
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Peerawat - Wongsurakul | -- | 2683 | 2022-05-10 09:08:26 | | | |
| 2 | Camila Xu | -1 word(s) | 2682 | 2022-05-10 09:42:51 | | | | |
| 3 | Camila Xu | Meta information modification | 2682 | 2022-05-12 07:50:51 | | |
Video Upload Options
Ethanol can be produced from sugary, starchy, and lignocellulosic feedstocks. Each feedstock requires different procedures for its conversion to fermentable sugar. Lignocellulosic biomass requires extra pretreatment compared to sugar and starch feedstocks to disrupt the structure and improve enzymatic hydrolysis efficiency. However, the greatest concern regarding the pretreatment process is inhibitor formation, which might retard enzymatic hydrolysis and fermentation. In addition to the inhibitors from pretreatment, chemicals used during the pretreatment and fermentation of byproducts may remain in the final product if they are not removed by ethanol distillation and dehydration. Maintaining the quality of ethanol during storage is another concerning issue. Initial impurities of ethanol being stored and its nature, including hygroscopic, high oxygen and carbon dioxide solubility, influence chemical reactions during the storage period and change ethanol’s characteristics (e.g., water content, ethanol content, acidity, pH, and electrical conductivity).
1. Introduction
| Specification | Unit | European Union |
USA | Brazil | Thailand | |||
|---|---|---|---|---|---|---|---|---|
| prEN 15376 | ASTM D-4806-16a |
ANP Resolution nº 19 | TIS 2324 | TIS 640-1 | TIS 640-2 | |||
| Ethanol type | - | - | Anhydrous | Denatured anhydrous |
Anhydrous | Denatured anhydrous |
Anhydrous | Anhydrous |
| Ethanol | % by volume | Min. | - | - | 98 | - | - | - |
| Ethanol and higher saturated alcohols | % by volume, (% by mass) |
Min. | (98.7) | 92.1 | (99.3) | 99 | 99.5 | 99.5 |
| Higher saturated mono-alcohols-C3-C5 | % by volume, (% by mass) |
Max. | (2) | - | 3 | 2 | - | - |
| Methanol | % by volume, (% by mass) |
Max. | (1) | 0.5 | 0.5 | 0.5 | 0.02 | 0.05 |
| Water content | % by volume, (% by mass) |
Max. | (0.3) | 1 | (0.7) | 0.3 | - | - |
| Density at 20 °C | kg/m3 | Max. | - | - | 791.5 | - | 790–793 | - |
| Total acidity (as acetic acid) | mg/L, (% by mass) | Max. | (0.007) | 56 (0.007) | 30 | 30 | 30 | (0.005) |
| Electrical conductivity | µS/m | Max. | - | - | 300 | 500 | - | - |
| pHe | - | - | 6.5~9.0 | - | 6.5~9.0 | - | - | |
| Copper | mg/kg, (mg/L) | Max. | 0.1 | 0.1 | 0.07 | 0.07 | - | - |
| Inorganic chloride | mg/kg, (mg/L) | Max. | 1.5 | 6.7 (5) | 1 | (20) | - | - |
| Solvent-washed gum | mg/100 mL | Max. | - | 5 | - | 5 | - | - |
| Sulfur | mg/kg, (ppm) | Max. | 10 | (30) | Report | - | - | - |
| Total sulfate | mg/kg | Max. | 3 | 4 | 4 | - | - | - |
| Phosphorus content | mg/L | Max. | 0.15 | - | - | - | - | - |
| Non-volatile material | mg/100 mL, (% by mass) |
Max. | 10 | - | 5 | - | 2.5 | (0.005) |
| Denaturant content | vol. % | Max. | - | 1.96~2.5 | - | - | - | - |
| Iron | mg/kg | Max. | - | - | 5 | - | - | - |
| Benzene | mL/kL | Max. | - | - | - | - | 2 | - |
| Acetaldehyde and acetal (as acetaldehyde) |
% by volume, (% by mass) |
Max. | - | - | - | - | 0.001 | (0.10) |
| Any other volatile impurity (as 4-methylpentan-2-ol) | mL/kL | Max. | - | - | - | - | 300 | - |
| Absorbance - Lower than 240 nm - 250 to 260 nm - 270 to 340 nm |
Max. | - | - | - | - | 0.4 0.3 0.1 |
- | |
| Sodium | % by mass | Max. | - | - | 0.0002 | - | - | - |
| Permanganate time | Minute | Min. | - | - | - | - | - | 15 |
| Aspect | - | Clear and colorless | Clear and colorless | Clear and no impurities | Clear, colorless and no visible suspended solids | Clear and colorless | Corresponding to ISO 2211 | |
2. Ethanol Production from Different Types of Feedstock
3. Impact of Different Feedstocks on Impurities in Fuel Ethanol
3.1. Conversion of Sugar-Containing Feedstock
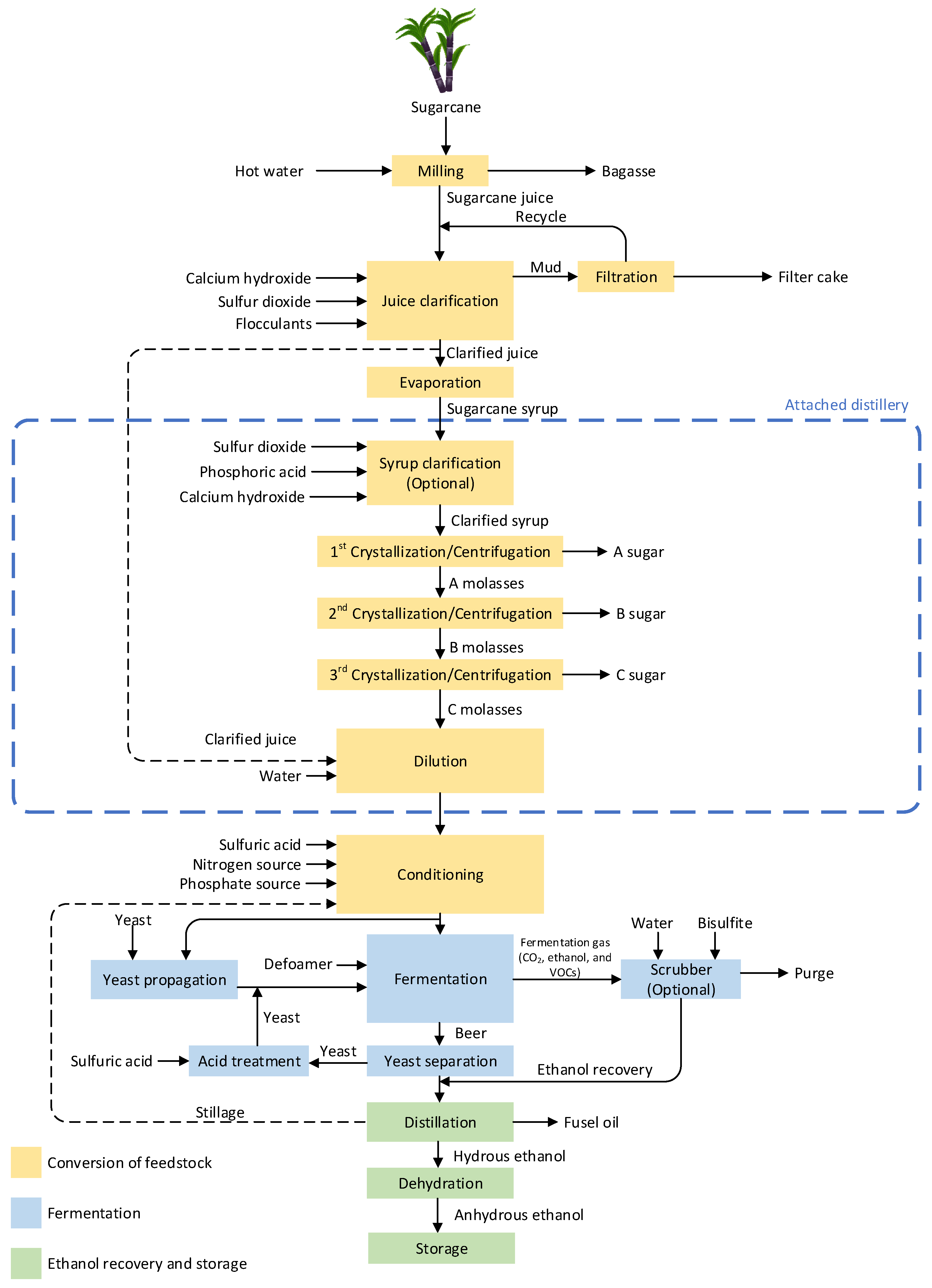 Figure 1. Type of sugarcane-derived ethanol production plant.
Figure 1. Type of sugarcane-derived ethanol production plant.3.2. Conversion of Starch-Containing Feedstocks
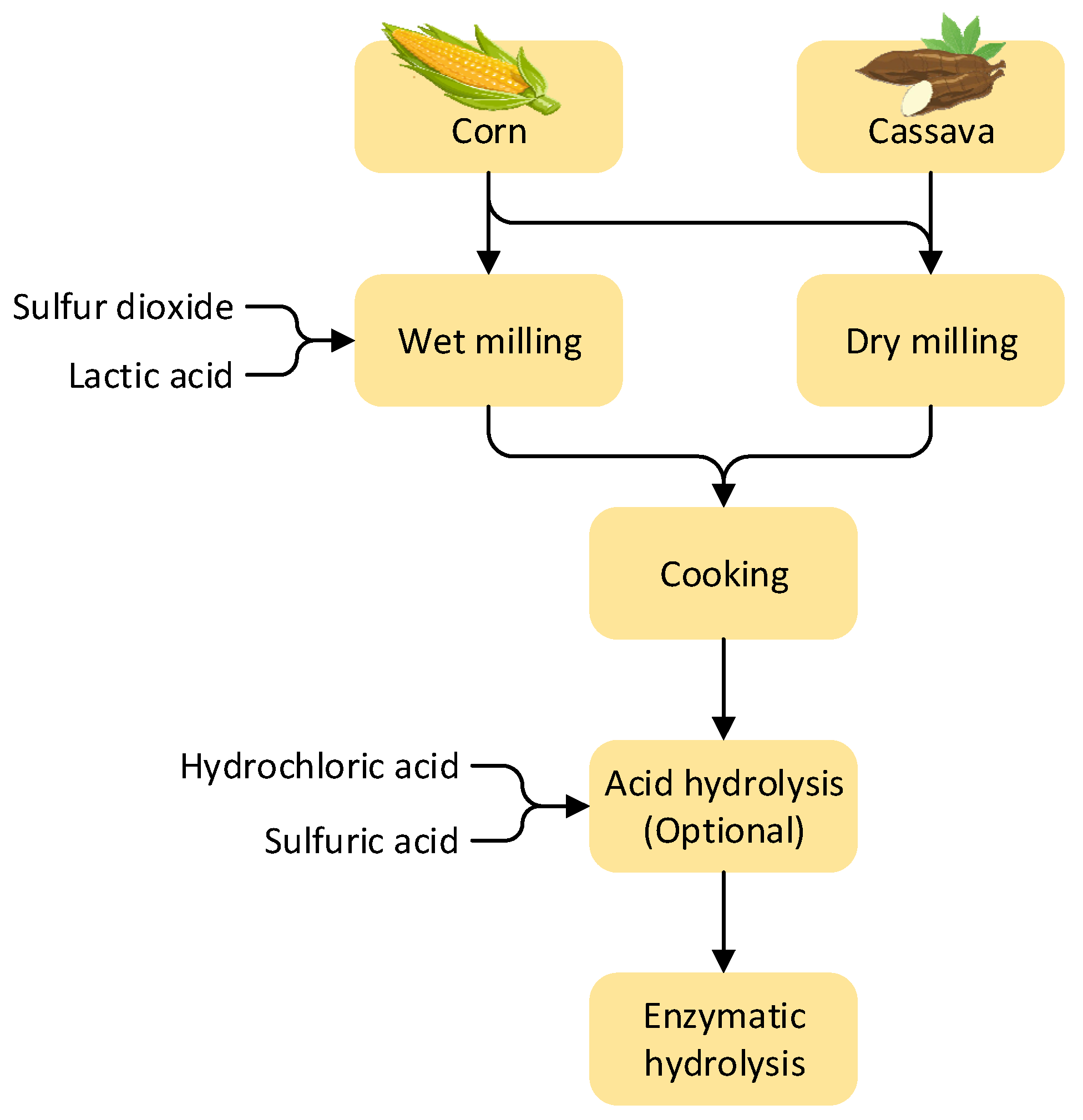 Figure 2. Conversion of starch-containing feedstock.
Figure 2. Conversion of starch-containing feedstock.4. Ethanol Recovery
4.1. Distillation Process
4.2. Stillage Recycles
4.3. The Fate of Electrolytes during Distillation
4.4. Dehydration Process
References
- Gallo, J.M.; Bueno, J.; Schuchardt, U. Catalytic transformations of ethanol for biorefineries. J. Braz. Chem. Soc. 2014, 25, 2229–2243.
- Galante-Fox, J.; Von Bacho, P.; Notaro, C.; Zizelman, J. E-85 fuel corrosivity: Effects on port fuel injector durability performance. SAE Trans. 2007, 116, 989–994.
- Sriroth, K.; Wanlapatit, S.; Piyachomkwan, K. Cassava bioethanol. In Bioethanol; IntechOpen: London, UK, 2012.
- SEWPAC. Regulation Impact Statement Fuel Quality Standard: Ethanol (E85) Automotive Fuel; Department of Sustainability, Environment, Water, Population and Communities: Canberra, Australia, 2012. Available online: https://parlinfo.aph.gov.au/parlInfo/download/publications/tabledpapers/HSTP012629_2010-13/upload_pdf/12629_2010-13.pdf;fileType=application%2Fpdf#search=%22publications/tabledpapers/HSTP012629_2010-13%22 (accessed on 7 January 2021).
- Cortez, L.A.B. Sugarcane Bioethanol: R&D for Productivity and Sustainability; Blucher: São Paulo, Brazil, 2010.
- TTF. White Paper on Internationally Compatible Biofuel Standards. Tripartite Task Force. 2007. Available online: https://www.nist.gov/document/biofuelsreportpdf (accessed on 15 December 2020).
- McCormick, R.L.; Alleman, T.; Yanowitz, J. Sulfate Salts in Gasoline and Ethanol Fuels—Historical Perspective and Analysis of Available Data; National Renewable Energy Lab. (NREL): Golden, CO, USA, 2017.
- Stepien, Z.; Krasodomski, W. Investigation into Engine Deposit-Forming Tendency due to Sulfate Salt Contamination of Gasoline–Ethanol Blends. Energy Fuels 2019, 33, 4244–4253.
- ASTM D5798-10a; Standard Specification for Fuel Ethanol (Ed70-Ed85) for Automotive Spark-Ignition Engines. ASTM International: West Conshohocken, PA, USA, 2011.
- Habe, H.; Shinbo, T.; Yamoto, T.; Sato, S.; Shimada, H.; Sakaki, K. Chemical analysis of impurities in diverse bioethanol samples. J. Jpn. Pet. Inst. 2013, 56, 414–422.
- Christensen, E.; Fioroni, G.M.; Kim, S.; Fouts, L.; Gjersing, E.; Paton, R.S.; McCormick, R.L. Experimental and theoretical study of oxidative stability of alkylated furans used as gasoline blend components. Fuel 2018, 212, 576–585.
- Haaz, E.; Fozer, D.; Toth, A.J. Development of Anhydrous Ethanol Purification: Reduction of Acetal Content and Vapor–Liquid Equilibrium Study of the Ethanol–Acetal Binary System. ACS Omega 2021, 6, 1289–1298.
- Naegeli, D.W.; Lacey, P.I.; Alger, M.J.; Endicott, D.L. Surface corrosion in ethanol fuel pumps. SAE Trans. 1997, 106, 564–571.
- Hoekman, S.K.; Broch, A. Investigation into Filter Plugging Due to Sulfate Salt Contamination of Ethanol, Gasoline, and Gasoline-Ethanol Blends; Coordinating Research Council, Inc.: Alpharetta, GA, USA, 2018.
- Styarini, D.; Aristiawan, Y.; Aulia, F.; Abimanyu, H.; Sudiyani, Y. Determination of organic impurities in lignocellulosic bioethanol product by GC-FID. Energy Procedia 2013, 32, 153–159.
- Sánchez, R.; Sánchez, C.; Lienemann, C.-P.; Todolí, J.-L. Metal and metalloid determination in biodiesel and bioethanol. J. Anal. At. Spectrom. 2015, 30, 64–101.
- Sánchez, C.; Santos, S.; Sánchez, R.; Lienemann, C.-P.; Todolí, J.-L. Profiling of Organic Compounds in Bioethanol Samples of Different Nature and the Related Fractions. ACS Omega 2020, 5, 20912–20921.
- RFA. Fuel Ethanol: Industry Guidelines, Specifications, and Procedures; Renewable Fuels Association: Washington, DC, USA, 2018.
- API 1626: 2010; Storing and Handling Ethanol and Gasoline-Ethanol Blends at Distribution Terminals and Service Stations. American Petroleum Institute: Washington, DC, USA, 1985.
- Costenoble, O. Worldwide Fuels Standards. In Overview of Specifications and Regulations on (Bio) Fuels; NEN–Netherlands Standardization Institute: Delft, The Netherlands, 2017.
- TIS 640-1; Ethanol for Pharmaceutical Use. Thai Industrial Standards Institute (TISI): Bangkok, Thailand, 2010.
- TIS 640-2; Ethanol for Industrial Use. Thai Industrial Standards Institute (TISI): Bangkok, Thailand, 2010.
- TIS 2324; Denatured Ethanol for Gasohol Production. Thai Industrial Standards Institute (TISI): Bangkok, Thailand, 2006.
- Vohra, M.; Manwar, J.; Manmode, R.; Padgilwar, S.; Patil, S. Bioethanol production: Feedstock and current technologies. J. Environ. Chem. Eng. 2014, 2, 573–584.
- Nguyen, T.L.T.; Gheewala, S.H. Life cycle assessment of fuel ethanol from cane molasses in Thailand. Int. J. Life Cycle Assess. 2008, 13, 301.
- Cardona, C.A.; Sanchez, O.J.; Gutierrez, L.F. Process Synthesis for Fuel Ethanol Production; CRC Press: Boca Raton, FL, USA, 2009.
- Modesto, M.; Nebra, S.A.; Zemp, R.J. Improving the Ethanol Production From Sugar Cane Biomass. In Proceedings of the ASME 8th Biennial Conference on Engineering Systems Design and Analysis, Torino, Italy, 4–7 July 2006; pp. 203–210.
- Dias, M.O.; Junqueira, T.L.; Cavalett, O.; Cunha, M.P.; Jesus, C.D.; Rossell, C.E.; Maciel Filho, R.; Bonomi, A. Integrated versus stand-alone second generation ethanol production from sugarcane bagasse and trash. Bioresour. Technol. 2012, 103, 152–161.
- Onuki, S.; Koziel, J.A.; Jenks, W.S.; Cai, L.; Grewell, D.; van Leeuwen, J. Taking ethanol quality beyond fuel grade: A review. J. Inst. Brew. 2016, 122, 588–598.
- de Castro, R.E.N.; de Brito Alves, R.M.; do Nascimento, C.A.O.; Giudici, R. Assessment of Sugarcane-Based Ethanol Production. In Fuel Ethanol Production from Sugarcane; IntechOpen: London, UK, 2018.
- de Souza Dias, M.O.; Maciel Filho, R.; Mantelatto, P.E.; Cavalett, O.; Rossell, C.E.V.; Bonomi, A.; Leal, M.R.L.V. Sugarcane processing for ethanol and sugar in Brazil. Environ. Dev. 2015, 15, 35–51.
- Onuki, S.; Koziel, J.A.; van Leeuwen, J.H.; Jenks, W.S.; Grewell, D.; Cai, L. Ethanol production, purification, and analysis techniques: A review. In Proceedings of the 2008 Providence, Providence, RI, USA, 29 June–2 July 2008; p. 1.
- Friedl, A. Bioethanol from Sugar and Starch. In Energy from Organic Materials (Biomass): A Volume in the Encyclopedia of Sustainability Science and Technology, 2nd ed.; Kaltschmitt, M., Ed.; Springer: New York, NY, USA, 2019; pp. 905–924.
- Stenberg, K.; Tengborg, C.; Galbe, M.; Zacchi, G.; Palmqvist, E.; Hahn-Hägerdal, B. Recycling of process streams in ethanol production from softwoods based on enzymatic hydrolysis. Appl. Biochem. Biotechnol. 1998, 70, 697.
- Stout, B.A. Handbook of Energy for World Agriculture; Elsevier: Amsterdam, The Netherlands, 2012.
- Nguyen, T.L.T.; Gheewala, S.H. Fuel ethanol from cane molasses in Thailand: Environmental and cost performance. Energy Policy 2008, 36, 1589–1599.
- Mangmeechai, A.; Pavasant, P. Water footprints of Cassava-and Molasses-based ethanol production in Thailand. Nat. Resour. Res. 2013, 22, 273–282.
- Schill, S.R. Sulfur Compounded in Ethanol Regulations. Ethanol Prod. Mag. 2016. Available online: http://www.ethanolproducer.com/articles/13513/sulfur-compounded-in-ethanol-regulations (accessed on 12 October 2020).
- Zhang, Q.; Du, J.; Jin, Y.; Zhao, Z.; Li, Y. SO2 reduction in distilled grape spirits by three methods. J. Inst. Brew. 2013, 119, 314–320.
- Edwards, T.J.; Newman, J.; Prausnitz, J.M. Thermodynamics of aqueous solutions containing volatile weak electrolytes. AIChE J. 1975, 21, 248–259.
- Huang, H.-J.; Ramaswamy, S.; Tschirner, U.W.; Ramarao, B. Separation and Purification processes for lignocellulose-to-bioalcohol production. In Bioalcohol Production; Elsevier: Amsterdam, The Netherlands, 2010; pp. 246–277.
- Ramos, W.B.; Figueiredo, M.F.; Brito, R.P. Optimization of extractive distillation process with a single column for anhydrous ethanol production. In Computer Aided Chemical Engineering; Elsevier: Amsterdam, The Netherlands, 2014; Volume 33, pp. 1411–1416.
- Ortuño-Boter, D.; Plesu, V.; Ruiz, A.E.B.; Ruiz, J.B.; Iancu, P.; Llorens, J. Enhanced Distillation Based on Feed Impurities. In Computer Aided Chemical Engineering; Elsevier: Amsterdam, The Netherlands, 2016; Volume 38, pp. 1923–1928.
- Morigami, Y.; Kondo, M.; Abe, J.; Kita, H.; Okamoto, K. The first large-scale pervaporation plant using tubular-type module with zeolite NaA membrane. Sep. Purif. Technol. 2001, 25, 251–260.





