
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Maria Isabel L. Soares | + 2573 word(s) | 2573 | 2022-02-25 15:24:37 | | | |
| 2 | Maria Isabel L. Soares | Meta information modification | 2573 | 2022-03-23 14:03:31 | | | | |
| 3 | Jessie Wu | -631 word(s) | 1942 | 2022-03-24 02:07:19 | | | | |
| 4 | Jessie Wu | -631 word(s) | 1942 | 2022-03-24 02:13:34 | | | | |
| 5 | Jessie Wu | -3 word(s) | 1939 | 2022-03-24 02:33:32 | | | | |
| 6 | Maria Isabel L. Soares | -315 word(s) | 1624 | 2022-03-24 10:42:22 | | | | |
| 7 | Jessie Wu | Meta information modification | 1624 | 2022-03-25 07:08:15 | | |
Video Upload Options
Diels–Alder cycloaddition reaction is one of the most powerful strategies for the construction of six-membered carbocyclic and heterocyclic systems, in most cases with high regio- and stereoselectivity. An insight into the most relevant advances on sustainable Diels–Alder reactions since 2010 is provided. Various environmentally benign solvent systems are discussed, namely bio-based derived solvents, polyethylene glycol, deep eutectic solvents, supercritical carbon dioxide, water and water-based aqueous systems.
1. Introduction
2. Bio-based solvents
Glycerol, the main by-product in the biodiesel industry, is a nontoxic, biodegradable, recyclable and inexpensive viscous liquid. These properties, allied with the high stability, biocompatibility and ability to dissolve organic compounds poorly miscible in water as well as inorganic compounds, make glycerol a valuable green solvent in synthetic organic chemistry [35][36][37].
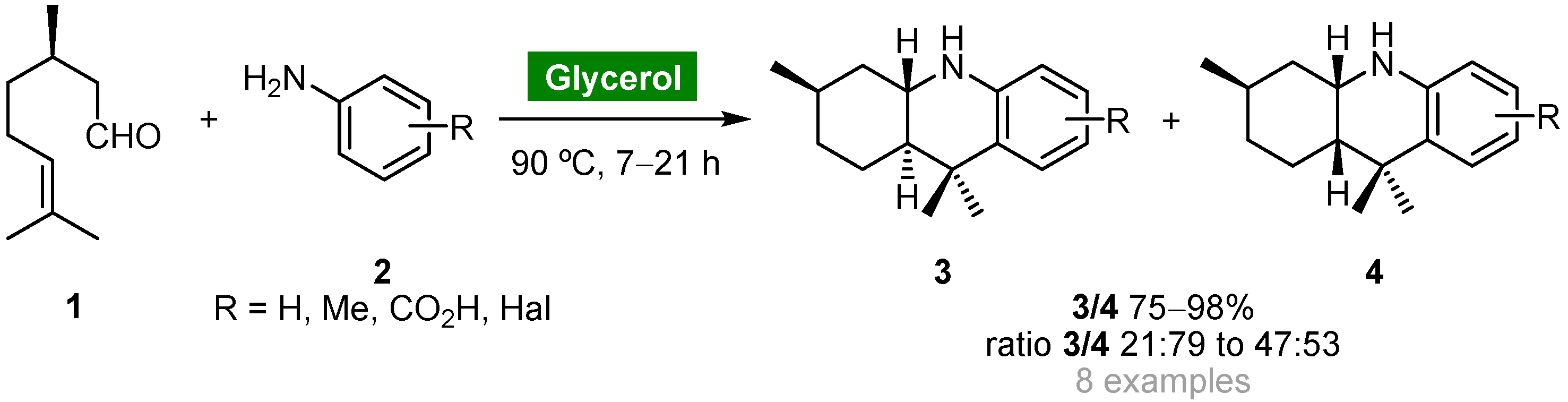
The three-component aza-Diels–Alder reaction of substituted anilines, aldehydes and electron-rich alkenes, also known as three-component imino-Diels–Alder reaction, or multicomponent Povarov reaction, is one of the most straightforward, efficient and atom-economical strategies towards complex cores starting from simple, inexpensive and available materials. Perin and coworkers explored the intramolecular version of this reaction for the catalyst-free synthesis of octahydroacridines starting from (R)-citronellal (1) and substituted arylamines 2 using glycerol as a recyclable and eco-friendly solvent (Scheme 1) [38]. Cycloadducts 3 and 4 were obtained as diastereoisomeric mixtures in good to high yields (75–98%) and moderate cis-selectivity when the reaction was carried out at 90 °C. Cycloadducts 3/4 (R = H) were obtained in lower yield (62%) using water as solvent, whereas the reaction carried out in organic solvents afforded the corresponding adducts in only trace amounts. Due to the insolubility of 3 and 4 in glycerol, products could be removed from the reaction medium by decantation, and the solvent could be reused for further aza-Diels–Alder reactions without loss of activity.
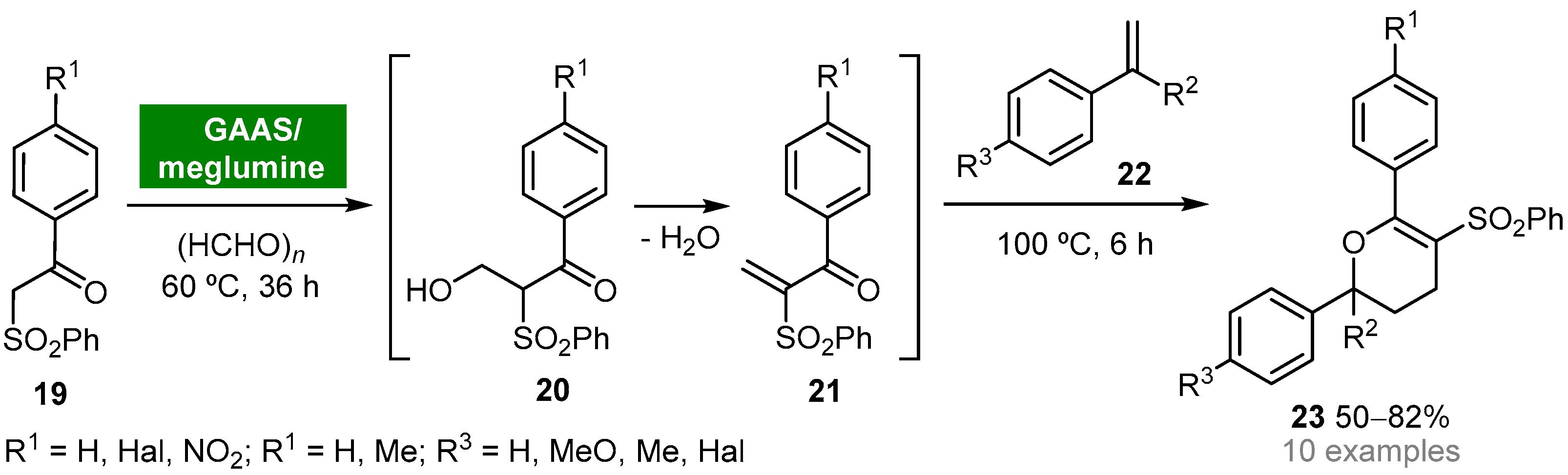
Gluconic acid (GA) can be obtained from biomass and possesses the ideal properties for being classified as a green and sustainable solvent (e.g., nontoxicity, biodegradability, recyclability, high boiling point, low vapor pressure) [39]. Due to the high solubility of gluconic acid in water, gluconic acid aqueous solutions (GAASs) have found wide application as solvent media for organic reactions, namely for the Knoevenagel condensation reaction. The Gu group reported the synthesis of 2H-pyrans by a one-pot multicomponent reaction between β-ketosulfones, formaldehyde and styrenes in a bio-based binary mixture solvent system composed of GAAS and a sugar-based organic base, meglumine [40]. The disclosed protocol involves the in situ generation of α-methylene-β-ketosulfones 21 through a Knoevenagel reaction of β-ketosulfones 19 and formaldehyde. Next, nucleophilic trap of 21 with styrenes 22 via oxa-Diels–Alder reaction afforded 2,6-diaryl-5-(phenylsulfonyl)-3,4-dihydro-2H-pyrans 23 in moderate to good yields (50–82%) (Scheme 2). The binary solvent system GAAS/meglumine proved to play a pivotal role in controlling the selectivity of the hydroxymethylation step. Moreover, the hydrophilic properties of bio-based solvent meglumine allowed it to be easily recycled and reused in the GAAS/meglumine system without significant loss of activity.
3. Polyethylene Glycol
Polyethylene glycol (PEG), HO–(CH2CH2O)n–H, is a biodegradable, nontoxic, odorless, neutral, nonvolatile and inexpensive water-soluble polymer that has found widespread application as a green reaction medium for several organic transformations [18]. The Kouznetsov group reported the diastereoselective synthesis of heterolignan-like 6,7-methylendioxy-tetrahydroquinolines via a BF3.OEt-catalyzed three-component Povarov reaction using clove bud essential oil as a renewable raw material and PEG-400 as green solvent (Scheme 3) [41]. Clove bud essential oil enriched with eugenol 24 (60.5%) was obtained by hydrodistillation of dried flower buds and then subjected to a solid base-catalyzed isomerization to give trans/cis-isoeugenol 25, which could be used as a dienophile in the multicomponent hetero-Diels–Alder reaction without further purification. The reaction of 25 with aldimines generated in situ from substituted benzaldehydes 27 and 3,4-(methylendioxy)aniline (26) afforded trans-2,4-diaryl-1,2,3,4-tetrahydroquinolines 28 as racemic mixtures in moderate yields (35–55%). The reaction with phthalaldehydic acid (29) afforded isoindolo[2,1-a]quinolin-11(5H)-one 30 via an intramolecular condensation of the initially generated NH-tetrahydroquinoline core with the o-carboxylic acid function leading to the formation of the γ-lactam ring. It is noteworthy that these reactions also worked using acetonitrile as solvent media; however, less solvent volume and reduced reaction times were required when using PEG-400.
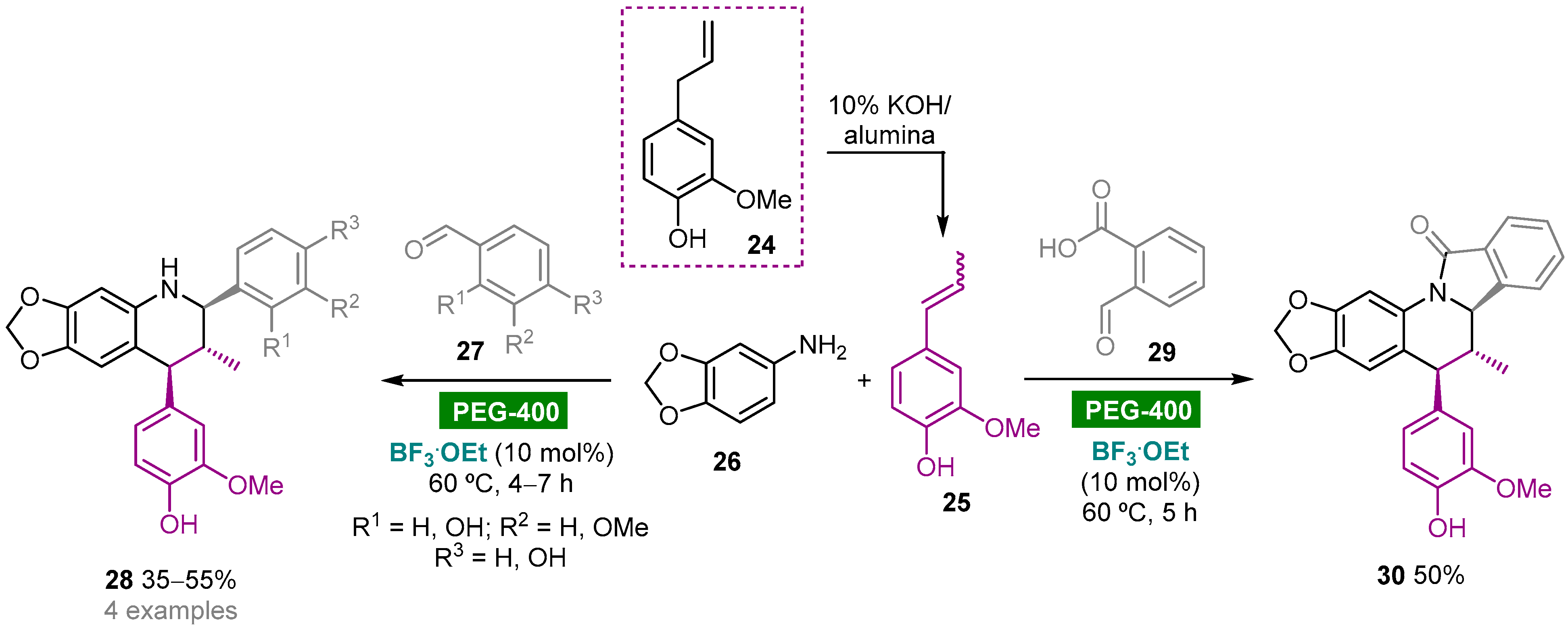
Scheme 3. BF3.OEt-catalyzed one-pot multicomponent aza-Diels–Alder reaction of 3,4-(methylendioxy)aniline, aromatic aldehydes and trans/cis-isoeugenol in PEG-400.
4. Organic Carbonates
Propylene carbonate (PC) is a polar aprotic solvent that can be obtained from propylene oxide and carbon dioxide, a renewable source of carbon, in a 100% atom economy reaction with relevance regarding the development of CO2 fixation processes. The noncorrosive, nontoxic, odorless and biodegradable properties of PC, allied with high boiling point, low vapor pressure and low cost, make this solvent a green and sustainable alternative to conventional organic solvents [42]. The Povarov reaction has also been explored using PC as an environmentally friendly solvent [43]. The one-pot iodine-catalyzed reaction of mono- or disubstituted anilines 2, aromatic aldehydes 5 and isoeugenol (45) carried out at room temperature using PC as solvent medium afforded functionalized tetrahydroquinolines 46 in good to high yields (77–95%) and high diastereoselectivity (dr up to >99:1) (Scheme 4). The same cycloadducts were obtained using organic solvents, albeit in low yields and requiring longer reaction times. It is noteworthy that, in general, products precipitated from the reaction medium and were purified by recrystallization.
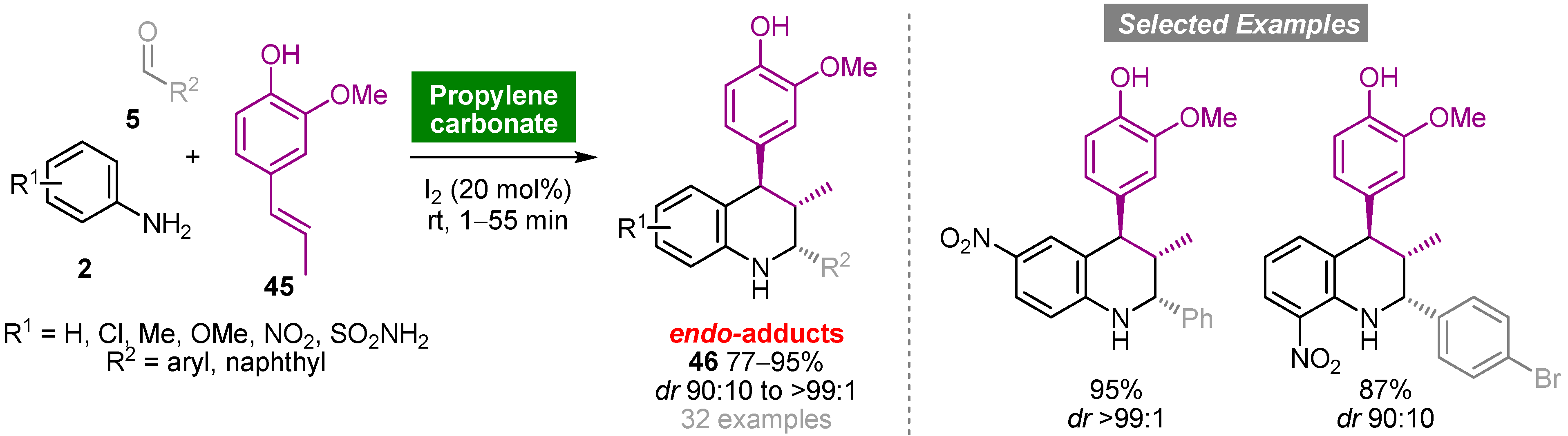
Scheme 4. Iodine-catalyzed one-pot multicomponent aza-Diels–Alder reaction of anilines, aromatic aldehydes and isoeugenol in propylene carbonate.
5. Deep Eutectic Solvents
First introduced by Abbot [44], deep eutectic solvents (DESs) are low melting mixtures obtained by combination of at least two components, a hydrogen bond acceptor (HBA), generally a quaternary ammonium or metal salt, and a hydrogen bond donor (HBD), to form a eutectic phase via hydrogen bond interactions. DESs are characterized by a melting point lower than those of the single components. The properties of DESs are very similar to those of room-temperature ionic liquids; however, the main difference from ionic liquids is that DESs also contain an organic molecular component, the HBD (e.g., urea, amide, polyol), generally as a major component. Due to their low vapor pressure, nonflammability, thermal and chemical stability, nontoxicity, biodegradability, recyclability and low price, DESs have emerged as green and sustainable media in different areas of chemical research [45][46][47][48], namely organic synthesis and catalysis [21][22][23].
Garcia-Álvarez’s group reported a one-pot tandem cycloisomerization/Diels–Alder reaction using a ChCl (choline chloride)-based eutectic mixture as solvent [49]. The protocol involves the in situ generation of furans 59 by cycloisomerization of (Z)-enynols 57 using a ChCl/Gly (1:2) eutectic mixture as solvent and bis(iminophosphorane)-Au(i) complex 58 as catalyst (Scheme 5). The Diels–Alder reaction of furans 59 with activated alkynes 60 afforded 7-oxanorbornadienes 61, whereas the reaction with activated alkenes, 55b or 62, afforded selectively exo-7-oxanorbornenes 63. The authors have demonstrated that complex 58 is crucial for the cycloisomerization step; however, it does not participate in the cycloaddition step.
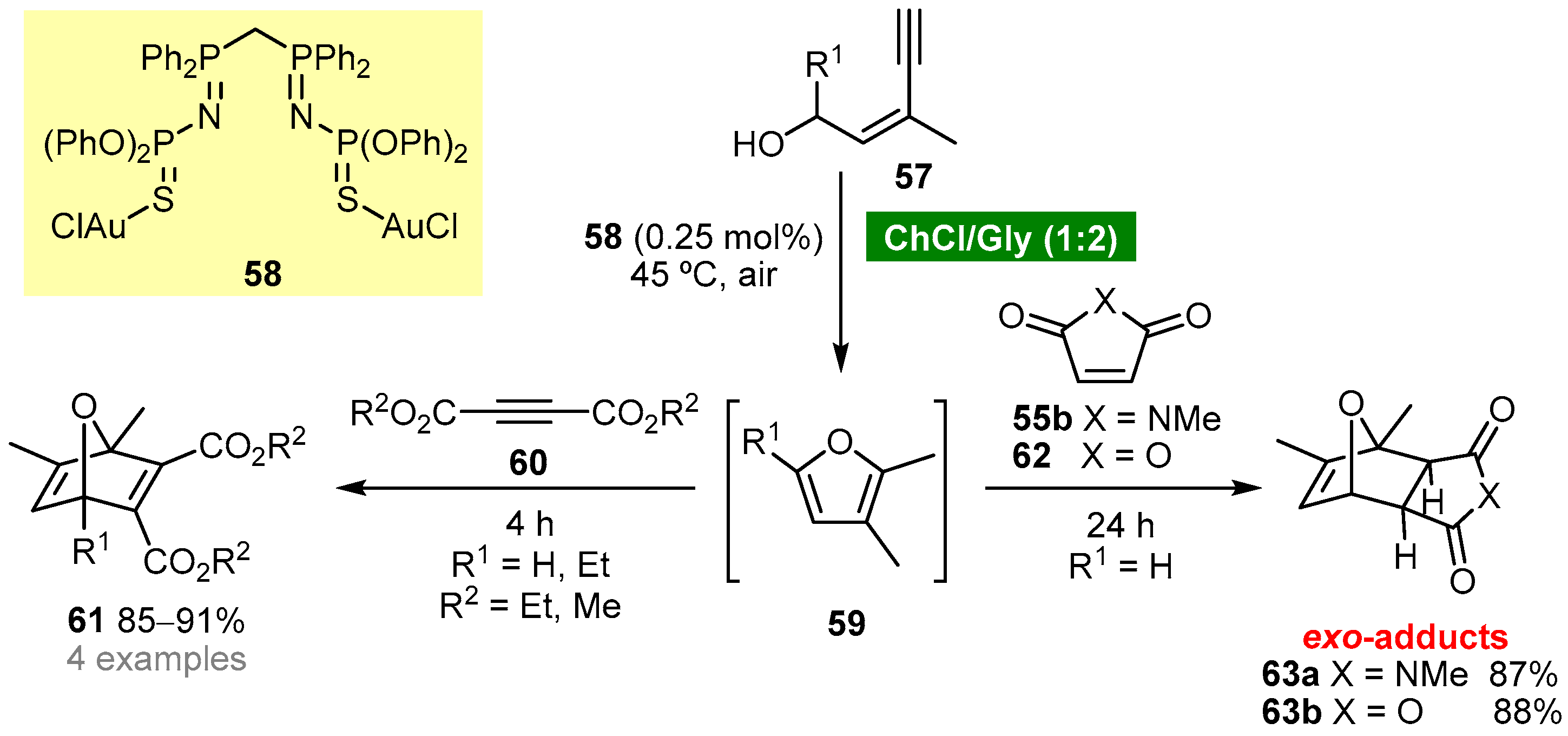
Scheme 5. One-pot tandem cycloisomerization/Diels–Alder reaction of (Z)-enynols in a ChCl/Gly deep eutectic solvent.
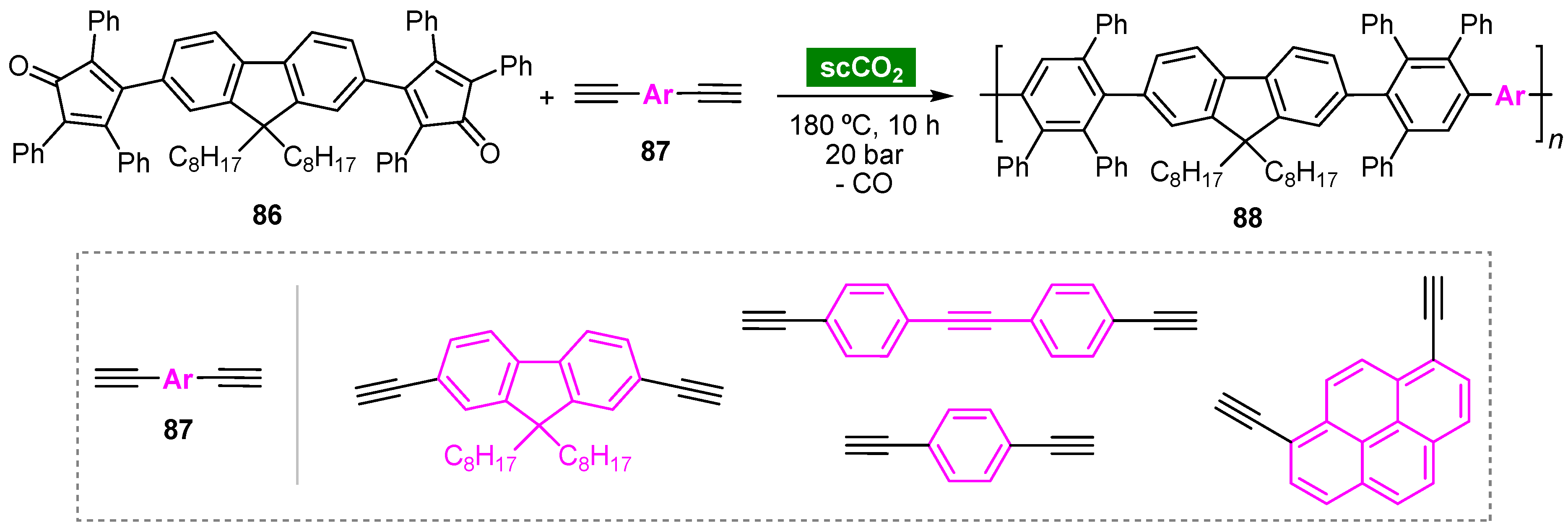
6. Supercritical Carbon Dioxide
References
- Ulf. Pindur; Gundula. Lutz; Christian. Otto; Acceleration and selectivity enhancement of Diels-Alder reactions by special and catalytic methods. Chemical Reviews 1993, 93, 741-761, 10.1021/cr00018a006.
- E. J. Corey; Catalytic Enantioselective Diels-Alder Reactions: Methods, Mechanistic Fundamentals, Pathways, and Applications. Angewandte Chemie International Edition 2002, 41, 1650-1667, 10.1002/1521-3773(20020517)41:10<1650::aid-anie1650>3.0.co;2-b.
- Pedro Merino; Raquel P. Herrera; Eugenia Marqués-López; Tomás Tejero; Enantioselective Organocatalytic Diels-Alder Reactions. Synthesis 2009, 2010, 1-26, 10.1055/s-0029-1217130.
- Víctor Laina‐Martín; Jose A. Fernández‐Salas; José Alemán; Organocatalytic Strategies for the Development of the Enantioselective Inverse‐electron‐demand Hetero‐Diels‐Alder Reaction. Chemistry – A European Journal 2021, 27, 12509-12520, 10.1002/chem.202101696.
- Bm Trost; The Atom Economy—A Search for Synthetic Efficiency. Science 1991, 254, 1471-1477, 10.1126/science.1962206.
- Hanno C. Erythropel; Julie B. Zimmerman; Tamara M. de Winter; Laurène Petitjean; Fjodor Melnikov; Chun Ho Lam; Amanda W. Lounsbury; Karolina E. Mellor; Nina Z. Janković; Qingshi Tu; et al.Lauren N. PincusMark M. FalinskiWenbo ShiPhilip CoishDesirée L. PlataPaul T. Anastas The Green ChemisTREE: 20 years after taking root with the 12 principles. Green Chemistry 2018, 20, 1929-1961, 10.1039/c8gc00482j.
- Roger A. Sheldon; Green solvents for sustainable organic synthesis: state of the art. Green Chem. 2005, 7, 267-278, 10.1039/b418069k.
- Martín Avalos; Reyes Babiano; Pedro Cintas; José L. Jiménez; Juan C. Palacios; Greener Media in Chemical Synthesis and Processing. Angewandte Chemie International Edition 2006, 45, 3904-3908, 10.1002/anie.200504285.
- Carlos Z. Andrade; Carlos Kleber Z. Andrade And Luana M. Alves; Environmentally Benign Solvents in Organic Synthesis: Current Topics. Current Organic Chemistry 2005, 9, 195-218, 10.2174/1385272053369178.
- Tom Welton; Solvents and sustainable chemistry. Proceedings of the Royal Society A: Mathematical, Physical and Engineering Sciences 2015, 471, 20150502-20150502, 10.1098/rspa.2015.0502.
- Coby Clarke; Wei-Chien Tu; Oliver Levers; Andreas Bröhl; Jason P. Hallett; Green and Sustainable Solvents in Chemical Processes. Chemical Reviews 2018, 118, 747-800, 10.1021/acs.chemrev.7b00571.
- Devendra S. Wagare; Sagar E. Shirsath; Mujahed Shaikh; Prashant Netankar; Sustainable solvents in chemical synthesis: a review. Environmental Chemistry Letters 2021, 19, 3263-3282, 10.1007/s10311-020-01176-6.
- K. Tanaka; F. Toda; Solvent-Free Organic Synthesis. Chemical Reviews 2000, 100, 1025-1074, 10.1021/cr940089p.
- Yanlong Gu; François Jérôme; Bio-based solvents: an emerging generation of fluids for the design of eco-efficient processes in catalysis and organic chemistry. Chem. Soc. Rev. 2013, 42, 9550-9570, 10.1039/c3cs60241a.
- Stefano Santoro; Francesco Ferlin; Lorenzo Luciani; Lutz Ackermann; Luigi Vaccaro; Biomass-derived solvents as effective media for cross-coupling reactions and C–H functionalization processes. Green Chemistry 2017, 19, 1601-1612, 10.1039/c7gc00067g.
- Magne O. Sydnes; Green Bio-Based Solvents in C-C Cross-Coupling Reactions. Current Green Chemistry 2019, 6, 96-104, 10.2174/2213346106666190411151447.
- Parthasarathy Gandeepan; Nikolaos Kaplaneris; Stefano Santoro; Luigi Vaccaro; Lutz Ackermann; Biomass-Derived Solvents for Sustainable Transition Metal-Catalyzed C–H Activation. ACS Sustainable Chemistry & Engineering 2019, 7, 8023-8040, 10.1021/acssuschemeng.9b00226.
- Ji Chen; Scott K. Spear; Jonathan G. Huddleston; Robin D. Rogers; Polyethylene glycol and solutions of polyethylene glycol as green reaction media. Green Chemistry 2005, 7, 64-82, 10.1039/b413546f.
- Jason P. Hallett; Tom Welton; Room-Temperature Ionic Liquids: Solvents for Synthesis and Catalysis. 2. Chemical Reviews 2011, 111, 3508-3576, 10.1021/cr1003248.
- Marcos A. P. Martins; Clarissa P. Frizzo; Dayse N. Moreira; Nilo Zanatta; Helio G. Bonacorso; Ionic Liquids in Heterocyclic Synthesis. Chemical Reviews 2008, 108, 2015-2050, 10.1021/cr078399y.
- Peng Liu; Jian-Wu Hao; Li-Ping Mo; Zhan-Hui Zhang; Recent advances in the application of deep eutectic solvents as sustainable media as well as catalysts in organic reactions. RSC Advances 2015, 5, 48675-48704, 10.1039/c5ra05746a.
- Sarita Khandelwal; Yogesh Kumar Tailor; Mahendra Kumar; Deep eutectic solvents (DESs) as eco-friendly and sustainable solvent/catalyst systems in organic transformations. Journal of Molecular Liquids 2016, 215, 345-386, 10.1016/j.molliq.2015.12.015.
- Diego A. Alonso; Alejandro Baeza; Rafael Chinchilla; Gabriela Guillena; Isidro M. Pastor; Diego J. Ramón; Deep Eutectic Solvents: The Organic Reaction Medium of the Century. European Journal of Organic Chemistry 2016, 2016, 612-632, 10.1002/ejoc.201501197.
- Gerd Kaupp; Reactions in Supercritical Carbon Dioxide. Angewandte Chemie International Edition 1994, 33, 1452-1455, 10.1002/anie.199414521.
- Eric J Beckman; Supercritical and near-critical CO2 in green chemical synthesis and processing. The Journal of Supercritical Fluids 2004, 28, 121-191, 10.1016/s0896-8446(03)00029-9.
- Phillip E. Savage; Sudhama Gopalan; Thamid I. Mizan; Christopher J. Martino; Eric E. Brock; Reactions at supercritical conditions: Applications and fundamentals. AIChE Journal 1995, 41, 1723-1778, 10.1002/aic.690410712.
- R. Scott Oakes; Anthony A. Clifford; Christopher M. Rayner; The use of supercritical fluids in synthetic organic chemistry. Journal of the Chemical Society, Perkin Transactions 1 2001, 2001, 917-941, 10.1039/b101219n.
- Peter Licence; Jie Ke; Maia Sokolova; Stephen K. Ross; Martyn Poliakoff; Chemical reactions in supercritical carbon dioxide: from laboratory to commercial plantThis work was presented at the Green Solvents for Catalysis Meeting held in Bruchsal, Germany, 13–16th October 2002.. Green Chemistry 2003, 5, 99-104, 10.1039/b212220k.
- Helen C. Hailes; Reaction Solvent Selection: The Potential of Water as a Solvent for Organic Transformations. Organic Process Research & Development 2006, 11, 114-120, 10.1021/op060157x.
- Arani Chanda; Valery V. Fokin; Organic Synthesis “On Water”. Chemical Reviews 2009, 109, 725-748, 10.1021/cr800448q.
- Manoj B. Gawande; Vasco D. B. Bonifácio; Rafael Luque; Paula S. Branco; Rajender S. Varma; Benign by design: catalyst-free in-water, on-water green chemical methodologies in organic synthesis. Chemical Society Reviews 2013, 42, 5522-5551, 10.1039/c3cs60025d.
- Richard N. Butler; Anthony G. Coyne; Organic synthesis reactions on-water at the organic–liquid water interface. Organic & Biomolecular Chemistry 2016, 14, 9945-9960, 10.1039/c6ob01724j.
- Taku Kitanosono; Koichiro Masuda; Pengyu Xu; Shu̅ Kobayashi; Catalytic Organic Reactions in Water toward Sustainable Society. Chemical Reviews 2017, 118, 679-746, 10.1021/acs.chemrev.7b00417.
- Kartick C. Bhowmick; Manisha Bihani; John C.-G. Zhao; Organocatalyzed Asymmetric Diels-Alder Reactions in Aqueous or Semi- Aqueous Media. Mini-Reviews in Organic Chemistry 2018, 15, 3-19, 10.2174/1570193x14666170518121235.
- Alba E. Díaz-Álvarez; Javier Francos; Beatriz Lastra-Barreira; Pascale Crochet; Victorio Cadierno; Glycerol and derived solvents: new sustainable reaction media for organic synthesis. Chemical Communications 2011, 47, 6208-6227, 10.1039/c1cc10620a.
- Alba Estrella Díaz Álvarez; Javier Francos; Pascale Veronique Crochet; Victorio Cadierno; Recent Advances in the Use of Glycerol as Green Solvent for Synthetic Organic Chemistry. Current Green Chemistry 2013, 1, 51-65, 10.2174/221334610101131218094907.
- Mohamed Abd-Elmonem; Ramadan A. Mekheimer; Alaa M. Hayallah; Fatma A. Abo Elsoud; Kamal U. Sadek; Recent Advances in the Utility of Glycerol as a Benign and Biodegradable Medium in Heterocyclic Synthesis. Current Organic Chemistry 2020, 23, 3226-3246, 10.2174/1385272823666191025150646.
- José E.R. Nascimento; Angelita Barcellos; Maraísa Sachini; Gelson Perin; Eder Joao Lenardao; Diego Alves; Raquel G. Jacob; Fabiana Missau; Catalyst-free synthesis of octahydroacridines using glycerol as recyclable solvent. Tetrahedron Letters 2011, 52, 2571-2574, 10.1016/j.tetlet.2011.03.045.
- Han Yin Lim; Anton V. Dolzhenko; Gluconic acid aqueous solution: A bio-based catalytic medium for organic synthesis. Sustainable Chemistry and Pharmacy 2021, 21, 100443, 10.1016/j.scp.2021.100443.
- Jie Yang; Haoquan Li; Minghao Li; Jiajian Peng; Yanlong Gu; Multicomponent Reactions of β-Ketosulfones and Formaldehyde in a Bio-Based Binary Mixture Solvent System Composed of Meglumine and Gluconic Acid Aqueous Solution. Advanced Synthesis & Catalysis 2012, 354, 688-700, 10.1002/adsc.201100799.
- Diego R. Merchan Arenas; Fernando A. Rojas Ruíz; Vladimir V. Kouznetsov; Highly diastereoselective synthesis of new heterolignan-like 6,7-methylendioxy-tetrahydroquinolines using the clove bud essential oil as raw material. Tetrahedron Letters 2011, 52, 1388-1391, 10.1016/j.tetlet.2011.01.083.
- Josué Sebastian Bello Forero; July A. Hernández Muñoz; Joel Jones Junior; Flavia M. Da Silva; Propylene Carbonate in Organic Synthesis: Exploring its Potential as a Green Solvent. Current Organic Synthesis 2016, 13, 834-846, 10.2174/1570179413999160211094705.
- Josué Sebastian Bello Forero; Erika M. De Carvalho; Joel J. Junior; Flavia M. Da Silva; Facile, Efficient Diastereoselective Synthesis of Tetrahydroquinoline Scaffolds Using Propylene Carbonate as an Eco-Friendly Solvent. Current Organic Synthesis 2015, 12, 102-107, 10.2174/1570179411666140722175810.
- Andrew P. Abbott; David Boothby; Glen Capper; David L. Davies; Raymond K. Rasheed; Deep Eutectic Solvents Formed between Choline Chloride and Carboxylic Acids: Versatile Alternatives to Ionic Liquids. Journal of the American Chemical Society 2004, 126, 9142-9147, 10.1021/ja048266j.
- Qinghua Zhang; Karine De Oliveira Vigier; Sébastien Royer; François Jérôme; Deep eutectic solvents: syntheses, properties and applications. Chem. Soc. Rev. 2012, 41, 7108-7146, 10.1039/c2cs35178a.
- Emma L. Smith; Andrew P. Abbott; Karl S. Ryder; Deep Eutectic Solvents (DESs) and Their Applications. Chemical Reviews 2014, 114, 11060-11082, 10.1021/cr300162p.
- Dongkun Yu; Zhimin Xue; Tiancheng Mu; Eutectics: formation, properties, and applications. Chemical Society Reviews 2021, 50, 8596-8638, 10.1039/d1cs00404b.
- Benworth B. Hansen; Stephanie Spittle; Brian Chen; Derrick Poe; Yong Zhang; Jeffrey M. Klein; Alexandre Horton; Laxmi Adhikari; Tamar Zelovich; Brian W. Doherty; et al.Burcu GurkanEdward J. MaginnArthur RagauskasMark DadmunThomas A. ZawodzinskiGary A. BakerMark E. TuckermanRobert F. SavinellJoshua R. Sangoro Deep Eutectic Solvents: A Review of Fundamentals and Applications. Chemical Reviews 2020, 121, 1232-1285, 10.1021/acs.chemrev.0c00385.
- C. Vidal; L. Merz; J. García-Álvarez; Deep eutectic solvents: biorenewable reaction media for Au(i)-catalysed cycloisomerisations and one-pot tandem cycloisomerisation/Diels–Alder reactions. Green Chemistry 2015, 17, 3870-3878, 10.1039/c5gc00656b.
- Charles A. Eckert; Barbara L. Knutson; Pablo G. DeBenedetti; Supercritical fluids as solvents for chemical and materials processing. Nature 1996, 383, 313-318, 10.1038/383313a0.
- Jocelyn Peach; Julian Eastoe; Supercritical carbon dioxide: a solvent like no other. Beilstein Journal of Organic Chemistry 2014, 10, 1878-1895, 10.3762/bjoc.10.196.
- M. L. Keshtov; E. I. Mal’Tsev; A. M. Lopatin; L. N. Nikitin; D. V. Marochkin; V. P. Perevalov; P. V. Petrovskii; A. R. Khokhlov; Photoluminescent phenylated polyfluorenes synthesized in an organic solvent and supercritical carbon dioxide. Polymer Science, Series B 2012, 54, 106-114, 10.1134/s1560090412020054.
- M. L. Keshtov; E. I. Mal’Tsev; A. M. Lopatin; L. N. Nikitin; D. V. Marochkin; V. P. Perevalov; P. V. Petrovskii; A. R. Khokhlov; Photoluminescent phenylated polyfluorenes synthesized in an organic solvent and supercritical carbon dioxide. Polymer Science, Series B 2012, 54, 106-114, 10.1134/s1560090412020054.





