Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Seon-Jin Choi | + 7721 word(s) | 7721 | 2022-01-17 07:51:17 | | | |
| 2 | Vivi Li | + 155 word(s) | 7876 | 2022-01-17 10:02:48 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Choi, S. Nanomaterials for IoT Sensing Platforms and Point-of-Care Applications. Encyclopedia. Available online: https://encyclopedia.pub/entry/18327 (accessed on 10 May 2026).
Choi S. Nanomaterials for IoT Sensing Platforms and Point-of-Care Applications. Encyclopedia. Available at: https://encyclopedia.pub/entry/18327. Accessed May 10, 2026.
Choi, Seon-Jin. "Nanomaterials for IoT Sensing Platforms and Point-of-Care Applications" Encyclopedia, https://encyclopedia.pub/entry/18327 (accessed May 10, 2026).
Choi, S. (2022, January 17). Nanomaterials for IoT Sensing Platforms and Point-of-Care Applications. In Encyclopedia. https://encyclopedia.pub/entry/18327
Choi, Seon-Jin. "Nanomaterials for IoT Sensing Platforms and Point-of-Care Applications." Encyclopedia. Web. 17 January, 2022.
Copy Citation
Herein, state-of-the-art research advances in South Korea regarding the development of chemical sensing materials and fully integrated Internet of Things (IoT) sensing platforms were comprehensively reviewed for verifying the applicability of such sensing systems in point-of-care testing (POCT). Various organic/inorganic nanomaterials were synthesized and characterized to understand their fundamental chemical sensing mechanisms upon exposure to target analytes. Moreover, the applicability of nanomaterials integrated with IoT-based signal transducers for the real-time and on-site analysis of chemical species was verified.
nanostructure
IoT
POCT
gas sensor
ion sensor
biosensor
1. Introduction
The development of high-performance chemical sensors has become increasingly important in recent years because of industrialization and the need to prevent widespread viral infections. Additionally, chemical sensors for biomarker detection in the human body are gaining considerable attention for the non-invasive diagnosis of diseases and the monitoring of health conditions in real-time [1][2]. Various analytes such as gases, ions, and biocomponents (e.g., glucose, viruses, and bacteria) can be detected using chemical sensors, thereby making such sensors suitable for applications in environmental monitoring and healthcare. To detect trace amounts of analytes, various sensing parameters, particularly high sensitivity and selectivity, must be considered. Moreover, the rapid detection of target analytes is essential for preventing the spread of hazardous chemical species and detecting abnormal health states within a short time. Furthermore, the miniaturization of sensor platforms with low power consumption is necessary for portable on-site detection and point-of-care testing (POCT) [3][4][5].
To develop next-generation chemical sensors, the development of novel sensing materials and their integration with sensing systems are desired (Figure 1). For the development of chemical sensing layers, several nanomaterials with structural engineering have been proposed to facilitate their large surface area and high porosity considering that the fundamental sensing mechanism is the result of surface chemical reactions [6][7][8][9]. For example, multidimensional nanostructures such as zero-dimensional (0D) nanoparticles [10], one-dimensional (1D) nanofibers [11], two-dimensional (2D) nanosheets [12][13], and three-dimensional (3D) nanocubes have been demonstrated to be effective sensing layers with high sensitivity [14][15]. In addition, attempts have been devoted toward the tuning of physical properties such as electrical conductivity and optical emission of nanomaterials by compositional and chemical reactivity modulations [16]. These nanomaterials serve as a transducer layer on a sensing substrate to produce distinguishable signal outputs owing to the changes in their electrical and optical properties. To enhance selectivity by inducing specific binding, various selectors (e.g., synthetic molecules [17], antibodies [18], enzymes [19], and DNAs/aptamers [20]) have been functionalized with nanomaterials [21]. Chemical interactions between analytes and selectors can be effectively transduced into electrical or optical signals through the transducer layer.
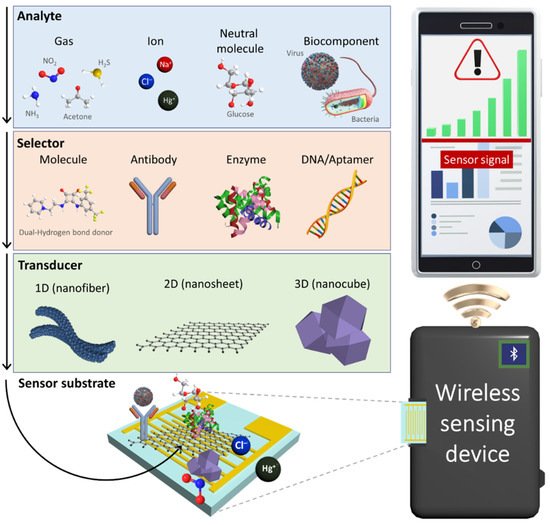
Figure 1. Schematic illustration of an IoT sensing platform comprising a chemical sensor, a wireless sensing device, and a smartphone for POCT application. Electrochemical sensors using nanomaterials consisting of selectors and transducers produce electrical signals upon chemical interactions with various analytes such gases, ions, neutral molecules, and biocomponents.
In sensing systems, sensing data are transmitted to a personal mobile device through wireless communication based on the Internet of Things (IoT) platform. Chemical sensors integrated with portable IoT devices have been employed for the real-time and on-site detection of target analytes in order to enable POCT applications [22][23]. For example, wearable sensor systems were fabricated by combining a transducer layer with a flexible substrate to quantitatively monitor chemical analytes in body fluids, and the measured sensing data were displayed on a smartphone [24][25][26][27]. The sensing systems assembled with nanomaterials can be further optimized for integration with IoT sensing platforms, depending on their applications and target analytes.
2. Gas Sensors
Nanomaterials integrated with IoT-based gas-sensing modules are gaining considerable attention for their use in monitoring hazardous environments, food freshness, and disease diagnosis [28][29]. For example, nitrogen dioxide (NO2) is a toxic gas emitted from automobiles and industrial plants that causes respiratory diseases under excess exposure [30]. In addition, highly sensitive gas sensors that detect various volatile organic compounds can be employed in diagnostic POCT applications [31]. For instance, the analysis of acetone concentration in exhaled breath can provide information about the metabolic state, such as body fat burning and diabetic symptoms [32][33].
Among the various types of gas sensors, chemiresistive gas sensors are suitable for integration with IoT sensing platforms because of their simple working principle, ease of fabrication, and low cost [34]. The performance of chemiresistive gas sensors depends on the microstructures of nanomaterials, considering that sensing signals are transduced by surface chemical reactions. To further improve the gas-sensing performance, microstructural and compositional modifications have been attempted using novel synthesis techniques [35]. In this section, we review the recent research progress in South Korea on the development of gas sensors using multidimensional nanocomposites, which can be integrated with IoT sensing platforms, and the use of such sensors in POCT applications.
1D nanostructures have been employed as gas-sensing layers owing to their large surface area and porosity, which allows for a high sensitivity [36][37][38][39]. Moreover, 1D nanomaterials with controlled structures and morphologies have been developed as gas-sensing layers, such as nanorods (NRs) [40], nanowires (NWs) [41], nanofibers (NFs) [42], and nanotubes (NTs) [43].
1D graphene fibers have become a research focus considering that fibrous structures with high mechanical strength and tensile modulus can be integrated with wearable chemical sensors for the on-site detection of gas species [44][45]. A sensor using nitrogen-doped reduced graphene oxide (nRGO) fibers functionalized with Pt nanoparticles (NPs) was developed for application in wearable humidity sensors [46]. The nRGO fiber was produced by a wet-spinning process followed by heat-treatment in a reducing ambient. Continuously aligned graphene oxide (GO) fibers were obtained by lyotropic liquid crystals (LCs) property during the wet-spinning process [47]. Specifically, a 1.8 wt% GO solution was coagulated in a CaCl2 solution, and the aligned GO sheets were precipitated into a continuous fiber structure under ejection through a syringe nozzle (Figure 2a). GO fibers with a diameter range of 50–80 μm were obtained after washing and drying. Subsequently, the GO fibers were annealed at 900 °C in a reducing atmosphere (H2/N2, 5%/95%, v/v) to form nRGO fibers, resulting in nitrogen doping and removal of oxygen functional groups [48]. The X-ray photoelectron spectroscopy (XPS) analysis revealed that the RGO fiber was reduced by the formation of C–N bond and removal of oxygen functional groups, which resulted in the improved electrical conductivity of nRGO.
To functionalize Pt NPs on the nRGO fiber, a hollow protein cage (i.e., apoferritin) was used as a template. Apoferritin is composed of peptide subunits with an inner cavity diameter of 8 nm and overall size of 13 nm [49]. The protein cage can encapsulate Pt ions inside the protein cavity, and the subsequent reduction process results in metallic Pt NPs encapsulated by protein cages. Optothermal sintering upon intense pulse light (IPL) irradiation was performed using a xenon flash lamp to remove protein templates and form Pt NPs on the nRGO fiber (Pt-nRGO) (Figure 2b).
A colorless polyimide (cPI) film was prepared as a substrate for the Pt-nRGO humidity sensor. As a precursor solution for cPI film, a polyamic acid (PAA) solution was first prepared by mixing 4,4-(hexafluoroisopropylidene)diphthalic anhydride and 3,3-diaminodiphenyl sulfone in N,N-dimethylacetamide. Next, the PAA solution was coated on a glass substrate by the screen-printing method followed by imidization at 100 °C, 200 °C, and 230 °C for 1 h at each temperature to form a cPI film. After patterning the sensing electrodes on the cPI film, the Pt-nRGO fiber was electrically connected between the two electrodes to measure resistance changes.
Humidity sensing properties were investigated by measuring the resistance transitions of Pt-nRGO fibers on a flexible cPI substrate at different relative humidity (RH) levels. The response of the sensors was calculated as ((RH–RD)/RD (%)), where RH and RD are the resistance upon exposure toward humid air (i.e., 6.1–99.9% RH) and baseline dry air (i.e., 2.6% RH), respectively. The pristine nRGO fiber exhibited responses of 0.27% at 6.1% RH and 3.53% at 66.4% RH. The Pt-nRGO fiber exhibited improved humidity responses of 0.32% at 6.1% RH and 4.51% at 66.4% RH. The improved humidity sensing properties of Pt-nRGO were mainly attributed to the uniform functionalization of Pt with a particle size of ~2 nm through catalytic water dissociation [50]. The Pt-nRGO fiber was integrated with a portable sensing module to demonstrate its applicability for the real-time and on-site detection of humidity changes under direct exposure to human exhaled breath (Figure 2c). Consistent response transitions of 0.86% were observed after repetitive injections of exhaled breath to the sensor for 2 s (Figure 2d). The stable sensing property of Pt-nRGO was mainly attributed to the favorable adsorption of water molecules owing to the presence of numerous defect sites at the domain boundary of nRGO and the catalytic effect of Pt NPs by dissociation of water molecules. Because of its unique fibrous structure and outstanding humidity sensing properties, Pt-nRGO can be applied for the detection of biomarkers in exhaled breath.
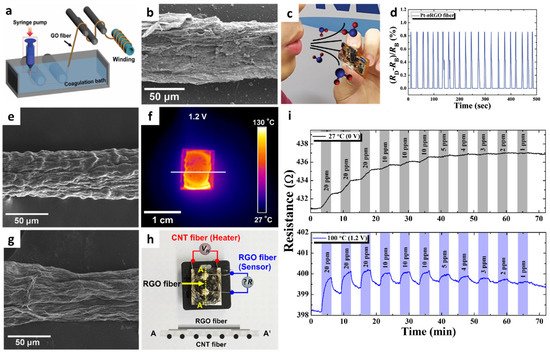
Figure 2. (a) Schematic illustration of the synthesis of a GO fiber via wet spinning. (b) SEM image of Pt-nRGO fiber after optothermal sintering upon IPL irradiation. (c) Exhaled breath injection to the Pt-nRGO fiber sensor integrated with a portable sensing module. (d) Real-time response transitions during breath humidity monitoring by using the sensing module. Reproduced with permission from Ref. [46] Copyright (2018), Wiley-VCH. (e) SEM image of the CNT fiber synthesized via wet spinning. (f) IR image of a CNT fiber-cPI film as a heater under an applied voltage of 1.2 V. (g) SEM image of an RGO fiber synthesized via wet-spinning. (h) Camera image of an all-carbon fiber-based sensor fabricated by the integration of an RGO fiber on a CNT fiber-cPI film with the schematic image of the cross-sectional structure. (i) Resistance transitions of an all-carbon fiber-based sensor at different operating temperatures (applied voltages). Reproduced with permission from Ref. [51] Copyright (2019), Elsevier.
Another promising application of 1D graphene fibers is environmental monitoring through the on-site detection of toxic gases such as NO2. However, graphene-based sensing layers suffer from incomplete recovery and drift in baseline resistance after exposure to NO2 as a result of the irreversible recovery process. To achieve reversible NO2 sensing, a flexible heating substrate was prepared by embedding carbon nanotube (CNT) fibers in a cPI film and integrating it with a graphene fiber [51]. Continuous CNT fibers were synthesized by the wet-spinning process, in which purified CNT powder dispersed in chlorosulfonic acid was ejected through a syringe nozzle in a coagulation bath. The CNT fiber exhibited a 1D structure with preferentially aligned CNTs along the axial direction (Figure 2e). A CNT fiber-embedded cPI (CNT fiber-cPI) film was fabricated by dispersing CNT fiber networks in PAA followed by the imidization process. The heating property of a CNT fiber-cPI heater was characterized by applying a voltage of 0–1.2 V to the CNT fiber-cPI film. Voltage-dependent current transitions were observed with an increase in the film temperature. The infrared image clearly shows the heating property of the CNT fiber-cPI heater with an operating temperature of 90.5 °C at an applied voltage of 1.2 V (Figure 2f).
For the sensing layer, RGO fibers were prepared by the wet-spinning process followed by thermal reduction. GO fibers were first produced by the wet-spinning process, similar to the synthesis of CNT fibers. Subsequently, heat-treatment was performed at 900 °C in a reducing atmosphere (H2/N2, 4%/96%) for 2 h to form RGO fibers. The continuous fibrous structure of RGO was maintained with RGO sheets aligned on the surface (Figure 2g). The RGO fiber was deposited on a CNT fiber-cPI film to produce an all-carbon fiber-based sensor (Figure 2h).
The sensing property of the all-carbon fiber-based sensor was investigated toward NO2 in the concentration range of 1–20 ppm under different operating temperatures controlled by the CNT fiber-cPI heater. (Figure 2i). Although the RGO fibers showed a noticeable response to 20 ppm NO2 at room temperature, the recovery was negligible, resulting in a severe drift in the sensor signal. In addition, the resistance changes of the RGO fibers were negligible at concentrations below 5 ppm at room temperature. On the other hand, further improved response and recovery properties were achieved when the operating temperature was increased to 100 °C (1.2 V). The theoretical detection limit was calculated to be 814 ppb at 100 °C, implying that the all-carbon fiber-based sensor can potentially detect NO2 at sub-ppm levels.
To quantitatively analyze the reversible NO2 reaction and recovery processes, adsorption and desorption kinetics were evaluated by calculating the reaction rate constants, i.e., the desorption rate constant (kdes) and adsorption rate constant (kads), based on the following equations [51][52][53]:
(1)
(2)
where S0 is the response when the analyte gas is removed, Smax is the maximum response toward the analyte gas, and Ca is the concentration of the analyte gas. A relatively low adsorption rate constant (kads = 2.48 × 10−2 ppm−1 s−1) was obtained at room temperature. Moreover, the negative desorption rate constant (kdes = −1.34 × 10−3 s−1) indicates negligible recovery upon exposure to air at room temperature. On the other hand, a 2.17-fold increase in response kinetics (kads = 5.37 × 10−2 ppm−1 s−1) was achieved by increasing the operating temperature to 100 °C. In particular, substantially improved recovery kinetics were achieved with a 9.22-fold enhancement in the desorption rate constant (kdes = 8.85 × 10−3 s−1) at 100 °C. This work paves the way for the development of next-generation chemical sensors using unique carbonaceous fibers as a sensing layer, as well as a heating element for the detection of toxic chemicals with improved reversibility.
The use of multi-compositional 1D structures is an effective way to improve the gas-sensing performance [54]. Incorporating heterogeneous sensing materials that combine metal oxides on a conductive carbon framework can facilitate the development of high-performance gas sensors through the activation of the physical/chemical adsorption properties of gas species [55][56]. Jang et al. fabricated porous RGO fibers functionalized with WO3 NRs by employing wet-spinning and solution-based self-assembly processes [57]. The formation of abundant pore sites on the GO fiber is advantageous for improving the gas response through the promotion of gas penetration and the acceleration of the surface reaction. Thus, tunicate cellulose nanofibers (TCNFs) were prepared by forming porous GO fibers [58]. During the wet-spinning process, TCNF and GO were wound into a fibrous structure (TCNF-GO) by exploiting the LC properties in an aqueous solution. As a result, a TCNF-GO fiber with a unique wrinkled surface morphology and well-distributed mesopores was obtained (Figure 3a). To form WO3 NRs on the porous RGO fiber (porous WO3 NRs-RGO), the solution-based self-assembly process was performed by inducing the adsorption of a tungsten precursor on a hydrophilic TCNF, resulting in the uniform distribution of the tungsten precursor on the TCNF-GO fiber [59]. After heat-treatment in an argon atmosphere at 700 °C, WO3 NRs were grown on RGO fibers with a mean width of 197 nm (Figure 3b).
Gas-sensing characterization of the WO3 NRs-RGO fiber was performed at 100 °C toward 5 ppm NO2, which revealed a high response (|Rgas–Rair|/Rair × 100 (%)) of 9.67%. In addition, notable selectivity toward NO2 was confirmed with minor responses (<2.45%) toward other interfering gases such as ethanol (C2H5OH), acetone (C3H6O), toluene (C7H8), hydrogen sulfide (H2S), and nitrogen monoxide (NO). The porous WO3 NRs-RGO fiber was integrated with a wristband-type sensing module to demonstrate its applicability in wearable sensors (Figure 3c). After the injection of 20 ppm NO2 for 10 cycles, consistent and reversible NO2 sensing properties were obtained with a response range of 2.25–2.75% at room temperature (Figure 3d). The improved NO2 sensing performance of the porous WO3 NRs-RGO fiber was mainly attributed to the heterojunction effect between the WO3 NRs and RGO fibers facilitating an effective surface reaction and the charge transduction properties [60].
Sacrificial templates can be utilized to form porous nanostructures and to transport catalytic NPs to the sensing layer. In particular, well-dispersed catalytic NPs with multiple compositions can be synthesized by encapsulating them in a sacrificial template. Kim et al. proposed a new approach to synthesize Pt-based bimetallic catalysts (PtM, M = Pd, Ru, and Ni) on mesoporous WO3 NFs by employing the encapsulating route by using apoferritin protein nanocages [61]. Highly dispersed bimetallic PtM NPs were obtained by the reduction of both Pt and metal (Pd, Ru, Ni) ions in the apoferritin hollow nanocage (PtM-apo), resulting in average particle size of less than 3 nm. The high-resolution TEM image of PtPd NPs showed an interplanar distance of 2.21 Å, implying the formation of an intermetallic PtPd compound (Figure 3e). Elemental distribution of PtM NPs was confirmed by energy-dispersive X-ray spectroscopy (EDS) mapping analysis; overlapping images for both Pt and Pd elements were observed for PtPd-apo (Figure 3f). On the other hand, PtNi-apo showed a scattered elemental distribution as a result of the difference in the reduction rate between Pt and Ni, leading to the increased size of PtNi-apo compared to that of PtPd-apo (Figure 3g).
The electrospinning process was performed to form WO3 NFs functionalized with PtM NPs (PtM-WO3 NFs) (Figure 3h). An electrospinning solution was prepared by dissolving tungsten precursor ((NH4)6H2W12O40·xH2O) and polyvinylpyrrolidone (PVP) in deionized (DI) water followed by the homogeneous dispersion of PtM-apo NPs to obtain the nanofibrous composite 1D structure of W precursor/PVP/PtM-apo. After heat-treatment at 600 °C, porous WO3 NFs functionalized with catalytic PtM NPs were obtained as a result of the decomposition of protein shells and the transfer of PtM NPs onto WO3 NFs.
Gas-sensing properties of the pristine WO3 NFs, Pt-WO3 NFs, and PtPd-WO3 NFs toward acetone were evaluated in the concentration range of 1–5 ppm at 300 °C. The gas response of PtPd-WO3 NFs (Rair/Rgas = 97.5) was substantially improved as compared to those of the pristine WO3 NFs (Rair/Rgas = 4.3) and Pt-WO3 NFs (Rair/Rgas = 24.9) toward 1 ppm acetone at 300 °C (Figure 3i). In addition, the PtNi-WO3 NFs exhibited a drastically improved gas response of 340 toward 1 ppm H2S at 300 °C. The drastic improvement in the H2S sensing performance was mainly attributed to the dual catalytic effect of PtNi-WO3 NFs induced by the phase separation between Pt and NiO after the calcination process. The PtPd-WO, PtRu-WO, and PtNi-WO3 NFs were combined as a sensor array to analyze cross-selectivity and exhaled breath patterns. Simulated exhaled breath composed of healthy human breath containing 1 ppm of acetone and H2S was exposed to a sensor array to demonstrate its applicability for the diagnosis of diabetes and halitosis, respectively. As a result, separated patterns depending on the exhaled breath composition were obtained by principal component analysis (PCA), which verified the applicability of PtM-WO3 NFs for non-invasive diagnosis through the detection of exhaled breath biomarkers (Figure 3j).
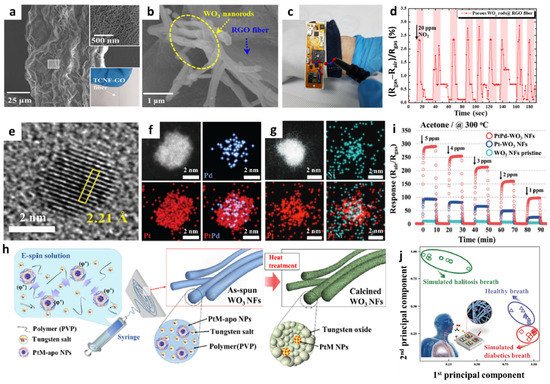
Figure 3. SEM images of (a) TCNF-GO and (b) porous WO3 NRs-RGO fibers. (c) Digital image of a portable sensing module loaded with porous WO3 NRs-RGO fibers. (d) Real-time NO2 sensing property using a portable sensing module. Reproduced with permission from Ref. [57] Copyright (2019), American Chemical Society. (e) High-resolution TEM image of PtPd-apo NPs. EDS elemental mapping images of (f) PtPd-apo NPs and (g) PtNi-apo NPs. (h) Schematic illustration of the electrospinning process for the synthesis of mesoporous WO3 NFs functionalized with PtM NPs by the apoferritin-encapsulating method. (i) Sensing properties of pristine WO3 NFs, Pt-WO3 NFs, and PtPd-WO3 NFs toward acetone in the concentration range of 1–5 ppm at 300 °C. (j) Pattern recognition of exhaled breath using sensor arrays, demonstrating their applicability for non-invasive POCT for diabetes and halitosis diagnosis. Reproduced with permission from Ref. [61] Copyright (2017), Wiley-VCH.
The formation of porous and hollow structures can offer high gas permeability and accelerate the surface reaction [62][63][64]. A new synthesis strategy was proposed by aligning 1D nanostructures on a flexible substrate [65]. Hollow nanowires (HNWs) of Pd–Ag composite were synthesized by lithographically patterned nanowire electrodeposition (LPNE) and the subsequent galvanic replacement reaction (GRR) on a flexible cPI film for a reversible hydrogen (H2) reaction. The aligned Ag NWs were formed on the cPI film attached to a glass substrate by using the LPNE method (Figure 4a). Specifically, the Ni film was deposited by thermal evaporation as an etch mask during the patterning process followed by spin-coating of the photoresist to form a line pattern. Next, the sample was immersed in an etchant solution to form a line pattern of Ni by undercut etching below the photoresist while forming trenches. Electrodeposition was performed to form Ag NWs on the surface of Ni using the trenches as a template followed by the removal of the photoresist and Ni. To induce the transformation of Ag NWs to Pd-functionalized Ag HNWs (Pd@Ag HNWs), the GRR was performed by immersing the electrodeposited Ag NWs into the aqueous Pd precursor for a certain duration. SEM analysis revealed the optimum GRR time, wherein well-aligned Pd@Ag HNWs were obtained after 17 h (Figure 4b). A porous structure was formed on the surface of the Pd@Ag HNWs during the GRR (Figure 4c). In addition, the Pd@Ag HNWs with the continuous hemitubular structure were obtained owing to the standard reduction potential difference between Pd and Ag, resulting in the dissolution of Ag NWs and the subsequent growth of metallic Pd (Figure 4d). XPS revealed the formation of the Pd–Ag alloy during the replacement reaction.
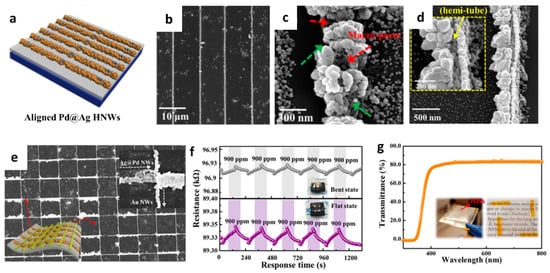
Figure 4. (a) Schematic illustration of Pd@Ag HNWs on a cPI film attached to a glass substrate. SEM images of the (b,c) Pd@Ag HNWs after the GRR for 17 h, (d) overturned Pd@Ag HNWs with a magnified image in the inset, and (e) grid-type NWs composed of Pd@Ag HNWs and Au NWs. (f) Resistance transitions of the heterogeneous Pd@Ag HNWs on a flexible cPI substrate toward 900 ppm H2 in flat and bent states. (g) The transmittance of the heterogeneous NWs composed of Pd@Ag HNWs and Au NWs. Reproduced with permission from Ref. [65] Copyright (2017), American Chemical Society.
3. Ion Sensors
The development of innovative ion sensor systems, including sensing materials, sensor substrates, and signal transduction techniques, enables real-time analysis through the rapid detection of analyte species, minimization of sensing platforms, and quantitative analysis of ion concentrations [66]. Advanced ion sensors can be integrated with IoT devices for developing portable and wearable sensing platforms [67]. Particularly, wearable sensing platforms have been developed for the analysis of biofluids, including sweat, considering their major advantages such as high efficiency for non-invasive healthcare monitoring and POCT [68][69].
Anion detection is gaining considerable attention in various fields, including healthcare, environmental monitoring, and biotechnology. For example, acetate (AcO−) is a metabolic switch that controls the rate of bacterial cell growth. In an abundant nutrient environment, bacterial cells such as Escherichia coli (E. coli) grow rapidly and excrete AcO− [70][71][72]. The bacterial cells switch to a slower growth rate when their nutrients are depleted in the environment to enhance survival. The accumulation of AcO− can inhibit cell growth and lower the productivity of recombinant proteins (e.g., synthetic insulin). Moreover, chloride (Cl−) in sweat is an important biomarker for the diagnosis of cystic fibrosis [73]. Increased Cl− concentration in the range of 60–150 mM in sweat is generally observed in cystic fibrosis patients, whereas the normal Cl− concentration range of a healthy individual is 10–40 mM [74].
Various receptors have been synthesized to detect anions, and their binding affinities toward specific anions have been evaluated. There has been particular interest in the design of receptor structures using dual-hydrogen bond donors such as urea, thiourea, deltamide, squaramide, and croconamide, considering their geometrical uniqueness for the binding of halides and Y-shaped oxoanions forming stable six- and eight-membered chelated structures, respectively [75][76]. Improvement in the anion-binding affinities of dual-hydrogen bond donors has been achieved by modulating N,N-substitutional functional groups. The anion-binding behavior of receptors comprising dual-hydrogen bond donors was characterized either by hydrogen bond interactions or deprotonation upon the injection of anions, depending on the acidity of the receptors.
Various signal transduction techniques have been employed to understand the anion-binding behavior, such as those utilizing optical, magnetic, and electrochemical signals. Among the electrochemical signal transduction techniques, chemiresistive-type anion sensors have attracted considerable attention because of their ability for the real-time detection of anions with rapid screening and potential for integration with wireless sensing modules (Table 1).
Recently, multiplexed chemiresistive anion sensors have been developed for the detection of AcO− facilitating deprotonation of dual-hydrogen bond donors and electrical transduction using single-walled carbon nanotubes (SWCNTs) [77]. To fabricate the anion sensor, poly(4-vinylpyridine) (P4VP)-wrapped SWCNTs (P4VP-SWCNT) were patterned by spray-coating method, followed by the non-covalent functionalization of selectors composed of squaramide-based dual-hydrogen bond donors (Figure 5a). Specifically, a homogeneous dispersion of SWCNTs was prepared in N,N-Dimethylformamide (DMF) by dissolving P4VP and wrapping the SWCNTs. To prepare the sensing substrate, parallel Au electrodes were patterned on a glass substrate by depositing Au/Cr layers using a thermal evaporator. Subsequently, the glass substrate was treated with 3-bromopropyltrichlorosilane to form bromo alkyl chains on the surface. The mechanically stable P4VP-SWCNT composite was anchored on a surface-treated glass substrate by a quaternization reaction, wherein SWCNT-wrapped P4VP was covalently linked to the surface by the reaction between the pyridyl groups of P4VP and the bromo alkyl chains on the glass substrate. To induce selective anion-binding interactions, squaramide-based selectors were functionalized on the P4VP-SWCNT with different electron-withdrawing 3,5-bis(trifluoromethyl)benzyl (1) and 3,5-bis(trifluoromethyl)phenyl (2) groups (Figure 5a). The model structures of (1) and (2) were synthesized by N,N-substitution of squaramide with cationic moieties (e.g., pyridinium) and electron-withdrawing groups to systemically investigate the binding affinities toward various anions such as AcO−, Cl−, bromide (Br−), and nitrate (NO3−).
The anion-binding properties of (1) and (2) were evaluated by UV-vis titrations upon the addition of AcO− in dimethyl sulfoxide (Figure 5b–c). For the model selector (1), minor shifts in the absorption band at 292 nm were observed upon the addition of AcO− up to 1 equivalent. A binding stoichiometry of 1:1 was confirmed by the Job curve, implying a hydrogen bond interaction between AcO− and (1). For (2), an increased absorption band at 386 nm and decreased absorption bands at 280, 325, and 343 nm were observed upon the addition of up to 1 equivalent AcO−. The characteristic absorption spectra of (2) indicate the occurrence of hydrogen bond interactions between AcO− and (2) with 1:1 binding stoichiometry at a low concentration (<1 equivalent) followed by the deprotonation of (2), resulting in the formation of a hydrogen-bond self-complex ((H(AcO)2)−) with 1:2 binding stoichiometry. The UV-vis titrations of (1) and (2) exhibited minor changes toward Cl−, Br−, and NO3−, which confirmed the occurrence of weak hydrogen bond interactions.
The anion-sensing properties of functional P4VP-SWCNTs with different electron-withdrawing groups, i.e., P4VP-(1)-SWCNT and P4VP-(2)-SWCNT, were evaluated for various anions such as AcO−, Cl−, Br−, and NO3−. A baseline solution of 10 μL acetonitrile was injected to establish the baseline resistance before the addition of the analyte solution. After stabilizing the sensor resistance, a 2 μL solution containing the target anion was injected to measure the resistance transitions. The sensor response was defined as the normalized resistance, i.e., (R–R0)/R0 (%), where R and R0 are the resistances upon the addition of the analyte solution and baseline solvent, respectively. The results revealed that P4VP-(1)-SWCNT exhibited a response of 7.34% toward AcO− at 16.7 mM, followed by Cl− > Br− > NO3−. An approximately 16-fold higher response was achieved using P4VP-(2)-SWCNT with a response of 120.27% upon the addition of 16.7 mM AcO−. In terms of the selectivity of P4VP-(2)-SWCNT, the highest response was obtained with AcO− followed by Br− > Cl− > NO3−.
Real-time wireless anion sensing was demonstrated using P4VP-(2)-SWCNT by integrating an anion sensor with a wireless sensor module (Figure 5d). The resistance changes were measured using the sensing module, and the sensing data were transmitted to a smartphone through near-field communication (NFC). Increasing response transitions were observed by increasing the AcO− concentrations in the range of 0.17–83.33 mM (Figure 5e). The detection limit was 0.17 mM with a response of 12.39%. The resistance transitions upon the addition of AcO− were mainly attributed to the internal charge transfer of (2) induced by the deprotonation of the squaramide. Increasing resistance transitions resulted from the negatively charged selector (2) after deprotonation, which traps hole carriers in the SWCNTs. The chemiresistive-sensing platform enables the real-time wireless anion detection of AcO− by integration with an IoT sensing module.
Chemiresistive ion sensors using SWCNTs with different functional components can be employed for the detection of different ionic species such as proton (H+). The development of pH sensors by monitoring H+ concentrations is important for applications in healthcare systems and for water quality monitoring. For example, the normal pH of the sweat of a healthy person is 4.0–6.8, whereas an increased pH level of >9 is observed in patients with cystic fibrosis [78][79]. Additionally, pH values can indicate exercise intensity and dehydration levels [80]. In this regard, the pH level of individuals is closely related to their health conditions. In terms of monitoring pH in the environment, lowering the pH levels in seawater results in ocean acidification, threatening marine organisms that use calcium carbonate for their structural components [81]. Therefore, simple and portable pH measurement systems with wireless data transmission modules must be developed for the continuous monitoring of health and environmental conditions.
A wireless pH sensing system was demonstrated by facilitating a screen-printed SWCNT-Nafion nanocomposite on a flexible PI film [82]. The SWCNT was mixed with Nafion-117 at a concentration of 5%, which is the optimal condition for the screen-printing process. Nafion is composed of a hydrophobic backbone and hydrophilic side chains with sulfonic acid moieties, resulting in high proton conductivity (Figure 5f) [83][84]. The SWCNT-Nafion nanocomposite with a thickness of 40 μm was screen-printed on the PI substrate followed by heat-treatment at 100 °C for 10 min in the air (Figure 5g). A polydimethylsiloxane (PDMS) layer was attached to the top of the SWCNT-Nafion nanocomposite to protect the electrical contacts from pH buffer solutions.
The material properties and wireless pH sensing performance of the SWCNT-Nafion nanocomposite films were investigated. From the XPS spectra, two major peaks at 283.9 and 290.9 eV were assigned to the carbon–carbon interactions from C–C sp2 and the carbon–fluorine interactions from CF2, respectively (Figure 5h). These peaks are attributed to the binding of the Nafion chains with the SWCNTs. The surface morphology of the SWCNT-Nafion nanocomposite film was investigated by SEM (Figure 5i). Dense SWCNT networks were formed after coating by the screen-printing method with multiple electrical conducting paths. Real-time wireless pH sensing in a river was demonstrated by integrating the SWCNT-Nafion sensor with a drone (Figure 5j). The resistance transition was measured by immersing the SWCNT-Nafion sensor in a river and indicated a pH value of 7.5 (Figure 5k). The measured data were transmitted to a smartphone via wireless communication, enabling water quality monitoring from remote locations. The sensing mechanism revealed that the formation of negatively charged OH− interacts with C–H bonds from the SWCNT-Nafion nanocomposite, wherein the majority of hole carriers in the SWCNTs are immobilized in a basic environment, leading to an increase in resistivity. On the other hand, H+ produced in an acidic environment binds to C–O bonds in the SWCNT-Nafion composite, which results in decreased resistivity because hole carriers are donated back to the SWCNTs.
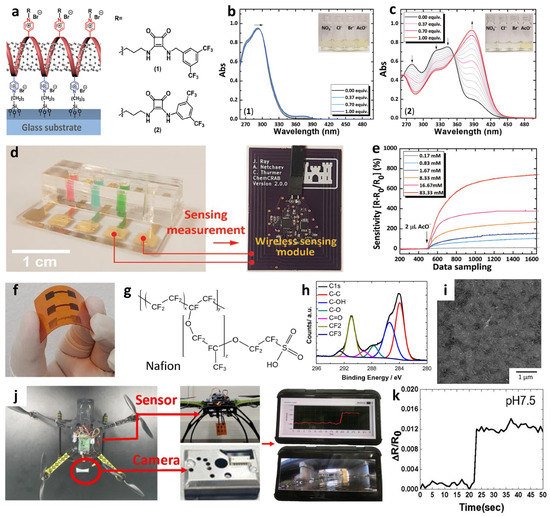
Figure 5. (a) Schematic illustration of an anion sensor with surface-anchored P4VP-SWCNT composite and anion selectors (1) and (2). UV-vis titrations of selector (b) (1) ([(1)] = 4.4 × 10−5 M) and (c) (2) ([(2)] = 4.5 × 10−5 M) upon the addition of up to 1 equivalent AcO−. (d) IoT-based anion-sensing platform composed of a sensor array and a wireless sensing module. (e) Real-time wireless detection of AcO− in the concentration range of 0.17–83.33 mM. Reprinted with permission from Ref. [77] Copyright (2019), Wiley-VCH. (f) Chemical structure of a Nafion. (g) Camera image of screen-printed SWCNT-Nafion composite on a PI substrate. (h) XPS spectra and (i) SEM image of the SWCNT-Nafion composite film. (j) Real-time wireless pH sensing system by integrating the SWCNT-Nafion film sensor with a drone and transmitting the sensing data to a smartphone. (k) Real-time wireless pH monitoring of a river. Reproduced with permission from Ref. [82] Copyright (2019), Elsevier.
Table 1. Recent development of chemiresistive ion sensors for IoT applications.
| Material | Response Definition | Response | Detection Limit | Testing Ambient | Target Ions | Applications | Ref. |
|---|---|---|---|---|---|---|---|
| SWCNT-P4VP-Squaramide | ΔR/R0 (%) | 120.27% @ 16.7 mM | 1.7 mM | Acetonitrile | CH3COO− | IoT sensor | [77] |
| SWCNT-Nafion | ΔR/R0 | ~0.2 @ pH 12 | - | H2O | H+ | IoT sensor | [82] |
| SWCNT-P4VP-Thiourea | ΔR/R0 (%) | 101.9% @ 16.7 mM | 0.17 mM | Acetonitrile | CH3COO− | - | [85] |
| SWCNT-P4VP-Croconamide | ΔR/R0 (%) | 140.91% @ 83.33 mM | 0.17 mM | Acetonitrile | CH3COO− | - | [86] |
As a different type of electrochemical ion sensor, potentiometric sensors that facilitate potential differences across selective ion-sensing membranes have been developed for the detection of cationic species integrated with a wireless sensing module for POCT applications. Potentiometric sensors are advantageous because of the simplicity of operation, low power consumption, and their potential for miniaturization [87][88][89]. Recently, wearable-type potentiometric ion sensors (WPISs) integrated with IoT sensing modules have gained significant attention for their applicability toward the real-time monitoring of ion concentration changes in body fluids for healthcare management, sports performance monitoring, and physiological analysis [90][91]. Additionally, they can be employed for the diagnosis of critical nervous disorders and heart failure by monitoring metabolic indicators such as Na+, K+, and pH [92][93]. The recent development of WPISs in South Korea is summarized in Table 2 for the detection of Na+, K+, and H+.
Table 2. Recent developments of potentiometric ion sensors for IoT applications.
| Material | Response | Dynamic Range | Testing Ambient | Target Ions | Response/Recovery Time | Applications | Ref. |
|---|---|---|---|---|---|---|---|
| PCSC-coated CFT | 60.7 ± 1.5 mV log[Na+]−1 54.8 ± 0.6 mV log[K+]−1 |
10−1–10−4 M | Body fluid (sweat) | Na+, K+ | 10–20 s | IoT wearable sensor | [94] |
| PCSC-coated CFT | 58.28 mV/pH | pH 3.89–10.09 | Body fluid (sweat) | H+ | 5 s | IoT wearable sensor | [95] |
| WO3 NFs/ CI-TPES |
−377.5 mV/pH (With differential amp.) |
pH 6.90–8.94 | Artificial seawater | H+ | - | Ocean acidification monitoring | [96] |
| Defect-free exfoliated graphene | 54.0 mV log [Na+]−1 | 10−1–10−4 M | Body fluid (sweat) | Na+ | 3.6 s | IoT wearable sensor | [97] |
| MWCNTs– MXene (Ti3C2TX) |
63 mV log [K+]−1 | 1–32 mM | Body fluid (sweat) | K+ | 2 s | IoT wearable sensor | [98] |
The development of a mechanically robust potentiometric wearable sensing platform is important, considering that the sensor is subjected to constant mechanical stress during human natural activities such as walking, running, and stretching [99][100][101][102]. To address this issue, fibrous wearable sensors with self-healing polymers (SHPs) have been proposed for the autonomous repair of damage [103][104][105][106][107]. Yoon et al. developed a wearable sweat sensor for the detection of Na+/K+ using SHPs, i.e., poly (1,4-cyclohexanedimethanol succinate-co-citrate) (PCSC)-coated carbon fiber thread (CFT) electrodes [94]. The self-healing polymer can be restored with >97.0% healing efficiency within 30 s at room temperature (Figure 6a).
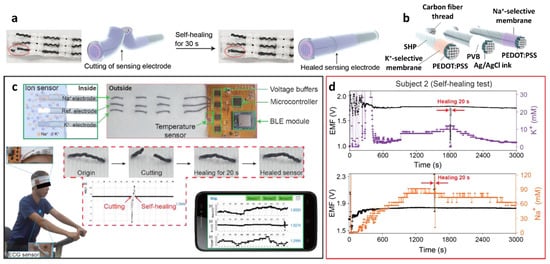
Figure 6. (a) Schematic illustrations and photographic images of the self-healing process of PCSC-coated CFT electrodes. (b) Schematic illustration of the PCSC-coated K+ ISE, reference electrode, and Na+ ISE. (c) Application of the self-healing ISE to the flexible ion-sensing platform, communicating with a wireless sensing device. Data acquisition from a headband-shaped sensor. (d) Simultaneous data acquisition of Na+ and K+ from sweat using the sensor platform during exercise. Cutting and healing processes occurred during signal collection. Reproduced with permission from Ref. [94] Copyright (2019), American Chemical Society.
4. Biosensors
Innovations in the development of biosensors integrated with biological elements and signal transducers enable the development of a new generation of sensor systems for POCT with rapid and precise detection of biological signals. Flexible and wearable sensor systems integrated with IoT sensing platforms have been developed to detect various biomolecules in the human body, such as glucose, lactate, uric acid, and bacteria (e.g., pathogenic Escherichia coli) [108][109][110]. In particular, novel glucose sensors are gaining considerable attention worldwide because of their applicability in the non-invasive diagnosis of diabetes mellitus through continuous glucose monitoring (CGM) [111][112][113]. Early diagnosis of diabetes by monitoring glucose levels is of significant importance, considering that the total global diabetes population is expected to increase by over 50% in 2045 compared to the estimated number of diabetes patients worldwide in 2017 [114]. Patients with diabetes experience uncontrolled blood glucose levels as a result of chronic hyperglycemia, causing various diabetic complications such as blindness, nerve damage, cardiovascular disease, and kidney failure [115]. Therefore, adequate medical treatment and prevention of diabetes should be achieved by continuous real-time monitoring of blood glucose levels.
Electrochemical glucose sensors have been widely utilized by facilitating enzymatic reactions for CGM [112]. In particular, the glucose oxidase (GOx) enzyme has been commonly employed owing to several advantages such as high specificity toward glucose, stability over various pH levels, and temperature changes [116]. The basic principle of glucose sensors using GOx is based on the oxidation of glucose via an enzymatic reaction that produces gluconic acid and hydrogen peroxide (H2O2) in the presence of oxygen, as shown in the following reaction [109]:
Glucose + H2O + O2 −→−− GOx Gluconic acid + H2O2
Toward the development of enzymatic glucose sensors using GOx, three generations have been established depending on the mechanism of charge transfer to the sensing electrode [109][111]. The first generation of glucose sensors indicates the amount of glucose oxidation as a result of an enzymatic reaction, which is monitored by measuring either oxygen consumption or H2O2 production. The first-generation glucose sensors exhibit major advantages such as simplicity and potential for miniaturization; thus, they can be applied for in vitro and in vivo clinical trials [117]. However, a high overpotential for the detection of H2O2 causes side reactions of electroactive species, resulting in low selectivity toward the target analyte. The second generation of glucose sensors involves the use of redox mediators with GOx, wherein the mediators interact directly with enzymes and an electrical current signal is generated upon the addition of glucose as a result of the redox reaction of the mediator. For the third generation of GOx-based glucose sensors, electron transfer occurs by direct interaction between the enzyme and the electrode without incorporating mediators. Generally, engineered enzymes are utilized to combine the electrode and GOx through structural modification, resulting in direct electron exchange. For example, GOx enzymes are coupled with porous polymeric membrane electrodes or nanostructured carbon nanotube electrodes to facilitate electron transfer [118][119][120][121].
Kang et al. demonstrated a wearable glucose-sensing system using GOx-Nafion-composite-functionalized SWCNTs, which can be categorized as a third-generation GOx-based glucose sensor [122]. The multilayered structure of GOx-Nafion-composite-functionalized SWCNTs on a flexible substrate was achieved by an all-solution process (Figure 7a). Specifically, a thin layer of PI with a thickness of 30 μm was coated on a Si wafer as a substrate, followed by the deposition of 1 μm of poly(methyl methacrylate) (PMMA) by spin-coating. Subsequently, the substrate was immersed in a 3-(aminopropyl)triethoxysilane (APTES) solution to form amine groups on the surface. A dispersion of SWCNTs (length ranging from 100 nm to 4 μm; diameter of 1.2–1.7 nm) in 1,2-dichlorobenzene (1 mg/100 mL) was deposited through spray-coating onto the APTES-modified PMMA/PI/Si substrate having a thickness of 3–7 nm followed by annealing at 150 °C for 30 min. Thus, dense SWCNT networks were formed as a result of Coulombic interactions between the SWCNTs and the amine groups from the APTES layer. Finally, a composite solution of GOx and Nafion-117 was covered on the SWCNT networks through spin-coating. The composite layers were detached from the Si substrate resulting in a flexible and wearable glucose sensor, which can be directly attached to the human skin to monitor glucose concentration using a smartphone in real-time (Figure 7a,b). A wearable glucose sensor system was established by integrating a small glucose sensor (1 cm × 1 cm in dimension) and an armband-type sensing module to transmit the sensing signal to a smartphone (Figure 7c,d).
Material characterization and glucose-sensing performance of the fabricated sensor were investigated (Figure 7e–g). The XPS survey analysis confirmed the surface functionalization of the SWCNT networks with the GOx-Nafion composites, wherein peaks related to fluoride, oxygen, and sulfur were observed as a result of surface functionalization (Figure 7e). On the other hand, the XPS survey spectrum of pristine SWCNTs exhibited no relevant peaks of GOx-nafion composites. Real-time wireless glucose-sensing properties of the wearable SWCNT-based glucose sensor systems were evaluated by monitoring the response transitions defined by A/A0, where A0 and A are the initial current before exposure to glucose and measured current after the injection of glucose, respectively. The results revealed that there is a sudden increase in the current of the SWCNTs functionalized with GOx-Nafion composites upon exposure to 50 μM glucose, whereas there were no changes in the current signal from the pristine SWCNTs (Figure 7f). The current response transitions upon successive injection of glucose were investigated in the range of 50 μM–1 mM (Figure 7g). Increasing current responses for the SWCNT-based glucose sensor functionalized with the GOx-Nafion composite were observed with continuously increasing glucose concentrations. The glucose-sensing mechanism is based on the conductance of SWCNT networks affected by the enzymatic oxidation of glucose by GOx. The fundamental principle of glucose oxidation can be explained by the formation of oxidized flavin adenine dinucleotide (FAD) as a sub-unit of the GOx enzyme from the reduced form of FAD (i.e., FADH2), while catalytically oxidizing glucose [123]. The increasing current upon the injection of glucose is mainly attributed to the direct electron transfer to the SWCNT networks during the oxidation of FAD [124].
Various flexible biosensor platforms for the detection of biological analytes have been developed on flexible substrates and applied for the point-of-care (POC) diagnosis [125][126]. For example, a flexible biosensor composed of a multilayered GOx/gold/MoS2/gold nanofilm on a PI substrate was demonstrated to be applicable for glucose detection [126]. The multilayer structure was fabricated by sputtering gold on the PI film and subsequently depositing MoS2 NPs through the spin-coating method. The gold sputtering process was performed again to form a gold/MoS2/gold nanofilm on a PI substrate with the dimension of 2.5 × 20 mm. To induce glucose-sensing properties, GOx was immobilized on a gold surface assisted by a chemical linker. The amperometric glucose-sensing result of the GOx/gold/MoS2/gold nanofilm revealed that a rapid increase in the current signal was obtained upon the addition of glucose with a limit of detection of 10 nM. The improved glucose-sensing response was mainly attributed to efficient electron transfer by the MoS2 NPs during catalytic glucose oxidation.
The development of a biosensing platform composed of a paper substrate is advantageous considering its major advantages such as simplicity of fabrication, low cost, and large-scale production of sensor devices [127]. Flexible biosensors with scalable and cost-effective strategies have been demonstrated using a disposable paper substrate. For example, a waste newspaper was employed as a sensor substrate for the detection of pathogenic Escherichia coli O157:H7 (E. coli O157:H7) using an electrochemical measurement technique [125]. The disposable paper was coated with parylene C (P-paper) to enhance its mechanical properties and increase its hydrophobicity while maintaining its porous nature [128]. After patterning the sensing electrodes on the P-paper, a self-assembled capture probe monolayer, i.e., single-strand probe DNA (ssDNA), was immobilized on a sensing electrode, followed by the formation of a 6-mercapto-1-hexanol (MCH) monolayer to block nonspecific binding to the bare gold electrode. Subsequently, a hybridization reaction was conducted by injecting synthetic cDNA or denatured amplicons of E. coli O157:H7 as a model foodborne pathogen (Figure 7h). CV and EIS were performed to investigate the step-by-step assembly process and the target cDNA detection capability. The CV characteristics of the aqueous 5 mM Fe(CN)63−/4− electrolyte solution revealed that the peak current was significantly decreased with an increase in peak-to-peak separation (ΔEP) from 110 mV for the bare Au electrode to 310 mV after the immobilization of the ssDNA probe and blocking with the MCH monolayer (Figure 7i). Further decreased peak current and increased ΔEP (330 mV) were achieved after hybridization with the target cDNA. EIS further confirmed the cDNA detection capability at different concentrations using P-paper-based sensors in the presence of Fe(CN)63−/4− as an indicator (Figure 7j). Nyquist plots revealed gradually increased charge transfer resistance (Rct) upon increasing the target cDNA concentration. This result indicates binding between the ssDNA probe and cDNA, which results in a negatively charged surface leading to the attenuation of electron transfer.
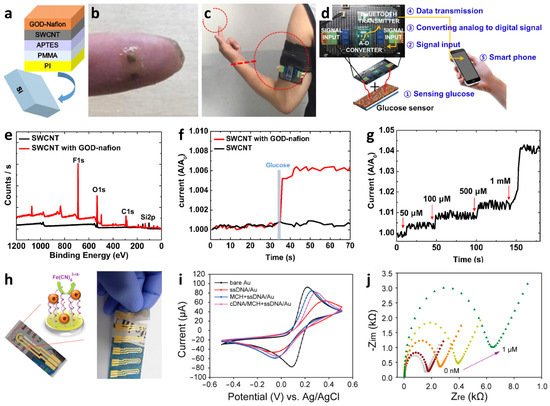
Figure 7. (a) Schematic illustration of a wearable SWCNT-based glucose sensor fabricated by an all-solution process. Camera images of (b) a wearable SWCNT-based glucose sensor on a finger and (c) integrated with a wearable sensing module. (d) Wearable glucose sensor system integrated with an IoT-based sensing module to transmit glucose-sensing data to a mobile device. (e) XPS spectra analysis to confirm the functionalization of the GOx-Nafion composite on an SWCNT film. (f) Real-time glucose-sensing property of pristine SWCNT and GOx-Nafion-composite-functionalized SWCNT upon exposure to 50 μM of glucose. (g) Real-time response changes of GOx-Nafion-composite-functionalized SWCNT upon successive addition of glucose ranging from 50 μM to 1 mM. Reprinted with permission from Ref. [122] Copyright (2019), Elsevier. (h) Disposable-paper-based electrochemical sensors after coating parylene C (P-paper) and electrodes for the detection of foodborne pathogens (i.e., cDNA of E. coli O157:H7). (i) CV curve to investigate the step-by-step assembly process of ssDNA probe immobilization, MCH blocking monolayer formation, and cDNA hybridization. (j) Nyquist plots of the paper-based sensor upon exposure to different concentrations of target cDNA. Reprinted with permission from Ref. [125] Copyright (2016), American Chemical Society.
As a different type of transduction mechanism, colorimetric sensing mechanism has been utilized to detect biomolecules and viruses because of their simple visual readout and their capability to rapidly screen multiple analytes with high portability [129][130][131][132][133][134]. Several studies have been conducted to fabricate colorimetric biosensors using a paper substrate paired with a smartphone-based reader for application in POCT [135][136][137][138][139]. A paper/soluble polymer hybrid-based biosensing platform was developed for the diagnosis of myocardial infarction by detecting human cardiac troponin I (cTnI) as a standard biomarker [135]. Among the various biosensing platforms for POCT applications, lateral flow assays (LFAs) are the most widely used because of their major advantages such as affordability, simplified device architecture, user-friendliness, ability for rapid detection, robustness, and long shelf life (~2 years) under ambient conditions [139][140][141][142]. However, because of the relatively low sensitivity of conventional LFAs, they cannot be effectively applied for biomarker detection in the concentration as low as sub-ng/mL. To overcome this limitation, a paper-based LFA with signal amplification (i.e., signal-amplification-based LFA) was proposed to facilitate biochemical reactions to further enhance sensitivity and promote quantitative analysis [143][144]. To this end, low-cost and mass-produced batch-type test strips were prepared to analyze cTnI, incorporated with a smartphone-based reader for high-performance POCT. The paper/polyvinyl alcohol (PVA) hybrid was patterned by dispensing the PVA solution on nitrocellulose (NC) membrane, which plays a key role in programmable fluid control and automated fluid switching (Figure 8a). The test proceeds with an assay followed by a signal readout using a smartphone. The assay was performed by the injection of a sample solution containing a cTnI biomarker to induce immunoreaction and a reagent solution to activate the amplification reaction within 20 min (Figure 8b). The mixture fluid injected through a reagent pad gradually dissolved the patterned PVA barrier, resulting in fluid switching from the sample fluid to the amplification fluid (Figure 8c). As a result, the test platform realized automated signal amplification reactions at the test and control lines. Various amplification techniques have been demonstrated such as Au-ion amplification, wherein gold ions (Au3+) were reduced to Au NPs in the presence of a reducing agent (H3NO), thereby generating amplified colorimetric signal changes. The enhanced colorimetric signal was proportional to the amount of reduced Au NPs, which were formed after the immunocomplex reaction. The intensity of color changes was measured using a smartphone reader after 20 min of the assay (Figure 8d). The result revealed excellent analytical sensitivity with a detection limit of 0.92 pg/mL cTnI and a coefficient of variation of <10% in serum or plasma samples comparable to those of commercially available standard analyzers, thereby demonstrating its potential application in POCT systems.
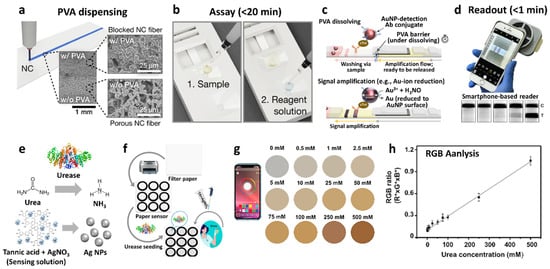
Figure 8. (a) Dispensing of PVA on a nitrocellulose (NC) membrane for the LFA test platform with SEM images. (b) LFA test process by the injection of sample and reagent solutions. (c) Schematic illustration of colorimetric Au-ion amplification facilitating automated reaction/fluid switching mechanism by dissolving the PVA barrier. (d) Colorimetric signal readout using a smartphone. Reprinted with permission from Ref. [135] Copyright (2020), American Chemical Society. (e) Schematic illustration of the mechanism of the colorimetric urea biosensors, generation of NH3 by an enzymatic reaction between urea and urease, and pH-responsive reduction of Ag+ ions to Ag NPs by tannic acid. (f) Schematic illustration of the fabrication of a colorimetric urea biosensor. (g) Analysis of colorimetric urea biosensor based on the calculated RGB ratio at different urea concentrations. (h) Smartphone-assisted RGB ratio measurement in the urea concentration range of 0–500 mM. Reprinted with permission from Ref. [136] Copyright (2021), Elsevier.
References
- Lim, J.W.; Kim, T.-Y.; Woo, M.-A. Trends in sensor development toward next-generation point-of-care testing for mercury. Biosens. Bioelectron. 2021, 183, 113228.
- Yoon, J.-W.; Lee, J.-H. Toward breath analysis on a chip for disease diagnosis using semiconductor-based chemiresistors: Recent progress and future perspectives. Lab Chip 2017, 17, 3537–3557.
- Sempionatto, J.R.; Jeerapan, I.; Krishnan, S.; Wang, J. Wearable chemical sensors: Emerging systems for on-body analytical chemistry. Anal. Chem. 2019, 92, 378–396.
- Justino, C.I.; Freitas, A.C.; Pereira, R.; Duarte, A.C.; Santos, T.A.R. Recent developments in recognition elements for chemical sensors and biosensors. TrAC Trends Anal. Chem. 2015, 68, 2–17.
- Majhi, S.M.; Mirzaei, A.; Kim, H.W.; Kim, S.S.; Kim, T.W. Recent advances in energy-saving chemiresistive gas sensors: A review. Nano Energy 2021, 79, 105369.
- Choi, S.-J.; Kim, I.-D. Recent developments in 2D nanomaterials for chemiresistive-type gas sensors. Electron. Mater. Lett. 2018, 14, 221–260.
- Yang, C.; Denno, M.E.; Pyakurel, P.; Venton, B.J. Recent trends in carbon nanomaterial-based electrochemical sensors for biomolecules: A review. Anal. Chim. Acta 2015, 887, 17–37.
- Yao, S.; Swetha, P.; Zhu, Y. Nanomaterial-enabled wearable sensors for healthcare. Adv. Healthc. Mater. 2018, 7, 1700889.
- Kim, S.-J.; Choi, S.-J.; Jang, J.-S.; Cho, H.-J.; Kim, I.-D. Innovative nanosensor for disease diagnosis. Acc. Chem. Res. 2017, 50, 1587–1596.
- Wang, Z.; Hu, T.; Liang, R.; Wei, M. Application of zero-dimensional nanomaterials in biosensing. Front. Chem. 2020, 8, 320.
- Choi, S.J.; Persano, L.; Camposeo, A.; Jang, J.S.; Koo, W.T.; Kim, S.J.; Cho, H.J.; Kim, I.D.; Pisignano, D. Electrospun nanostructures for high performance chemiresistive and optical sensors. Macromol. Mater. Eng. 2017, 302, 1600569.
- Choi, S.-J.; Kim, I.-D.; Park, H.J. 2D layered Mn and Ru oxide nanosheets for real-time breath humidity monitoring. Appl. Surf. Sci. 2022, 573, 151481.
- Liu, X.; Ma, T.; Pinna, N.; Zhang, J. Two-dimensional nanostructured materials for gas sensing. Adv. Funct. Mater. 2017, 27, 1702168.
- Jeong, J.-M.; Yang, M.; Kim, D.S.; Lee, T.J.; Choi, B.G. High performance electrochemical glucose sensor based on three-dimensional MoS2/graphene aerogel. J. Colloid Interface Sci. 2017, 506, 379–385.
- Choi, S.-J.; Choi, H.-J.; Koo, W.-T.; Huh, D.; Lee, H.; Kim, I.-D. Metal–organic framework-templated PdO-Co3O4 nanocubes functionalized by SWCNTs: Improved NO2 reaction kinetics on flexible heating film. ACS Appl. Mater. Interfaces 2017, 9, 40593–40603.
- Kwon, O.S.; Song, H.S.; Park, T.H.; Jang, J. Conducting nanomaterial sensor using natural receptors. Chem. Rev. 2018, 119, 36–93.
- Fang, Y.; Deng, Y.; Dehaen, W. Tailoring pillararene-based receptors for specific metal ion binding: From recognition to supramolecular assembly. Coord. Chem. Rev. 2020, 415, 213313.
- Durkin, T.J.; Barua, B.; Savagatrup, S. Rapid detection of sepsis: Recent advances in biomarker sensing platforms. ACS Omega 2021, 6, 31390–31395.
- Kucherenko, I.; Soldatkin, O.; Dzyadevych, S.; Soldatkin, A. Electrochemical biosensors based on multienzyme systems: Main groups, advantages and limitations—A review. Anal. Chim. Acta 2020, 1111, 114–131.
- Li, F.; Yu, Z.; Han, X.; Lai, R.Y. Electrochemical aptamer-based sensors for food and water analysis: A review. Anal. Chim. Acta 2019, 1051, 1–23.
- Naresh, V.; Lee, N. A review on biosensors and recent development of nanostructured materials-enabled biosensors. Sensors 2021, 21, 1109.
- Mohammadniaei, M.; Nguyen, H.V.; Tieu, M.V.; Lee, M.-H. 2D materials in development of electrochemical point-of-care cancer screening devices. Micromachines 2019, 10, 662.
- Sivakumar, R.; Lee, N.Y. Recent progress in smartphone-based techniques for food safety and the detection of heavy metal ions in environmental water. Chemosphere 2021, 275, 130096.
- Zhao, J.; Lin, Y.; Wu, J.; Nyein, H.Y.Y.; Bariya, M.; Tai, L.-C.; Chao, M.; Ji, W.; Zhang, G.; Fan, Z. A fully integrated and self-powered smartwatch for continuous sweat glucose monitoring. ACS Sens. 2019, 4, 1925–1933.
- Bandodkar, A.J.; Gutruf, P.; Choi, J.; Lee, K.; Sekine, Y.; Reeder, J.T.; Jeang, W.J.; Aranyosi, A.J.; Lee, S.P.; Model, J.B. Battery-free, skin-interfaced microfluidic/electronic systems for simultaneous electrochemical, colorimetric, and volumetric analysis of sweat. Sci. Adv. 2019, 5, eaav3294.
- Sempionatto, J.R.; Nakagawa, T.; Pavinatto, A.; Mensah, S.T.; Imani, S.; Mercier, P.; Wang, J. Eyeglasses based wireless electrolyte and metabolite sensor platform. Lab Chip 2017, 17, 1834–1842.
- Park, J.; Kim, J.; Kim, S.-Y.; Cheong, W.H.; Jang, J.; Park, Y.-G.; Na, K.; Kim, Y.-T.; Heo, J.H.; Lee, C.Y. Soft, smart contact lenses with integrations of wireless circuits, glucose sensors, and displays. Sci. Adv. 2018, 4, eaap9841.
- Feng, S.B.; Farha, F.; Li, Q.J.; Wan, Y.L.; Xu, Y.; Zhang, T.; Ning, H.S. Review on Smart gas sensing technology. Sensors 2019, 19, 3790.
- Kim, E.; Lee, S.; Kim, J.H.; Kim, C.; Byun, Y.T.; Kim, H.S.; Lee, T. Pattern recognition for selective odor detection with gas sensor arrays. Sensors 2012, 12, 16262–16273.
- Speizer, F.E.; Ferris, B.; Bishop, Y.M.M.; Spengler, J. Respiratory-disease rates and pulmonary-function in children associated with NO2 exposure. Am. Rev. Respir. Dis 1980, 121, 3–10.
- Das, S.; Pal, M. Non-invasive monitoring of human health by exhaled breath analysis: A comprehensive review. J. Electrochem. Soc. 2020, 167, 037562.
- Tassopoulos, C.N.; Barnett, D.; Russell Fraser, T. Breath-acetone and blood-sugar measurements in diabetes. Lancet 1969, 293, 1282–1286.
- Zhou, X.; Xue, Z.; Chen, X.; Huang, C.; Bai, W.; Lu, Z.; Wang, T. Nanomaterial-based gas sensors used for breath diagnosis. J. Mater. Chem. B 2020, 8, 3231–3248.
- Baharuddin, A.A.; Ang, B.C.; Haseeb, A.; Wong, Y.C.; Wong, Y.H. Advances in chemiresistive sensors for acetone gas detection. Mater. Sci. Semicond. Process. 2019, 103, 104616.
- Wang, H.; Ma, J.; Zhang, J.; Feng, Y.; Vijjapu, M.T.; Yuvaraja, S.; Surya, S.G.; Salama, K.N.; Dong, C.; Wang, Y.; et al. Gas sensing materials roadmap. J. Phys. Condens. Matter 2021, 33, 303001.
- Zhao, S.; Shen, Y.; Yan, X.; Zhou, P.; Yin, Y.; Lu, R.; Han, C.; Cui, B.; Wei, D. Complex-surfactant-assisted hydrothermal synthesis of one-dimensional ZnO nanorods for high-performance ethanol gas sensor. Sens. Actuators B Chem. 2019, 286, 501–511.
- Al-Hazeem, N.Z.; Ahmed, N.M.; Matjafri, M.Z.; Bououdina, M. Hydrogen gas sensor based on nanofibers TiO2-PVP thin film at room temperature prepared by electrospinning. Microsyst. Technol. 2021, 27, 293–299.
- Li, P.; Zhang, Z.; Zhuang, Z.; Guo, J.; Fang, Z.; Fereja, S.L.; Chen, W. Pd-doping-induced oxygen vacancies in one-dimensional tungsten oxide nanowires for enhanced acetone gas sensing. Anal. Chem. 2021, 93, 7465–7472.
- Wang, Z.; Zhu, L.; Sun, S.; Wang, J.; Yan, W. One-dimensional nanomaterials in resistive gas sensor: From material design to application. Chemosensors 2021, 9, 198.
- Moon, D.-B.; Bag, A.; Lee, H.-B.; Meeseepong, M.; Lee, D.-H.; Lee, N.-E. A stretchable, room-temperature operable, chemiresistive gas sensor using nanohybrids of reduced graphene oxide and zinc oxide nanorods. Sens. Actuators B Chem. 2021, 345, 130373.
- Shaalan, N.M.; Yamazaki, T.; Kikuta, T. Effect of micro-electrode geometry on NO2 gas-sensing characteristics of one-dimensional tin dioxide nanostructure microsensors. Sens. Actuators B Chem. 2011, 156, 784–790.
- Moon, J.; Park, J.-A.; Lee, S.-J.; Zyung, T.; Kim, I.-D. Pd-doped TiO2 nanofiber networks for gas sensor applications. Sens. Actuators B Chem. 2010, 149, 301–305.
- Feng, C.; Kou, X.; Liao, X.; Sun, Y.; Lu, G. One-dimensional Cr-doped NiO nanostructures serving as a highly sensitive gas sensor for trace xylene detection. RSC Adv. 2017, 7, 41105–41110.
- Li, W.; Chen, R.; Qi, W.; Cai, L.; Sun, Y.; Sun, M.; Li, C.; Yang, X.; Xiang, L.; Xie, D.; et al. Reduced graphene oxide/mesoporous ZnO NSs hybrid fibers for flexible, stretchable, twisted, and wearable NO2 e-textile gas sensor. ACS Sens. 2019, 4, 2809–2818.
- Xu, Z.; Gao, C. Graphene fiber: A new trend in carbon fibers. Mater. Today 2015, 18, 480–492.
- Choi, S.-J.; Yu, H.; Jang, J.-S.; Kim, M.-H.; Kim, S.-J.; Jeong, H.S.; Kim, I.-D. Nitrogen-doped single graphene fiber with platinum water dissociation catalyst for wearable humidity sensor. Small 2018, 14, 1703934.
- Cong, H.-P.; Ren, X.-C.; Wang, P.; Yu, S.-H. Wet-spinning assembly of continuous, neat and macroscopic graphene fibers. Sci. Rep. 2012, 2, 613.
- Du, D.; Li, P.; Ouyang, J. Nitrogen-doped reduced graphene oxide prepared by simultaneous thermal reduction and nitrogen doping of graphene oxide in air and its application as an electrocatalyst. ACS Appl. Mater. Interfaces 2015, 7, 26952–26958.
- Jutz, G.; Van Rijn, P.; Santos Miranda, B.; Böker, A. Ferritin: A versatile building block for bionanotechnology. Chem. Rev. 2015, 115, 1653–1701.
- Fajin, J.L.; Bruix, A.; Cordeiro, M.N.; Gomes, J.R.; Illas, F. Density functional theory model study of size and structure effects on water dissociation by platinum nanoparticles. J. Chem. Phys. 2012, 137, 034701.
- Choi, S.J.; Lee, D.M.; Yu, H.; Jang, J.S.; Kim, M.H.; Kang, J.Y.; Jeong, H.S.; Kim, I.D. All-carbon fiber-based chemical sensor: Improved reversible NO2 reaction kinetics. Sens. Actuators B Chem. 2019, 290, 293–301.
- Choi, S.J.; Kim, S.J.; Jang, J.S.; Lee, J.H.; Kim, I.D. Silver nanowire embedded colorless polyimide heater for wearable chemical sensors: Improved reversible reaction kinetics of optically reduced graphene oxide. Small 2016, 12, 5826–5835.
- Eom, W.; Jang, J.S.; Lee, S.H.; Lee, E.; Jeong, W.; Kim, I.D.; Choi, S.J.; Han, T.H. Effect of metal/metal oxide catalysts on graphene fiber for improved NO2 sensing. Sens. Actuators B Chem. 2021, 344.
- Liu, J.; Zhang, L.; Cheng, B.; Fan, J.; Yu, J. A high-response formaldehyde sensor based on fibrous Ag-ZnO/In2O3 with multi-level heterojunctions. J. Hazard. Mater. 2021, 413, 125352.
- Liu, A.; Lv, S.; Jiang, L.; Liu, F.; Zhao, L.; Wang, J.; Hu, X.; Yang, Z.; He, J.; Wang, C.; et al. The gas sensor utilizing polyaniline/MoS2 nanosheets/SnO2 nanotubes for the room temperature detection of ammonia. Sens. Actuators B Chem. 2021, 332, 129444.
- Mishra, R.K.; Murali, G.; Kim, T.-H.; Kim, J.H.; Lim, Y.J.; Kim, B.-S.; Sahay, P.P.; Lee, S.H. Nanocube In2O3@RGO heterostructure based gas sensor for acetone and formaldehyde detection. RSC Adv. 2017, 7, 38714–38724.
- Jang, J.S.; Yu, H.; Choi, S.J.; Koo, W.T.; Lee, J.; Kim, D.H.; Kang, J.Y.; Jeong, Y.J.; Jeong, H.; Kim, I.D. Heterogeneous metal oxide-graphene thorn-bush single fiber as a freestanding chemiresistor. ACS Appl. Mater. Interfaces 2019, 11, 10208–10217.
- Cho, S.Y.; Yu, H.; Choi, J.; Kang, H.; Park, S.; Jang, J.S.; Hong, H.J.; Kim, I.D.; Lee, S.K.; Jeong, H.S.; et al. Continuous meter-scale synthesis of weavable tunicate cellulose/carbon nanotube fibers for high-performance wearable sensors. ACS Nano 2019, 13, 9332–9341.
- Cheng, Q.; Ye, D.; Chang, C.; Zhang, L. Facile fabrication of superhydrophilic membranes consisted of fibrous tunicate cellulose nanocrystals for highly efficient oil/water separation. J. Membr. Sci. 2017, 525, 1–8.
- Nakate, U.T.; Ahmad, R.; Patil, P.; Wang, Y.; Bhat, K.S.; Mahmoudi, T.; Yu, Y.T.; Suh, E.-K.; Hahn, Y.-B. Improved selectivity and low concentration hydrogen gas sensor application of Pd sensitized heterojunction n-ZnO/p-NiO nanostructures. J. Alloys Compd. 2019, 797, 456–464.
- Kim, S.J.; Choi, S.J.; Jang, J.S.; Cho, H.J.; Koo, W.T.; Tuller, H.L.; Kim, I.D. Exceptional high-performance of pt-based bimetallic catalysts for exclusive detection of exhaled biomarkers. Adv. Mater. 2017, 29, 1700737.
- Zou, Y.; Chen, S.; Sun, J.; Liu, J.; Che, Y.; Liu, X.; Zhang, J.; Yang, D. Highly efficient gas sensor using a hollow SnO2 microfiber for triethylamine detection. ACS Sens. 2017, 2, 897–902.
- Cha, J.-H.; Choi, S.-J.; Yu, S.; Kim, I.-D. 2D WS2-edge functionalized multi-channel carbon nanofibers: Effect of WS2 edge-abundant structure on room temperature NO2 sensing. J. Mater. Chem. A 2017, 5, 8725–8732.
- Han, D.; Ji, Y.; Gu, F.; Wang, Z. Cobalt oxide nanorods with special pore structure for enhanced ethanol sensing performance. J. Colloid Interface Sci. 2018, 531, 320–330.
- Jang, J.S.; Qiao, S.; Choi, S.J.; Jha, G.; Ogata, A.F.; Koo, W.T.; Kim, D.H.; Kim, I.D.; Penner, R.M. Hollow Pd–Ag composite nanowires for fast responding and transparent hydrogen sensors. ACS Appl. Mater. Interfaces 2017, 9, 39464–39474.
- Jo, S.; Sung, D.; Kim, S.; Koo, J. A review of wearable biosensors for sweat analysis. Biomed. Eng. Lett. 2021, 11, 117–129.
- Kaisti, M.; Boeva, Z.; Koskinen, J.; Nieminen, S.; Bobacka, J.; Levon, K. Hand-held transistor based electrical and multiplexed chemical sensing system. Acs Sens. 2016, 1, 1423–1431.
- Kang, S.G.; Song, M.S.; Kim, J.W.; Lee, J.W.; Kim, J. Near-field communication in biomedical applications. Sensors 2021, 21, 703.
- Baker, L.B.; Model, J.B.; Barnes, K.A.; Anderson, M.L.; Lee, S.P.; Lee, K.A.; Brown, S.D.; Reimel, A.J.; Roberts, T.J.; Nuccio, R.P. Skin-interfaced microfluidic system with personalized sweating rate and sweat chloride analytics for sports science applications. Sci. Adv. 2020, 6, eabe3929.
- Eiteman, M.A.; Altman, E. Overcoming acetate in Escherichia coli recombinant protein fermentations. Trends Biotechnol. 2006, 24, 530–536.
- Wolfe, A.J. The acetate switch. Microbiol. Mol. Biol. Rev. 2005, 69, 12–50.
- Dittrich, C.R.; Vadali, R.V.; Bennett, G.N.; San, K.Y. Redistribution of metabolic fluxes in the central aerobic metabolic pathway of E. coli mutant strains with deletion of the ackA-pta and poxB pathways for the synthesis of isoamyl acetate. Biotechnol. Prog. 2005, 21, 627–631.
- Xu, G.; Cheng, C.; Yuan, W.; Liu, Z.Y.; Zhu, L.H.; Li, X.T.; Lu, Y.L.; Chen, Z.T.; Liu, J.L.; Cui, Z.; et al. Smartphone-based battery-free and flexible electrochemical patch for calcium and chloride ions detections in biofluids. Sens. Actuators B Chem. 2019, 297, 126743.
- Choi, D.-H.; Li, Y.; Cutting, G.R.; Searson, P.C. A wearable potentiometric sensor with integrated salt bridge for sweat chloride measurement. Sens. Actuators B Chem. 2017, 250, 673–678.
- Chen, L.J.; Berry, S.N.; Wu, X.; Howe, E.N.W.; Gale, P.A. Advances in anion receptor chemistry. Chem 2020, 6, 61–141.
- Amendola, V.; Bergamaschi, G.; Boiocchi, M.; Fabbrizzi, L.; Milani, M. The squaramide versus urea contest for anion recognition. Chem.-Eur. J. 2010, 16, 4368–4380.
- Choi, S.J.; Yoon, B.; Ray, J.D.; Netchaev, A.; Moores, L.C.; Swager, T.M. Chemiresistors for the real-time wireless detection of anions. Adv. Funct. Mater. 2020, 30, 1907087.
- Manjakkal, L.; Dang, W.T.; Yogeswaran, N.; Dahiya, R. Textile-based potentiometric electrochemical pH sensor for wearable applications. Biosensors 2019, 9, 14.
- Promphet, N.; Rattanawaleedirojn, P.; Siralertmukul, K.; Soatthiyanon, N.; Potiyaraj, P.; Thanawattano, C.; Hinestroza, J.P.; Rodthongkum, N. Non-invasive textile based colorimetric sensor for the simultaneous detection of sweat pH and lactate. Talanta 2019, 192, 424–430.
- Qin, Y.H.; Kwon, H.J.; Howlader, M.M.R.; Deen, M.J. Microfabricated electrochemical pH and free chlorine sensors for water quality monitoring: Recent advances and research challenges. RSC Adv. 2015, 5, 69086–69109.
- Dore, J.E.; Lukas, R.; Sadler, D.W.; Church, M.J.; Karl, D.M. Physical and biogeochemical modulation of ocean acidification in the central North Pacific. Proc. Natl. Acad. Sci. USA 2009, 106, 12235–12240.
- Jeon, J.Y.; Kang, B.C.; Ha, T.J. Flexible pH sensors based on printed nanocomposites of single-wall carbon nanotubes and nafion. Appl. Surf. Sci. 2020, 514, 145956.
- Liu, Y.-L.; Su, Y.-H.; Chang, C.-M.; Wang, D.-M.; Lai, J.-Y. Preparation and applications of Nafion-functionalized multiwalled carbon nanotubes for proton exchange membrane fuel cells. J. Mater. Chem. 2010, 20, 4409–4416.
- Siu, A.; Schmeisser, J.; Holdcroft, S. Effect of water on the low temperature conductivity of polymer electrolytes. J. Phys. Chem. B 2006, 110, 6072–6080.
- Choi, S.J.; Yoon, B.; Lin, S.B.; Swager, T.M. Functional single-walled carbon nanotubes for anion sensing. ACS Appl. Mater. Interfaces 2020, 12, 28375–28382.
- Yoon, B.; Choi, S.J. Selective acetate recognition and sensing using SWCNTs functionalized with croconamides. Sens. Actuators B Chem. 2021, 346, 130461.
- Parrilla, M.; Cuartero, M.; Crespo, G.A. Wearable potentiometric ion sensors. TrAC Trend Anal. Chem. 2019, 110, 303–320.
- Cuartero, M.; Parrilla, M.; Crespo, G.A. Wearable potentiometric sensors for medical applications. Sensors 2019, 19, 363.
- Bandodkar, A.J.; Wang, J. Non-invasive wearable electrochemical sensors: A review. Trends Biotechnol. 2014, 32, 363–371.
- Xu, J.N.; Zhang, Z.; Gan, S.Y.; Gao, H.; Kong, H.J.; Song, Z.Q.; Ge, X.M.; Bao, Y.; Niu, L. Highly stretchable fiber-based potentiometric ion sensors for multichannel real-time analysis of human sweat. ACS Sens. 2020, 5, 2834–2842.
- An, Q.B.; Gan, S.Y.; Xu, J.N.; Bao, Y.; Wu, T.S.; Kong, H.J.; Zhong, L.J.; Ma, Y.M.; Song, Z.Q.; Niu, L. A multichannel electrochemical all-solid-state wearable potentiometric sensor for real-time sweat ion monitoring. Electrochem. Commun. 2019, 107, 106553.
- Gao, W.; Emaminejad, S.; Nyein, H.Y.Y.; Challa, S.; Chen, K.V.; Peck, A.; Fahad, H.M.; Ota, H.; Shiraki, H.; Kiriya, D.; et al. Fully integrated wearable sensor arrays for multiplexed in situ perspiration analysis. Nature 2016, 529, 509.
- Wu, W.W.; Haick, H. Materials and wearable devices for autonomous monitoring of physiological markers. Adv. Mater. 2018, 30.
- Yoon, J.H.; Kim, S.M.; Eom, Y.; Koo, J.M.; Cho, H.W.; Lee, T.J.; Lee, K.G.; Park, H.J.; Kim, Y.K.; Yoo, H.J.; et al. Extremely fast self-healable bio-based supramolecular polymer for wearable real-time sweat-monitoring sensor. ACS Appl. Mater. Interfaces 2019, 11, 46165–46175.
- Yoon, J.H.; Kim, S.M.; Park, H.J.; Kim, Y.K.; Oh, D.X.; Cho, H.W.; Lee, K.G.; Hwang, S.Y.; Park, J.; Choi, B.G. Highly self-healable and flexible cable-type pH sensors for real-time monitoring of human fluids. Biosens. Bioelectron. 2020, 150, 111946.
- Choi, S.J.; Savagatrup, S.; Kim, Y.; Lang, J.H.; Swager, T.M. Precision pH sensor based on WO3 nanofiber-polymer composites and differential amplification. ACS Sens. 2019, 4, 2593–2598.
- Kim, D.S.; Jeong, J.M.; Park, H.J.; Kim, Y.K.; Lee, K.G.; Choi, B.G. Highly concentrated, conductive, defect-free graphene ink for screen-printed sensor application. Nano-Micro Lett. 2021, 13, 87.
- Zhang, S.P.; Abu Zahed, M.; Sharifuzzaman, M.; Yoon, S.; Hui, X.; Barman, S.C.; Sharma, S.; Yoon, H.S.; Park, C.; Park, J.Y. A wearable battery-free wireless and skin-interfaced microfluidics integrated electrochemical sensing patch for on-site biomarkers monitoring in human perspiration. Biosens. Bioelectron. 2021, 175, 112844.
- Kim, D.H.; Lu, N.S.; Ma, R.; Kim, Y.S.; Kim, R.H.; Wang, S.D.; Wu, J.; Won, S.M.; Tao, H.; Islam, A.; et al. Epidermal electronics. Science 2011, 333, 838–843.
- Yeo, W.H.; Kim, Y.S.; Lee, J.; Ameen, A.; Shi, L.K.; Li, M.; Wang, S.D.; Ma, R.; Jin, S.H.; Kang, Z.; et al. Multifunctional epidermal electronics printed directly onto the skin. Adv. Mater. 2013, 25, 2773–2778.
- Trung, T.Q.; Lee, N.E. Flexible and stretchable physical sensor integrated platforms for wearable human-activity monitoring and personal healthcare. Adv. Mater. 2016, 28, 4338–4372.
- Huynh, T.P.; Sonar, P.; Haick, H. Advanced materials for use in soft self-healing devices. Adv. Mater. 2017, 29, 1604973.
- Thakur, V.K.; Kessler, M.R. Self-healing polymer nanocomposite materials: A review. Polymer 2015, 69, 369–383.
- Li, J.J.; Geng, L.F.; Wang, G.; Chu, H.J.; Wei, H.L. Self-healable gels for use in wearable devices. Chem. Mater. 2017, 29, 8932–8952.
- Luo, C.S.; Wan, P.B.; Yang, H.; Shah, S.A.A.; Chen, X.D. Healable transparent electronic devices. Adv. Funct. Mater. 2017, 27, 1606339.
- Chen, D.D.; Wang, D.R.; Yang, Y.; Huang, Q.Y.; Zhu, S.J.; Zheng, Z.J. Self-healing materials for next-generation energy harvesting and storage devices. Adv. Energy Mater. 2017, 7, 1700890.
- Zou, Z.N.; Zhu, C.P.; Li, Y.; Lei, X.F.; Zhang, W.; Xiao, J.L. Rehealable, fully recyclable, and malleable electronic skin enabled by dynamic covalent thermoset nanocomposite. Sci. Adv. 2018, 4, eaaq0508.
- Kim, J.; Campbell, A.S.; de Avila, B.E.F.; Wang, J. Wearable biosensors for healthcare monitoring. Nat. Biotechnol. 2019, 37, 389–406.
- Lee, H.; Hong, Y.J.; Baik, S.; Hyeon, T.; Kim, D.H. Enzyme-based glucose sensor: From invasive to wearable device. Adv. Healthc. Mater. 2018, 7, 1701150.
- Shin, H.; Seo, H.; Chung, W.G.; Joo, B.J.; Jang, J.; Park, J.U. Recent progress on wearable point-of-care devices for ocular systems. Lab Chip 2021, 21, 1269–1286.
- Tang, L.; Chang, S.J.; Chen, C.J.; Liu, J.T. Non-invasive blood glucose monitoring technology: A review. Sensors 2020, 20, 6925.
- Teymourian, H.; Barfidokht, A.; Wang, J. Electrochemical glucose sensors in diabetes management: An updated review (2010–2020). Chem. Soc. Rev. 2020, 49, 7671–7709.
- Nichols, S.P.; Koh, A.; Storm, W.L.; Shin, J.H.; Schoenfisch, M.H. Biocompatible materials for continuous glucose monitoring devices. Chem. Rev. 2013, 113, 2528–2549.
- Cho, N.H.; Shaw, J.E.; Karuranga, S.; Huang, Y.; Fernandes, J.D.D.; Ohlrogge, A.W.; Malanda, B. IDF diabetes atlas: Global estimates of diabetes prevalence for 2017 and projections for 2045. Diabetes Res. Clin. Pract. 2018, 138, 271–281.
- Harding, J.L.; Pavkov, M.E.; Magliano, D.J.; Shaw, J.E.; Gregg, E.W. Global trends in diabetes complications: A review of current evidence. Diabetologia 2019, 62, 3–16.
- Heller, A.; Feldman, B. Electrochemical glucose sensors and their applications in diabetes management. Chem. Rev. 2008, 108, 2482–2505.
- Ciobanu, M.; Taylor, D.E.; Wilburn, J.P.; Cliffel, D.E. Glucose and lactate biosensors for scanning electrochemical microscopy imaging of single live cells. Anal. Chem. 2008, 80, 2717–2727.
- Soylemez, S.; Yoon, B.; Toppare, L.; Swager, T.M. Quaternized polymer-single-walled carbon nanotube scaffolds for a chemiresistive glucose sensor. ACS Sens. 2017, 2, 1123–1127.
- Fang, L.; Liang, B.; Yang, G.; Hu, Y.C.; Zhu, Q.; Ye, X.S. A needle-type glucose biosensor based on PANI nanofibers and PU/E-PU membrane for long-term invasive continuous monitoring. Biosens. Bioelectron. 2017, 97, 196–202.
- Chen, D.J.; Wang, C.; Chen, W.; Chen, Y.Q.; Zhang, J.X.J. PVDF-Nafion nanomembranes coated microneedles for in vivo transcutaneous implantable glucose sensing. Biosens. Bioelectron. 2015, 74, 1047–1052.
- Wang, L.Y.; Xie, S.L.; Wang, Z.Y.; Liu, F.; Yang, Y.F.; Tang, C.Q.; Wu, X.Y.; Liu, P.; Li, Y.J.; Saiyin, H.; et al. Functionalized helical fibre bundles of carbon nanotubes as electrochemical sensors for long-term in vivo monitoring of multiple disease biomarkers. Nat. Biomed. Eng. 2020, 4, 159–171.
- Kang, B.C.; Park, B.S.; Ha, T.J. Highly sensitive wearable glucose sensor systems based on functionalized single-wall carbon nanotubes with glucose oxidase-nafion composites. Appl. Surf. Sci. 2019, 470, 13–18.
- Yoo, E.H.; Lee, S.Y. Glucose biosensors: An overview of use in clinical practice. Sensors 2010, 10, 4558–4576.
- Gupta, S.; Murthy, C.N.; Prabha, C.R. Recent advances in carbon nanotube based electrochemical biosensors. Int. J. Biol. Macromol. 2018, 108, 687–703.
- Yang, M.H.; Jeong, S.W.; Chang, S.J.; Kim, K.H.; Jang, M.; Kim, C.H.; Bae, N.H.; Sim, G.S.; Kang, T.; Lee, S.J.; et al. Flexible and disposable sensing platforms based on newspaper. ACS Appl. Mater. Interfaces 2016, 8, 34978–34984.
- Yoon, J.; Lee, S.N.; Shin, M.K.; Kim, H.W.; Choi, H.K.; Lee, T.; Choi, J.W. Flexible electrochemical glucose biosensor based on GOx/gold/MoS2/gold nanofilm on the polymer electrode. Biosens. Bioelectron. 2019, 140, 83–89.
- Trieu, P.T.; Lee, N.Y. Paper-based all-in-one origami microdevice for nucleic acid amplification testing for rapid colorimetric identification of live cells for point-of-care testing. Anal. Chem. 2019, 91, 11013–11022.
- You, J.B.; Yoo, Y.; Oh, M.S.; Im, S.G. Simple and reliable method to incorporate the janus property onto arbitrary porous substrates. ACS Appl. Mater. Inter. 2014, 6, 4005–4010.
- Trinh, T.N.D.; La, H.C.; Lee, N.Y. Fully integrated and foldable microdevice encapsulated with agarose for long-term storage potential for point-of-care testing of multiplex foodborne pathogens. ACS Sens. 2019, 4, 2754–2762.
- Noh, J.Y.; Yoon, S.W.; Kim, Y.; Lo, T.V.; Ahn, M.J.; Jung, M.C.; Le, T.B.; Na, W.; Song, D.; Le, V.P.; et al. Pipetting-based immunoassay for point-of-care testing: Application for detection of the influenza A virus. Sci. Rep. 2019, 9, 16661.
- Kim, S.H.; Woo, H.C.; Kim, M.H. Solid-phase colorimetric sensing probe for bromide based on a tough hydrogel embedded with silver nanoprisms. Anal. Chim. Acta 2020, 1131, 80–89.
- Shaban, S.M.; Moon, B.S.; Kim, D.H. Selective and sensitive colorimetric detection of p-aminophenol in human urine and paracetamol drugs based on seed-mediated growth of silver nanoparticles. Environ. Technol. Innov. 2021, 22, 101517.
- Tran, V.K.; Gupta, P.K.; Park, Y.; Son, S.E.; Hur, W.; Lee, H.B.; Park, J.Y.; Kim, S.N.; Seong, G.H. Functionalized bimetallic IrPt alloy nanoparticles: Multi-enzyme mimics for colorimetric and fluorometric detection of hydrogen peroxide and glucose. J. Taiwan Inst. Chem. E 2021, 120, 336–343.
- Lee, J.I.; Jang, S.C.; Chung, J.; Choi, W.K.; Hong, C.; Ahn, G.R.; Kim, S.H.; Lee, B.Y.; Chung, W.J. Colorimetric allergenic fungal spore detection using peptide-modified gold nanoparticles. Sens. Actuators B Chem. 2021, 327, 128894.
- Han, G.R.; Koo, H.J.; Ki, H.; Kim, M.G. Paper/Soluble polymer hybrid-based lateral flow biosensing platform for high-performance point-of-care testing. ACS Appl. Mater. Interfaces 2020, 12, 34564–34575.
- Choi, C.K.; Shaban, S.M.; Moon, B.S.; Pyun, D.; Kim, D.H. Smartphone-assisted point-of-care colorimetric biosensor for the detection of urea via pH-mediated AgNPs growth. Anal. Chim. Acta 2021, 1170, 338630.
- Son, S.U.; Seo, S.B.; Jane, S.; Choi, J.; Lim, J.W.; Lee, D.K.; Kim, H.; Seo, S.; Kang, T.; Jung, J.; et al. Naked-eye detection of pandemic influenza a (pH1N1) virus by polydiacetylene (PDA)-based paper sensor as a point-of-care diagnostic platform. Sens. Actuators B Chem. 2019, 291, 257–265.
- Tawfik, S.M.; Elmasry, M.R.; Sharipov, M.; Azizov, S.; Lee, C.H.; Lee, Y.I. Dual emission nonionic molecular imprinting conjugated polythiophenes-based paper devices and their nanofibers for point-of-care biomarkers detection. Biosens. Bioelectron. 2020, 160, 112211.
- Han, K.N.; Choi, J.S.; Kwon, J. Three-dimensional paper-based slip device for one-step point-of-care testing. Sci. Rep. 2016, 6, 25710.
- Posthuma-Trumpie, G.A.; Korf, J.; van Amerongen, A. Lateral flow (immuno) assay: Its strengths, weaknesses, opportunities and threats. A literature survey. Anal. Bioanal. Chem. 2009, 393, 569–582.
- Li, F.; You, M.L.; Li, S.X.; Hu, J.; Liu, C.; Gong, Y.; Yang, H.Y.; Xu, F. Paper-based point-of-care immunoassays: Recent advances and emerging trends. Biotechnol. Adv. 2020, 3, 107442.
- Kim, K.; Kashefi-Kheyrabadi, L.; Joung, Y.; Kim, K.; Dang, H.J.; Chavan, S.G.; Lee, M.H.; Choo, J. Recent advances in sensitive surface-enhanced Raman scattering-based lateral flow assay platforms for point-of-care diagnostics of infectious diseases. Sens. Actuators B Chem. 2021, 329, 129214.
- Hang, G.R.; Ki, H.; Kim, M.G. Automated, universal, and mass-producible paper-based lateral flow biosensing platform for high-performance point-of-care testing. ACS Appl. Mater. Interfaces 2020, 12, 1885–1894.
- Kim, K.; Joung, H.A.; Han, G.R.; Kim, M.G. An immunochromatographic biosensor combined with a water-swellable polymer for automatic signal generation or amplification. Biosens. Bioelectron. 2016, 85, 422–428.
More
Information
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.5K
Revisions:
2 times
(View History)
Update Date:
17 Jan 2022
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





