Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Amir Syahir | + 1934 word(s) | 1934 | 2021-12-14 07:25:49 | | | |
| 2 | Catherine Yang | Meta information modification | 1934 | 2021-12-24 02:13:28 | | | | |
| 3 | Catherine Yang | Meta information modification | 1934 | 2021-12-28 01:38:34 | | | | |
| 4 | Catherine Yang | Meta information modification | 1934 | 2021-12-28 01:38:57 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Syahir, A. CD44. Encyclopedia. Available online: https://encyclopedia.pub/entry/17535 (accessed on 22 May 2026).
Syahir A. CD44. Encyclopedia. Available at: https://encyclopedia.pub/entry/17535. Accessed May 22, 2026.
Syahir, Amir. "CD44" Encyclopedia, https://encyclopedia.pub/entry/17535 (accessed May 22, 2026).
Syahir, A. (2021, December 24). CD44. In Encyclopedia. https://encyclopedia.pub/entry/17535
Syahir, Amir. "CD44." Encyclopedia. Web. 24 December, 2021.
Copy Citation
CD44, a non-kinase cell surface transmembrane glycoprotein, has been widely implicated as a cancer stem cell (CSC) marker in several cancers. Cells overexpressing CD44 possess several CSC traits, such as self-renewal and epithelial-mesenchymal transition (EMT) capability, as well as a resistance to chemo- and radiotherapy.
CD44
regulation
tumourigenesis
1. CD44 Structure and Isoforms
The full-length CD44 gene comprises 20 exons, with the constant exons 1–5 and 16–20 encoding the N-terminal and C-terminal domains respectively, which are homologous domains shared by all CD44 family members [1]. The smallest and the most expressed CD44 isoform is the CD44 standard (CD44s), constructed of ten constant exons with no variant exons [2]. The other isoform, the CD44 variant (CD44v), differs from CD44s by the insertion or excision of alternatively spliced exons between the N-terminal and C-terminal domains [3]. Tolg et al. [4] confirmed that besides ten constant exons, the mouse and rat genome has at least ten variant exons, all of which can be combined alternatively into CD44 mRNA. They suggested that the variant exons be numbered by the exon code v1 to v10. Screaton et al. [5] described the structure of the human CD44 gene, reporting that it contains 19 exons crossing some 50 kilobases of DNA with ten constant exons and nine variant exons coded v2–v10 [6][7]. CD44v isoforms may contain a single variant exon such as CD44v3 or CD44v6, or a combination of variant exons such as CD44v3–v7 and CD44v8-v10. Individual cells can continually alter the splicing of CD44 pre-mRNA, resulting in many possible combinations of these variant exons, giving the potential for great diversity [3].
The CD44 protein has four primary characteristic regions: the extracellular region, the stem region (standard stem region and/or variable stem region), the transmembrane region (TM), and the short C-terminal intracellular/cytoplasmic (CP) region [8]. The extracellular part consists of seven extracellular domains (1–5, 6 and 7 of the constant exons) including N-terminal domains (ligand-binding region). The stem region (alternative splicing area) has an insertion of one or more of the variant exons between exon 5 and exon 6. The transmembrane region is encoded by a single exon (exon 8), whereas the cytoplasmic region is encoded by exon 10 or exon 9. However, exon 9 is spliced out in almost all CD44 cDNA isoforms [3][9]. Several isoforms of the human CD44 molecule are associated with tumour progression and stemness in various cancers, such as breast cancer [10], gliomas [11][12], head and neck squamous cell carcinoma [13], pancreatic cancer [14][15], prostate cancer [16] and colorectal cancer [17][18] (Figure 1 and Table 1). The complexity of the CD44 protein is further augmented by post-translational modifications including variance glycosylation with O-glycans, N-glycans and glycosaminoglycans, such as chondroitin sulphate and heparan sulphate [8]. Due to these side-chain attachments, the conserved format of CD44 (37 kDa) is enlarged to 80–100 kDa with some isoforms surpassing 200 kDa due to a high level of glycosylation [3]. An illustration of CD44 protein structure is shown in Figure 2.
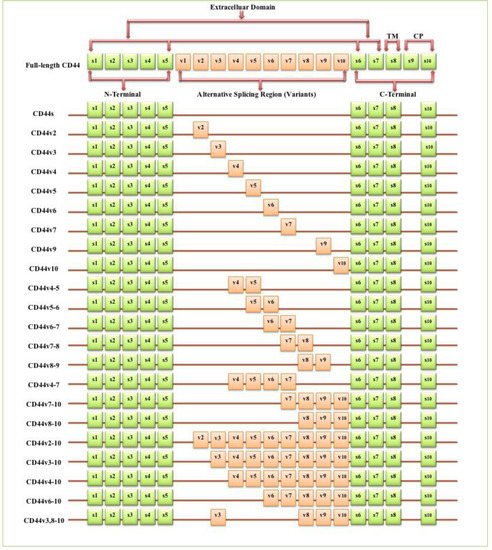
Figure 1. Schematic diagram of the mouse CD44 gene and most CD44 isoforms involved in cancer progression. The full-length CD44 gene contains 20 exons in mice and 19 exons in humans, with the constant exons 1–5 and 16–20 encoding the N-terminal and the C-terminal domains. CD44 standard (CD44s) is encoded by these ten constant exons and contains no variant exons, whereas the CD44 variant (CD44v) is produced by the alternative splicing of a variable insertion of nine extra exons in humans or ten extra exons in mice. These extra exons are exons 6-15, typically identified as (v1 to v10) in mice and the exons 7-15 identified as (v2 to v10) in humans and are located between the N-terminal and C-terminal domains. CD44v can contain one or multiple variant exons and exon 19 is spliced out in all CD44 isoforms. Abbreviations: CD44s, CD44 standard; CD44v, CD44 variant; s, standard; v, variant; TM, transmembrane; CP, cytoplasmic. Green boxes refer to the constant/standard exons. Orange boxes refer to the variant exons.
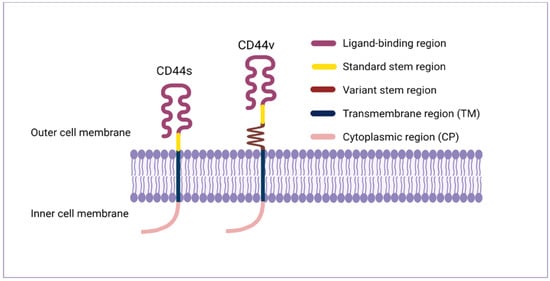
Figure 2. CD44 protein structure. The CD44 protein has four primary regions: the extracellular region consists of seven extracellular domains including N-terminal domains (ligand-binding region), the stem region (variable stem region and/or standard stem region) which is the alternative splicing area containing an insertion of one or more variant exons, the transmembrane region (TM), and the C-terminal cytoplasmic (CP) region.
Table 1. CD44 isoforms relevant to cancer progression. Abbreviations: CSCs, cancer stem cells; EMT, epithelial–mesenchymal transition; DFS, disease-free survival; OS, overall survival; TNM stage, tumour (T), node (N), and metastasis (M) stage; FIGO stage, the international federation of gynaecology and obstetrics stage; NHL, Non-Hodgkin’s lymphoma; HPV, human papillomavirus; MAPK, mitogen-activated protein kinase.
| CD44 Isoform | Association in Cancer Progress | Cancer Type | Ref |
|---|---|---|---|
| CD44, non-specified | Tumour cell aggregation, metastasis | Breast cancer | [10] |
| CD44, non-specified | Adhesion, migration, invasion | Glioblastoma | [11][12] |
| CD44, non-specified | Angiogenesis | Head and neck squamous carcinoma | [13] |
| CD44, non-specified | Invasion, metastasis, EMT, cancer progression, poor prognosis | Pancreatic cancer | [14][15] |
| CD44, non-specified | Proliferation, migration, invasion | Prostate Cancer | [16] |
| CD44, non-specified | Metastasis, poor differentiation, invasion | Colorectal cancer | [17][18] |
| CD44s | Tumour initiation, CSCs traits induction | Breast cancer | [19] |
| CD44s | Metastasis | Breast cancer | [20] |
| CD44s | EMT regulation, cancer progression | Breast cancer | [21] |
| CD44s | Poor DFS, poor OS, invasion, EMT | Hepatocellular carcinoma | [22] |
| CD44s | Invasion, metastasis, EMT, poor differentiation, chemotaxis |
Gallbladder cancer | [23] |
| CD44s | Proliferation, invasion, migration, EMT, stemness | Prostate cancer | [24] |
| CD44s | EMT, invasion, metastasis, chemoresistance | Pancreatic ductal adenocarcinoma | [25] |
| CD44s | EMT, radio-resistance | Pancreatic cancer | [26] |
| CD44v2 | Poor OS, advanced cancer stage | Colorectal cancer | [27] |
| CD44v2 | Poor OS, invasion | Pancreatic cancer | [28] |
| CD44v3 | Poor OS, invasion, metastasis | Oral squamous carcinoma | [29] |
| CD44v3 | Stem cells self-renewal | Myeloid leukaemia | [30] |
| CD44v3 | Metastasis | Colorectal adenocarcinoma | [31] |
| CD44v4 | Proliferation, migration, radio-resistance | Head and neck squamous carcinoma |
[32] |
| CD44v5 | High histological grade, poor differentiation, poor OS |
Hepatocellular carcinoma | [33] |
| CD44v6 | Tumour budding, invasion, metastasis | Oral squamous carcinoma | [34] |
| CD44v6 | Proliferation, invasion, adhesion, metastasis, EMT, chemo/radio-resistance |
Prostate cancer | [35] |
| CD44v6 | Local recurrence, invasion, metastasis | Tongue squamous carcinoma | [36] |
| CD44v6 | Tumour budding, locoregional failure (metastasis, local recurrence) | Colorectal cancer | [37] |
| CD44v6 | Proliferation, migration, radio-resistance | Head and neck squamous carcinoma |
[32] |
| CD44v6 | Metastasis | Colorectal adenocarcinoma | [31] |
| CD44v6 | Poor OS, invasion | Pancreatic cancer | [28] |
| CD44v6 | High histological grade, poor differentiation, poor OS |
Hepatocellular carcinoma | [33] |
| CD44v6 | Invasion, metastasis, poor OS, TNM stage | Pancreatic cancer | [38] |
| CD44v6 | FIGO stage, poor prognosis | Cervical cancer | [39] |
| CD44v6 | Metastasis, self-adhesion of aggressive NHL cells | Non-Hodgkin’s lymphoma | [40] |
| CD44v6 | Infiltration, metastasis | Oesophageal squamous carcinoma | [41] |
| CD44v6 | Proliferation, myofibroblastic differentiation | Gastric cancer | [42] |
| CD44v7 | Proliferation, migration, radio-resistance | Head and neck squamous carcinoma |
[32] |
| CD44v9 | Increased tumourigenicity | Gallbladder cancer | [23] |
| CD44v9 | Invasion, metastasis, poor OS, TNM stage | Pancreatic cancer | [38] |
| CD44v9 | Proliferation, invasion, migration, EMT | Cholangiocarcinoma | [43] |
| CD44v9 | Invasion, migration, worse prognosis | Bladder cancer | [44] |
| CD44v10 | High histological grade, poor differentiation, poor OS |
Hepatocellular carcinoma | [33] |
| CD44v10 | Histological grade, clinical and pathological stage, poor survival |
Renal carcinoma | [45] |
| CD44v10 | Migration, metastasis, promote tumourigenesis |
Breast cancer | [46][47] |
| CD44v4-5 | Infiltration, metastasis | Oesophageal squamous carcinoma | [41] |
| CD44v4-5 | Poor differentiation | Non-small cell lung carcinoma | [48] |
| CD44v5-6 | Proliferation, KRAS/MAPK signalling, promoting tumour development | Lung adenocarcinoma | [49] |
| CD44v6-7 | Metastasis | Pancreatic adenocarcinoma | [2] |
| CD44v7-8 | High histological grade, poor differentiation, poor OS |
Hepatocellular carcinoma | [33] |
| CD44v7-8 | FIGO stage, poor prognosis | Cervical cancer | [39] |
| CD44v7-8 | Invasion, high-risk HPV infection | Uterine cervical squamous carcinoma |
[50] |
| CD44v8-9 | Proliferation, KRAS/MAPK signalling, promoting tumour development | Lung adenocarcinoma | [49] |
| CD44v4-7 | Metastasis | Pancreatic adenocarcinoma | [2] |
| CD44v7-10 | Invasion | Prostate cancer | [51] |
| CD44v8-10 | Migration, metastasis, sphere formation | Breast cancer | [52] |
| CD44v8-10 | Tumour initiation, CSCs traits induction | Gastric cancer | [53] |
| CD44v8-10 | Metastasis | Lung cancer | [54] |
| CD44v8-10 | Metastasis, relapse | Gastric cancer | [55] |
| CD44v8-10 | Poor prognosis, chemo/radio-resistance | Oesophageal squamous carcinoma | [56] |
| CD44v8-10 | Chemoresistance | Urothelial cancer | [57] |
| CD44v2-10 | CSCs traits induction, tumour subtype, oncogenic signalling pathways | Breast cancer | [58] |
| CD44v3-10 | CSCs traits induction, tumour subtype, oncogenic signalling pathways | Breast cancer | [58] |
| CD44v3-10 | Metastasis, self-adhesion of aggressive NHL cells | Non-Hodgkin’s lymphoma | [40] |
| CD44v4-10 | Tumour initiation, wild-type phenotype | Intestinal cancer | [6] |
| CD44v6-10 | Metastasis, self-adhesion of aggressive NHL cells | Non-Hodgkin’s lymphoma | [40] |
| CD44v6-10 | Metastasis, relapse | Gastric cancer | [55] |
| CD44v3, 8-10 | Metastasis, relapse | Gastric cancer | [55] |
| CD44v3, 8-10 | Metastasis, migration | Breast cancer | [59] |
2. CD44 Expression in Normal Cells
CD44 is significantly expressed in lymphocytes, smooth muscle, fibroblasts and various types of epithelia and is involved in lymphocyte homing, cell adhesion and aggregation, cell migration, leukocyte activation, lymphopoiesis and myelopoiesis, angiogenesis and cytokine release [3][60]. CD44s was initially isolated from haematopoietic cells even though it is expressed in several other tissues including the liver, lung, pancreas, skin and central nervous system [3]. CD44s is expressed in adult tissues and embryo tissues from day 9.5 post coitum, whereas numerous isoforms of CD44v show a highly specialised expression pattern and are already in the egg cylinder at day 6.5 of development [61]. In contrast to CD44s, CD44v isoforms distribution is more restricted to a selected range of cells during specific stages of activation, maturation or development including macrophages, activated lymphocytes, keratinocytes and some epithelial cells such as in the stomach, bladder and uterine cervix [3] and many carcinomas. In normal tissues, CD44 isoforms play a role in hyaluronic acid (HA) metabolism regulation, whereby loss of CD44 expression disrupts HA metabolism and impairs hair regrowth, wound healing and keratinocyte proliferation [62].
3. CD44 Expression in Tumours
Numerous studies indicated that lymphoma, breast, colon and endometrial cancer have elevated levels of CD44 mRNA [60]. Increasing evidence also suggests that CD44 is extensively overexpressed in other cancer types including gallbladder, prostate, ovarian, oral squamous cell carcinoma and gastric cancer, correlating with aggressive biological behaviour and a poor prognosis [63]. The role of CD44 in tumours is not well defined, however, elevated levels of CD44 are associated with numerous malignant tumours. The physiological functions of CD44 indicate that it is involved in the metastasis of tumours [3]. For instance, lung adenocarcinoma cells show a high expression of CD44v, which correlates with enhanced CSCs characteristics, proliferation and resistance to chemotherapeutics [64], whereas these variants, especially CD44v6, are closely related to metastasis of pancreatic carcinoma cells [60]. Many studies have investigated CD44 expression levels in several cancers in comparison to their adjacent normal tissues and explored the relationship with tumour progression and clinicopathological outcomes by mining various publicly available databases, including The Cancer Genome Atlas (TCGA), Tumour Immune Estimation Resource (TIMER) database, Oncomine database, Gene Expression Profiling Interactive Analysis (GEPIA), In silico Transcriptomics (IST) database, R2 online database, SAGE Genie tools, and Human Gene Expression Map (HGEM) (Table 2 and Figure 3).
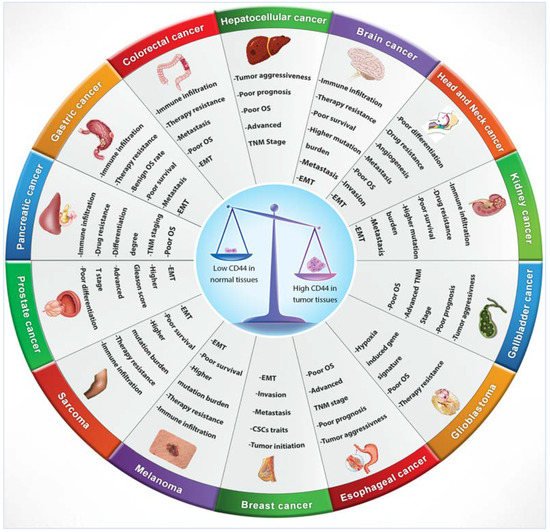
Figure 3. CD44 distribution in normal versus cancerous tissues and its correlation with clinical outcomes.
Table 2. Low and high CD44 expression in normal and tumour tissues respectively and association with clinical outcomes.
| Cancer Type | Correlation with Clinical Outcomes | Public Database | Reference |
|---|---|---|---|
| Gallbladder cancer, hepatocellular carcinoma, cholangiocarcinoma | Poor prognosis, advanced TNM stage, poor OS, aggressive tumour behaviour (proliferation, migration, invasion, clonogenicity) | TCGA database | [63] |
| Colon cancer, gastric cancer, brain cancer, stomach cancer, pancreatic cancer, liver cancer | Benign OS rate in gastric cancer, poor OS in colon cancer, TNM staging, differentiation degree, and poor survival in pancreatic cancer | SAGE Genie and Oncomine database | [65][66] |
| Head and neck squamous carcinoma | Poor OS, poor differentiation, angiogenesis, immune regulation, invasion | TCGA database | [67] |
| Head and neck squamous carcinoma | Pro-angiogenetic phenotype | TCGA database | [13] |
| Prostate cancer | Advanced T stage, higher Gleason score, poor differentiation | TCGA database | [68] |
| Colon adenocarcinoma | Therapy resistance | TCGA database and GEPIA | [69] |
| Head and neck squamous carcinoma, acute myeloid leukaemia (AML), lung carcinoma | Not specified | IST database and HGEM database | [70] |
| Glioblastoma | Poor OS, hypoxia-induced gene signature | TCGA database | [71] |
| Glioblastoma | Poor OS, therapy resistance | R2 online database | [72] |
| Invasive ductal breast carcinoma | Invasion, metastasis | TCGA database | [73] |
| Brain and CNS cancer, colorectal cancer, melanoma, sarcoma, gastric cancer, head and neck carcinoma, kidney cancer, oesophageal cancer, cholangiocarcinoma, pancreatic cancer | EMT, drug resistance, metastasis, immune infiltration and suppression features, poor survival, higher mutation burden, afflict older patients | Oncomine database and TIMER database | [74] |
References
- Loh, T.J.; Moon, H.; Cho, S.; Jang, H.; Liu, Y.C.; Tai, H.; Jung, D.W.; Williams, D.R.; Kim, H.R.; Shin, M.G.; et al. CD44 alternative splicing and hnRNP A1 expression are associated with the metastasis of breast cancer. Oncol. Rep. 2015, 34, 1231–1238.
- Rall, C.J.; Rustgi, A.K. CD44 isoform expression in primary and metastatic pancreatic adenocarcinoma. Cancer Res. 1995, 55, 1831–1835.
- Mishra, M.N.; Chandavarkar, V.; Sharma, R.; Bhargava, D. Structure, function and role of CD44 in neoplasia. J. Oral Maxillofac. Pathol. 2019, 23, 267–272.
- Tölg, C.; Hofmann, M.; Herrlich, P.; Ponta, H. Splicing choice from ten variant exons establishes CD44 variability. Nucleic Acids Res. 1993, 21, 1225–1229.
- Screaton, G.R.; Bell, M.V.; Jackson, D.G.; Cornelis, F.B.; Gerth, U.; Bell, J.I. Genomic structure of DNA encoding the lymphocyte homing receptor CD44 reveals at least 12 alternatively spliced exons. Proc. Natl. Acad. Sci. USA 1992, 89, 12160–12164.
- Zeilstra, J.; Joosten, S.P.; van Andel, H.; Tolg, C.; Berns, A.; Snoek, M.; Van De Wetering, M.; Spaargaren, M.; Clevers, H.; Pals, S.T. Stem cell CD44v isoforms promote intestinal cancer formation in Apc(min) mice downstream of Wnt signaling. Oncogene 2014, 33, 665–670.
- Goodison, S.; Urquidi, V.; Tarin, D. CD44 cell adhesion molecules. Mol. Pathol. 1999, 52, 189–196.
- Chen, K.L.; Li, D.; Lu, T.X.; Chang, S.W. Structural Characterization of the CD44 Stem Region for Standard and Cancer-Associated Isoforms. Int. J. Mol. Sci. 2020, 21, 336.
- Misra, S.; Hascall, V.C.; Markwald, R.R.; Ghatak, S. Interactions between Hyaluronan and Its Receptors (CD44, RHAMM) Regulate the Activities of Inflammation and Cancer. Front. Immunol. 2015, 6, 201.
- Liu, X.; Taftaf, R.; Kawaguchi, M.; Chang, Y.F.; Chen, W.; Entenberg, D.; Zhang, Y.; Gerratana, L.; Huang, S.; Patel, D.B.; et al. Homophilic CD44 Interactions Mediate Tumor Cell Aggregation and Polyclonal Metastasis in Patient-Derived Breast Cancer Models. Cancer Discov. 2019, 9, 96–113.
- Wolf, K.J.; Shukla, P.; Springer, K.; Lee, S.; Coombes, J.D.; Choy, C.J.; Kenny, S.J.; Xu, K.; Kumar, S. A mode of cell adhesion and migration facilitated by CD44-dependent microtentacles. Proc. Natl. Acad. Sci. USA 2020, 117, 11432–11443.
- Hassn Mesrati, M.; Behrooz, A.B.; Abuhamad, A.Y.; Syahir, A. Understanding Glioblastoma Biomarkers: Knocking a Mountain with a Hammer. Cells 2020, 9, 1236.
- Ludwig, N.; Szczepanski, M.J.; Gluszko, A.; Szafarowski, T.; Azambuja, J.H.; Dolg, L.; Gellrich, N.-C.; Kampmann, A.; Whiteside, T.L.; Zimmerer, R.M. CD44(+) tumor cells promote early angiogenesis in head and neck squamous cell carcinoma. Cancer Lett. 2019, 467, 85–95.
- Gzil, A.; Zarębska, I.; Bursiewicz, W.; Antosik, P.; Grzanka, D.; Szylberg, Ł. Markers of pancreatic cancer stem cells and their clinical and therapeutic implications. Mol. Biol. Rep. 2019, 46, 6629–6645.
- Durko, L.; Wlodarski, W.; Stasikowska-Kanicka, O.; Wagrowska-Danilewicz, M.; Danilewicz, M.; Hogendorf, P.; Strzelczyk, J.; Malecka-Panas, E. Expression and Clinical Significance of Cancer Stem Cell Markers CD24, CD44, and CD133 in Pancreatic Ductal Adenocarcinoma and Chronic Pancreatitis. Dis. Markers 2017, 2017, 3276806.
- Li, W.; Qian, L.; Lin, J.; Huang, G.; Hao, N.; Wei, X.; Wang, W.; Liang, J. CD44 regulates prostate cancer proliferation, invasion and migration via PDK1 and PFKFB4. Oncotarget 2017, 8, 65143–65151.
- Wang, Z.; Tang, Y.; Xie, L.; Huang, A.; Xue, C.; Gu, Z.; Wang, K.; Zong, S. The Prognostic and Clinical Value of CD44 in Colorectal Cancer: A Meta-Analysis. Front. Oncol. 2019, 9, 309.
- Holah, N.S.; Aiad, H.A.; Asaad, N.Y.; Elkhouly, E.A.; Lasheen, A.G. Evaluation of the role of CD44 as a cancer stem cell marker in colorectal carcinoma: Immunohistochemical study. Menoufia Med. J. 2017, 30, 174–183.
- Zhang, H.; Brown, R.L.; Wei, Y.; Zhao, P.; Liu, S.; Liu, X.; Deng, Y.; Hu, X.; Zhang, J.; Gao, X.D.; et al. CD44 splice isoform switching determines breast cancer stem cell state. Genes Dev. 2019, 33, 166–179.
- Zhao, P.; Xu, Y.; Wei, Y.; Qiu, Q.; Chew, T.L.; Kang, Y.; Cheng, C. The CD44s splice isoform is a central mediator for invadopodia activity. J. Cell Sci. 2016, 129, 1355–1365.
- Brown, R.L.; Reinke, L.M.; Damerow, M.S.; Perez, D.; Chodosh, L.A.; Yang, J.; Cheng, C. CD44 splice isoform switching in human and mouse epithelium is essential for epithelial-mesenchymal transition and breast cancer progression. J. Clin. Investig. 2011, 121, 1064–1074.
- Mima, K.; Okabe, H.; Ishimoto, T.; Hayashi, H.; Nakagawa, S.; Kuroki, H.; Watanabe, M.; Beppu, T.; Tamada, M.; Nagano, O.; et al. CD44s regulates the TGF-β-mediated mesenchymal phenotype and is associated with poor prognosis in patients with hepatocellular carcinoma. Cancer Res. 2012, 72, 3414–3423.
- Miwa, T.; Nagata, T.; Kojima, H.; Sekine, S.; Okumura, T. Isoform switch of CD44 induces different chemotactic and tumorigenic ability in gallbladder cancer. Int. J. Oncol. 2017, 51, 771–780.
- Chen, Q.; Gu, M.; Cai, Z.k.; Zhao, H.; Sun, S.C.; Liu, C.; Zhan, M.; Chen, Y.B.; Wang, Z. TGF-β1 promotes epithelial-to-mesenchymal transition and stemness of prostate cancer cells by inducing PCBP1 degradation and alternative splicing of CD44. Cell. Mol. Life Sci. 2021, 78, 949–962.
- Zhao, S.; Chen, C.; Chang, K.; Karnad, A.; Jagirdar, J.; Kumar, A.P.; Freeman, J.W. CD44 Expression Level and Isoform Contributes to Pancreatic Cancer Cell Plasticity, Invasiveness, and Response to Therapy. Clin. Cancer Res. 2016, 22, 5592–5604.
- Tsubouchi, K.; Minami, K.; Hayashi, N.; Yokoyama, Y.; Mori, S.; Yamamoto, H.; Koizumi, M. The CD44 standard isoform contributes to radioresistance of pancreatic cancer cells. J. Radiat. Res. 2017, 58, 816–826.
- Ozawa, M.; Ichikawa, Y.; Zheng, Y.W.; Oshima, T.; Miyata, H.; Nakazawa, K.; Guan, H.B.; Shiozawa, M.; Akaike, M.; Watanabe, K.; et al. Prognostic significance of CD44 variant 2 upregulation in colorectal cancer. Br. J. Cancer 2014, 111, 365–374.
- Gotoda, T.; Matsumura, Y.; Kondo, H.; Saitoh, D.; Shimada, Y.; Kosuge, T.; Kanai, Y.; Kakizoe, T. Expression of CD44 variants and its association with survival in pancreatic cancer. Jpn. J. Cancer Res. 1998, 89, 1033–1040.
- Todoroki, K.; Ogasawara, S.; Akiba, J.; Nakayama, M.; Naito, Y.; Seki, N.; Kusukawa, J.; Yano, H. CD44v3+/CD24- cells possess cancer stem cell-like properties in human oral squamous cell carcinoma. Int. J. Oncol. 2016, 48, 99–109.
- Holm, F.; Hellqvist, E.; Mason, C.N.; Ali, S.A.; Delos-Santos, N.; Barrett, C.L.; Chun, H.J.; Minden, M.D.; Moore, R.A.; Marra, M.A.; et al. Reversion to an embryonic alternative splicing program enhances leukemia stem cell self-renewal. Proc. Natl. Acad. Sci. USA 2015, 112, 15444–15449.
- Bánky, B.; Rásó-Barnett, L.; Barbai, T.; Tímár, J.; Becságh, P.; Rásó, E. Characteristics of CD44 alternative splice pattern in the course of human colorectal adenocarcinoma progression. Mol. Cancer 2012, 11, 83.
- Spiegelberg, D.; Kuku, G.; Selvaraju, R.; Nestor, M. Characterization of CD44 variant expression in head and neck squamous cell carcinomas. Tumour Biol. 2014, 35, 2053–2062.
- Endo, K.; Terada, T. Protein expression of CD44 (standard and variant isoforms) in hepatocellular carcinoma: Relationships with tumor grade, clinicopathologic parameters, p53 expression, and patient survival. J. Hepatol. 2000, 32, 78–84.
- Okuyama, K.; Fukushima, H.; Naruse, T.; Yanamoto, S.; Tsuchihashi, H.; Umeda, M. CD44 Variant 6 Expression and Tumor Budding in the Medullary Invasion Front of Mandibular Gingival Squamous Cell Carcinoma Are Predictive Factors for Cervical Lymph Node Metastasis. Pathol. Oncol. Res. 2019, 25, 603–609.
- Ni, J.; Cozzi, P.J.; Hao, J.L.; Beretov, J.; Chang, L.; Duan, W.; Shigdar, S.; Delprado, W.J.; Graham, P.H.; Bucci, J.; et al. CD44 variant 6 is associated with prostate cancer metastasis and chemo-/radioresistance. Prostate 2014, 74, 602–617.
- Yanamoto, S.; Yamada, S.; Takahashi, H.; Naruse, T.; Matsushita, Y.; Ikeda, H.; Shiraishi, T.; Seki, S.; Fujita, S.; Ikeda, T.; et al. Expression of the cancer stem cell markers CD44v6 and ABCG2 in tongue cancer: Effect of neoadjuvant chemotherapy on local recurrence. Int. J. Oncol. 2014, 44, 1153–1162.
- Masaki, T.; Goto, A.; Sugiyama, M.; Matsuoka, H.; Abe, N.; Sakamoto, A.; Atomi, Y. Possible contribution of CD44 variant 6 and nuclear beta-catenin expression to the formation of budding tumor cells in patients with T1 colorectal carcinoma. Cancer 2001, 92, 2539–2546.
- Li, Z.; Chen, K.; Jiang, P.; Zhang, X.; Li, X.; Li, Z. CD44v/CD44s expression patterns are associated with the survival of pancreatic carcinoma patients. Diagn. Pathol. 2014, 9, 79.
- Kainz, C.; Kohlberger, P.; Tempfer, C.; Sliutz, G.; Gitsch, G.; Reinthaller, A.; Breitenecker, G. Prognostic value of CD44 splice variants in human stage III cervical cancer. Eur. J. Cancer 1995, 31, 1706–1709.
- Yakushijin, Y.; Steckel, J.; Kharbanda, S.; Hasserjian, R.; Neuberg, D.; Jiang, W.; Anderson, I.; Shipp, M.A. A directly spliced exon 10-containing CD44 variant promotes the metastasis and homotypic aggregation of aggressive non-Hodgkin’s lymphoma. Blood 1998, 91, 4282–4291.
- Li, D.M.; Li, S.S.; Zhang, Y.H.; Zhang, H.J.; Gao, D.L.; Wang, Y.X. Expression of human chorionic gonadotropin, CD44v6 and CD44v4/5 in esophageal squamous cell carcinoma. World J. Gastroenterol. 2005, 11, 7401–7404.
- Lourenço, B.N.; Springer, N.L.; Ferreira, D.; Oliveira, C.; Granja, P.L.; Fischbach, C. CD44v6 increases gastric cancer malignant phenotype by modulating adipose stromal cell-mediated ECM remodeling. Integr. Biol. 2018, 10, 145–158.
- Suwannakul, N.; Ma, N.; Midorikawa, K.; Oikawa, S.; Kobayashi, H.; He, F.; Kawanishi, S.; Murata, M. CD44v9 Induces Stem Cell-Like Phenotypes in Human Cholangiocarcinoma. Front. Cell Dev. Biol. 2020, 8, 417.
- Kobayashi, K.; Matsumoto, H.; Matsuyama, H.; Fujii, N.; Inoue, R.; Yamamoto, Y.; Nagao, K. Clinical significance of CD44 variant 9 expression as a prognostic indicator in bladder cancer. Oncol. Rep. 2016, 36, 2852–2860.
- Li, N.; Tsuji, M.; Kanda, K.; Murakami, Y.; Kanayama, H.; Kagawa, S. Analysis of CD44 isoform v10 expression and its prognostic value in renal cell carcinoma. BJU Int. 2000, 85, 514–518.
- Iida, N.; Bourguignon, L.Y. Coexpression of CD44 variant (v10/ex14) and CD44S in human mammary epithelial cells promotes tumorigenesis. J. Cell. Physiol. 1997, 171, 152–160.
- Iida, J.; Clancy, R.; Dorchak, J.; Somiari, R.I.; Somiari, S.; Cutler, M.L.; Mural, R.J.; Shriver, C.D. DNA aptamers against exon v10 of CD44 inhibit breast cancer cell migration. PLoS ONE 2014, 9, e88712.
- Tran, T.A.; Kallakury, B.V.; Sheehan, C.E.; Ross, J.S. Expression of CD44 standard form and variant isoforms in non-small cell lung carcinomas. Hum. Pathol. 1997, 28, 809–814.
- Zhao, P.; Damerow, M.S.; Stern, P.; Liu, A.H.; Sweet-Cordero, A.; Siziopikou, K.; Neilson, J.R.; Sharp, P.A.; Cheng, C. CD44 promotes Kras-dependent lung adenocarcinoma. Oncogene 2013, 32, 5186–5190.
- Dall, P.; Hekele, A.; Ikenberg, H.; Göppinger, A.; Bauknecht, T.; Pfleiderer, A.; Moll, J.; Hofmann, M.; Ponta, H.; Herrlich, P. Increasing incidence of CD44v7/8 epitope expression during uterine cervical carcinogenesis. Int. J. Cancer 1996, 69, 79–85.
- Omara-Opyene, A.L.; Qiu, J.; Shah, G.V.; Iczkowski, K.A. Prostate cancer invasion is influenced more by expression of a CD44 isoform including variant 9 than by Muc18. Lab. Investig. 2004, 84, 894–907.
- Hiraga, T.; Nakamura, H. Comparable roles of CD44v8-10 and CD44s in the development of bone metastases in a mouse model. Oncol. Lett. 2016, 12, 2962–2969.
- Lau, W.M.; Teng, E.; Chong, H.S.; Lopez, K.A.; Tay, A.Y.; Salto-Tellez, M.; Shabbir, A.; So, J.B.; Chan, S.L. CD44v8-10 is a cancer-specific marker for gastric cancer stem cells. Cancer Res. 2014, 74, 2630–2641.
- Yae, T.; Tsuchihashi, K.; Ishimoto, T.; Motohara, T.; Yoshikawa, M.; Yoshida, G.J.; Wada, T.; Masuko, T.; Mogushi, K.; Tanaka, H.; et al. Alternative splicing of CD44 mRNA by ESRP1 enhances lung colonization of metastatic cancer cell. Nat. Commun. 2012, 3, 883.
- Choi, E.S.; Kim, H.; Kim, H.P.; Choi, Y.; Goh, S.H. CD44v8-10 as a potential theranostic biomarker for targeting disseminated cancer cells in advanced gastric cancer. Sci. Rep. 2017, 7, 4930.
- Kagami, T.; Yamade, M.; Suzuki, T.; Uotani, T.; Tani, S.; Hamaya, Y.; Iwaizumi, M.; Osawa, S.; Sugimoto, K.; Baba, S.; et al. High expression level of CD44v8-10 in cancer stem-like cells is associated with poor prognosis in esophageal squamous cell carcinoma patients treated with chemoradiotherapy. Oncotarget 2018, 9, 34876–34888.
- Hagiwara, M.; Kikuchi, E.; Tanaka, N.; Kosaka, T.; Mikami, S.; Saya, H.; Oya, M. Variant isoforms of CD44 involves acquisition of chemoresistance to cisplatin and has potential as a novel indicator for identifying a cisplatin-resistant population in urothelial cancer. BMC Cancer 2018, 18, 113.
- Olsson, E.; Honeth, G.; Bendahl, P.O.; Saal, L.H.; Gruvberger-Saal, S.; Ringnér, M.; Vallon-Christersson, J.; Jönsson, G.; Holm, K.; Lövgren, K.; et al. CD44 isoforms are heterogeneously expressed in breast cancer and correlate with tumor subtypes and cancer stem cell markers. BMC Cancer 2011, 11, 418.
- Bourguignon, L.Y.; Zhu, H.; Shao, L.; Zhu, D.; Chen, Y.W. Rho-kinase (ROK) promotes CD44v (3,8-10)-ankyrin interaction and tumor cell migration in metastatic breast cancer cells. Cell Motil. Cytoskelet. 1999, 43, 269–287.
- Fujita, N.; Yaegashi, N.; Ide, Y.; Sato, S.; Nakamura, M.; Ishiwata, I.; Yajima, A. Expression of CD44 in normal human versus tumor endometrial tissues: Possible implication of reduced expression of CD44 in lymph-vascular space involvement of cancer cells. Cancer Res. 1994, 54, 3922–3928.
- Ruiz, P.; Schwärzler, C.; Günthert, U. CD44 isoforms during differentiation and development. BioEssays 1995, 17, 17–24.
- Yu, Q.; Stamenkovic, I. Localization of matrix metalloproteinase 9 to the cell surface provides a mechanism for CD44-mediated tumor invasion. Genes Dev. 1999, 13, 35–48.
- He, Y.; Xue, C.; Yu, Y.; Chen, J.; Chen, X.; Ren, F.; Ren, Z.; Cui, G.; Sun, R. CD44 is overexpressed and correlated with tumor progression in gallbladder cancer. Cancer Manag. Res. 2018, 10, 3857–3865.
- Nishino, M.; Ozaki, M.; Hegab, A.E.; Hamamoto, J.; Kagawa, S.; Arai, D.; Yasuda, H.; Naoki, K.; Soejima, K.; Saya, H.; et al. Variant CD44 expression is enriching for a cell population with cancer stem cell-like characteristics in human lung adenocarcinoma. J. Cancer 2017, 8, 1774–1785.
- Xia, P.; Xu, X.Y. Prognostic significance of CD44 in human colon cancer and gastric cancer: Evidence from bioinformatic analyses. Oncotarget 2016, 7, 45538–45546.
- Li, X.P.; Zhang, X.W.; Zheng, L.Z.; Guo, W.J. Expression of CD44 in pancreatic cancer and its significance. Int. J. Clin. Exp. Pathol. 2015, 8, 6724–6731.
- Mishra, A.; Sriram, H.; Chandarana, P.; Tanavde, V.; Kumar, R.V.; Gopinath, A.; Govindarajan, R.; Ramaswamy, S.; Sadasivam, S. Decreased expression of cell adhesion genes in cancer stem-like cells isolated from primary oral squamous cell carcinomas. Tumour Biol. 2018, 40, 1010428318780859.
- Carneiro, I.; Quintela-Vieira, F.; Lobo, J.; Moreira-Barbosa, C.; Menezes, F.D.; Martins, A.T.; Oliveira, J.; Silva, R.; Jerónimo, C.; Henrique, R. Expression of EMT-Related Genes CAMK2N1 and WNT5A is increased in Locally Invasive and Metastatic Prostate Cancer. J. Cancer 2019, 10, 5915–5925.
- Dzobo, K.; Senthebane, D.A.; Ganz, C.; Thomford, N.E. The significance of Cancer stem cell markers’ gene expression and Relevance for Survival Outcomes. OMICS J. Integr. Biol. 2020. Preprint.
- Rajarajan, A.; Stokes, A.; Bloor, B.K.; Ceder, R.; Desai, H.; Grafström, R.C.; Odell, E.W. CD44 expression in oro-pharyngeal carcinoma tissues and cell lines. PLoS ONE 2012, 7, e28776.
- Pietras, A.; Katz, A.M.; Ekström, E.J.; Wee, B.; Halliday, J.J.; Pitter, K.L.; Werbeck, J.L.; Amankulor, N.M.; Huse, J.T.; Holland, E.C. Osteopontin-CD44 signaling in the glioma perivascular niche enhances cancer stem cell phenotypes and promotes aggressive tumor growth. Cell Stem Cell 2014, 14, 357–369.
- Wang, F.; Zheng, Z.; Guan, J.; Qi, D.; Zhou, S.; Shen, X.; Wang, F.; Wenkert, D.; Kirmani, B.; Solouki, T.; et al. Identification of a panel of genes as a prognostic biomarker for glioblastoma. EBioMedicine 2018, 37, 68–77.
- Liu, M.Y.; Su, H.; Huang, H.L.; Chen, J.Q. Cancer stem-like cells with increased expression of NY-ESO-1 initiate breast cancer metastasis. Oncol. Lett. 2019, 18, 3664–3672.
- Dzobo, K.; Sinkala, M. The Cancer Stem Cell Marker CD44 Play Key Roles in Immune Suppression/Evasion, Drug Resistance, Epithelial-Mesenchymal Transition, and Metastasis in Several Human Cancers. OMICS J. Integr. Biol. 2021, 2021, 2021010490.
More
Information
Subjects:
Anatomy & Morphology
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
2.0K
Entry Collection:
Biopharmaceuticals Technology
Revisions:
4 times
(View History)
Update Date:
28 Dec 2021
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





