, P.J. Use of a Porcine Collagen Matrix as an Alternative to Autogenous Tissue for Grafting Oral Soft Tissue Defects. J. Oral Maxillofac. Surg. 2010, 68, 1463–1470.
- Zhang, J.; Wang, L.; Zhu, M.; Wang, L.; Xiao, N.; Kong, D. Wet-spun poly (ε-caprolactone) microfiber scaffolds for oriented growth and infiltration of smooth muscle cells. Mater. Lett. 2014, 132, 59–62.
- Smirani, R.; Rémy, M.; Devillard, R.; Naveau, A. Engineered Prevascularization for Oral Tissue Grafting: A Systematic Review. Tissue Eng. Part B Rev. 2020, 26, 383–398.
- Bottino, M.C.; Thomas, V.; Schmidt, G.; Vohra, Y.K.; Chu, T.M.G.; Kowolik, M.J.; Janowski, G.M. Recent advances in the development of GTR/GBR membranes for periodontal regeneration—A materials perspective. Dent. Mater. 2012, 28, 703–721.
- Moharamzadeh, K.; Brook, I.M.; Van Noort, R.; Scutt, A.M.; Thornhill, M.H. Tissue-engineered oral mucosa: A review of the scientific literature. J. Dent. Res. 2007, 86, 115–124.
- Schroeder, H.E.; Listgarten, M.A. The gingival tissues: The architecture of periodontal protection. Periodontol. 2000 1997, 13, 91–120.
- Jati, A.S.; Furquim, L.Z.; Consolaro, A. Gingival recession: Its causes and types, and the importance of orthodontic treatment. Dental Press J. Orthod. 2016, 21, 18–29.
- Mahdavishahri, N.; Matin, M.M.; Fereidoni, M.; Yarjanli, Z.; Rad, S.A.B.; Ahmadi, S.K. In vitro assay of human gingival scaffold in differentiation of rat’s bone marrow mesenchymal stem cells to keratinocystes. Iran. J. Basic Med. Sci. 2012, 15, 1185–1190.
- Kaur, P.; Kakar, V. Collagen: Role in oral tissues: A review. Int. J. Sci. Res. 2014, 3, 273–276.
- Vindin, H.; Mithieux, S.M.; Weiss, A.S. Elastin architecture. Matrix Biol. 2019, 84, 4–16.
- Schoen, F.J.; Mitchell, R. Tissues, the Extracellular Matrix, and Cell Biomaterial Interactions, 3rd ed.; Ratner, B.D., Hoffman, A.S., Schoen, F.J., Lemons, J.E., Eds.; Academic Press: Oxford, UK, 2013.
- Hughes, C.C.W. Endothelial-stromal interactions in angiogenesis. Curr. Opin. Hematol. 2008, 15, 204–209.
- Pitaru, S.; Melcher, A.H. Orientation of gingival fibroblasts and newly-synthesized collagen fibers in vitro: Resemblance to transseptal and dento-gingival fibers. J. Periodontal Res. 1983, 18, 483–500.
- Bullon, P.; Fioroni, M.; Goteri, G.; Rubini, C.; Battino, M. Immunohistochemical analysis of soft tissues in implants with healthy and peri-implantitis condition, and aggressive periodontitis. Clin. Oral Implant. Res. 2004, 15, 553–559.
- Yoshida, S.; Noguchi, K.; Imura, K.; Miwa, Y.; Sunohara, M.; Sato, I. A morphological study of the blood vessels associated with periodontal probing depth in human gingival tissue. Okajimas Folia Anat. Jpn. 2011, 88, 103–109.
- Le, N.M.; Song, S.; Zhou, H.; Xu, J.; Li, Y.; Sung, C.E.; Sadr, A.; Chung, K.H.; Subhash, H.M.; Kilpatrick, L.; et al. A noninvasive imaging and measurement using optical coherence tomography angiography for the assessment of gingiva: An in vivo study. J. Biophotonics 2018, 11, e201800242.
- Mikecs, B.; Vág, J.; Gerber, G.; Molnár, B.; Feigl, G.; Shahbazi, A. Revisiting the vascularity of the keratinized gingiva in the maxillary esthetic zone. BMC Oral Health 2021, 21, 160.
- DiPietro, L.A. Angiogenesis and wound repair: When enough is enough. J. Leukoc. Biol. 2016, 100, 979–984.
- Molnár, E.; Molnár, B.; Lohinai, Z.; Tóth, Z.; Benyó, Z.; Hricisák, L.; Windisch, P.; Vág, J. Evaluation of Laser Speckle Contrast Imaging for the Assessment of Oral Mucosal Blood Flow following Periodontal Plastic Surgery: An Exploratory Study. Biomed Res. Int. 2017, 2017, 4042902.
- Patel, M.; Nixon, P.J.; Chan, M.F.W. Gingival recession: Part 3. Surgical management using free grafts and guided tissue regeneration. Nat. Publ. Gr. 2011, 211, 353–358.
- Schmitt, C.M.; Tudor, C.; Kiener, K.; Wehrhan, F.; Schmitt, J.; Eitner, S.; Agaimy, A.; Schlegel, K.A. Vestibuloplasty: Porcine Collagen Matrix Versus Free Gingival Graft: A Clinical and Histologic Study. J. Periodontol. 2013, 84, 914–923.
- Bassetti, R.G.; Stähli, A.; Bassetti, M.A.; Sculean, A. Soft tissue augmentation around osseointegrated and uncovered dental implants: A systematic review. Clin. Oral Investig. 2017, 21, 53–70.
- Trivedi, S.R.; Bhavsar, N.V.; Dulani, K.; Trivedi, R. Clinical evaluation of subepithelial connective tissue graft and guided tissue regeneration for treatment of Miller’s class 1 gingival recession (comparative, split mouth, six months study). J. Clin. Exp. Dent. 2014, 6, 218–224.
- Dragan, I.F.; Hotlzman, L.P.; Karimbux, N.Y.; Morin, R.A.; Bassir, S.H. Clinical Outcomes of Comparing Soft Tissue Alternatives to Free Gingival Graft: A Systematic Review and Meta-Analysis. J. Evid. Based Dent. Pract. 2017, 17, 370–380.
- Smith, P.C.; Martínez, C.; Martínez, J.; Mcculloch, C.A. Role of fibroblast populations in periodontal wound healing and tissue remodeling. Front. Physiol. 2019, 10, 270.
- Sudbeck, B.D.; Pilcher, B.K.; Welgus, H.G.; Parks, W.C. Induction and repression of collagenase-1 by keratinocytes is controlled by distinct components of different extracellular matrix compartments. J. Biol. Chem. 1997, 272, 22103–22110.
- Zhang, C.; Lim, J.; Liu, J.; Ponugoti, B.; Alsadun, S.; Tian, C.; Vafa, R.; Graves, D.T. FOXO1 expression in keratinocytes promotes connective tissue healing. Sci. Rep. 2017, 7, 42834.
- Dugina, V.; Fontao, L.; Chaponnier, C.; Vasiliev, J.; Gabbiani, G. Focal adhesion features during myofibroblastic differentiation are controlled by intracellular and extracellular factors. J. Cell Sci. 2001, 114, 3285–3296.
- Levy, L.; Broad, S.; Diekmann, D.; Evans, R.D.; Watt, F.M. β1 Integrins regulate keratinocyte adhesion and differentiation by distinct mechanisms. Mol. Biol. Cell 2000, 11, 453–466.
- Groeger, S.E.; Meyle, J. Epithelial barrier and oral bacterial infection. Periodontol. 2000 2015, 69, 46–67.
- Narayan Biswal, B.; Narayan Das, S.; Kumar Das, B.; Rath, R. Alteration of cellular metabolism in cancer cells and its therapeutic. J. Oral Maxillofac. Pathol. 2017, 21, 244–251.
- Yoshizawa, M.; Koyama, T.; Kojima, T.; Kato, H.; Ono, Y.; Saito, C. Keratinocytes of tissue-engineered human oral mucosa promote re-epithelialization after intraoral grafting in athymic mice. J. Oral Maxillofac. Surg. 2012, 70, 1199–1214.
- Izumi, K.; Feinberg, S.E.; Terashi, H.; Marcelo, C.L. Evaluation of transplanted tissue-engineered oral mucosa equivalents in severe combined immunodeficient mice. Tissue Eng. 2003, 9, 163–174.
- McGuire, M.K.; Scheyer, E.T. Randomized, Controlled Clinical Trial to Evaluate a Xenogeneic Collagen Matrix as an Alternative to Free Gingival Grafting for Oral Soft Tissue Augmentation. J. Periodontol. 2014, 85, 1333–1341.
- Vallecillo, C.; Toledano-Osorio, M.; Vallecillo-Rivas, M.; Toledano, M.; Osorio, R. In Vitro biodegradation pattern of collagen matrices for soft tissue augmentation. Polymers 2021, 13, 2633.
- Caballé-Serrano, J.; Zhang, S.; Ferrantino, L.; Simion, M.; Chappuis, V.; Bosshardt, D.D. Tissue response to a porous collagen matrix used for soft tissue augmentation. Materials 2019, 12, 3721.
- Tavelli, L.; McGuire, M.K.; Zucchelli, G.; Rasperini, G.; Feinberg, S.E.; Wang, H.L.; Giannobile, W.V. Extracellular matrix-based scaffolding technologies for periodontal and peri-implant soft tissue regeneration. J. Periodontol. 2020, 91, 17–25.
- Wei, P.-C.; Laurell, L.; Geivelis, M.; Lingen, M.W.; Maddalozzo, D.; Increased, A.; Gingiva, A. Acellular Dermal Matrix Allografts to Achieve Increased Attached Gingiva. Part 1. A Clinical Study. J. Periodontol. 2000, 71, 1297–1305.
- Romeu, D.; De Resende, B.; Luiz, S.; Greghi, A.; Siqueira, A.F.; Augusto, C.; Benfatti, M.; Damante, C.A.; Schutzer, M.; Zangrando, R.; et al. Acellular dermal matrix allograft versus free gingival graft: A histological evaluation and split-mouth randomized clinical trial. Clin. Oral Investig. 2019, 23, 539–550.
More
Information
Subjects:
Materials Science, Biomaterials
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.6K
Revisions:
3 times
(View History)
Update Date:
17 May 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





