
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Marco F. L. Lemos | + 3809 word(s) | 3809 | 2021-12-02 04:11:19 | | | |
| 2 | Beatrix Zheng | + 1204 word(s) | 5013 | 2021-12-16 02:50:15 | | | | |
| 3 | Beatrix Zheng | Meta information modification | 5013 | 2021-12-17 15:32:02 | | |
Video Upload Options
Fungal phytopathogens are a growing problem all over the world; their propagation causes significant crop losses, affecting the quality of fruits and vegetables, diminishing the availability of food, leading to the loss of billions of euros every year. To control fungal diseases, the use of synthetic chemical fungicides is widely applied; these substances are, however, environmentally damaging. Marine algae, one of the richest marine sources of compounds possessing a wide range of bioactivities, present an eco-friendly alternative in the search for diverse compounds with industrial applications.
1. Introduction
Plant pests pose a paramount problem that has been increasing in recent years. The exact production losses due to these phytopathogens are hard to quantify but it is estimated that plant pests account for 20–40% of annual crop production losses [1][2], at a cost of more than 185 billion euros [3]. Included among these pests, fungal pathogens are one of the most damaging agents in plants, accounting for the devastation of myriad fruits and crops, which results in vast economic losses [4], and ultimately reduces food availability for a continuously increasing world population [5][6]. In fact, diseases provoked by fungi or related microorganisms have already caused starvation scenarios, such as the Irish Potato Famine in the 19th century, caused by a fungal-like oomycete, which led to a million of deaths, mass emigration, and economical and political crisis in Ireland [7][8]. Phytopathogenic fungi were also responsible for the baring of landscapes caused by Dutch elm blight and chestnut blight [8] and the complete ruin of 30% of world food crops in 2012 [3]. Currently, it is predicted that phytopathogenic fungi are responsible for about 80% of plant diseases [9][10][11], for which the absence of control can lead to disastrous global crop losses [6][12]. Even the remaining crops, potentially infected but without symptoms, can raise concerns about consumption safety [13]. Moreover, current and forecasted climatic change scenarios, leading to the increase of temperature and humidity, are crucial conditions promoting the dispersion and development of phytopathogenic fungi, giving cause for extra concerns [12][14].
The regular application of agrochemicals with antimicrobial properties is the most effective method against these microbial phytopathogens, but it is expensive and environmentally harmful, prevailing in the ecosystem and damaging it [15][16]. Every year, farmers spend more than 6 billion euros on such products to control the microbial infections, which represents a quarter of the costs for agricultural purposes [17]. For sustainability reasons, novel alternative methods have been sought that will have the same effectiveness, improve agricultural techniques, and enhance food production, ensuring the quality and security of food [18]. Several techniques and methodologies have been tested to minimize plant and financial losses either by directly targeting the microbial phytopathogens or by preventive measures, conferring resistance to the plant hosts. The laboratory manipulation of synthetic compounds to increase the effectiveness of products [19] or the introduction of “site-specific fungicides” [20] to control the most problematic and common microbial pathogens, have been suggested. Nevertheless, these products remain inefficient due to the great genetic resources and adaptative abilities of phytopathogens, which allow them to acquire resistance and overcome the efficiency of these types of products [20][21]. The biocontrol technique, characterized by the introduction of an antagonist microbial organism, harmless to the host but damaging for the phytopathogen [14], has been tested in vitro [2][22][23][24][25][26] and shown a great potential in field applications [2]. This methodology is characterized by the absence of chemicals, providing a viable and sustainable agriculture [27]. Although some limitations associated with the establishment and maintenance of biocontrol agents have been identified [2], including their interaction with the plant microbial community [28], the continuous stress conditions provoked in the host plant, the inconsistent results among tests [14][29][30], and the poor effectiveness compared to chemical fungicides, are factors which could and should be improved [29][30][31]. Though their potential can be enhanced through their combination with chemical interventions [28][32], this fails to solve the harm these compounds pose to the environment. The exploitation of genetic manipulation to alter the plant host genome with the insertion of resistance genes [33] was quickly shown to be ineffective against non-target phytopathogenic microorganisms and/or the emergence of new microbial races [15]. Therefore, the continuous search for biodegradable natural compounds, eco-friendly and effective against phytopathogenic microorganisms, is paramount [34], promising as it does to enhance food production and ensure the quality and security of agricultural products [18].
Marine habitats have been increasingly investigated due to the potential of bioactive products synthesized by the micro- and macro-organisms inhabiting them [35] being used in medicine and industry [36]. Seaweeds are one of the most attractive sources of bioactive substances due to their unique and diversified production of phenolic compounds, polysaccharides, fatty acids, and pigments. It is known that macroalgal applications have the potential to go beyond the ongoing uses in cosmetics, agricultural fertilizers, and the food industry [37]. Marine algae have revealed interesting compounds with antibiotic activity against pathogenic bacteria and fungi. Polysaccharides, polyphenols, carotenoids, proteins, peptides, sterols, terpenes, and fatty acids, among others, are the main constituents of algae that are associated with the antimicrobial properties of seaweed extracts [38][39][40]. Moreover, some of these algae compounds are capable of stimulating the natural defences of plants and promoting their resistance against microbial attacks, exhibiting a priming potential [39][41].
Considering the problems referred to above and the constant reduction of the effectiveness of available eco-friendly methodologies, given the promising results of in vitro assays, macroalgae constitute a source of diverse and natural compounds with antimicrobial potential against phytopathogenic fungi. Given this framework, the present review focuses on the potential of macroalgae-derived products, aiming to combine the available information regarding the potential/activity of fungal phytopathogen inhibition, while trying to clarify/link some “compound mode-of-action” and provide help and insights for future research into antimicrobial products derived from seaweeds.
2. Macroalgae Potential in the Eradication of Fungal Infections in Plants
2.1. Phytopathogenic Fungi
| Fungal Genera | Host Plant | References |
|---|---|---|
| Alternaria | Fruit plants, such as tomato (Lycopersicon esculentum) and apple (Malus domestica) | [49][57][58][59] |
| Aspergillus | Seeds, nuts, and fruits of a wide range of plant species | [57][58][60][61][62] |
| Botrytis | Wide range of plant hosts | [57][63][64] |
| Colletotrichum | Mediterranean plants and trees (fruits), tropical species and vegetables | [42][47][65][66][67][68][69] |
| Fusarium | The broad range of hosts include mono- and dicotyledons in greenhouses, cereals crops, and other plant species, such as tomato, upland cotton (Gossypium hirsutum), banana (Musa sp.), and plants belonging to the Brassicaceae family | [42][52][57][63][64][70][71][72][73][74][75] |
| Penicillium | Fruits and vegetables | [57][58][76][77] |
| Puccinia | Wheat crops (Triticum aestivum) | [42][47][64][78] |
| Rhizoctonia | Root pathogen of a wide range of hosts, including tomato, soybean (Glycine max), pepper (Capsicum annuum), eggplant (Solanum melongena), watermelon (Citrullus lanatus), upland cotton, sunflower (Helianthus annuus), rice (Oryza sativa), and potato (Solanum tuberosum) |
[32][57][71][72][73][74][75][79][80] |
| Rhizopus | Brassicaceae plants | [57][70] |
2.2. Macroalgae Potential against Phytopathogenic Fungi
2.2.1. In vitro Antifungal Potential
| Phytopathogenic Fungi | Host Species | Methodology | Reference |
|---|---|---|---|
| Alternaria alternata | Hormophysa cuneiformis | Agar diffusion assay/Broth microdilution assay | [56] |
| Ulva lactuca | Disc diffusion technique | [58] | |
| Aspergillus fumigatus | Anthophycus longifolius | Well diffusion technique | [51] |
| Osmundea pinnatifida | Radial growth inhibition | [117] | |
| Aspergillus niger | Anthophycus longifolius | Well diffusion technique | [51] |
| Ulva lactuca | Disc diffusion technique | [58] | |
| Aspergillus terreus | Anthophycus longifolius | Well diffusion technique | [51] |
| Botrytis cinerea | Dictyopteris polypodioides | Agar diffusion technique | [63] |
| Cladosporium herbarum | Hormophysa cuneiformis | Agar diffusion assay/Broth microdilution assay | [56] |
| Colletotrichum acutatum | Dictyota dichotoma | Disc diffusion technique | [65] |
| Dictyota implexa | Disc diffusion technique | [65] | |
| Dictyota spiralis | Disc diffusion technique | [65] | |
| Colletotrichum falcatum | Caulerpa racemosa | Poisoned food technique | [66] |
| Hydropuntia edulis | Poisoned food technique | [66] | |
| Sargassum myricocystum * | Poisoned food technique | [66] | |
| Colletotrichum gloeosporioides | Hypnea musciformis | Disc diffusion technique | [67][68] |
| Kappaphycus alvarezii | Poisoned food technique | [69] | |
| Laurencia dendroidea | Disc diffusion technique | [67] | |
| Ochtodes secundiramea | Disc diffusion technique | [67][68] | |
| Palisada flagellifera | Disc diffusion technique | [68] | |
| Pterocladiella capillacea | Disc diffusion technique | [67] | |
| Colletotrichum musae | Hypnea musciformis | Poisoned food technique | [67] |
| Laurencia dendroidea | Poisoned food technique | [67] | |
| Ochtodes secundiramea | Poisoned food technique | [67] | |
| Padina gymnospora | Poisoned food technique | [67] | |
| Pterocladiella capillacea | Poisoned food technique | [67] | |
| Fusarium culmorum | Fucus vesiculosus | Inhibition of mycelial growth/Macroconidia germination inhibition | [57] |
| Fusarium graminearum | Dictyopteris polypodioides | Agar diffusion technique | [63] |
| Fusarium moniliforme | Botryocladia leptopoda | Test tube in agar | [96] |
| Dictyota hauckiana | Test tube in agar | [96] | |
| Fusarium oxysporum | Asparagopsis taxiformis | Well diffusion technique | [118] |
| Calliblepharis floresii * | Poisoned food technique | [52] | |
| Caulerpa chemnitzia | Poisoned food technique | [52] | |
| Caulerpa racemosa | Poisoned food technique | [52] | |
| Caulerpa scalpelliformis | Poisoned food technique | [52] | |
| Caulerpa taxifolia | Poisoned food technique | [52] | |
| Centroceras sp. | Poisoned food technique | [52] | |
| Ceramium sp. | Poisoned food technique | [52] | |
| Chaetomorpha antennina | Poisoned food technique | [52] | |
| Codium indicum | Poisoned food technique | [52] | |
| Dictyopteris polypodioides | Agar diffusion technique | [63] | |
| Dictyota dicotoma | Poisoned food technique | [52] | |
| Gelidium pulchrum | Poisoned food technique | [52] | |
| Gracilaria corticata | Poisoned food technique | [52] | |
| Halimeda tuna | Poisoned food technique/Field studies | [52][71] | |
| Halymenia porphyriformis | Poisoned food technique | [52] | |
| Hormophysa cuneiformis | Agar diffusion assay/Broth microdilution assay | [56] | |
| Hypnea musciformis | Poisoned food technique | [52] | |
| Jania pedunculata var. adhaerens | Poisoned food technique | [52] | |
| Jolyna laminariodes | Poisoned food technique | [52] | |
| Melanothamnus afaqhusainii | Poisoned food technique/Field studies | [52][72] | |
| Neoporphyra perforata | Poisoned food technique | [52] | |
| Osmundea pinnatifida | Poisoned food technique | [52] | |
| Padina boergesenii | Disc diffusion technique | [119] | |
| Padina tetrastromatica | Poisoned food technique | [52][71] | |
| Polycladia indica | Poisoned food technique/Disc diffusion technique | [52][71][72][109] | |
| Polycladia myrica | Disc diffusion technique | [119] | |
| Sargassum aquifolium | Poisoned food technique | [52] | |
| Sargassum cinereum | Disc diffusion technique | [119] | |
| Sargassum ilicifolium | Disc diffusion technique | [109] | |
| Sargassum tenerrimum | Poisoned food technique | [52] | |
| Sargassum wightii | Poisoned food technique | [52] | |
| Scinaia huismanii | Poisoned food technique | [52] | |
| Spatoglossum asperum | Disc diffusion assay | [120] | |
| Steochospermum polypolides * | Poisoned food technique | [52] | |
| Udotea sp. | Poisoned food technique | [52] | |
| Ulva rigida | Poisoned food technique | [52] | |
| Valaniopsis sp. * | Poisoned food technique | [52] | |
| Fusarium oxysporum albedinis | Dictyota dichotoma | Disc diffusion technique | [65] |
| Dictyota implexa | Disc diffusion technique | [65] | |
| Dictyota spiralis | Disc diffusion technique | [65] | |
| Fusarium oxysporum dianthi | Dictyota dichotoma | Disc diffusion technique | [65] |
| Dictyota implexa | Disc diffusion technique | [65] | |
| Dictyota spiralis | Disc diffusion technique | [65] | |
| Fusarium oxysporum f.sp. udum | Caulerpa racemosa | Poisoned food technique | [105] |
| Hydropuntia edulis | Poisoned food technique | [105] | |
| Sargassum myricocystum * | Poisoned food technique | [105] | |
| Fusarium oxysporum lycopersici | Dictyota dichotoma | Disc diffusion technique | [65] |
| Dictyota implexa | Disc diffusion technique | [65] | |
| Dictyota spiralis | Disc diffusion technique | [65] | |
| Fusarium solani | Botryocladia leptopoda | Test tube in agar | [96] |
| Caulerpa racemosa | Test tube in agar | [96] | |
| Caulerpa taxifolia | Test tube in agar | [96] | |
| Champia compressa | Test tube in agar | [96] | |
| Codium indicum | Test tube in agar | [96] | |
| Gracilaria corticata | Test tube in agar | [96] | |
| Hypnea musciformis | Test tube in agar | [96] | |
| Hypnea valentiae | Test tube in agar | [96] | |
| Osmundea pinnatifida | Test tube in agar | [96] | |
| Padina antillarum | Test tube in agar | [96] | |
| Sarconema filiforme | Test tube in agar | [96] | |
| Sargassum ilicifolium | Test tube in agar | [96] | |
| Sargassum vulgare | Test tube in agar | [96][121] | |
| Solieria robusta | Test tube in agar/Field studies | [71][74][96][121] | |
| Spatoglossum asperum | Disc diffusion assay | [120] | |
| Stoechospermum polypodioides | Test tube in agar/Field studies | [71][74][96] | |
| Ulva lactuca | Test tube in agar | [96] | |
| Fusarium sp. | Anthophycus longifolius | Well diffusion technique | [51] |
| Ganoderma boninense | Caulerpa lamourouxii | Poisoned food technique | [122] |
| Caulerpa racemosa | Poisoned food technique | [122] | |
| Halimeda macrophysa | Poisoned food technique | [122] | |
| Sargassum oligocystum | Poisoned food technique | [122] | |
| Geotrichum sp. | Dictyopteris polypodioides | Agar diffusion technique | [63] |
| Macrophomina phaseolina | Calliblepharis floresii * | Poisoned food technique | [52] |
| Caulerpa racemosa | Poisoned food technique | [52] | |
| Caulerpa taxifolia | Poisoned food technique | [52] | |
| Centroceras sp. | Poisoned food technique | [52] | |
| Ceramium sp. | Poisoned food technique | [52] | |
| Chaetomorpha antennina | Poisoned food technique | [52] | |
| Codium indicum | Poisoned food technique | [52] | |
| Dictyota dicotoma | Poisoned food technique | [52] | |
| Gelidium pulchrum | Poisoned food technique | [52] | |
| Gracilaria corticata | Poisoned food technique | [52] | |
| Halymenia porphyriformis | Poisoned food technique | [52] | |
| Hypnea musciformis | Poisoned food technique | [52] | |
| Jania pedunculata var. adhaerens | Poisoned food technique | [52] | |
| Jolyna laminariodes | Poisoned food technique | [52] | |
| Melanothamnus afaqhusainii | Poisoned food technique | [52] | |
| Neoporphyra perforata | Poisoned food technique | [52] | |
| Osmundea pinnatifida | Poisoned food technique | [52] | |
| Padina tetrastromatica | Poisoned food technique | [52] | |
| Macrophomina phaseolina | Polycladia indica | Poisoned food technique/Disc diffusion technique | [52][109] |
| Sargassum aquifolium | Poisoned food technique | [52] | |
| Sargassum ilicifolium | Disc diffusion technique | [109] | |
| Sargassum tenerrimum | Poisoned food technique | [52] | |
| Sargassum wightii | Poisoned food technique | [52] | |
| Scinaia huismanii | Poisoned food technique | [52] | |
| Spatoglossum asperum | Disc diffusion assay | [120] | |
| Stoechospermum polypodioides | Poisoned food technique | [52] | |
| Udotea sp. | Poisoned food technique | [52] | |
| Ulva rigida | Poisoned food technique | [52] | |
| Valaniopsis sp. * | Poisoned food technique | [52] | |
| Mucor sp. | Champia compressa | Test tube in agar | [96] |
| Hypnea musciformis | Test tube in agar | [96] | |
| Sargassum boveanum | Test tube in agar | [96] | |
| Sargassum ilicifolium | Test tube in agar | [96] | |
| Ulva lactuca | Test tube in agar | [96] | |
| Penicillium expansum | Ulva lactuca | Disc diffusion technique | [58] |
| Penicillium sp. | Dictyota dichotoma | Disc diffusion technique | [123] |
| Ulva lactuca | Disc diffusion technique | [123] | |
| Penicillum digitatum | Hormophysa cuneiformis | Agar diffusion assay/Broth microdilution assay | [56] |
| Phialophora cinerescens | Dictyota dichotoma | Disc diffusion technique | [65] |
| Dictyota implexa | Disc diffusion technique | [65] | |
| Dictyota spiralis | Disc diffusion technique | [65] | |
| Phoma tracheiphila | Dictyota dichotoma | Disc diffusion technique | [65] |
| Dictyota implexa | Disc diffusion technique | [65] | |
| Dictyota spiralis | Disc diffusion technique | [65] | |
| Pseudocercospora fijiensis | Halymenia floresii | Minimum inhibitory concentration | [94] |
| Pyricularia oryzae | Rhodomela confervoides | Spore spreading method | [95] |
| Symphyocladia latiuscula | Spore spreading method | [95] | |
| Rhizoctonia solani | Calliblepharis floresii | Poisoned food technique | [52] |
| Centroceras sp. | Poisoned food technique | [52] | |
| Ceramium sp. | Poisoned food technique | [52] | |
| Chaetomorpha antennina | Poisoned food technique | [52] | |
| Codium indicum | Poisoned food technique | [52] | |
| Dictyopteris undulata | Fungitoxic activity | [92] | |
| Gelidium pulchrum | Poisoned food technique | [52] | |
| Gracilaria corticata | Poisoned food technique | [52] | |
| Halymenia porphyriformis | Poisoned food technique | [52] | |
| Hypnea musciformis | Poisoned food technique | [52] | |
| Jania pedunculata var. adhaerens | Poisoned food technique | [52] | |
| Melanothamnus afaqhusainii | Poisoned food technique | [52] | |
| Neoporphyra perforata | Poisoned food technique | [52] | |
| Osmundea pinnatifida | Poisoned food technique | [52] | |
| Padina tetrastromatica | Poisoned food technique | [52] | |
| Polycladia indica | Poisoned food technique | [52] | |
| Sargassum aquifolium | Poisoned food technique | [52][71][74] | |
| Sargassum tenerrimum | Poisoned food technique | [52][71] | |
| Rhizoctonia solani | Sargassum wightii | Poisoned food technique | [52] |
| Spatoglossum asperum | Disc diffusion assay/Field studies | [73][120] | |
| Stoechospermum polypodioides | Poisoned food technique/Field studies | [52][71][74] | |
| Udotea sp. | Poisoned food technique | [52] | |
| Ulva rigida | Poisoned food technique | [52] | |
| Valaniopsis sp. * | Poisoned food technique | [52] | |
| Dictyota dichotoma | Disc diffusion technique/Spore germination | [79] | |
| Padina gymnospora | Disc diffusion technique/Spore germination | [79] | |
| Sargassum muticum | Disc diffusion technique/Spore germination | [79] | |
| Sargassum tenerrimum | Disc diffusion technique/Spore germination | [79] | |
| Sargassum wightii | Disc diffusion technique/Spore germination | [79] | |
| Sclerotinia sclerotiorum | Dictyopteris undulata | Fungitoxic activity | [92] |
| Sclerotium rolfsii | Dictyopteris undulata | Fungitoxic activity | [92] |
| Verticillium dahliae | Cystoseira humilis var. myriophylloides | Poisoned food technique | [93] |
| Dictyopteris polypodioides | Agar diffusion technique | [63] | |
| Fucus spiralis | Poisoned food technique | [93] |
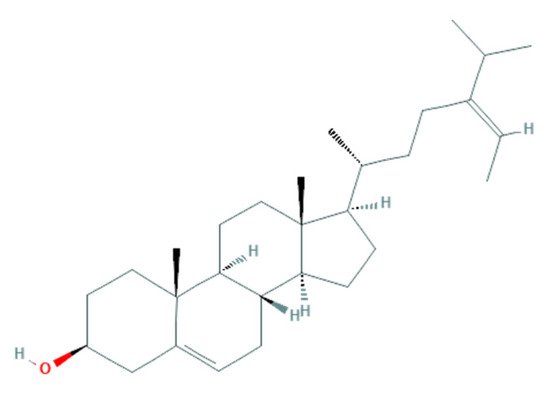
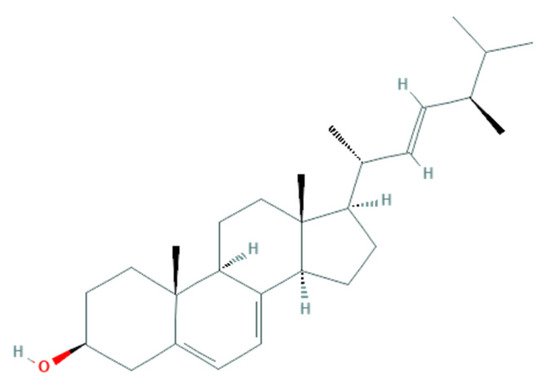
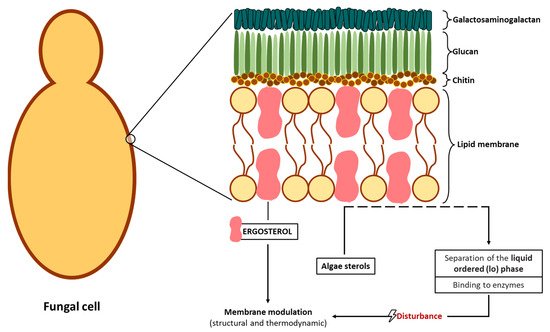
2.2.2. Potential Antifungal Mechanisms
References
- Savary, S.; Ficke, A.; Aubertot, J.-N.; Hollier, C. Crop Losses Due to Diseases and Their Implications for Global Food Production Losses and Food Security. Food Secur. 2012, 4, 519–537.
- Rahman, S.F.S.A.; Singh, E.; Pieterse, C.M.J.; Schenk, P.M. Emerging Microbial Biocontrol Strategies for Plant Pathogens. Plant Sci. 2017, 267, 102–111.
- Tleuova, A.B.; Wielogorska, E.; Talluri, V.S.S.L.P.; Štěpánek, F.; Elliott, C.T.; Grigoriev, D.O. Recent Advances and Remaining Barriers to Producing Novel Formulations of Fungicides for Safe and Sustainable Agriculture. J. Control. Release 2020, 326, 468–481.
- Kannan, V.R.; Bastas, K.K.; Devi, R.S. Scientific and Economic Impact of Plant Pathogenic Bacteria. In Sustainable Approaches to Controlling Plant Pathogenic Bacteria; CRC Press: Boca Raton, FL, USA, 2015; pp. 369–392.
- Savary, S.; Willocquet, L.; Pethybridge, S.J.; Esker, P.; McRoberts, N.; Nelson, A. The Global Burden of Pathogens and Pests on Major Food Crops. Nat. Ecol. Evol. 2019, 3, 430–439.
- Strange, R.N.; Scott, P.R. Plant Disease: A Threat to Global Food Security. Annu. Rev. Phytopathol. 2005, 43, 83–116.
- Mokyr, J.; Gráda, C.Ó. What Do People Die of during Famines: The Great Irish Famine in Comparative Perspective. Eur. Rev. Econ. Hist. 2002, 6, 339–363.
- Fisher, M.C.; Henk, D.A.; Briggs, C.J.; Brownstein, J.S.; Madoff, L.C.; McCraw, S.L.; Gurr, S.J. Emerging Fungal Threats to Animal, Plant and Ecosystem Health. Nature 2012, 484, 186–194.
- Narayanasamy, P. Diagnosis of Bacterial Diseases of Plants. In Microbial Plant Pathogens-Detection and Disease Diagnosis: Bacterial and Phytoplasmal Pathogens; Springer: Dordrecht, The Netherlands, 2011; Volume 2, pp. 233–246.
- Shuping, D.S.S.; Eloff, J.N. The Use of Plants to Protect Plants and Food Against Fungal Pathogens: A Review. Afr. J. Tradit. Complement. Altern. Med. 2017, 14, 120–127.
- El Hussein, A.A.; Alhasan, R.E.M.; Abdelwahab, S.A.; El Siddig, M.A. Isolation and Identification of Streptomyces Rochei Strain Active against Phytopathogenic Fungi. Br. Microbiol. Res. J. 2014, 4, 1057–1068.
- Zabiák, A.; Károlyi, G.M.; Sándor, E. Investigation of Host-Specificity of Phytopathogenic Fungi Isolated from Woody Plants. Acta Agrar. Debreceniensis 2020, 1, 155–160.
- Kainz, K.; Bauer, M.A.; Madeo, F.; Carmona-Gutierrez, D. Fungal Infections in Humans: The Silent Crisis. Microb. Cell 2020, 7, 143–145.
- Coque, R.; Álvarez-Pérez, M.; Cobos, R.; González-García, S.; Ibáñez, A.M.; Galán, A.D.; Calvo-Peña, C. Advances in the Control of Phytopathogenic Fungi That Infect Crops through Their Root System. Adv. Appl. Microbiol. 2020, 111, 1–48.
- Cornelissen, B.J.C.; Melchers, L.S. Strategies for Control of Fungal Diseases with Transgenic Plants. Plant Physiol. 1993, 101, 709–712.
- De Waard, M.A.; Georgopoulos, S.G.; Hollomon, D.W.; Ishii, H.; Leroux, P.; Ragsdale, N.N.; Schwinn, F.J. Chemical Control of Plant Diseases: Problems and Prospects. Annu. Rev. Phytopathol. 1993, 31, 403–421.
- Hamed, S.M.; Abd El-Rhman, A.A.; Abdel-Raouf, N.; Ibraheem, I.B.M. Role of Marine Macroalgae in Plant Protection & Improvement for Sustainable Agriculture Technology. Beni-Suef Univ. J. Basic Appl. Sci. 2018, 7, 104–110.
- Zhang, Z.; Chen, Y.; Li, B.; Chen, T.; Tian, S. Reactive Oxygen Species: A Generalist in Regulating Development and Pathogenicity of Phytopathogenic Fungi. Comput. Struct. Biotechnol. J. 2020, 18, 3344–3349.
- Badosa, E.; Ferre, R.; Planas, M.; Feliu, L.; Montesinos, E.; Cabrefiga, J.; Bardajı, E.; Besalu, E. A Library of Linear Undecapeptides with Bactericidal Activity against Phytopathogenic Bacteria. Peptides 2007, 28, 2276–2285.
- Hahn, M. The Rising Threat of Fungicide Resistance in Plant Pathogenic Fungi: Botrytis as a Case Study. J. Chem. Biol. 2014, 7, 133–141.
- Ma, Z.; Michailides, T.J. Advances in Understanding Molecular Mechanisms of Fungicide Resistance and Molecular Detection of Resistant Genotypes in Phytopathogenic Fungi. Crop Prot. 2005, 24, 853–863.
- Lorito, M.; Mach, R.L.; Sposato, P.; Strauss, J.; Peterbauer, C.K.; Kubicek, C.P. Mycoparasitic Interaction Relieves Binding of the Cre1 Carbon Catabolite Repressor Protein to Promoter Sequences of the ech42 (Endochitinase-Encoding) Gene in Trichoderma Harzianum. Proc. Natl. Acad. Sci. USA 1996, 93, 14868–14872.
- Lorito, M.; Harman, G.E.; Hayes, C.K.; Broadway, R.M.; Tronsmo, A.; Woo, S.L.; Di Pietro, A. Chinolytic Enzymes Produced by Trichoderma Harzianum: Antifungal Activity of Purified Endochitinase and Chitobiosidase. Mol. Plant Pathol. 1993, 83, 302–307.
- Neeraja, C.; Anil, K.; Purushotham, P.; Suma, K.; Sarma, P.; Moerschbacher, B.M.; Podile, A.R. Biotechnological Approaches to Develop Bacterial Chitinases as a Bioshield against Fungal Diseases of Plants. Crit. Rev. Biotechnol. 2010, 30, 231–241.
- Chandra, H.; Kumari, P.; Bisht, R.; Prasad, R.; Yadav, S. Plant Growth Promoting Pseudomonas aeruginosa from Valeriana wallichii Displays Antagonistic Potential against Three Phytopathogenic Fungi. Mol. Biol. Rep. 2020, 47, 6015–6026.
- Lorito, M.; Peterbauer, C.; Hayes, C.K.; Harman, G.E. Synergistic Interaction between Fungal Cell Wall Degrading Enzymes and Different Antifungal Compounds Enhances Inhibition of Spore Germination. Microbiology 1994, 140, 623–629.
- Azcón-Aguilar, C.; Barea, J.M. Arbuscular Mycorrhizas and Biological Control of Soil-Borne Plant Pathogens—An Overview of the Mechanisms Involved. Mycorrhiza 1997, 6, 457–464.
- O’Brien, P.A. Biological Control of Plant Diseases. Australas. Plant Pathol. 2017, 46, 293–304.
- Lorito, M.; Woo, S.L.; Fernandez, I.G.; Colucci, G.; Harman, G.E.; Pintor-Toro, J.A.; Filippone, E.; Muccifora, S.; Lawrence, C.B.; Zoina, A.; et al. Genes from Mycoparasitic Fungi as a Source for Improving Plant Resistance to Fungal Pathogens. Proc. Natl. Acad. Sci. USA 1998, 95, 7860–7865.
- Vidaver, A.K. Prospects for Control of Phytopathogenic Bacteria by Bacteriophages and Bacteriocins. Annu. Rev. Phytopathol. 1976, 14, 451–465.
- Alamri, S.; Hashem, M.; Mostafa, Y.S. in vitro and in vivo Biocontrol of Soil-Borne Phytopathogenic Fungi by Certain Bioagents and Their Possible Mode of Action. Biocontrol Sci. 2012, 17, 155–167.
- Samsatly, J.; Copley, T.R.; Jabaji, S.H. Antioxidant Genes of Plants and Fungal Pathogens Are Distinctly Regulated during Disease Development in Different Rhizoctonia solani Pathosystems. PLoS ONE 2018, 13, e0192682.
- DeGray, G.; Rajasekaran, K.; Smith, F.; Sanford, J.; Daniell, H. Expression of an Antimicrobial Peptide via the Chloroplast Genome to Control Phytopathogenic Bacteria and Fungi. Plant Physiol. 2001, 127, 852–862.
- Pathma, J.; Kennedy, R.K.; Bhushan, L.S.; Shankar, B.K.; Thakur, K. Microbial Biofertilizers and Biopesticides: Nature’s Assets Fostering Sustainable Agriculture. In Recent Developments in Microbial Technologies; Springer: Singapore, 2021; pp. 39–69.
- Kiuru, P.; D’Auria, M.V.; Muller, C.D.; Tammela, P.; Vuorela, H.; Yli-Kauhaluoma, J. Exploring Marine Resources for Bioactive Compounds. Planta Med. 2014, 80, 1234–1246.
- Nawaz, A.; Chaudhary, R.; Shah, Z.; Dufossé, L.; Fouillaud, M.; Mukhtar, H.; Haq, I.U. An Overview on Industrial and Medical Applications of Bio-Pigments Synthesized by Marine Bacteria. Microorganisms 2021, 9, 11.
- Milledge, J.J.; Nielsen, B.V.; Bailey, D. High-Value Products from Macroalgae: The Potential Uses of the Invasive Brown Seaweed, Sargassum Muticum. Rev. Environ. Sci. Biotechnol. 2016, 15, 67–88.
- Shannon, E.; Abu-Ghannam, N. Antibacterial Derivatives of Marine Algae: An Overview of Pharmacological Mechanisms and Applications. Mar. Drugs 2016, 14, 81.
- Rizzo, C.; Genovese, G.; Morabito, M.; Faggio, C.; Pagano, M.; Spanò, A.; Zammuto, V.; Minicante, S.A.; Manghisi, A.; Cigala, R.M.; et al. Potential Antibacterial Activity of Marine Macroalgae against Pathogens Relevant for Aquaculture and Human Health. J. Pure Appl. Microbiol. 2017, 11, 1695–1706.
- Besednova, N.N.; Andryukov, B.G.; Zaporozhets, T.S.; Kryzhanovsky, S.P.; Kuznetsova, T.A.; Fedyanina, L.N.; Makarenkova, I.D.; Zvyagintseva, T.N. Algae Polyphenolic Compounds and Modern Antibacterial Strategies: Current Achievements and Immediate Prospects. Biomedicines 2020, 8, 342.
- Khan, W.; Rayirath, U.P.; Subramanian, S.; Jithesh, M.N.; Rayorath, P.; Hodges, D.M.; Critchley, A.T.; Craigie, J.S.; Norrie, J.; Prithiviraj, B. Seaweed Extracts as Biostimulants of Plant Growth and Development. J. Plant Growth Regul. 2009, 28, 386–399.
- Brauer, V.S.; Rezende, C.P.; Pessoni, A.M.; De Paula, R.G.; Rangappa, K.S.; Nayaka, S.C.; Gupta, V.K.; Almeida, F. Antifungal Agents in Agriculture: Friends and Foes of Public Health. Biomolecules 2019, 9, 521.
- Dyakov, Y.T.; Zinovyeva, S.V. Plant Parasite Microorganisms. In Comprehensive and Molecular Phytopathology; Elsevier: Amsterdam, The Netherlands, 2007; pp. 19–47.
- Mendgen, K.; Hahn, M. Plant Infection and the Establishment of Fungal Biotrophy. Trends Plant Sci. 2002, 7, 352–356.
- Stuiver, M.H.; Custers, J.H.H.V. Engineering Disease Resistance in Plants. Nature 2001, 411, 865–868.
- Jayawardena, R.S.; Hyde, K.D.; McKenzie, E.H.C.; Jeewon, R.; Phillips, A.J.L.; Perera, R.H.; de Silva, N.I.; Maharachchikumburua, S.S.N.; Samarakoon, M.C.; Ekanayake, A.H.; et al. One Stop Shop III: Taxonomic Update with Molecular Phylogeny for Important Phytopathogenic Genera: 51–75 (2019); Springer: Dordrecht, The Netherlands, 2019; Volume 98.
- Marin-Felix, Y.; Groenewald, J.Z.; Cai, L.; Chen, Q.; Marincowitz, S.; Barnes, I.; Bensch, K.; Braun, U.; Camporesi, E.; Damm, U.; et al. Genera of Phytopathogenic Fungi: GOPHY 1. Stud. Mycol. 2017, 86, 99–216.
- Marin-Felix, Y.; Hernández-Restrepo, M.; Wingfield, M.J.; Akulov, A.; Carnegie, A.J.; Cheewangkoon, R.; Gramaje, D.; Groenewald, J.Z.; Guarnaccia, V.; Halleen, F.; et al. Genera of Phytopathogenic Fungi: GOPHY 2. Stud. Mycol. 2019, 92, 47–133.
- Marin-Felix, Y.; Hernández-Restrepo, M.; Iturrieta-González, I.; García, D.; Gené, J.; Groenewald, J.Z.; Cai, L.; Chen, Q.; Quaedvlieg, W.; Schumacher, R.K.; et al. Genera of Phytopathogenic Fungi: GOPHY 3. Stud. Mycol. 2019, 94, 1–124.
- Masuda, M.; Abe, T.; Sato, S.; Suzuki, T.; Suzuki, M. Diversity of Halogenated Secondary Metabolites in the Red Alga Laurencia Nipponica (Rhodomelaceae, Ceramiales). J. Phycol. 1997, 33, 196–208.
- Malini, M.; Ponnanikajamideen, M.; Malarkodi, C.; Rajeshkumar, S. Explore the Antimicrobial Potential from Organic Solvents Extract of Brown Seaweed (Sargassum longifolium) Alleviating to Pharmaceuticals. Int. J. Pharm. Res. 2014, 6, 28–35.
- Khan, S.A.; Abid, M.; Hussain, F. Antifungal Activity of Aqueous and Methanolic Extracts of Some Seaweeds against Common Soil-Borne Plant Pathogenic Fungi. Pak. J. Bot. 2017, 49, 1211–1216.
- Pohl, C.H.; Kock, J.L.F.; Thibane, V.S. Antifungal Free Fatty Acids: A Review. Sci. Against Microb. Pathog. Curr. Res. Technol. Adv. 2011, 3, 61–71.
- Avis, T.J.; Bélanger, R.R. Specificity and Mode of Action of the Antifungal Fatty Acid Cis-9-Heptadecenoic Acid Produced by Pseudozyma flocculosa. Appl. Environ. Microbiol. 2001, 67, 956–960.
- Desbois, A.P.; Smith, V.J. Antibacterial Free Fatty Acids: Activities, Mechanisms of Action and Biotechnological Potential. Appl. Microbiol. Biotechnol. 2010, 85, 1629–1642.
- Mohamed, S.S.; Saber, A.A. Antifungal Potential of the Bioactive Constituents in Extracts of the Mostly Untapped Brown Seaweed Hormophysa cuneiformis from The Egyptian Coastal Waters. Egypt. J. Bot. 2019, 59, 695–708.
- Tyśkiewicz, K.; Tyśkiewicz, R.; Konkol, M.; Rój, E.; Jaroszuk-Ściseł, J.; Skalicka-Woźniak, K. Antifungal Properties of Fucus vesiculosus L. Supercritical Fluid Extract Against Fusarium culmorum and Fusarium oxysporum. Molecules 2019, 24, 3518.
- Vehapi, M.; Koçer, A.T.; Yılmaz, A.; Özçimen, D. Investigation of the Antifungal Effects of Algal Extracts on Apple-infecting Fungi. Arch. Microbiol. 2020, 202, 455–471.
- Ali, N.; Ramkissoon, A.; Ramsubhag, A.; Jayaraj, J. Ascophyllum Extract Application Causes Reduction of Disease Levels in Field Tomatoes Grown in a Tropical Environment. Crop Prot. 2016, 83, 67–75.
- D’Mello, J.P.F.; Macdonald, A.M.C.; Postel, D.; Dijksma, W.T.P.; Dujardin, A.; Placinta, C.M. Pesticide Use and Mycotoxin Production in Fusarium and Aspergillus Phytopathogens. Eur. J. Plant Pathol. 1998, 104, 741–751.
- Windham, G.L.; Williams, W.P.; Davis, F.M. Effects of the Southwestern Corn Borer on Aspergillus flavus Kernel Infection and Aflatoxin Accumulation in Maize Hybrids. Plant Dis. 1999, 83, 535–540.
- Kelley, R.Y.; Williams, W.P.; Mylroie, J.E.; Boykin, D.L.; Harper, J.W.; Windham, G.L.; Ankala, A.; Shan, X. Identification of Maize Genes Associated with Host Plant Resistance or Susceptibility to Aspergillus flavus Infection and Aflatoxin Accumulation. PLoS ONE 2012, 7, 12.
- Belattmania, Z.; Reani, A.; Barakate, M.; Zrid, R.; Elatouani, S.; Hassouani, M.; Eddaoui, A.; Bentiss, F.; Sabour, B. Antimicrobial, Antioxidant and Alginate Potentials of Dictyopteris polypodioides (Dictyotales, Phaeophyceae) from the Moroccan Atlantic Coast. Der Pharma Chem. 2016, 8, 216–226.
- Dean, R.; Van Kan, J.A.L.; Pretorius, Z.A.; Hammond-Kosack, K.E.; Di Pietro, A.; Spanu, P.D.; Rudd, J.J.; Dickman, M.; Kahmann, R.; Ellis, J.; et al. The Top 10 Fungal Pathogens in Molecular Plant Pathology. Mol. Plant Pathol. 2012, 13, 414–430.
- Moreau, J.; Pesando, D.; Bernard, P.; Caram, B.; Pionnat, J.C. Seasonal Variations in the Production of Antifungal Substances by Some Dictyotales (Brown Algae) from the French Mediterranean Coast. Hydrobiologia 1988, 162, 157–162.
- Ambika, S.; Sujatha, K. Antifungal Activity of Aqueous and Ethanol Extracts of Seaweeds against Sugarcane Red Rot Pathogen (Colletotrichum falcatum). Sci. Res. Essays 2015, 10, 232–235.
- Machado, L.P.; Matsumoto, S.T.; Jamal, C.M.; Silva, M.B.; da Cruz Centeno, D.; Neto, P.C.; Carvalho, L.R.; Yokoya, N.S. Chemical Analysis and Toxicity of Seaweed Extracts with Inhibitory Activity against Tropical Fruit Anthracnose Fungi. J. Sci. Food Agric. 2013, 94, 1739–1744.
- Machado, L.P.; Matsumoto, S.T.; Cuzzuol, G.R.F.; Oliveira, L.F.G., Jr. Influence of Laboratory Cultivation on Species of Rhodophyta Physiological Evaluations and Antifungal Activity against Phytopathogens. Rev. Ciênc. Agron. 2014, 45, 52–61.
- Mani, S.D.; Nagarathnam, R. Sulfated Polysaccharide from Kappaphycus Alvarezii (Doty) Doty Ex P.C. Silva Primes Defense Responses against Anthracnose Disease of Capsicum annuum Linn. Algal Res. 2018, 32, 121–130.
- Ishimoto, H.; Fukushi, Y.; Yoshida, T.; Tahara, S. Rhizopus and Fusarium Are Selected as Dominant Fungal Genera in Rhizospheres of Brassicaceae. J. Chem. Ecol. 2000, 26, 2387–2399.
- Ehteshamul-Haque, S.; Baloch, G.N.; Sultana, V.; Ara, J.; Tariq, R.M.; Athar, M. Impact of Seaweeds on Fluorescent Pseudomonas and Their Role in Suppressing the Root Diseases of Soybean and Pepper. J. Appl. Bot. Food Qual. 2013, 86, 126–132.
- Baloch, G.N.; Tariq, S.; Ehteshamul-Haque, S.; Athar, M.; Sultana, V.; Ara, J. Management of Root Diseases of Eggplant and Watermelon with the Application of Asafoetida and Seaweeds. J. Appl. Bot. Food Qual. 2013, 86, 138–142.
- Sultana, V.; Ehteshamul-Haque, S.; Ara, J.; Athar, M. Effect of Brown Seaweeds and Pesticides on Root Rotting Fungi and Root-Knot Nematode Infecting Tomato Roots. J. Appl. Bot. Food Qual. 2009, 83, 50–53.
- Sultana, V.; Tariq, S.; Hira, K.; Tariq, A.; Ara, J.; Tariq, R.M.; Ehteshamul-Haque, S. Seaweed Bio-Fertilizer for the Management of Root Rotting Fungi and Root Knot Nematodes Affecting Cotton Crop. Pak. J. Bot. 2018, 50, 2409–2412.
- Sultana, V.; Baloch, G.N.; Ara, J.; Ehteshamul-Haque, S.; Tariq, R.M.; Athar, M. Seaweeds as Alternative to Chemical Pesticides for the Management of Root Diseases of Sunflower and Tomato. J. Appl. Bot. Food Qual. 2011, 84, 162–168.
- Vitoratos, A.; Bilalis, D.; Karkanis, A.; Efthimiadou, A. Antifungal Activity of Plant Essential Oils Against Botrytis cinerea, Penicillium italicum and Penicillium digitatum. Not. Bot. Horti Agrobot. Cluj-Napoca 2013, 41, 86–92.
- Neri, F.; Mari, M.; Brigati, S. Control of Penicillium expansum by Plant Volatile Compounds. Plant Pathol. 2006, 55, 100–105.
- Ivanov, S.; Miteva, L.; Alexieva, V.; Karjin, H.; Karanov, E. Alterations in Some Oxidative Parameters in Susceptible and Resistant Wheat Plants Infected with Puccinia recondita f. Sp. tritici. J. Plant Physiol. 2005, 162, 275–279.
- Graff, K.H.; Raj, T.S. Effect of Sargassum tenerrimum on Controlling Sheath Blight of Rice Caused by Rhizoctonia solani Kuhn. Plant Arch. 2019, 19, 1132–1135.
- Cordovez, V.; Mommer, L.; Moisan, K.; Lucas-Barbosa, D.; Pierik, R.; Mumm, R.; Carrion, V.J.; Raaijmakers, J.M. Plant Phenotypic and Transcriptional Changes Induced by Volatiles from the Fungal Root Pathogen Rhizoctonia solani. Front. Plant Sci. 2017, 8, 1262.
- Jiménez, E.; Dorta, F.; Medina, C.; Ramírez, A.; Ramírez, I.; Peña-Cortés, H. Anti-Phytopathogenic Activities of Macro-Algae Extracts. Mar. Drugs 2011, 9, 739–756.
- Robles-Centeno, P.O.; Ballantine, D.L.; Gerwick, W.H. Dynamics of Antibacterial Activity in Three Species of Caribbean Marine Algae as a Function of Habitat and Life History. Hydrobiologia 1996, 326–327, 457–462.
- Ballantine, D.L.; Gerwick, W.H.; Velez, S.M.; Alexander, E.; Guevara, P. Antibiotic Activity of Lipid-Soluble Extracts from Caribbean Marine Algae. Hydrobiologia 1987, 151–152, 463–469.
- Khaleafa, A.F.; Kharboush, M.A.M.; Metwalli, A.; Mohsen, A.F.; Serwi, A. Antibiotic (Fungicidal) Action from Extracts of Some Seaweeds. Bot. Mar. 1975, XVIII, 163–165.
- Kumar, K.A.; Rengasamy, R. Evaluation of Antibacterial Potential of Seaweeds Occurring along the Coast of Tamil Nadu, India against the Plant Pathogenic Bacterium Xanthomonas oryzae Pv. oryzae (Ishiyama) Dye. Bot. Mar. 2000, 43, 409–415.
- Rao, P.P.S.; Rao, P.S.; Karmarkar, S.M. Antibacterial Substances from Brown Algae II. Efficiency of Solvents in the Evaluation of Antibacterial Substances from Sargassum Johnstonü Setchell et Gardner. Bot. Mar. 1986, XXIX, 503–507.
- Lakhdar, F.; Boujaber, N.; Oumaskour, K.; Assobhei, O.; Etahiri, S. Inhibitive Activity of 17 Marine Algae from the Coast of El Jadida-Morocco against Erwinia chrysanthemi. Int. J. Pharm. Pharm. Sci. 2015, 7, 376–380.
- Kamenarska, Z.; Serkedjieva, J.; Najdenski, H.; Stefanov, K.; Tsvetkova, I.; Dimitrova-Konaklieva, S.; Popov, S. Antibacterial, Antiviral, and Cytotoxic Activities of Some Red and Brown Seaweeds from the Black Sea. Bot. Mar. 2009, 52, 80–86.
- Caccamese, S.; Azzolina, R.; Furnari, G.; Cormaci, M.; Grasso, S. Antimicrobial and Antiviral Activities of Some Marine Algae from Eastern Sicily. Bot. Mar. 1981, XXIV, 365–367.
- Kumar, K.A.; Rengasamy, R. Antibacterial Activities of Seaweed Extracts/Fractions Obtained through a TLC Profile against the Phytopathogenic Bacterium Xanthomonas oryzae Pv. oryzae. Bot. Mar. 2000, 43, 417–421.
- Rao, P.S.; Parekh, K.S. Antibacterial Activity of Indian Seaweed Extracts. Bot. Mar. 1981, XXIV, 577–582.
- Fenical, W.; Sims, J.J. Zonarol and Isozonarol, Fungitoxic Hydroquinones from the Brown Seaweed Dictyopteris zonarioides. J. Org. Chem. 1973, 38, 2383–2386.
- Esserti, S.; Smaili, A.; Rifai, L.A.; Koussa, T.; Makroum, K.; Belfaiza, M.; Kabil, E.M.; Faize, L.; Burgos, L.; Alburquerque, N.; et al. Protective Effect of Three Brown Seaweed Extracts against Fungal and Bacterial Diseases of Tomato. J. Appl. Phycol. 2017, 29, 1081–1093.
- Gómez-Hernández, M.; Rodríguez-García, C.M.; Peraza-Echeverría, L.; Peraza-Sánchez, S.R.; Torres-Tapia, L.W.; Pérez-Brito, D.; Vargas-Coronado, R.F.; Cauich-Rodríguez, J.V. In Vitro Antifungal Activity Screening of Beach-Cast Seaweeds Collected in Yucatan, Mexico. J. Appl. Phycol. 2021, 33, 1229–1237.
- Zhang, Y.; Han, J.; Mu, J.; Feng, Y.; Gu, X.; Ji, Y. Bioactivity and Constituents of Several Common Seaweeds. Chin. Sci. Bull. 2013, 58, 2282–2289.
- Rizvi, M.A.; Shameel, M. Pharmaceutical Biology of Seaweeds from the Karachi Coast of Pakistan Pharmaceutical. Pharm. Biol. 2005, 43, 97–107.
- Ali, N.; Farrell, A.; Ramsubhag, A.; Jayaraman, J. The Effect of Ascophyllum nodosum Extract on the Growth, Yield and Fruit Quality of Tomato Grown under Tropical Conditions. J. Appl. Phycol. 2016, 28, 1353–1362.
- Xiao, C.L.; Mackenzie, S.J.; Legard, D.E. Genetic and Pathogenic Analyses of Colletotrichum gloeosporioides Isolates from Strawberry and Noncultivated Hosts. Phytopathology 2004, 94, 446–453.
- Viswanathan, R.; Samiyappan, R. Induced Systemic Resistance by Fluorescent Pseudomonads against Red Rot Disease of Sugarcane Caused by Colletotrichum falcatum. Crop Prot. 2002, 21, 1–10.
- Qureshi, M.A.; Afghan, S. Sugarcane Cultivation in Pakistan; Pakistan Society of Sugar Technologists: Sindh, Pakistan, 2005.
- Paul, V.J.; Hay, M.E.; Duffy, J.E.; Fenical, W.; Gustafson, K. Chemical Defense in the Seaweed Ochtodes secundiramea (Montagne) Howe (Rhodophyta): Effects of Its Monoterpenoid Components upon Diverse Coral-Reef Herbivores. J. Exp. Mar. Biol. Ecol. 1987, 114, 249–260.
- Peres, J.C.F.; de Carvalho, L.R.; Gonçalez, E.; Berian, L.O.S.; Felicio, J.D. Evaluation of Antifungal Activity of Seaweed Extracts. Ciênc. Agrotecnol. 2012, 36, 294–299.
- Yoshie-Stark, Y.; Hsieh, Y.-P.; Takeshi, S. Distribution of Flavonoids and Related Compounds from Seaweeds in Japan. J. Tokyo Univ. Fish. 2003, 89, 1–6.
- Walters, D.; Raynor, L.; Mitchell, A.; Walker, R.; Walker, K. Antifungal Activities of Four Fatty Acids against Plant Pathogenic Fungi. Mycopathologia 2004, 157, 87–90.
- Ambika, S.; Sujatha, K. Comparative Studies on Brown, Red and Green Alga Seaweed Extracts for Their Antifungal Activity against Fusarium oxysporum f.Sp. udum in Pigeon Pea Var. CO (Rg)7 (Cajanus cajan (L.) Mills.). J. Biopestic. 2014, 7, 167–176.
- Bennamara, A.; Abourriche, A.; Berrada, M.; Charrouf, M.; Chaib, N.; Boudouma, M.; Garneau, F.X. Methoxybifurcarenone: An Antifungal and Antibacterial Meroditerpenoid from the Brown Alga Cystoseira tamariscifolia. Phytochemistry 1999, 52, 37–40.
- Vahdani, M.; Faridi, P.; Zarshenas, M.M.; Javadpour, S.; Abolhassanzadeh, Z.; Moradi, N.; Bakzadeh, Z.; Karmostaji, A.; Mohagheghzadeh, A.; Ghasemi, Y. Major Compounds and Antimicrobial Activity of Essential Oils from Five Iranian Endemic Medicinal Plants. Pharmacogn. J. 2011, 3, 48–53.
- Altieri, C.; Bevilacqua, A.; Cardillo, D.; Sinigaglia, M. Antifungal Activity of Fatty Acids and Their Monoglycerides against Fusarium Spp. in a Laboratory Medium. Int. J. Food Sci. Technol. 2009, 44, 242–245.
- Ambreen, A.; Khan, H.; Tariq, A.; Ruqqia, A.; Sultana, V.; Ara, J. Evaluation of Biochemical Component and Antimicrobial Activity of Some Seaweeeds Occurring at Karachi Coast. Pak. J. Bot. 2012, 44, 1799–1803.
- Islam, M.S.; Haque, M.S.; Islam, M.M.; Emdad, E.M.; Halim, A.; Hossen, Q.M.M.; Hossain, M.Z.; Ahmed, B.; Rahim, S.; Rahman, M.S.; et al. Tools to Kill: Genome of One of the Most Destructive Plant Pathogenic Fungi Macrophomina phaseolina. BMC Genom. 2012, 13, 1–16.
- Shafique, H.A.; Sultana, V.; Ehteshamul-Haque, S.; Athar, M. Management of Soil-Borne Diseases of Organic Vegetables. J. Plant Prot. Res. 2016, 56, 221–230.
- Agarwal, P.; Patel, K.; Das, A.K.; Ghosh, A. Insights into the Role of Seaweed Kappaphycus alvarezii Sap towards Phytohormone Signalling and Regulating Defence Responsive Genes in Lycopersicon esculentum. Environ. Boil. Fishes 2016, 28, 2529–2537.
- Nelson, W.R.; Van Staden, J. Aminocyclopropane-1-Carboxylic Acid in Seaweed Concentrate. Bot. Mar. 1985, XXVIII, 415–417.
- Kajiwara, T.; Matsui, K.; Akakabe, Y.; Murakawa, T.; Arai, C. Antimicrobial Browning-Inhibitory Effect of Flavor Compounds in Seaweeds. J. Appl. Bot. Food Qual. 2006, 18, 413–422.
- Kubo, I.; Himejima, M.; Tsujimoto, K.; Muroi, H.; Ichikawa, N. Antibacterial Activity of Crinitol and Its Potentiation. J. Nat. Prod. 1992, 55, 780–785.
- National Center for Biotechnology Information. PubChem Compound Summary for CID 6441081, Crinitol. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Crinitol (accessed on 2 October 2021).
- Silva, P.; Fernandes, C.; Barros, L.; Ferreira, I.C.F.R.; Pereira, L.; Gonçalves, T. The Antifungal Activity of Extracts of Osmundea pinnatifida, an Edible Seaweed, Indicates Its Usage as a Safe Environmental Fungicide or as a Food Additive Preventing Post-Harvest Fungal Food Contamination. Food Funct. 2018, 9, 6187–6195.
- Manilal, A.; Sujith, S.; Kiran, G.S.; Selvin, J.; Shakir, C.; Gandhimathi, R.; Lipton, A.P. Antimicrobial Potential and Seasonality of Red Algae Collected from the Southwest Coast of India Tested against Shrimp, Human and Phytopathogens. Ann. Microbiol. 2009, 59, 207–219.
- El-sheekh, M.M.; Mousa, A.S.H.; Farghl, A.A.M. Biological Control of Fusarium Wilt Disease of Tomato Plants Using Seaweed Extracts. Arab. J. Sci. Eng. 2020, 45, 4557–4570.
- Ara, J.; Sultana, V.; Qasim, R.; Ehteshamul-Haque, S.; Ahmad, V.U. Biological Activity of Spatoglossum asperum: A Brown Alga. Phyther. Res. 2005, 19, 618–623.
- Sultana, V.; Baloch, G.N.; Ambreen; Ara, J.; Tariq, M.R.; Ehteshamul-Haque, S. Comparative Efficacy of a Red Alga Solieria robusta, Chemical Fertilizers and Pesticides in Managing the Root Diseases and Growth of Soybean. Pak. J. Bot. 2011, 43, 1–6.
- Aziz, S.D.A.; Jafarah, N.F.; Sabri, S.; Wahab, M.A.A.; Yusof, Z.N.B. Antifungal Activities against Oil Palm Pathogen Ganoderma boninense from Seaweed Sources. Asia-Pacific J. Mol. Biol. Biotechnol. 2019, 27, 75–83.
- Zouaoui, B.; Ghalem, B.R. The Phenolic Contents and Antimicrobial Activities of Some Marine Algae from the Mediterranean Sea (Algeria). Russ. J. Mar. Biol. 2017, 43, 491–495.
- Ghannoum, M.A.; Rice, L.B. Antifungal Agents: Mode of Action, Mechanisms of Resistance, and Correlation of These Mechanisms with Bacterial Resistance. Clin. Microbiol. Rev. 1999, 12, 501–517.
- Martins, V.D.P.; Dinamarco, T.M.; Curti, C.; Uyemura, S.A. Classical and Alternative Components of the Mitochondrial Respiratory Chain in Pathogenic Fungi as Potential Therapeutic Targets. J. Bioenerg. Biomembr. 2011, 43, 81–88.
- Lopes, G.; Pinto, E.; Andrade, P.B.; Valentão, P. Antifungal Activity of Phlorotannins against Dermatophytes and Yeasts: Approaches to the Mechanism of Action and Influence on Candida albicans Virulence Factor. PLoS ONE 2013, 8, e72203.
- Avis, T.J. Antifungal Compounds That Target Fungal Membranes: Applications in Plant Disease Control. Can. J. Plant Pathol. 2007, 29, 323–329.
- Hajlaou, M.R.; Traquair, J.A.; Jarvis, W.R.; Bélanger, R.R. Antifungal Activity of Extracellular Metabolites Produced by Sporothrix flocculosa. Biocontrol Sci. Technol. 1994, 4, 229–237.
- Ohvo-Rekilä, H.; Ramstedt, B.; Leppimäki, P.; Slotte, J.P. Cholesterol Interactions with Phospholipids in Membranes. Prog. Lipid Res. 2002, 41, 66–97.
- Thibane, V.S.; Kock, J.L.F.; Ells, R.; van Wyk, P.W.J.; Pohl, C.H. Effect of Marine Polyunsaturated Fatty Acids on Biofilm Formation of Candida albicans and Candida dubliniensis. Mar. Drugs 2010, 8, 2597–2604.
- Stengel, D.B.; Connan, S. Natural Products from Marine Algae: Methods and Protocols. In Natural Products From Marine Algae; Humana Press: New York, NY, USA, 2015; Volume 1308, pp. 1–439.





