Environmental changes caused by human activities alter the water, energy, and carbon cycles in the Amazon region. This has resulted in biological changes across several plant species, some of which are used in both regional and global trade and represent important sources of food and income for people. Reports from local people and scientific studies point to the effects of deforestation, forest degradation, and climate change on native plant species. Indeed, people who are typically dependent on natural resources and ecosystem services are the most threatened by plant species productivity and geographical distribution changes. However, there is a lack of scientific literature concerning the effects of environmental changes on plant species and forest-dependent communities in the Amazon region.
1. Introduction
The change in plant species composition in degraded areas is well established by studies conducted in the Amazon and includes increased mortality of wet-affiliated stems and increased small-seeded plant species
[1][2]. In addition, droughts have become more intense in the region
[3][4], and the ensuing regional hydrological cycle changes, in turn, alter plant ecophysiology and ecosystem productivity
[5][6][7]. In the context of climate change
[8], ecological niche modeling has estimated a decrease in the geographical distribution of native species that are highly embedded in the region’s economy
[9]. For example, the area of occurrence of the
Bertholletia excelsa Bonpl. (commonly known as Brazil nut) could decrease by 25% by 2050, affecting 2239 extractive families of non-timber forest products (NTFP) who live in protected areas
[9]. This may, in turn, potentially result in lower wood and seed density
[2], as changes in species composition are reducing the density of trees and palms widely employed by local populations
[10].
The researchers aimed to broadly characterize environmental changes in the Amazon to assess why the livelihoods of people who depend on forest products are increasingly threatened. The researchers specifically review the effects of deforestation, forest fragmentation, selective logging, forest fire, droughts, global warming, and possible “savannization” on both the productivity and geographical distribution of plant species. To exemplify this, the researchers prepared a list of 30 native plant species negatively impacted by environmental changes, alongside a list of 30 native species that thrive in deforested and degraded lands. Maps of the distribution of typical savannah species found in the Brazilian Amazon are presented for nine species. The researchers also discuss estimates of the annual number of trees and palms that are being destroyed by deforestation in the Amazon and potential economic losses. The conclusions are multidisciplinary and combine botany, ecology, economy, forestry, and sustainability science, and will offer an overview of recent trends and highlight future strategies for improving agroforestry systems.
It is important to understand the interactions between environmental changes and plant species to collectively create strategies to drive a future agroforestry system agenda focused on people, business, and forest restoration. The plant diversity in the Amazon is the highest in the world, with an estimated 15,000 trees and palms species living alongside 30 million people
[11]. The Amazon rainforest is located in South America, spread over 6.7 million square kilometers (km
2), encompassing Brazil, Bolivia, Colombia, Ecuador, French Guiana, Guyana, Peru, Suriname, and Venezuela. Hence, such studies are crucial for understanding the rapid land degradation currently taking place in the Amazon region and the consequences for social and economic activities
[12][13][14].
2. Current Insights
2.1. Deforestation and Forest Degradation Implications to Forest Dependent Communities
Deforestation reduces the livelihoods of forest-dependent communities in the Amazon by abruptly eliminating about 500 million trees and palms each year
[1][15][16]. Forest fragmentation increases plant mortality along the edges
[17][18]. Local climate change, like increased surface air temperature and decreased humidity, influences the ecophysiology of many species
[17]. The physical and biological changes associated with deforestation and forest fragmentation cause widespread loss of animal disperses and pollinators and affect flower, fruit, and seed development
[19][20][21].
Selective logging has reduced the supply of forest-dependent communities in the Amazon. However, some organizations argue that selective logging in the Amazon can take place with reduced impacts
[22]. These arguments underpin the Brazilian legislation that grants areas of public Amazonian forest to private, selective logging companies through a bidding process
[23]. However, severe and significant selective logging affects the forest structure, microclimate, and plant species composition
[24][25][26][27][28]. In fact, people living in the Amazon who depend on the forest for income report negative effects of selective logging on NTFP collection in countries such as Brazil
[29], Bolivia
[30], and Guyana
[31]. Similar reports are noted in the tropical forests of Costa Rica, Nicaragua, Cameroon, and Malaysia
[24]. Therefore, granting public Amazonian forests for selective logging presents both a social and economic threat to communities extracting NTFPs.
Forest fires affect NTFP extraction activities and the cultivation of native species by destroying vegetation and temporarily or permanently interrupting the supply of forest products
[32][33][34]. In addition, forest fires increase the risk of infrastructure damage and forest structure
[35], species composition, and microclimate modifications
[36][37]. Historical data and future projections indicate increased forest fires in the Amazon region
[38][39][40]. Therefore, without forest fire prevention and adaptation plans, livelihoods activities linked to forest products from species intolerant to recurrent fires may be reduced.
2.2. The Effects of Climate Change on Plant Species
Droughts in the Amazon region are increasing in intensity and frequency
[3][4]. The negative effects of drought on tree mortality and fruiting can last up to two or more years after the water deficit period
[41]. However, the species mentioned in
Table 1 are found in both deforested and abandoned areas, where the microclimate is drier and may be better adapted to the more frequent droughts predicted in the future for the region
[42]. Droughts lead to more forest fires
[4][43], and the species listed in
Table 1 generally occur in both deforested and abandoned areas that are frequently affected by fires
[44][45].
Table 1. Examples of plant species undergoing population increases in deforested and degraded lands in the Amazon.
| Botanical Family |
Species Name |
Scientific References |
| Annonaceae |
Guatteria punctata (Aubl.) R.A.Howard |
[46] |
| Arecaceae |
Attalea speciosa Mart. ex Spreng. |
[47] |
| Cannabaceae |
Trema micrantha (L.) Blume |
[48] |
| Dilleniaceae |
Curatella americana L. |
[47] |
| Euphobiaceae |
Croton diasii Pires ex Secco & P.E.Berry |
[48] |
| Euphobiaceae |
Crotonmatourensis Aubl. |
[46] |
| Euphorbiaceae |
Sapium marmieri Huber |
[47] |
| Fabaceae |
Apuleia leiocarpa (Vogel) J.F.Macbr. |
[47] |
| Fabaceae |
Inga thibaudiana DC. |
[46] |
| Hypericaceae |
Vismia amazonica Ewan |
[44] |
| Hypericaceae |
Vismia bemerguii M.E.Berg |
[44] |
| Hypericaceae |
Vismia cauliflora A.C.Sm. |
[44] |
| Hypericaceae |
Vismia cayennensis (Jacq.) Pers. |
[44] |
| Hypericaceae |
Vismia guianensis (Aubl.) Choisy |
[44] |
| Hypericaceae |
Vismia japurensis Reichardt |
[44] |
| Malpighiaceae |
Byrsonima duckeana W.R.Anderson |
[46] |
| Malpighiaceae |
Byrsonima stipulacea A.Juss. |
[44] |
| Malvaceae |
Eriotheca longipedicellata (Ducke) A.Robyns |
[48] |
| Melastomataceae |
Bellucia grossularioides (L.) Triana |
[46] |
| Melastomataceae |
Bellucia imperialis Saldanha & Cogn. |
[46] |
| Rubiaceae |
Coutarea hexandra (Jacq.) K.Schum. |
[47] |
| Rutaceae |
Zanthoxylum rhoifolium Lam. |
[48] |
| Salicaceae |
Banara guianensis Aubl. |
[48] |
| Salicaceae |
Caseariadecandra Jacq. |
[48] |
| Salicaceae |
Casearia sylvestris Sw. |
[47] |
| Solanaceae |
Solanum crinitum Lam. |
[48] |
| Urticaceae |
Cecropia purpurascens C.C.Berg |
[44] |
| Urticaceae |
Cecropia sciadophylla Mart. |
[46] |
| Urticaceae |
Pourouma apiculata Spruce ex Benoist |
[46] |
| Vochysiaceae |
Erisma uncinatum Warm. |
[47] |
Global warming appears to be a key long-term threat to the Amazonian forest products by changing the geographic distribution of plant species
[49][9]. It reduces flowers’ exposure to pollinators and alters physiological processes associated with protein, oil, and vegetable fat syntheses
[6][50]. Pollination and protein and lipid production are essential for seed formation
[51][20].
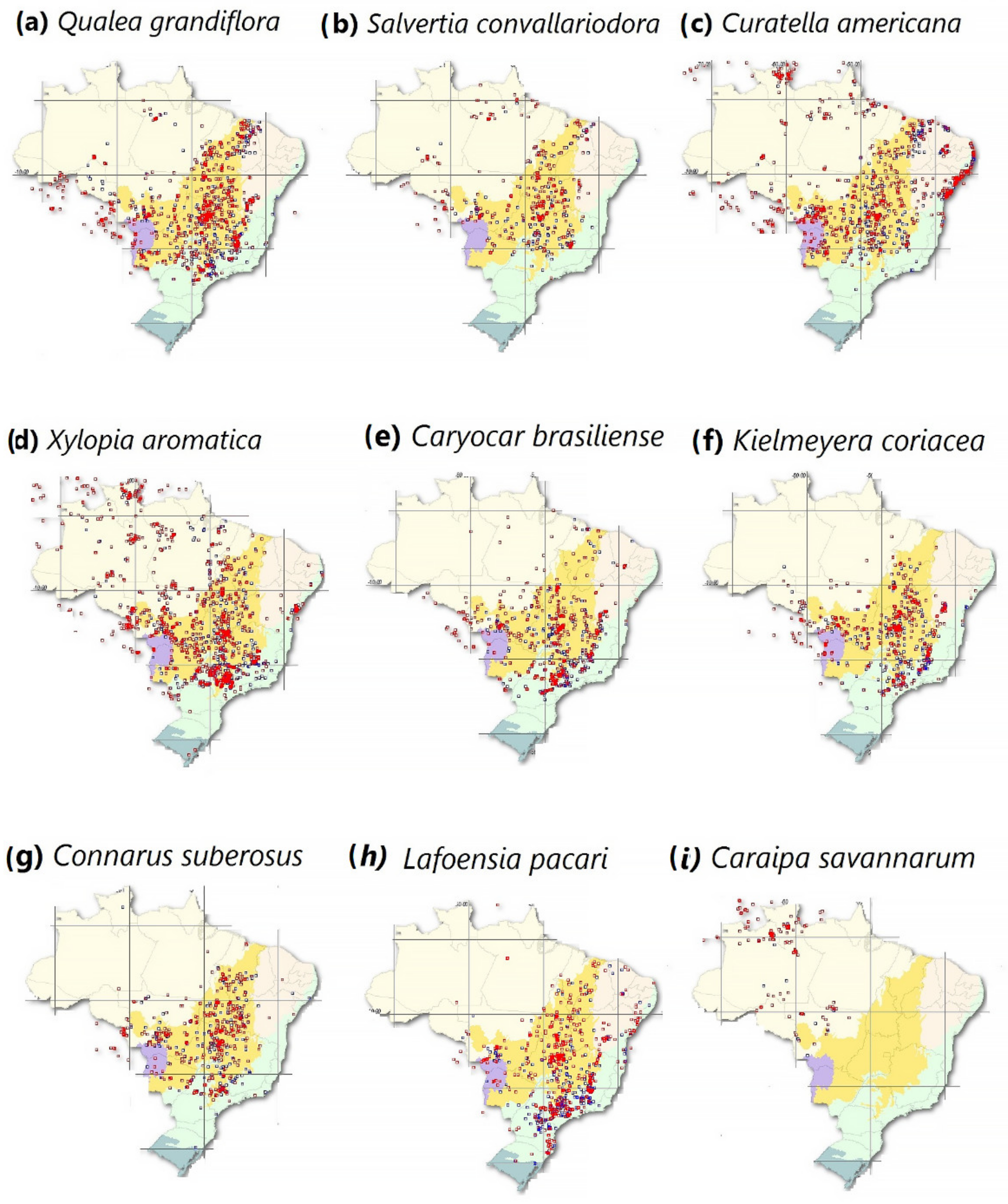
Increased reports of certain species, such as
C. savannarum and
Cecropia [1][52], may be evidence of a regional climatic shift that favors the replacement of typical humid climate-adapted species with typical savannah species
[53][54]. In fact, although poorly studied, changes in species composition are considered a slow and gradual process
[1][55]. The Amazon region vulnerable to “savannization” is characterized by an open ombrophilous forest type
[54][56]. To a large extent, this region is located in the southern Brazilian Amazon forest, encompassing municipalities in Acre, south Amazonas, Rondônia, north Mato Grosso, and southern Pará
[12][56]. Therefore, typical savannah species populations, such as those in
Figure 1, may increase in the southern and eastern regions of the Brazilian Amazon, where the environment is drier, warmer and where frequent forest fires occur.
Figure 1. The geographical distribution of typical savannah species that are found in the Brazilian Amazon. The points displayed on the maps represent original species geographic coordinates (red) and per occurrence municipality in Brazil (blue). Reprinted with permission from ref.
[57]. 2021 speciesLink network.
2.3. Potential Reduction of Native Amazonian Plants and Annual Range of Economic Losses
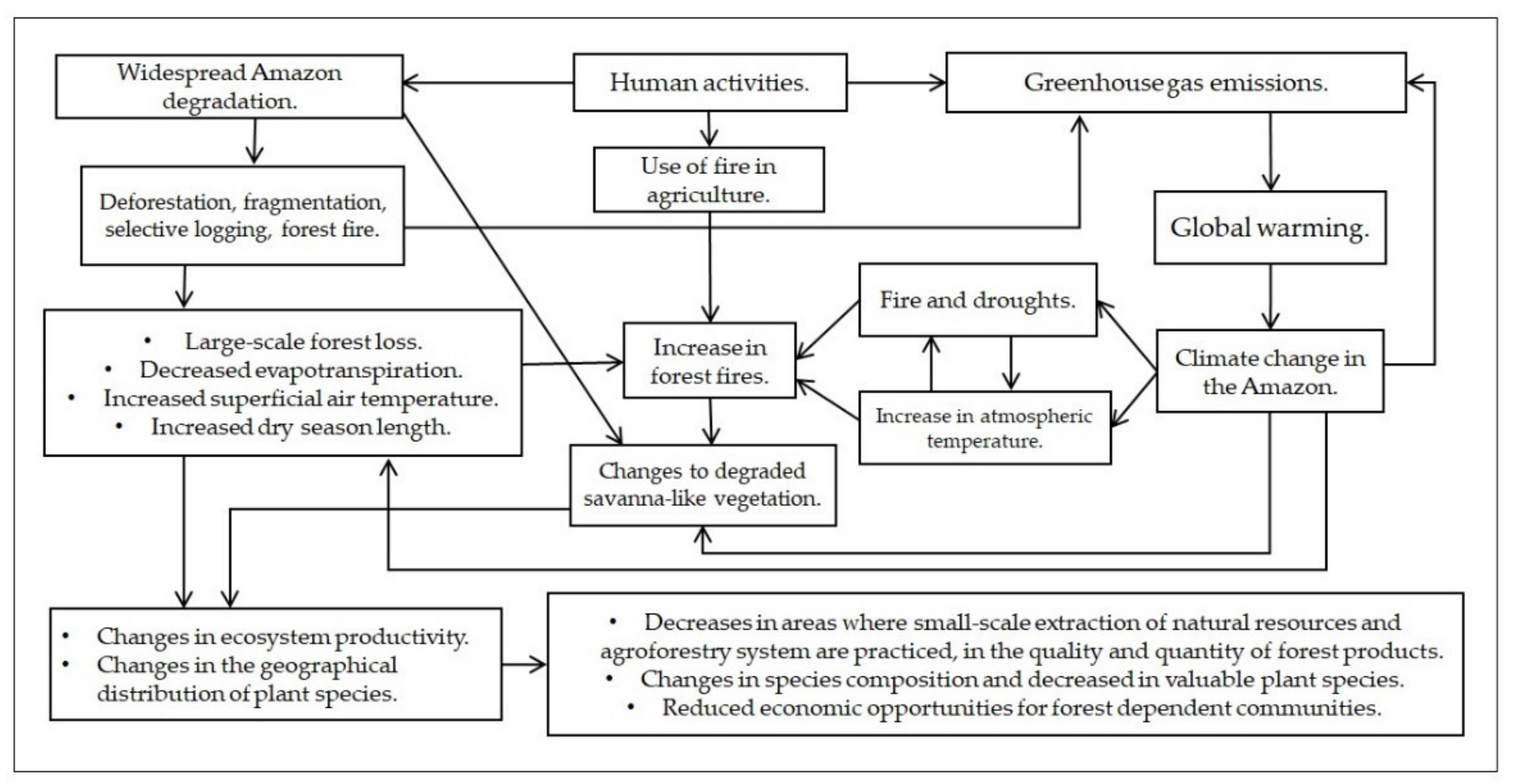
There will be a reduction of native Amazonian species widely employed by local populations due to environmental changes, both local and globally, in turn leading to decreased supply, diversity, quality, and amount of forest products. This suggests that without the restoration of deforested and degraded forests, the food, well-being, and income obtained by communities from native forest products in the Amazon will be severely impacted (
Figure 2). Other human activity effects that alter ecosystem productivity and geographical distribution of plant species in the Amazon are noted, in addition to those reviewed herein, such as extreme flooding, over-exploitation, invasive species, and loss dispersers
[58][59][60]. Species that dominate the forest succession in deforested and abandoned lands are not economically exploited in the region (as listed in
Table 1), with the exception of palms.
Figure 2. Schematic representation of plant responses to human environmental changes and livelihoods effects in forest-dependent communities.
Palm species are currently the most important in the Amazon NTFP economy
[61][62][63], comprising the botanical Arecaceae family, and occur widely in undisturbed forests
[64]. Since they are resistant to forest fires and extreme droughts, they are also present during different vegetation succession phases in both deforested and abandoned areas
[47][36][65]. This indicates that food provisioning and income from palm species may be less impacted by some of the environmental changes discussed in this entry
[47][36][65]. Therefore, an increase in palm density may indicate a future with more palms in the southern and eastern regions of the Amazon.
Finally, a decline of valuable species has been observed mainly in the eastern, central, and southern regions of the Brazilian Amazon
[10][29][24][13][20][66][67][27]. In fact, the value generated in the Brazilian Amazon from NTFPs is approximately US$2 billion year
−1 [68][62][69]. After deducting collection and transport costs, Peters et al. (1989) estimated that US$422 ha
−1 year
−1 was obtained for NTFP alone
[70]. As more environmental products and services are included in estimates of tropical forest value potential, the global average is US$5264 ha
−1 year
−1 [71]. Based on these estimates and the 16-year average Amazon deforestation rate (about 1,668,600 hectares per year
[72]), the loss of native species’ economic importance ranges between US$704,149,200 year
−1 and US$8,783,510,400 year
−1.
2.4. Potential for the Improvement of Agroforestry Systems Strategies and Advancement of Stakeholder Engagement Approaches
Agroforestry systems combine the management of native and exotic species simultaneously in the same land. For example, native plants highly present in agroforestry systems in Amazon are acai berry (
Euterpe oleracea Mart. and
E. precatoria), andiroba (
C. guianensis), buriti (
Mauritia flexuosa L.), bacuri (
Platonia insignis Mart.), cacau (
Theobroma cacao L.), Brazil nut (
B. excelsa), tonka bean (
D. odorata), cupuassu (
Theobroma grandiflorum (Willd. ex Spreng.) K.Schum.), pupunha (
Bactris gasipaes Kunth.), and taperebá (
Spondias mombin L.), while common exotic species are coconut (
Cocos nucifera L.), coffee (
Coffea arabica L. and
Coffea canephora Pierre ex A.Froehner.), palm oil
(Elaeis guineenses Jacq.), mango (
Mangifera indica L.), banana (
Musa × paradisiaca L.), lemon, orange, tangerine (
Citrus spp.), and black pepper (
Piper nigrum L.). Agroforestry systems in the Amazon are more productive and biodiverse compared to monocultures and can also include animal management, such as cattle, chickens, ducks, and pigs, without having to deforest down huge forest areas
[10][73][74][75].
Agroforestry system costs and benefits in the Amazon are promising to public and private investments, especially concerning forest restoration. The average investment required for forest restoration through the agroforestry system method ranges between US$2500 and US$7000 per hectare
[73][76][77], with internal return rates ranging from 10 to 111%, and payback between 2 and 13 years
[76][77]. Income generation with NTFPs obtained from agroforestry systems has varied between US$400 and US$800 ha
−1 year
−1 [77]. In fact, native species NTFPs are generating around US$1 billion/year
−1 in municipalities in the state of Pará, with an estimated economic value of US$32 billion by 2040 if investments in science and technology are made
[69]. Furthermore, forest restoration through the agroforestry systems has resulted in three times more aboveground biomass compared to the average in degraded and abandoned areas
[16][78], consequently comprising a Reducing Emissions from Deforestation and Forest Degradation (REDD+) strategy
[79].
The demand from industrial activities for NTFP is to increase the quality and quantity of raw material to the supply chain
[80]. Companies in the cosmetics, food, chemical, and perfume industries are already being supplied with NTFP from Amazonian plants
[80][61]. The technological and scientific challenges, however, are significant. For example, only 15% of municipalities in the Brazilian Amazon have the industrial infrastructure to produce raw materials, such as fats, oils, and pulp, for the national and global markets
[10]. In addition, technical assistance investments are crucial for businesses and people who depend on NTFP in the Amazon, but the number of establishments that receive technical assistance ranges between 1 and 22% in the different regions
[69]. Therefore, science and technology investments are considered promising in implementing agroforestry systems recovering deforested and degraded areas, which could engage companies that use NTPF due to supply chain advantages.
Science and technology investments are still low in the Amazon. For example, the Brazilian National Bank for Economic and Social Development (BNDES) is the largest public resource investor for the development of NTFP supply chains in the Brazilian Amazon, investing an average of US$10 million year
−1 [81]. The largest private investor is located in the cosmetic company, averaging US$50 million year
−1 [61]. In addition, deforested and abandoned lands are rarely being restored, with native species presenting economic value
[82], and NTFPs are rapidly reducing due to deforestation, forest degradation, and climate change
[10]. Therefore, new investments are essential for people, businesses, and sustainable value-chains in the Amazon, which will strengthen entrepreneurship, innovations, and startups
[80][12][83][84][85].
The species lists presented herein can aid in the species selection phase for agroforestry system implementation and can be used as references for stakeholders. Both species lists contain empirical references to changes in species composition due to human activities, which can be revised and enhanced in future studies. Furthermore, other species that may increase in agroforestry systems in the Amazon comprise those typically employed as NTFP in the savannah vegetation region and present in the Amazon, such as the araticum (Annona crassiflora Mart.), cashew (Anacardium occidentale L.), macauba (Acrocomia aculeata (Jacq.) Lodd. ex Mart.) e pequi (Caryocar brasiliense Cambess.). A research agenda on species increasing in degraded land can augment the social and economic importance of these plant species.