Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Li, S. Bamboo Node’s Vascular Bundle. Encyclopedia. Available online: https://encyclopedia.pub/entry/17479 (accessed on 01 June 2026).
Li S. Bamboo Node’s Vascular Bundle. Encyclopedia. Available at: https://encyclopedia.pub/entry/17479. Accessed June 01, 2026.
Li, Shan. "Bamboo Node’s Vascular Bundle" Encyclopedia, https://encyclopedia.pub/entry/17479 (accessed June 01, 2026).
Li, S. (2021, December 23). Bamboo Node’s Vascular Bundle. In Encyclopedia. https://encyclopedia.pub/entry/17479
Li, Shan. "Bamboo Node’s Vascular Bundle." Encyclopedia. Web. 23 December, 2021.
Copy Citation
The vascular bundle is an important structural unit that determines the growth and properties of bamboo. A high-resolution X-ray microtomography (μCT) was used to observe and reconstruct a three-dimensional (3D) morphometry model of the vascular bundle of the Qiongzhuea tumidinoda node due to its advantages of quick, nondestructive, and accurate testing of plant internal structure.
Qiongzhuea tumidinoda
structure of bamboo node
three-dimensional reconstruction
1. Introduction
The node is the basic characteristic of bamboo plants and plays a key role in the overall performance of the hollow structure of bamboo culms [1][2]. The vascular bundle serves as a mechanical support to improve the mechanical properties of bamboo [2][3] and perform the function of transporting water and nutrients [4]. Numerous reports are available on the microstructure of the bamboo culm, mainly on the internodes [5][6][7]. However, the research concerning the structure of bamboo nodes due to its disordered and relatively complicated arrangement of vascular bundles is limited. The research on the structure of bamboo nodes began in the 1960s [8] and mainly relied on optical microscopes. In the late 20th century, the researches related to the anatomy of bamboo nodes gradually increased [4][9][10][11]. Grossor and Liese [11] studied the characteristics of vascular bundles of bamboo nodes through enlarged and spliced images of microsections. Xiong et al. studied the shape of bamboo nodes and conducted a preliminary discussion on the 3D characteristics of vascular bundles [10][12]. Ding and Liese [4] reconstructed a complex 3D structure of vascular bundles of bamboo nodes by observing continuous longitudinal and transverse slices. Currently, the research of the bamboo microstructure mainly relies on two-dimensional (2D) image characterization techniques such as optical microscope and field emission scanning electron microscope. However, the aforementioned methods damaged the samples, leading to the inaccurate results. The complexity and fragility of sample preparation with traditional techniques make it difficult to meet the requirements of nondestructive, high-efficiency visualization. Therefore, a higher-resolution nondestructive testing method is needed.
With the continuous development of research methods, X-ray microscopy has strong penetrating power, high resolution, and nondestructive property, resulting in fast detection and intuitive results, and indicating advantages of bamboo node analysis. Peng et al. [13] and Xiang et al. [14] used X-ray microtomography (μCT) as a tool for 2D and 3D complex network structures of the vascular bundle in the bamboo node. Huang et al. quantified the difference in the density distribution and the porosity of bamboo nodes based on 2D images [15][16]. Palombinifl et al. [17][18][19] analyzed the relative density and cell shape of different tissues of a cutting region of bamboo nodes, evaluated the mechanical behavior of parenchyma by finite element analysis, and first obtained the 3D reconstruction model of the complex vascular bundle system of monocot nodes. The volume, porosity, relative density, and distribution of nodes were also characterized. The aforementioned research by X-ray technique had two limitations: (1) The conducting tissue and the fibers of the completed node’s vascular bundle were not separated to separately observe the morphology of them. (2) The complex distribution of transverse vascular bundles in the diaphragm was neglected.
Qiongzhuea tumidinoda Hsueh et T.P.Yi. belongs to the genus Qiongzhuea in the Gramineae family, with an enlarged node and a peculiar-shaped culm. The species reaches approximately 2.5–6 m in height, and its culm reaches 1–3 cm in diameter. Q. tumidinoda, only found in a restricted area in S.W. Sichuan and N.E. Yunnan, is a rare and endangered bamboo species, which is a Class 3 protected plant on the national key protection list in China. Among them, especially in Daguan County, Yunnan province, this bamboo accounts for more than 50% of the total global resources, with 0.014 million hectares. The bamboo culm is mainly used for crafts and production of round bamboo furniture with extremely high economic and ornamental value. However, this bamboo at more than 5-years of age will gradually begin to wither and die. The most obvious feature is that the node automatically disconnects. Furthermore, the culm is more prone to regular fracture at the nodal ridge of the node, even less than 5-year-old bamboo. The presence of the bamboo node makes it possible for the lateral transport of water and nutrients [4], and there is little research on what happened to its structure before the node fell off.
2. Materials
Five 2-year-old Q. tumidinoda bamboo plants with good growth and straight stems were collected from Yinji Village, Daguan County, Yunnan Province (103°99′ N, 28°13′ E). The culm was cut every 2 m from the base to the top of it, marked, and transported to the laboratory. After natural air drying, complete bamboo nodes with 5.74% moisture content from the middle of the culm were selected (Figure 1b) and were ready to be used for the subsequent experiments. The selected node was approximately 1.83 m of the culm, which was 3.7 m in height and 1.2 cm in diameter.

Figure 1. Preparation of μCT sample. (a) Bamboo. (b) Sampling location (white wireframe area).
3. Image Processing and 3D Reconstruction
Images were processed and analyzed with the open-source Fiji software (National Institutes of Health, Bethesda, MD, USA) and the AVIZO software (FEI Hillsboro, OR, USA), the processing steps are shown in Figure 2. The slices were combined and reconstructed from the projections with a noniterative filtered backprojection algorithm in NRecon software and then exported as a stack of TIFF images. A total of 1290 slices with serial numbers 329–1618 were obtained. The original TIFF stack (Figure 2a) was first denoised with the adaptive median filtering technique combined with the non-local mean filtering algorithm (Figure 2b), and the contrast was adjusted (Figure 2c). Figure 2a1,b1,c1 demonstrates details of the same slice before and after noise reduction and contrast adjustment, respectively. The denoised intensity images had clear boundaries for easy segmentation.

Figure 2. Image processing of μCT. (a) Original μCT slice and its details (a1). (b) Filter processing and its details (b1). (c) Contrast adjusting and its details (c1). (d) Segmentation of the conducting tissue (yellow), fibers (purple), parenchyma (green), and air (red). (e) Binary images of fibers. (f) Binary images of conducting tissue.
For a better visualization in addition to automatic recognition of different tissues in the nodal region using the machine learning segmentation plug-in [20] in ImageJ (FIJI is just ImageJ), the mask of each tissue type with threshold segmentation were corrected manually to obtain binarized images of fibers and conducting tissues (Figure 2e,f). At last, the AVIZO software was used to generate the skeleton and the surface mesh of fibers and conducting tissue. The skeleton was obtained by calculating a distance map of the segmented image and then by thinning the binary image such that a final string of connected voxels remained. The voxel skeleton was then converted into a Spatial Graph object. The distance to the nearest boundary was stored at every point in the Spatial Graph object as a thickness attribute [21]. The surface mesh model was used to generate a smoother surface to compensate for the information loss caused by the resampling process [22]. Some slices (slice 339, slice 387, slice 579, slice 669, slice 849, slice 944, slice 1129, slice 1429) appeared as new and particular features relative to its bottom slice, which were used for observing the morphology of them.
4. Morphology of the Bamboo Node
The morphological diagram of the node of Q. tumidinoda is shown in Figure 3a, which consists of a sheath scar, nodal ridge, diaphragm, and the intra-node between the nodal ridge and sheath scar. The rare swollen node was seen in Q. tumidinoda bamboo, the nodal ridge was more swollen than the sheath scar. This might be related to the different speeds of cell division and growth around the intercalary meristem [12].
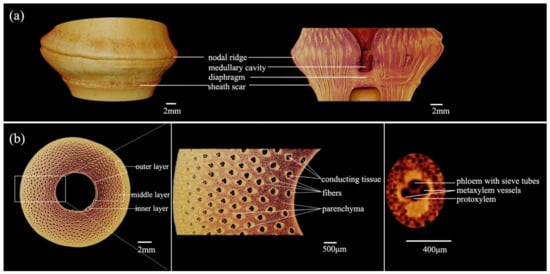
Figure 3. Morphology of the node in Q. tumidinoda. (a) Longitudinal section view. (b) Cross-section and details.
The cross-section of bamboo was divided into the following three parts according to the position in the bamboo culm: outer layer, middle layer, and inner layer (Figure 3b). It was also classified into parenchyma, fibers, and conducting tissue (metaxylem vessels, phloem with sieve tubes, protoxylem) by tissue type. The vascular bundle was mainly composed of conducting tissue and fibers. The conducting tissue transported water and nutrients from the roots to different parts of the bamboo. The fibers were relatively hard and provided mechanical support, while the parenchyma with a thin cell wall served as a reservoir and a buffer, they cooperated to enhance the elasticity of the bamboo culm [23].
No other solid part was observed in the nodal area except in the diaphragm from the cross-sectional view of a bamboo node with different heights (Figure 4a1–h1). Only the diaphragm was solid in the node, indicating disagreement with the previous results [24] and that the solid area appeared repeatedly in the node of Q. tumidinoda. This might be caused by the difference in the environment and position of the culm.
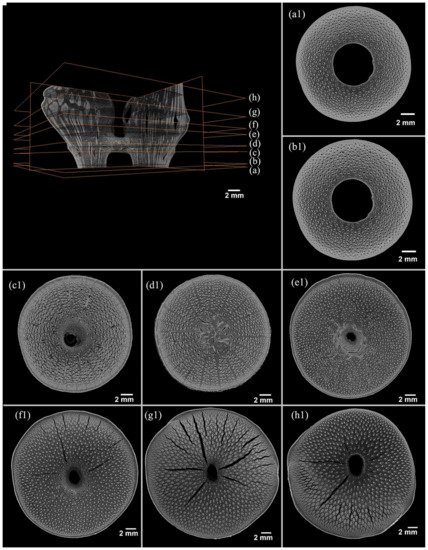
Figure 4. X-ray microtomography slices of the Q. tumidinoda node. The sequence number of the transversal slices is 329–1618 from the lower end to the upper end (details of a–h present in a1–h1). (a1) Slice 339, end of the node. (b1) Slice 387, lower end of the node. (c1) Slice 579, lower end of the sheath scar. (d1) Slice 669, middle of the diaphragm. (e1) Slice 849, upper end of the diaphragm. (f1) Slice 944, lower end of the nodal ridge. (g1) Slice 1129, middle of the nodal ridge, and (h1) Slice 1429, upper end of the nodal ridge.
References
- Liu, S.; Tong, Z.; Tang, Z.; Yang, L.; Zhang, Z. Bionic design modification of non-convex multi-corner thin-walled columns for improving energy absorption through adding bulkheads. Thin Wall Struct. 2015, 88, 70–81.
- Zou, M.; Xu, S.; Wei, C.; Wang, H.; Liu, Z. A bionic method for the crashworthiness design of thin-walled structures inspired by bamboo. Thin Wall Struct. 2016, 101, 222–230.
- Wang, F.; Shao, Z.; Wu, Y.; Wu, D. The Toughness Contribution of Bamboo Node to the Mode I Interlaminar Fracture Toughness of Bamboo. Wood Sci. Technol. 2014, 48, 1257–1268.
- Ding, Y.L.; Liese, W. On the nodal structure of bamboo. Bamboo Res. 1995, 14, 24–32.
- Burgert, I. Exploring the Micromechanical Design of Plant Cell Walls. Am. J. Bot. 2006, 93, 1391–1401.
- Wansi, F.; Zhangrong, Z.; Wang, H.; Jianbo, Z. Research on finite element model for parallel to bamboo culms axial shear. Appl. Mech. Mater. 2014, 477, 986–989.
- Ghysels, P.; Samaey, G.; Tijskens, B.; Liedekerke, P.V.; Ramon, H.; Roose, D. Multi-scale simulation of plant tissue deformation using a model for individual cell mechanics. Phys. Biol. 2009, 6, 016009.
- Lee, C.L.; Chin, T.C. Anatomical Studies Of Some Chinese Bamboos. J. Integr. Plant Biol. 1960, 9, 76–97.
- Ding, Y.L.; Fang, R.W.; Huang, J.S. Development and Ultrastructure of the Phloem Ganglion in Bamboo Node. Acta Bot. Sin. 2000, 42, 1009–1013.
- Xiong, W.Y.; Qiao, S.Y.; Li, Y.F. The Anatomical Structure Of Culms Of Phyllostachys pubescens Mazel ex H.de Lehaie. Acta Bot. Sin. 1980, 22, 343–348.
- Grosser, D.; Liese, W. On the Anatomy of Asian Bamboos, with Special Reference to Their Vascular Bundles. Wood Sci. Technol. 1971, 5, 290–312.
- Xiong, W.Y.; Ding, Z.F.; Li, Y.F. Intercalary Meristem And Internodal Elongation Of Bamboo Plants. Sci. Silvae Sin. 1980, 16, 81–89.
- Peng, G.Y.; Jiang, Z.H.; Liu, X.E.; Fei, B.H.; Yang, S.M. Detection of complex vascular system in bamboo node by X-ray μct imaging technique. Holzforschung 2014, 68, 223–227.
- Xiang, E.; Yang, S.; Cao, C.; Liu, X.; Peng, G.; Shang, L.; Tian, G.; Ma, Q.; Ma, J. Visualizing complex anatomical structure in bamboo nodes based on x-ray microtomography. J. Renew. Mater. 2021, 9, 1531–1540.
- Huang, P.; Chang, W.S.; Ansell, M.P.; Chew, Y.; Shea, A. Density distribution profile for internodes and nodes of phyllostachys edulis (moso bamboo) by computer tomography scanning. Constr. Build. Mater. 2015, 93, 197–204.
- Huang, P.; Chang, W.S.; Ansell, M.P.; John, C.; Shea, A. Porosity estimation of phyllostachys edulis (moso bamboo) by computed tomography and backscattered electron imaging. J. Wood Sci. Technol. 2017, 51, 11–27.
- Palombini, F.L.; Kindlein, W.; De Oliveira, B.F.; Ernesto, D.A.M.J. Bionics and design: 3d microstructural characterization and numerical analysis of bamboo based on x-ray microtomography. Mater. Charact. 2016, 120, 357–368.
- Palombini, F.L.; Nogueira, F.M.; Junior, K.W.; Paciornik, S.; Mariath, J.E.D.A.; Olivwira, B.F.D. Biomimetic Systems and Design in the 3D Characterization of the Complex Vascular System of Bamboo Node Based on X-Ray Microtomography and Finite Element Analysis. J. Mater. Res. 2020, 35, 842–854.
- Palombini, F.L.; Lautert, E.L.; Mariath, J.; Oliveira, B. Combining numerical models and discretizing methods in the analysis of bamboo parenchyma using finite element analysis based on x-ray microtomography. Wood Sci. Technol. 2020, 54, 161–186.
- Ignacio, A.C.; Verena, K.; Curtis, R.; Eliceiri, K.W.; Johannes, S.; Albert, C.; Seung, H.S. Trainable weka segmentation: A machine learning tool for microscopy pixel classification. Bioinformatics. Bioinformatics 2017, 33, 2424–2426.
- Fouard, C.; Malandain, G.; Prohaska, S.; Westerhoff, M. Blockwise processing applied to brain microvascular network study. IEEE Trans. Med. Imaging 2006, 25, 1319–1328.
- Hege, H.C.; Stalling, D.; Seebass, M.; Zockler, M. A Generalized Marching Cubes Algorithm Based on Non-Binary Classifications; Preprint SC 97-05; ZIB: Berlin, Germany, 1997.
- Wen, T.H.; Zhou, W.W. A Report on the Anatomy of the Vascular Bundle of Bamboos from China. Bamboo Res. 1984, 3, 1–21.
- Peng, G.Y. Study on Detection of Structure Characteristics for Wood and Bamboo Using Computed Tomography. Ph.D. Thesis, Chinese Academy of Forestry, Beijing, China, 2010.
More
Information
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
4.5K
Revisions:
2 times
(View History)
Update Date:
23 Dec 2021
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





