Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | ANDREA MONTAGNANI | + 852 word(s) | 852 | 2021-12-03 02:59:19 | | | |
| 2 | Camila Xu | Meta information modification | 852 | 2021-12-13 04:41:45 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Montagnani, A. RAASi Therapy. Encyclopedia. Available online: https://encyclopedia.pub/entry/17011 (accessed on 10 May 2026).
Montagnani A. RAASi Therapy. Encyclopedia. Available at: https://encyclopedia.pub/entry/17011. Accessed May 10, 2026.
Montagnani, Andrea. "RAASi Therapy" Encyclopedia, https://encyclopedia.pub/entry/17011 (accessed May 10, 2026).
Montagnani, A. (2021, December 12). RAASi Therapy. In Encyclopedia. https://encyclopedia.pub/entry/17011
Montagnani, Andrea. "RAASi Therapy." Encyclopedia. Web. 12 December, 2021.
Copy Citation
RAASi therapy (ACEi, ARB, MRA, and ARNi therapy) is the cornerstone therapy for patients with heart failure with reduced ejection fraction (HFrEF).
hyperkalemia
Patiromer
sodium zirconium cyclosilicate
1. Description of the Condition
RAASi therapy (ACEi, ARB, MRA, and ARNi therapy) is the cornerstone therapy for patients with heart failure with reduced ejection fraction (HFrEF). International guidelines recommend RAAS inhibitors because of strong evidence [1][2]. Even so, there are some barriers to the implementation of this therapy, including hypotension, kidney dysfunction, and hyperkalemia (HK) [3]. HK is defined as high serum potassium (K+) levels. There is no universal cutoff for HK, but a level of 5.0 mEq/L is common [4]. HK is a life-threatening condition that leads to significant morbidity and mortality. A recent evaluation of medical records demonstrated an increase in all-cause mortality both with K+ values below the normal range and values higher than 5.5–6.0 mEq/L, with a clear effect of comorbidities, such as diabetes, heart failure, and CKD, considered individually or together [5]. In three pivotal clinical studies examining the efficacy of RAAS inhibitors in heart failure, HK occurred in these patients at a non-negligible frequency, although patients with a high risk of HK (i.e., with impaired renal function) were excluded from the studies [6][7][8].
The latest ESC guidelines recommend that RAAS inhibitors should be administered at the maximum recommended dose in order to achieve a positive effect on mortality in patients with HFrEF [1]. The protective effect of RAAS inhibitors on mortality in patients with heart failure is maximized when used in combination, but at the same time, a higher risk of HK, and therefore a worse outcome, is caused by using a combination with a sub-optimal dose of RAASi. In addition, patients who appear to benefit from the administration of RAAS inhibitors also have a higher risk of HK [9]. Recently, Rossignol et al. published data from 9222 patients with chronic heart failure in the ESC-HFA-EORP Heart Failure Long-Term Registry, confirming the association between HK and mortality. However, when the same data were adjusted for RAAS inhibitor discontinuation, HK was no longer associated with mortality, suggesting that HK may be a risk marker for RAAS inhibitor discontinuation rather than a risk factor for poorer outcomes [10]. In an unpublished survey that the Federation of Hospital Internists (FADOI) carried out among approximately 300 Italian internists in 2020, RAAS inhibitors were discontinued by 80% of physicians when potassium was more than 5.5 mEq/L or down-titrated when potassium serum values were higher than 5.0 mEq/L. This conservative and cautious approach seems to be common since HK is correlated with higher mortality; however, it induces a lack of optimization of therapy in patients with heart failure and/or CKD who are treated with RAAS inhibitors, with a consequent increase in mortality [11].
2. Description of the Intervention
Two new drugs have been developed to treat HK: Patiromer and sodium zirconium cyclosilicate (SZC). Patiromer is a spherical, non-resorbed, metal-free, cross-linked fluoroacrylate polymer [12]. Sodium zirconium cyclosilicate is a non-absorbed, non-polymeric inorganic powder with a uniform microporous structure [13].
3. How the Intervention Might Work
Most potassium is eliminated through the kidneys, but 5–10% is eliminated in the colon. Both Patiromer and SZC remove potassium by exchanging cations (calcium and sodium for Patiromer and SZC, respectively) for potassium in the gastrointestinal tract, binding potassium and increasing its fecal excretion. Patiromer was developed to provide a higher potassium binding capacity compared with the polystyrene sulfonate polymer. It has improved physical properties, such as a low swell ratio that allows for minimal water uptake, and, importantly, Patiromer uses calcium rather than sodium as the exchange cation. Patiromer was completely ionized for optimal ion exchange at the physiological pH of the large intestine, where the potassium concentration in the gastrointestinal tract was the highest [12]. Sodium zirconium cyclosilicate is highly selective for potassium ions in vitro, even in the presence of other cations, such as calcium and magnesium. Sodium zirconium cyclosilicate traps potassium throughout the gastrointestinal tract (GI) and reduces the concentration of free potassium in the GI lumen, thereby lowering serum potassium and increasing fecal potassium excretion to resolve HK [13].
4. Effects of Interventions
NPBs seemed to have an effect on the optimization of MRA therapy, with a risk ratio (95% CI) of 1.24 (1.09, 1.42); there was no heterogeneity (I2 = 0%, moderate certainty evidence). Patiromer seemed to have an effect on MRA optimization; in two studies with 232 patients, the risk ratio (95% CI) was 1.25 (1.08, 1.45) with I2 = 0 (high certainty evidence). ZSC seemed to have no effect on MRA optimization, as shown by one study with 176 patients (risk ratio (95% CI)), 1.19 (0.89, 1.59) (low certainty evidence)) (Figure 1). The subgroup analysis did not show heterogeneity between the subgroups (I2 = 0%).
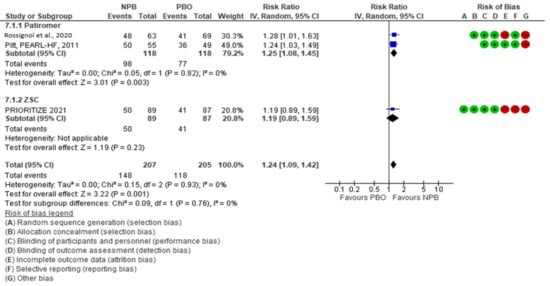
Figure 1. MRA optimization.
We conducted a sensitivity analysis of the MRA optimization outcome. The fixed effects model did not change the magnitude or direction of effect: the NPB RR (95% CI) was 1.25 (1.09, 1.42) (Figure 2); the same was observed for the exclusion of the study with a high risk of bias, with an RR (95% CI) of 1.25 (1.08, 1.45).
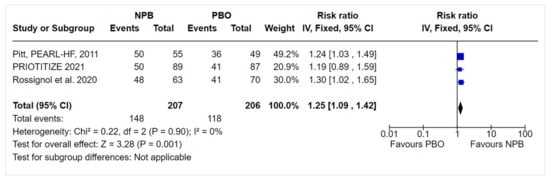
Figure 2. Sensitivity analysis.
Optimization of ACEi/ARB therapy: There was only one study with Patiromer; it did not seem to have an effect on ACEi/ARB optimization; in 49 patients, the risk ratio (95% CI) was 1.20 (0.85, 1.67) (low certainty evidence).
Optimization of ARNi therapy: No data were available.
References
- Ponikowski, P.; Voors, A.A.; Anker, S.D.; Bueno, H.; Cleland, J.; Coats, A.; Falk, V.; González-Juanatey, J.R.; Harjola, V.P.; Jankowska, E.A.; et al. 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: The Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC) Developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur. Heart J. 2016, 37, 2129–2200.
- Yancy, C.W.; Jessup, M.; Bozkurt, B.; Butler, J.; Casey, D.E., Jr.; Colvin, M.M.; Drazner, M.H.; Filippatos, G.S.; Fonarow, G.C.; Givertz, M.M.; et al. 2017 ACC/AHA/HFSA Focused Update of the 2013 ACCF/AHA Guideline for the Management of Heart Failure: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Failure Society of America. Circulation 2017, 136, e137–e161.
- Epstein, M.; Reaven, N.L.; Funk, S.E.; McGaughey, K.J.; Oestreicher, N.; Knispel, J. Evaluation of the treatment gap between clinical guidelines and the utilization of renin-angiotensin-aldosterone system inhibitors. Am. J. Manag. Care 2015, 21, S212–S220.
- Rosano, G.; Tamargo, J.; Kjeldsen, K.P.; Lainscak, M.; Agewall, S.; Anker, S.D.; Ceconi, C.; Coats, A.; Drexel, H.; Filippatos, G.; et al. Expert consensus document on the management of hyperkalaemia in patients with cardiovascular disease treated with renin angiotensin aldosterone system inhibitors: Coordinated by the Working Group on Cardiovascular Pharmacotherapy of the European Society. Eur. Heart J. Cardiovasc. Pharmacother. 2018, 4, 180–188.
- Collins, A.J.; Pitt, B.; Reaven, N.; Funk, S.; McGaughey, K.; Wilson, D.; Bushinsky, D.A. Association of serum potassium with all-cause mortality in patients with and without heart failure, chronic kidney disease, and/or diabetes. Am. J. Nephrol. 2017, 46, 213–221.
- Pitt, B.; Zannad, F.; Remme, W.J.; Cody, R.; Castaigne, A.; Perez, A.; Palensky, J.; Wittes, J. The effect of spironolactone on morbidity and mortality in patients with severe heart failure. Randomized Aldactone Evaluation Study Investigators. N. Engl. J. Med. 1999, 341, 709–717.
- Zannad, F.; McMurray, J.J.; Krum, H.; van Veldhuisen, D.J.; Swedberg, K.; Shi, H.; Vincent, J.; Pocock, S.J.; Pitt, B.; EMPHASIS-HF Study Group EMPHASIS-HF Study Group. Eplerenone in patients with systolic heart failure and mild symptoms. N. Engl. J. Med. 2011, 364, 11–21.
- Pitt, B.; Remme, W.; Zannad, F.; Neaton, J.; Martinez, F.; Roniker, B.; Bittman, R.; Hurley, S.; Kleiman, J.; Gatlin, M.; et al. Eplerenone, a Selective Aldosterone Blocker, in Patients with Left Ventricular Dysfunction after Myocardial Infarction. N. Engl. J. Med. 2003, 348, 1309–1321.
- Savarese, G.; Carrero, J.J.; Pitt, B.; Anker, S.D.; Rosano, G.; Dahlström, U.; Lund, L.H. Factors associated with underuse of mineralocorticoid receptor antagonists in heart failure with reduced ejection fraction: An analysis of 11 215 patients from the Swedish Heart Failure Registry. Eur. J. Heart Fail. 2018, 20, 1326–1334.
- Rossignol, P.; Lainscak, M.; Crespo-Leiro, M.G.; Laroche, C.; Piepoli, M.F.; Filippatos, G.; Rosano, G.; Savarese, G.; Anker, S.D.; Seferovic, P.M.; et al. Unravelling the interplay between hyperkalaemia, renin–angiotensin–aldosterone inhibitor use and clinical outcomes. Data from 9222 chronic heart failure patients of the ESC-HFA-EORP Heart Failure Long-Term Registry. Eur. J. Heart Fail. 2020, 22, 1378–1389.
- Aldahl, M.; Jensen, A.C.; Davidsen, L.; Eriksen, M.A.; Møller Hansen, S.; Nielsen, B.J.; Krogager, M.L.; Køber, L.; Torp-Pedersen, C.; Søgaard, P. Associations of serum potassium levels with mortality in chronic heart failure patients. Eur. Heart J. 2017, 38, 2890–2896.
- Li, L.; Harrison, S.D.; Cope, M.J.; Park, C.; Lee, L.; Salaymeh, F.; Madsen, D.; Benton, W.W.; Berman, L.; Buysse, J. Mechanism of Action and Pharmacology of Patiromer, a Nonabsorbed Cross-Linked Polymer That Lowers Serum Potassium Concentration in Patients With Hyperkalemia. J. Cardiovasc. Pharmacol. Ther. 2016, 21, 456–465.
- Stavros, F.; Yang, A.; Leon, A.; Nuttall, M.; Rasmussen, H.S. Characterization of structure and function of ZS-9, a K+ selective ion trap. PLoS ONE 2014, 9, e114686.
More
Information
Subjects:
Health Care Sciences & Services
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
2.2K
Entry Collection:
Hypertension and Cardiovascular Diseases
Revisions:
2 times
(View History)
Update Date:
13 Dec 2021
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





