2.2.1. In vitro Antifungal Potential
The potential of activities presented by the metabolites produced by seaweed is influenced by a myriad of combined environmental [
81,
82] and biological [
83,
84,
85] factors of the algae species involved, in addition to the methodology adopted for the recovery of the diverse bioactive compounds [
85,
86,
87,
88,
89,
90,
91]. The antifungal potential/activity of the macroalgae follows the same pattern.
An overwhelming majority of studies reporting antifungal activity/potential come from brown algae, followed by the green and red algae (extensively reported in the
Supplementary Material; Tables S1–S12). Additionally, there are studies demonstrating an exclusive antifungal activity from brown macroalgae against fungi species (
Table 2).
Botrytis cinerea [
63],
Cladosporium herbarum [
56],
Geotrichum sp. [
63],
Phialophora cinerescens,
Phoma tracheiphila [
65],
Sclerotinia sclerotiorum, Sclerotium rolfsii [
92], and
Verticillium dahliae [
63,
93] are some examples of fungi that only presented susceptibility to algae extracts belonging to the class Phaeophyceae. Exceptions were found in the species
Colletotrichum gloeosporioides,
Pseudocercospora fijiensis [
94], and
Pyricularia oryzae [
95], which were only inhibited by red algae, a group also possessing a large amount of diverse relevant compounds [
96]. The genus
Alternaria is one of the most prevalent phytopathogenic groups, responsible for soft-rotting infections and
Alternaria blight in apple trees and tomato plants, respectively, leading to important fruit losses [
58,
97]. In addition to this genus,
Penicillium expansum and
Aspergillus niger are also soft-rotting devastating fungi for a large range of fruits and vegetables. In a work performed by Vehapi, the in vitro antifungal potential of a green alga,
Ulva lactuca, was demonstrated, suggesting the presence of polyphenols responsible for the oxidation of important elements present in
Alternaria alternata and
P. expansum [
58].
Colletotrichum is one of the most devastating genera of phytopathogenic fungi, due to its cross-infection capacity affecting a large range of hosts, including fruit trees (tropical and Mediterranean species), vegetables, and one of the most economically important plants, sugarcane [
42,
47,
65,
66,
67,
68,
69]. The enormous losses caused in strawberry cultures are noticeable [
98]. Moreau and colleagues reported significant inhibitory activity exhibited by hexane extracts of brown algae,
Dictyota dichotoma and
Dilophus spiralis, against
Colletotrichum acutatum [
65]. This species can damage the fruit (black spot) and root (necrosis and crown rot) of strawberry, pepper, eggplant, tomato, and beans. Additionally,
Colletotrichum falcatum, a causative agent of red rot in sugarcane, is responsible for losses of hundreds of million dollars every year [
99,
100]. Ambika and Sujatha [
66] tested the susceptibility of this fungus to the aqueous and ethanolic extracts of
Sargassum myricocystum,
Gracilaria edulis, and
Caulerpa racemosa, and observed higher antifungal activity in brown algae, corroborating their higher potential. The ethanolic solvent used promoted the extraction of lipophilic compounds from macroalgae that are known for their antifungal activity. Also present in brown algae is a subgroup of phenolic compounds, the flavonoids, possessing a wide range of bioactivities, antifungal activity among them [
66]. Rhodophyta algae also exhibited antifungal activity against the agents responsible for anthracnose,
Colletotrichum species, in tropical crops [
67] and
Capsicum annuum plants [
69]. The high inhibition of red algae observed against
C. gloeosporioides and
Colletotrichum musae can be related to the natural compounds produced by algae as a defence mechanism against microbial attack [
101,
102]. The sessile characteristic of the algae leads to the production of phenols [
103] and terpenes (di-, sesquiterpenes) [
102], including halogenated monoterpenes, [
101] to self-protect under stress conditions [
68], and other compounds, such as fatty acids [
104], to which can be attributed antifungal activity against phytopathogenic fungi [
102]. Moreover, Mani and Nagarathnam demonstrated the capacity of ƙ-carrageenan, a polysaccharide produced by the Rhodophyta group, to alter the membrane permeability of
C.
gloeosporioides, an antifungal mechanism that can suppress their development [
69].
The genus
Fusarium is the most devastating soil-borne agent for several crops, and is known to produce toxins that are prejudicial for animals and in plants to be responsible for fusarium wilting, snow mold, the whitening of ears in crops, and root rot diseases [
52,
57]. Although the majority of studies focus on the evaluation of algae extracts as antifungal agents against two persistent phytopathogenic species,
Fusarium oxysporum and
Fusarium solani (
Table 2), which are involved in vascular bundle wilt with incidence in various economically relevant plants, such as eggplant, watermelon [
72], pigeon pea [
105], sunflower, and tomato [
75], there are also a high number of studies reporting the potential of algae extracts tested against a wide range of other
Fusarium species [
51,
57,
63,
96].
Diverse macroalgae species belonging to red, green, and brown macroalgae have been investigated for their antifungal potential against
Fusarium species, and their potential has been observed in in vitro assays, as well as in field and in greenhouse conditions [
71]. Rizvi and Shameel reported a higher susceptibility to methanolic extracts produced by Chlorophyta, Phaeophyta, and Rhodophyta in
F. solani, while
F. moniliforme was only inhibited by methanolic extracts from brown and red alga,
Dictyota hauckiana and
Botryocladia leptopoda, respectively, showing a different interaction between extracts and fungal species [
96]. In another work, Tyśkiewicz and colleagues presented the antifungal activity of an aqueous extract, obtained by supercritical carbon dioxide extraction from
Fucus vesiculosus, as a potential antifungal agent and/or fungistatic due to the complete degradation of macroconidia of
F. oxysporum and
F. culmorum [
57] observed in in vitro tests. Such results are extremely important since these globally spread species are very persistent in soil, making their elimination much more challenging.
Malini [
51] tested different promising organic solvents to extract bioactive compounds possessing antimicrobial activity. Their antifungal potential was confirmed, and all the organic extracts of
Anthophycus longifolius (then identified as
Sargassum longifolium) were able to inhibit the growth of
Fusarium sp., chloroform highlighted as the most effective solvent [
51]. A diversified range of different compounds was identified in this extract, namely proteins, phenolic compounds, alkaloids, coumarin, and sugars [
51]. Some of these compounds, such as phenolic compounds, in addition to terpenoids, a class of organic compounds usually abundant in brown algae, are commonly reported to possess antifungal activity [
63] against phytopathogenic fungi belonging to the
Fusarium genus [
106]. Additionally, the high antifungal activity of the chloroform extract of
Hormophysa cuneiformis and the methanolic extract of
Polycladia myrica (then named as
Cystoseira myrica) and
Sargassum cinereum against
Fusarium spp. have been associated with their richness in fatty acids, including saturated (lauric acid, palmitic, myristic, and stearic), monounsaturated, and polyunsaturated fatty acids (arachidonic, dihomo-γ-linolenic, and cis-11,14-eicosadienoic) [
56], as well as to the presence of essential oils with antimicrobial activities already described [
107]. Specifically, some of these acid compounds were tested against
Fusarium spp., and lauric, myristic, and palmitic acids demonstrated moderate inhibitory activity [
108]. In the study of Ambreen et al. [
109], the presence of polyunsaturated esters was found to be responsible for the antifungal activity of an ethanolic extract of
Sargassum ilicifolium against
F. oxysporum by disrupting its membrane.
In parallel with the
Fusarium genus, several studies have been developed to combat the propagation of phytopathogenic
Macrophomina phaseolina [
110], since this species is known to cause significant damages in food crops, including plants used in human diets [
14,
52,
111,
112]. Khan and colleagues found a general inhibitory activity against this species in the extracts of green, brown, and red algae [
52]. However, a higher activity from the aqueous and methanolic extracts obtained from
Sargassum tenerrinum was registered. Despite the common existence of some differences between algae species from the same genus [
52],
Sargassum ilicifolium [
109],
S. swartzii [
71], and
S. binderi [
74] have also demonstrated potential to inhibit
M. phaseolina growth. Among brown algae, relevant inhibitory activity was also revealed by
Cystoseira indica [
109],
Dictyota indica,
Padina tetrastomatica,
S. polypodioides (previously identified as
S. marginatum) [
71],
Stokeyia indica, and
Spatoglossum variabile [
72,
74]. As reported above for
Fusarium, the brown algae extracts seem to be more effective than the remaining algae groups, which may be due to the presence of polyphenols [
52] and/or 1-aminocyclopropane-1-carboxylic acid [
71,
113], which may also be the reason for their activity against
M. phaseolina. The effectiveness of the dry powder obtained from
Melanothamnus afaqhusainii [
72,
74] and
S. robusta [
71] demonstrated the potential of red algae in planta assays. The potential of the Rhodophyta group was also confirmed in vitro, namely, with
Centroceras sp.,
Ceramium sp.,
Gelidium pulchrumi,
Gracilaria corticate,
Halymenia porphyriformis,
Hypnea musciformis,
Jania pedunculata var.
adhaerens,
Neoporphyra perforate, and
Osmundea pinnatifida [
52], which presented antifungal activity against
M. phaseolina. Though to a lesser extent, the antifungal activity of green algae against this fungus species was also demonstrated in vitro with
C. racemosa,
C. taxifolia,
Chaetomorpha antennina,
Codium indicum,
Udotea sp., and
Ulva rigida [
52], and also in planta using dry powder
Rhizoclonium implexum and
H. tuna [
71,
74]. Some of the compounds associated with the antifungal activity from macroalgae extracts are the volatile compounds in the essential oils [
72], namely alcohols, aldehydes, carboxylic acids, ketones, esters, and hydrocarbons [
114].
Similar to the studies performed with
Fusarium species and
M. phaseolina, Khan [
52] also tested a diverse set of algae extracts against the growth of the soil-borne fungus
Rhizoctonia solani [
52]. Susceptibility to red, green, and brown algae was observed, but to a lesser extent than when the extracts were obtained using water instead of methanol [
52]. Curiously, for some of the macroalgae, inhibitory activity was observed only with the methanolic extracts. The suppression of this fungus was influenced by the different compounds, which resulted from the use of different solvents during the macroalgae extraction procedure, highlighting the type of extraction as a major factor in obtaining antifungal compounds, with the methanolic extracts presenting an overall higher activity [
52]. In the same study, a predominance of brown algae exhibiting antifungal activity (
Table 2) was observed. This is in agreement with the high diversity of classes of compounds typically found in brown algae, confirming their compositional diversity and revealing their antifungal bioactivities [
52]. This capacity is usually associated with phenolic compounds, specifically phlorotannins, which are very abundant in Phaeophyceae algae, and also with crinitol, an acyclic diterpene alcohol already described with antimicrobial activity against a wide range of microorganisms [
115,
116]. Recently, the chemical characterization by gas chromatography coupled to mass spectroscopy (GC–MS), of a brown alga extract,
Sargassum tenerrimum, possessing inhibitory activity against spore germination/growth of
R. solani, demonstrated a high abundance of n-hexadecanoic acid [
79]. Considering the vestigial quantities of the remaining compounds analysed, the antifungal activity presented by
S. tenerrimum was associated with this saturated long-chain fatty acid. However, this does not remove the need for more compositional tests with the remaining algae that also presented activity [
79], as well as the isolation and analysis of specific compounds, to unravel the molecular mechanism underlying the antifungal activity of macroalgae extracts. Promising results were also obtained in planta with the crude algae and extracts obtained from a diverse group of green, red, and brown seaweeds against
R. solani infection of soybean and pepper plants [
71], eggplant, watermelon [
72], cotton crops [
74], sunflower, and tomato plants [
75].
Table 2. Compilation of the best antifungal activities of macroalgae extracts against phytopathogenic fungi obtained using in vitro methodologies (summary of the information available in Scopus up until 25 February 2021). Detailed information regarding the results obtained for each study can be consulted in
Supplementary Tables S1–S10. * Algae species not found in the Algaebase database.
2.2.2. Potential Antifungal Mechanisms
The mode of action of antifungal compounds extracted by macroalgae is still poorly understood. Generally, a fungus can be affected by compounds directly targeting the cell wall or membrane, two important components that contact with the exterior environment, or intracellular organelles, such as nucleic acids or mitochondria. Antifungal agents that enter into the cell can disrupt protein synthesis by their interaction with nucleic acids [
124], as well as disturb the homeostasis and stability of the cell by interfering with the mitochondrial respiratory chain [
125,
126].
An important target usually affected by commercial antifungal products is the fungal membrane [
124,
126]. The cell membrane is a primary and crucial component for guaranteeing cellular stability in a fungal organism [
53]. Abnormalities and events occurring at the membrane level can disturb cell stability, leading to the reduction of cell lifespan [
127]. Fatty acids are a vast and diversified group of compounds present in macroalgae and have been mentioned several times throughout this work due to their antifungal potential. The unique composition of fatty acids, characterized by the presence of a carboxyl group at one end and a methyl group at the other chain end, allows their insertion into the fungal membrane, promoting an increase of fluidity and, consequently, their permeability, modifying their conformational organization and culminating in cell death [
54]. This antifungal mechanism was demonstrated by Hajlaou and colleagues against relevant fungal species, such as
Cladosporium cucumerinum,
B. cinerea and
Fusarium oxysporum f.sp.
radicislycopersici [
128], affecting conidia germination and fungal biomass production.
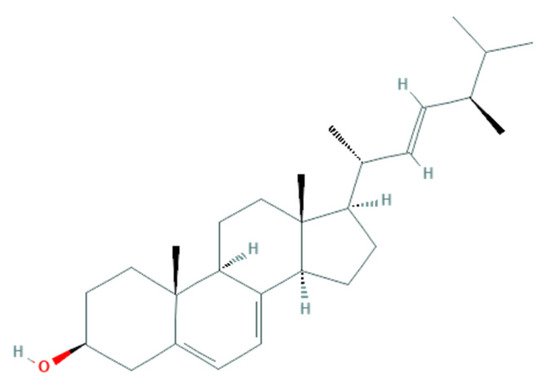
Another antifungal mechanism proposed is related to sterol present in the fungal membrane. Some algae compounds have the capacity to interact/inhibit sterol synthesis [
124]. One example is observed with the algae-based products of
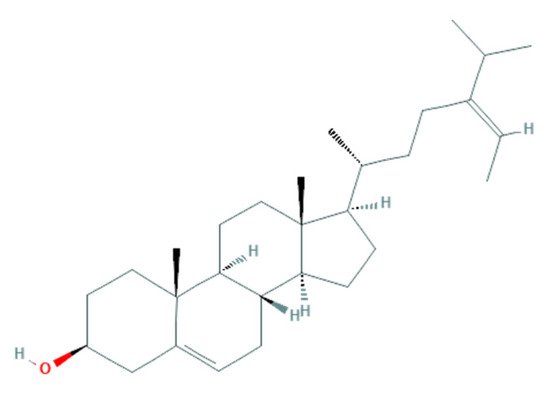
F. vesiculosus, presenting a high content of fucosterol, a natural sterol isolated from brown algae, known to possess fungistatic and antifungal activity against
F. culmorum [
57]. The similarity of this algae-derived sterol (
Figure 1) with ergosterol (
Figure 2) (a sterol in the fungal membrane, responsible for stability) allows the interaction of fucosterol with fungal membrane modulators to disturb their normal regulation (
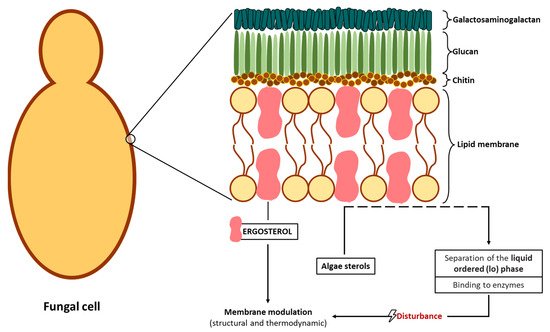
Figure 3) [
129], as well as the increase of the fluidity of the membrane components [
53].
Figure 3. Schematic representation of a possible interaction of algae sterols in the fungal membrane.
Another antifungal mechanism is demonstrated by
Candida spp. [
130] against filamentous phytopathogenic fungi. This action is related to the chemical characterization of unsaturated fatty acids (defined by one or more C=C bond/s), which can improve the antifungal action of these compounds. This property is associated with the easy incorporation of polyunsaturated lipids into the fungal membrane, which also contributes to the destabilization of cell structure, triggering events of oxidative stress [
128] known to act against several species of phytopathogenic fungi, such as
Alternaria solani,
A. niger,
B. cinerea,
C. cucumerinum,
F. oxysporum, and
Rh. solani [
53].
Another important group of compounds presenting antifungal potential are phenolic compounds. Among them, phlorotannins are highlighted as one of the relevant antifungal compounds of brown algae, as presented above. However, the antifungal mechanism of these compounds has only been clarified for yeast species [
126,
131].