Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Matteo Mario Carlà | -- | 2006 | 2022-04-01 19:41:36 | | | |
| 2 | Catherine Yang | Meta information modification | 2006 | 2022-04-02 03:21:27 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Carlà, M.M.; Boselli, F.; Giannuzzi, F.; Gambini, G.; Caporossi, T.; , .; Kilian, R.; Rizzo, S. IOCT-Assisted Endothelial Keratoplasty. Encyclopedia. Available online: https://encyclopedia.pub/entry/21297 (accessed on 24 May 2026).
Carlà MM, Boselli F, Giannuzzi F, Gambini G, Caporossi T, , et al. IOCT-Assisted Endothelial Keratoplasty. Encyclopedia. Available at: https://encyclopedia.pub/entry/21297. Accessed May 24, 2026.
Carlà, Matteo Mario, Francesco Boselli, Federico Giannuzzi, Gloria Gambini, Tomaso Caporossi, , Raphael Kilian, Stanislao Rizzo. "IOCT-Assisted Endothelial Keratoplasty" Encyclopedia, https://encyclopedia.pub/entry/21297 (accessed May 24, 2026).
Carlà, M.M., Boselli, F., Giannuzzi, F., Gambini, G., Caporossi, T., , ., Kilian, R., & Rizzo, S. (2022, April 01). IOCT-Assisted Endothelial Keratoplasty. In Encyclopedia. https://encyclopedia.pub/entry/21297
Carlà, Matteo Mario, et al. "IOCT-Assisted Endothelial Keratoplasty." Encyclopedia. Web. 01 April, 2022.
Copy Citation
iOCT serves as a guidance tool for key surgical processes in endothelial keratoplasty, from scoring the Descemet membrane to guaranteeing graft apposition at the conclusion of the procedure. For this reason, both Descemet stripping anterior endothelial keratoplasty (DSAEK) and Descemet membrane endothelial keratoplasty (DMEK) are made easier by intraoperative OCT.
anterior segment iOCT
intraoperative optical coherence tomography
Dsaek
Dmek
Endothelial keratoplasty
1. iOCT-Aided DSAEK
The first results of handheld intraoperative AS-OCT during DSAEK surgery were reported in 2010, when Knecht et al. highlighted the role of this technique to detect that there was still separation between the donor and host cornea at the conclusion of the procedure in two patients out of six, with an average width of interface fluid of 40 µm, despite following several published guidelines for good graft adhesion during surgery [1]. Other case studies have employed iOCT to demonstrate interface fluid outflow after corneal stab wounds [2][3], with Ide et al. focusing on the study of intraoperative fluid dynamics [4].
The PIONEER study was fundamental to highlight the utility of a microscope-mounted iOCT in different fields of ophthalmic surgery [5]. In DSAEK, iOCT allowed for clear visibility of fluid at the graft/host interface, in order to grant the best graft apposition, with the operation to be prolonged until optimum fluid evacuation was attained. On the first postoperative day, 3% (5/143) of the grafts dislocated. In all cases, a surgeon feedback form was completed and resulted in a 64% rate of uncertainty of graft apposition from the surgeon. Thanks to iOCT, persistent fluid was found in 48% of those cases, necessitating repeated procedures. Overall, in 65/135 (48%) eyes, iOCT determined surgical strategy changes, due to the evidence of persisting interface fluid necessitating further interventions, which was reported also in 29% of cases in which the surgeon believed the graft was completely apposed [5]. Hallahan et al. exploited the same PIONEER study cohort to analyze intraoperative interface fluid dynamics and correlate fluid parameters with graft non-adherence during the first week. iOCT measurements showed that total fluid volume was significantly higher when comparing completely or partially non-adherent grafts (0.22 and 0.17 µm2) with grafts that had no post-operative interface fluid (0.05 µm2). Similarly, the maximum area of fluid in the final iOCT also differed with the same trends (0.07 and 0.06 µm2 in eyes with subsequent displaced grafts and partial nonadherence grafts vs. 0.03 µm2 in eyes with totally adhered grafts) [6]. Despite the fact that there were no significant variations in results between the full and partial dislocation groups, the complete dislocation group tended to have higher interface fluid levels. Within the first week after surgery, many iOCT parameters were linked to whole and partial graft nonadherence, such as increased final interface fluid volume and area, maximum isolated interface fluid pocket volume, and mean and maximum interface fluid thickness, with volumetric and area assessments of final interface fluid more strongly associated with adherence graft rates rather than linear parameters. At last, there were no interface fluid characteristics linked to graft rebubbling and regrafting [6].
Successive studies were reported with the use of Mi-OCT, which offers better image stabilization compared to microscope-mounted or handheld iOCT in previous studies. Steverink et al. reported a case series of 8 eyes undergoing Mi-OCT-assisted DSAEK, which allowed the surgeon to identify persisting fluid interface in six cases, successfully managed with corneal swiping with a subsequent decrease in maximal interface width [7].
The most important research regarding microscope-integrated iOCT led to the publication of the DISCOVER study. In this research, Mi-OCT was reported to provide valuable feedback in 88.5% of cases, with interface fluid extent evaluation the most-cited area of iOCT feedback. The surgeon, by means of standardized questionnaires, declared the graft to be clinically apposed in 68.3% of patients in the DSAEK subgroup. In 46 cases (54.8%), however, residual interface fluid was seen on iOCT, thus permitting rescue procedures [8]. Further analyses were conducted with the same cohort of the DISCOVER study, with Ehlers et al. reporting that Mi-OCT aided decision-making during DSAEK surgery in 41% of cases, in which further maneuvers were necessary based on iOCT. When the surgeon thought the graft was entirely apposed clinically, iOCT revealed residual fluid in 19% of instances. Conversely, iOCT indicated full apposition in 47% of instances where the surgeon did not believe the graft was completely apposed, saving time and needless adjustments during surgery [9] (Figure 1).
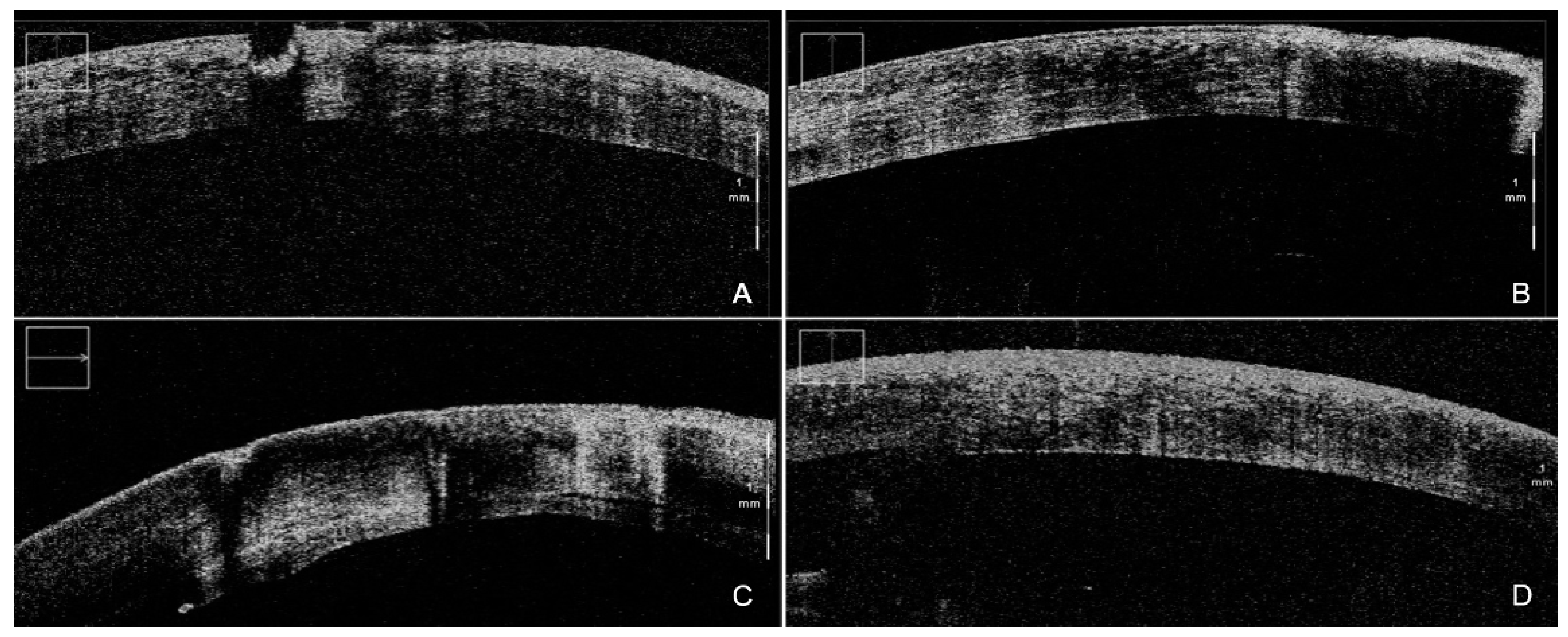
Figure 1. iOCT-assisted DSAEK performed by an expert surgeon. During DM stripping, iOCT aided debris visualization (A) in order to obtain the smoothest possible dissection plane (B). After the implantation of the graft, iOCT was able to identify even the smallest persisting interface fluid (C), thus allowing for complete apposition of the graft, clearly visible at the end of the surgery (D). iOCT = intra-operative optical coherence tomography; DSAEK = Descemet stripping automated endothelial keratoplasty; DM = Descemet membrane.
A recent retrospective study by Asif et al. focused on Mi-OCT-assisted DSAEK in congenital hereditary endothelial dystrophy (CHED) [10]. Corneal transplantation in infants may be difficult owing to a variety of ocular characteristics, including a tiny eyeball, a very low anterior chamber, enhanced positive posterior pressure, poor scleral stiffness, phakic status, and limited intraocular space. These variables may increase the likelihood of problems [11]. Moreover, CHED patients often have poor visibility due to corneal edema and stronger adherence of the DM to the underlying stroma, in contrast to decompensated corneas of Fuchs’ endothelial dystrophy [12]. This might cause DM residues to remain in the graft, obstructing graft apposition. iOCT proved useful in these conditions, thereby improving the outcomes of the procedure. Among the 39 eyes undergoing DSAEK, the graft was attached intraoperatively in all cases, while graft detachment was seen in the early postoperative phase in 4 eyes, requiring a rebubbling procedure [10].
2. iOCT-Aided DMEK
For the treatment of corneal endothelial disorders, Descemet membrane endothelial keratoplasty (DMEK) outperforms Descemet stripping automated endothelial keratoplasty (DSAEK). DMEK, in particular, has been demonstrated to speed up visual rehabilitation, improve visual acuity, and reduce corneal rejection [13][14][15][16]. Despite its benefits, DMEK necessitates the acquisition of new skills by the surgeon, which may result in a high learning curve [14][17]. DMEK scroll orientation may be seen and identified in real time using iOCT, avoiding the use of potentially hazardous exterior markers (such as the “S” stamp or peripheral notches), and may be useful in cases of corneal opacity or advanced corneal edema when vision is impaired [18][19][20].
Saad et al., in 2015, were the first to describe iOCT-assisted DMEK in a prospective case series of 14 eyes. Using the iOCT integrated into a surgical microscope (RESCAN 700; Carl Zeiss Meditec), which offered an 840-nm central wavelength that performed 27,000 A scans per second, graft orientation was correctly evaluated in all cases. The success rate was 100%, with only one patient needing rebubbling at 1 week. The mean unfolding time calculated from the moment the graft was inserted into the anterior chamber to the time the air bubble was injected into the anterior chamber, including the time spent looking at and evaluating the OCT pictures, was reported to be 6.1 min, faster when compared to the 8.9 min needed by the same surgeon without this technology [19]. Even in difficult situations with severe corneal edema, when direct visibility of the graft was hindered, OCT pictures aided graft orientation assessment, suggesting that live OCT aid may also assist in broadening DMEK indications in severe corneal edemas. Moreover, in the instance where the graft was in the incorrect orientation, OCT pictures enabled the surgeon to confirm graft rotation [19].
The aforementioned DISCOVER study evaluated the role of iOCT even in DMEK procedures. In the report on the first eight eyes, iOCT-assisted DMEK allowed completion of intraoperative graft attachments in seven eyes, while one graft exhibited a linear region of non-adherence matching a large posterior stromal irregularity, clearly visible on iOCT during descemetorhexis. The median “unscrolling time” was 6 min and 15 s, and the graft orientation was rapidly identifiable in 100% of cases [20].
These results were confirmed at 3-year follow-up in the DISCOVER study, in which surgeons reported iOCT-enabled graft orientation feedback in 63% of instances in DMEK, based on the scrolling arrangement of the tissue. Moreover, iOCT was commonly used to establish orientation even before viewing of the orienting “S” stamp, thus allowing surgeons to abandon the use of this stamp [8]. In addition, following first implantation, the surgeons reported clinical graft apposition in 54 patients (90%). Intraoperative imaging revealed persisting interface fluid in 7% of cases, allowing for further corneal sweeping before the surgery was completed. In this research, the trend of preferred iOCT visualization was analyzed: surgeons used the external screen in 30% of cases in the first year, the heads-up display in the oculars in 25% of cases, and both kinds of vision in 42% of instances. In years 2–3, a substantial change toward using the screen occurred in 82% of instances, compared to oculars in 10% and both systems in 8% [8].
In the last update of the DISCOVER study, those findings were confirmed. Patel et al. noted that iOCT offered helpful real-time feedback in all instances (100%) and did not interfere with the surgical operation in any manner [21]. In all instances, the iOCT picture on the linked external video display was preferred, because of the smaller, lower-definition picture on the inside display. iOCT also confirmed its effectiveness by allowing for quick and safe detection of tissue orientation in 99% of cases, thus avoiding other reported marking techniques [22][23]. Moreover, average unscrolling time of 4.4 ± 4.1 min was competitive when compared to reported times for S-stamped grafts (5.7 ± 3.9 min) and for non-stamped grafts (6.4 ± 4.4 min) [20][24]. The rebubbling rate reported in this study was 6.4%, suggesting that iOCT-assisted DMEK may yield equivalent or better early outcomes than previous reported outcomes, with a mean rebubbling rate of 28.8% reported in a recent thorough literature analysis across 17 DMEK case series without iOCT support [25].
iOCT favorability was also claimed by Muijzer et al., who reported recent experience with 22 iOCT-aided DMEKs with a reduced over-pressurization time (<2 min, compared to the >12 min of the standard procedure) [26]. Thanks to the capacity of iOCT in reducing unscrolling times, as reported by previous studies, they decided to avoid over-pressurizing the globe for an extended duration during surgery, since a high intra-ocular pressure (IOP) may harm corneal endothelial cells and the retinal nerve fiber layer [27][28]. Refraining from over-pressurization enabled reducing the entire surgical time by 24% without endangering its safety or compromising clinical outcomes. Moreover, iOCT provided the surgeon with useful information, including an unclear graft orientation in 12 cases and interface fluid and minor graft detachments in 8 instances. Ultimately, iOCT altered the surgical decision-making process in 42% of cases, and its role in the management of graft misorientation or in the prevention of unnecessary manipulation was crucial [26].
A recent prospective study was conducted by Sharma et al., specifically targeting 25 cases of corneal decompensation with poor visualization due to pseudophakic bullous keratopathy, Fuchs endothelial corneal dystrophy, failed graft or iridocorneal endothelial (ICE) syndrome that underwent Mi-OCT-guided DMEK [29]. Prior to descemetorhexis, regions of lacking DM in the host cornea were identified thanks to Mi-OCT, preventing needless DM scraping, which might have resulted in postoperative stromal haze. After completing the descemetorhexis, retained DM tags, otherwise undetectable, were detected on Mi-OCT in 92% of instances (23 eyes) and immediately removed using micro-vitreoretinal forceps and a vitrectomy cutter in all of these patients. In the case of ICE syndrome, Mi-OCT guided evaluation and release of peripheral and mid-peripheral anterior synechiae. In this research, Mi-OCT was able to identify the position and form of the DM roll in the cartridge in every case, with the majority of the instances having a double scroll (76%), and minor percentages of tight scroll and trifold. Following graft implantation, the correct orientation was always detectable, even in cases of severe stromal haze, thus overcoming the shortfalls of the numerous techniques previously proposed in cases of poor visualization, such as chandelier or trans-corneal illumination [30][31][32]. iOCT proved useful to assess whether or not to try to unfold the peripheral DM folds based on their precise position and extent, avoiding inadvertent damage to the graft. This study reported graft attachment success rates of 72% at day 3, with 16% of eyes requiring rebubbling, leading to a 100% rate of attachment at the 6-month follow-up, thus underlining how Mi-OCT assistance allowed for the safe and effective conduct of the procedure, otherwise rendered unsuitable for DMEK due to the presence of severe corneal edema and poor visibility of the anterior segment details [29].
References
- Knecht, P.B.; Kaufmann, C.; Menke, M.N.; Watson, S.L.; Bosch, M.M. Use of intraoperative fourier-domain anterior segment optical coherence tomography during descemet stripping endothelial keratoplasty. Am. J. Ophthalmol. 2010, 150, 360–365.e2.
- Miyakoshi, A.; Ozaki, H.; Otsuka, M.; Hayashi, A. Efficacy of Intraoperative Anterior Segment Optical Coherence Tomography during Descemet’s Stripping Automated Endothelial Keratoplasty. ISRN Ophthalmol. 2014, 2014, 562062.
- Wylegala, E.; Nowinska, A.K.; Wroblewska-Czajka, E.; Janiszewska, D. Donor disc attachment assessment with intraoperative spectral optical coherence tomography during descemet stripping automated endothelial keratoplasty. Indian J. Ophthalmol. 2013, 61, 511–513.
- Ide, T.; Wang, J.H.; Tao, A.Z.; Leng, T.; Kymionis, G.D.; O’Brien, T.P.; Yoo, S.H. Intraoperative Use of Three-Dimensional Spectral-Domain Optical Coherence Tomography. Ophthal. Surg. Las. Imaging Retin. 2010, 41, 250–254.
- Ehlers, J.P.; Dupps, W.J.; Kaiser, P.K.; Goshe, J.; Singh, R.P.; Petkovsek, D.; Srivastava, S.K. The Prospective Intraoperative and Perioperative Ophthalmic ImagiNg with Optical CoherEncE TomogRaphy (PIONEER) Study: 2-year results. Am. J. Ophthalmol. 2014, 158, 999–1007.
- Hallahan, K.M.; Cost, B.; Goshe, J.M.; Dupps, W.J., Jr.; Srivastava, S.K.; Ehlers, J.P. Intraoperative Interface Fluid Dynamics and Clinical Outcomes for Intraoperative Optical Coherence Tomography-Assisted Descemet Stripping Automated Endothelial Keratoplasty from the PIONEER Study. Am. J. Ophthalmol. 2017, 173, 16–22.
- Steverink, J.G.; Wisse, R.P.L. Intraoperative optical coherence tomography in descemet stripping automated endothelial keratoplasty: Pilot experiences. Int. Ophthalmol. 2017, 37, 939–944.
- Ehlers, J.P.; Modi, Y.S.; Pecen, P.E.; Goshe, J.; Dupps, W.J.; Rachitskaya, A.; Sharma, S.; Yuan, A.; Singh, R.; Kaiser, P.K.; et al. The DISCOVER Study 3-Year Results: Feasibility and Usefulness of Microscope-Integrated Intraoperative OCT during Ophthalmic Surgery. Ophthalmology 2018, 125, 1014–1027.
- Ehlers, J.P.; Goshe, J.; Dupps, W.J.; Kaiser, P.K.; Singh, R.P.; Gans, R.; Eisengart, J.; Srivastava, S.K. Determination of feasibility and utility of microscope-integrated optical coherence tomography during ophthalmic surgery: The DISCOVER Study RESCAN Results. JAMA Ophthalmol. 2015, 133, 1124–1132.
- Asif, M.I.; Bafna, R.K.; Sharma, N.; Kaginalkar, A.; Sinha, R.; Agarwal, T.; Maharana, P.K.; Kaur, M.; Taank, P.; Titiyal, J.S. Microscope Integrated Optical Coherence Tomography Guided Descemet Stripping Automated Endothelial Keratoplasty in Congenital Hereditary Endothelial Dystrophy. Clin. Ophthalmol. 2021, 15, 3173–3181.
- Vanathi, M.; Panda, A.; Vengayil, S.; Chaudhuri, Z.; Dada, T. Pediatric keratoplasty. Surv. Ophthalmol. 2009, 54, 245–271.
- Pineda, R.; Jain, V.; Shome, D.; Hunter, D.C.; Natarajan, S. Descemet’s stripping endothelial keratoplasty: Is it an option for congenital hereditary endothelial dystrophy? Int. Ophthalmol. 2010, 30, 307–310.
- Melles, G.R.; Ong, T.S.; Ververs, B.; van der Wees, J. Descemet membrane endothelial keratoplasty (DMEK). Cornea 2006, 25, 987–990.
- Tourtas, T.; Laaser, K.; Bachmann, B.O.; Cursiefen, C.; Kruse, F.E. Descemet membrane endothelial keratoplasty versus descemet stripping automated endothelial keratoplasty. Am. J. Ophthalmol. 2012, 153, 1082–1090.e1082.
- Price, M.O.; Giebel, A.W.; Fairchild, K.M.; Price, F.W., Jr. Descemet’s membrane endothelial keratoplasty: Prospective multicenter study of visual and refractive outcomes and endothelial survival. Ophthalmology 2009, 116, 2361–2368.
- Anshu, A.; Price, M.O.; Price, F.W. Risk of Corneal Transplant Rejection Significantly Reduced with Descemet’s Membrane Endothelial Keratoplasty. Ophthalmology 2012, 119, 536–540.
- Terry, M.A. Endothelial keratoplasty: Why aren’t we all doing Descemet membrane endothelial keratoplasty? Cornea 2012, 31, 469–471.
- Steven, P.; Le Blanc, C.; Velten, K.; Lankenau, E.; Krug, M.; Oelckers, S.; Heindl, L.M.; Gehlsen, U.; Huttmann, G.; Cursiefen, C. Optimizing Descemet Membrane Endothelial Keratoplasty Using Intraoperative Optical Coherence Tomography. JAMA Ophthalmol. 2013, 131, 1135–1142.
- Saad, A.; Guilbert, E.; Grise-Dulac, A.; Sabatier, P.; Gatinel, D. Intraoperative OCT-Assisted DMEK: 14 Consecutive Cases. Cornea 2015, 34, 802–807.
- Cost, B.; Goshe, J.M.; Srivastava, S.; Ehlers, J.P. Intraoperative optical coherence tomography-assisted descemet membrane endothelial keratoplasty in the DISCOVER study. Am. J. Ophthalmol. 2015, 160, 430–437.
- Patel, A.S.; Goshe, J.M.; Srivastava, S.K.; Ehlers, J.P. Intraoperative Optical Coherence Tomography-Assisted Descemet Membrane Endothelial Keratoplasty in the DISCOVER Study: First 100 Cases. Am. J. Ophthalmol. 2020, 210, 167–173.
- Bachmann, B.O.; Laaser, K.; Cursiefen, C.; Kruse, F.E. A method to confirm correct orientation of descemet membrane during descemet membrane endothelial keratoplasty. Am. J. Ophthalmol. 2010, 149, 922–925.e922.
- Veldman, P.B.; Dye, P.K.; Holiman, J.D.; Mayko, Z.M.; Sales, C.S.; Straiko, M.D.; Galloway, J.D.; Terry, M.A. The S-stamp in Descemet Membrane Endothelial Keratoplasty Safely Eliminates Upside-down Graft Implantation. Ophthalmology 2016, 123, 161–164.
- Sáles, C.S.; Terry, M.A.; Veldman, P.B.; Mayko, Z.M.; Straiko, M.D. Relationship between tissue unscrolling time and endothelial cell loss. Cornea 2016, 35, 471–476.
- Deng, S.X.; Lee, W.B.; Hammersmith, K.M.; Kuo, A.N.; Li, J.Y.; Shen, J.F.; Weikert, M.P.; Shtein, R.M. Descemet Membrane Endothelial Keratoplasty: Safety and Outcomes: A Report by the American Academy of Ophthalmology. Ophthalmology 2018, 125, 295–310.
- Muijzer, M.B.; Soeters, N.; Godefrooij, D.A.; van Luijk, C.M.; Wisse, R.P.L. Intraoperative Optical Coherence Tomography-Assisted Descemet Membrane Endothelial Keratoplasty: Toward More Efficient, Safer Surgery. Cornea 2020, 39, 674–679.
- Janson, B.J.; Alward, W.L.; Kwon, Y.H.; Bettis, D.I.; Fingert, J.H.; Provencher, L.M.; Goins, K.M.; Wagoner, M.D.; Greiner, M.A. Glaucoma-associated corneal endothelial cell damage: A review. Surv. Ophthalmol. 2018, 63, 500–506.
- Fortune, B.; Yang, H.; Strouthidis, N.G.; Cull, G.A.; Grimm, J.L.; Downs, J.C.; Burgoyne, C.F. The effect of acute intraocular pressure elevation on peripapillary retinal thickness, retinal nerve fiber layer thickness, and retardance. Investig. Ophthalmol. Vis. Sci. 2009, 50, 4719–4726.
- Sharma, N.; Sahay, P.; Maharana, P.K.; Kumar, P.; Ahsan, S.; Titiyal, J.S. Microscope Integrated Intraoperative Optical Coherence Tomography-Guided DMEK in Corneas with Poor Visualization. Clin. Ophthalmol. 2020, 14, 643–651.
- Jacob, S.; Agarwal, A.; Agarwal, A.; Narasimhan, S.; Kumar, D.A.; Sivagnanam, S. Endoilluminator-assisted transcorneal illumination for Descemet membrane endothelial keratoplasty: Enhanced intraoperative visualization of the graft in corneal decompensation secondary to pseudophakic bullous keratopathy. J. Cataract Refract. Surg. 2014, 40, 1332–1336.
- Shimizu, T.; Hayashi, T.; Yuda, K.; Tsuchiya, A.; Oyakawa, I.; Mizuki, N.; Kato, N. Chandelier Illumination for Descemet Membrane Endothelial Keratoplasty. Cornea 2017, 36, 1155–1157.
- Kobayashi, A.; Yokogawa, H.; Yamazaki, N.; Masaki, T.; Sugiyama, K. The use of endoillumination probe-assisted Descemet membrane endothelial keratoplasty for bullous keratopathy secondary to argon laser iridotomy. Clin. Ophthalmol. 2015, 9, 91.
More
Information
Subjects:
Ophthalmology
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
593
Revisions:
2 times
(View History)
Update Date:
02 Apr 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





