Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Riccardo Narducci | + 1820 word(s) | 1820 | 2021-11-17 11:05:39 | | | |
| 2 | Yvaine Wei | -23 word(s) | 1797 | 2021-11-29 03:46:25 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Narducci, R. Anion Exchange Membranes with Inorganic Fillers. Encyclopedia. Available online: https://encyclopedia.pub/entry/16423 (accessed on 26 March 2026).
Narducci R. Anion Exchange Membranes with Inorganic Fillers. Encyclopedia. Available at: https://encyclopedia.pub/entry/16423. Accessed March 26, 2026.
Narducci, Riccardo. "Anion Exchange Membranes with Inorganic Fillers" Encyclopedia, https://encyclopedia.pub/entry/16423 (accessed March 26, 2026).
Narducci, R. (2021, November 26). Anion Exchange Membranes with Inorganic Fillers. In Encyclopedia. https://encyclopedia.pub/entry/16423
Narducci, Riccardo. "Anion Exchange Membranes with Inorganic Fillers." Encyclopedia. Web. 26 November, 2021.
Copy Citation
Anion exchange membrane fuel cells (AEMFC) are clean energy conversion devices that are an attractive alternative to the more common proton exchange membrane fuel cells (PEMFCs), because they present, among others, the advantage of not using noble metals like platinum as catalysts for the oxygen reduction reaction. The AEMs are the central element of many technologically relevant devices, first and foremost alkaline membrane fuel cells (FC). These fuel cells can significantly reduce the amount of noble metal catalysts for the oxygen reduction reaction (ORR) and may represent the future of FC development.
AEMFCs
carbon nanotubes
LDH
silica
graphene
MOF
PVA
PPO
QCDs
1. 1D Materials
1.1. Titanate (TNTs) and Halloysite Nanotubes (HNTs)
Titanate nanotubes (TNTs) were first described in the 1990s. These nanostructured tubular materials present excellent characteristics and performances [1]. Among them, titanium dioxide TiO2 NT exhibit high chemical stability and catalytic activity, a high specific surface area, strong metal–support interaction, and good performances in alkaline and acidic environments. TiO2 NT have a one-dimensional (1D) structure and exists as bundled tubes with a length between 1 and 220 µm and a diameter between 30 and 80 nm. The morphology of TNT depends on which synthesis method was used, including electrochemical anodization [2][3], a template-assisted method, sol–gel chemistry [4], etc. given all these characteristics, among other applications, TNT are used as filler in fuel cell membranes [1].
One of the earliest examples of composite AEM with TNT was presented in 2018 by Elumalai and Sangeetha [5]. The same authors incorporated TNT covalently linked with an imidazolium-based ionic liquid (IL) into quaternized polysulfone (QPSU) with triethylamine (TEA) [6]. The presence of the IL increased the IEC; the best conductivity was observed for 5 wt% IL-TNT (21 mS cm−1, 30 °C), due to the uniform distribution of the filler without any cluster formation. The same composite membrane reached a maximum tensile strength (TS) of 43 MPa and a fuel cell power density of 302 mW cm−2 at 60 °C under 80% RH. Quaternized TNT (QTNT) were also incorporated in quaternized poly(arylene ether ketone) (QPAEK) [7].
Halloysite nanotubes (HNTs), natural two-layered aluminosilicate clay minerals, were quaternized (QHNTs) with imidazolium groups bearing different functionalities (butyl, decyl, carbethoxyl, and benzyl moieties) and then embedded into a chitosan (CS) matrix [8]. Figure 1 shows the TEM images of HNTs and QHNTs. HNTs presented a clear tubular structure while after modification QHNTs showed a well-defined polymeric layer.

Figure 1. TEM images of (a) halloysite nanotubes and (b) quaternized halloysite nanotubes. Reproduced with permission from Ref. [8].
1.2. Carbon Nanotubes (CNTs)
Carbon nanotubes are a relaxation of the spherical structure of fullerene rolled up on itself, obtaining the typical cylindrical structure. They can be divided into two types:
-
SWCNT (Single-Walled Carbon NanoTubes) consist of a single graphitic sheet wound on itself.
- MWCNT (Multi-Walled Carbon NanoTubes) are formed by several sheets coaxially wound one on top of the other.
2. 2D Materials
2.1. Layered Double Hydroxides (LDHs)
Layered double hydroxides (LDHs) are inorganic lamellar ionic materials belonging to the group of anionic clays and their synthesis has a low cost. The structure of LDH is based on Mg(OH)2 brucite-type blocks where the replacement of M2+ with M3+ cations gives positively charged layers, balanced by mobile anions in the interlayer, which can be reversibly inserted. The lamellae are linked by Van der Waals forces. LDHs with an acceptable anionic conductivity and excellent stability in alkaline media can be used successfully as filler in AEM [9]. Well dispersed LDHs increase the mechanical properties of the matrix and can help to mitigate the loss of conductivity observed in the case of non-conducting fillers.
2.1.1. Poly(vinyl alcohol) (PVA) and Chitosan (CS)
One of the first attempts to use LDH in a composite date back to 2012 when Zhao et al. dispersed LDH in crosslinked PVA [10]. SEM images showed a good homogeneity without aggregates or chunks until 30 wt% of LDH (30LDH). FC with a PVA/20LDH membrane achieved a maximum power density of 82 mW cm−2 at 80 °C.
Exfoliated LDH nanosheets, obtained via the filtration process, were inserted in quaternized PVA [11]. The conductivity of the LDH(NO3−) membrane after exfoliation and restacking was around 200 mS cm−1 at 80 °C, which seems surprisingly high especially for nitrate ions and also higher than the OH− form. The improvement of conductivity was attributed to the exposed surfaces of the nanosheets, the enhanced water uptake, and long-range ordered ionic channels. For the composite with 10 wt% of PVA, (LDH(NO3−)/QPVA-10) was reached at the same temperature 172.1 mS cm−1.
Other examples concern the use of mixed membranes composed of QCS and PVA; they hosted LDH intercalated with glycine betaine [12], an LDH flower-like hierarchical structure wrapped on SiO2 nanospheres (LDH@SiO2) [13], and carbon nanotubes coated with LDH (LDH@CNTs) [14]. In the last example, LDH nanosheets were anchored on carbon nanotubes by a NH4F-assisted in situ coating method (Figure 2) [14].

Figure 2. Preparation of LDH coated carbon nanotubes (LDH@CNTs). Reproduced with permission from Ref. [14].
2.1.2. Polysulfone (PSU)
An attempt to use an inexpensive commercial polymer was proposed in 2017 by Di Vona et al. [9]. The composites were prepared from QPSU grafted with TMA or 1,4-diazabicyclo[2.2.2]octane (DABCO) and 14 wt% of LDH, synthesized by the urea method with a composition Mg0.62Al0.38(OH)2(Cl)0.38·0.6H2O. The ionic conductivity at 60 °C as a function of RH is reported in Figure 3: composite membranes showed lower conductivity values due to the lower water uptake, while polymers with TMA were more conductive because of the higher water content. The mechanical properties in fully humidified conditions were improved, with a three-fold increase in Young’s modulus for composites (~600 MPa vs. ~200 MPa). The membranes were treated in alkaline conditions at 60 °C without losing their properties.
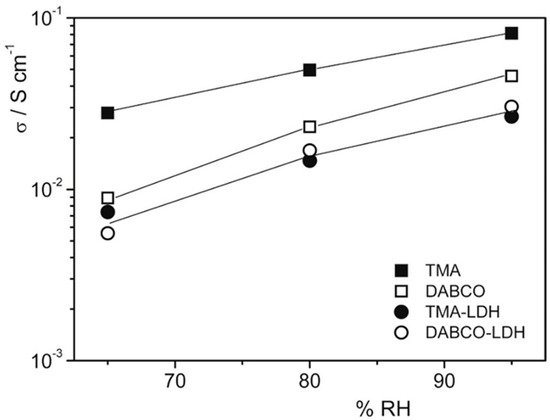
Figure 3. Conductivity measurements at 60 °C as a function of RH% of membranes after treatment at 25 °C in 2 M KOH for 24 h. Reproduced with permission from Ref. [9].
2.2. MXenes
MXenes are a family of 2D layered materials which include transition metal nitrides, carbides, and carbonitrides produced by selective exfoliation of MAX phases, where “M” refers to early d-block transition metals, “A” to groups 13 and 14 (main sp elements) and “X” to carbon or nitrogen atoms [15]. MXenes present several properties, such as hydrophilicity, fast ion transport and intercalation, large surface area, etc., provoking interest in many scientific applications including FC.
3. 3D Materials
3.1. Silica and Silicates
Silicon dioxide and silicates are the most abundant classes of minerals in nature, and their cost is practically negligible. Phyllosilicates, such as montmorillonite and palygorskite, are hydrated silicates of aluminium and/or magnesium with the ability to exchange ions. Notable successes were obtained by using Si and its derivatives as fillers in proton exchange membranes [16][17]; later their use was extended to AEM. Sol–gel silica composites were described in a recent review by Sgreccia et al. [18].
3.1.1. Poly(vinylidene fluoride) (PVDF) and Poly(vinyl alcohol) (PVA)
One of the first examples was proposed by Zuo et al. who prepared a composite with TMA-PVDF and SiO2 [19]. The membrane with 2 wt% of SiO2 showed the best specific conductivity (3 mS cm−1). More recently silica-coated PVDF (SiO2@PVDF) electrospun nanofibers were quaternized and inserted in QCS [20]. Tests in alkaline DMFC, performed at various MeOH concentrations, showed a maximum power density at 80 °C up to 99 mW cm−2 (2 M MeOH), presenting only 4% of performance loss after 100 h in chronoamperometry test.
PVA was often used as matrix for silica hybrids [21][22]. A DMAFC maximum power density (97 mW cm−2, 80 °C with 2 M MeOH + 6 M KOH) was achieved with quaternized PVA and 5 wt% of nanosized (14 nm) fumed silica (FS) due to its higher free volume and hydroxide ion transfer phenomenon [23]. A different approach was proposed by Lu et al. where cellulose nanocrystal (CNC)-based composite films were mixed with 40% of hydrophobic binder (PVA : silica gel = 1:2) [24]. Due to the hydrophilicity and dimensional stability of CNCs, membranes exhibited high water uptake (~80%) but low water swelling (~5%).
3.1.2. Aromatic Polymers
Aromatic polymers were also a preferred choice in the building of silica-containing membranes for fuel cells, diffusion dialysis (DD), and redox flow batteries. Pan et al. in 2015 improved the efficiency of acid recovery by DD with the fabrication of a quaternized PPO/SiO2 hybrid material (QPPO–SiO2) obtained by electrospinning and post-treatment (solvent fumigation and hot-press) [25]. The main fiber diameters were between 200–300 nm with an average of ~250 nm as shown in Figure 4.
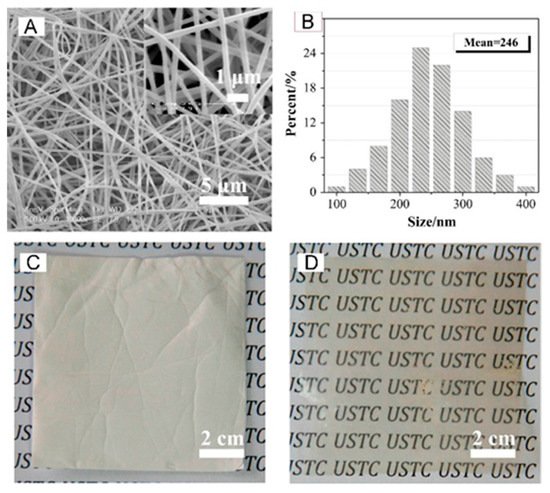
Figure 4. SEM images (A) and fiber diameter distribution (B) of QPPO–SiO2; optical photographs of nanofiber mats (C) and membrane (D) after hot-press. Reproduced with permission from Ref. [25].
3.2. Metal Oxides and Derivatives
One of the major uses of metal oxides as filler in AEM is in improving the mechanical properties of soft polymers such as PVA. Various oxides are used, including Al2O3, a well known hard material characterized by poor thermal and electrical conductivity, TiO2, non-expensive and non-toxic with high chemical stability, and ZrO2, a polymorphous crystalline oxide with high ionic conductivity when doped with acceptor cations and low electronic conductivity. The chosen matrices were mainly PVA and aromatic polymers.
3.2.1. Aluminum Oxides
PVA quaternized with glycidytrimethyl ammonium chloride and 10 wt% Al2O3 (QPVA/Al2O3) displayed in DMAFC a maximum power densities of 36 mW cm−2 with 4 M KOH + 4 M CH3OH, as shown in Figure 5 [26]. Although SEM images indicated some chunks and aggregates randomly distributed on the surface, the storage modulus slightly increased with respect to pure PVA (172 vs. 151 MPa at 100 °C).
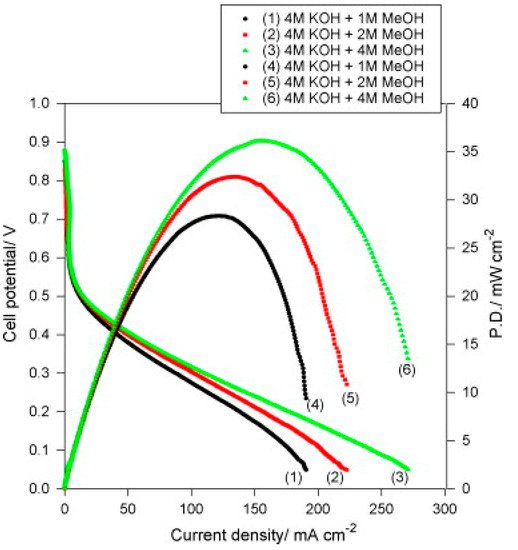
Figure 5. DMFC curves of QPVA/10 wt% Al2O3 composite membrane with various fuels (4 M KOH + x M CH3OH) at 25 °C and in ambient air. Reproduced with permission from Ref. [26].
3.2.2. Titanium Dioxide and Titanates
Many aromatic polymers were used as host matrices: PSU and quaternized polystyrene-block-poly(ethylene-ran-butylene)-block-polystyrene (PSEBS) [27], TMA-PSU [28], vinylbenzyl chloride-divinylbenzene copolymers (AEH) [29], PSU and TMA-PPO [30].
The effect of the doping with hydrophilic (tri(hydroxymethyl)propane, TMP) and hydrophobic (polymethyl-hydrosiloxane, PMHS) TiO2 nanoparticles was studied using DABCO-PSU as the matrix [31]. The DMA and DSC studies showed a Tg around 250 °C, due to a partial crystallization of the polymer, with a significant decrease in the contact angle (67°) for composites with hydrophilic character. The ionic conductivity was higher for the hydrophobic filler (PMHS-TiO2) related to a more homogeneous dispersion, as observed by AFM images.
Some examples of composites concern the use of ionic liquids and nanoparticles with the aim of simultaneously increasing conductivity and mechanical properties. 1-Methy-3-methylimidazolium IL and nano-TiO2 were mixed to TEA-PPO [32]. The conductivity increased with the amount of IL and TiO2 and the best mechanical properties were reached for 15 wt% IL and 1 wt% of TiO2 with Young’s modulus of 921 MPa and 6% of elongation at break. The degradation rates of the ion conductivity measured after treatment in 4 M NaOH solution at RT for 280 h, showed for 1 wt% of TiO2 a reduction of around 30% instead of 80% without the nanofiller, confirming the role of TiO2 in stabilizing the IL in the membranes [32]. The same authors inserted methyl, ethyl, hydroxyethyl IL and TiO2 into a TEA–PPO matrix [33]. The stability tests, in 4 M NaOH at RT for 288 h, revealed for the ethyl derivative a 92% retention of conductivity while the pristine membrane retained 78%.
References
- Abdullah, M.; Kamarudin, S. Titanium dioxide nanotubes (TNT) in energy and environmental applications: An overview. Renew. Sustain. Energy Rev. 2017, 76, 212–225.
- Premchand, Y.D.; Djenizian, T.; Vacandio, F.; Knauth, P. Fabrication of self-organized TiO2 nanotubes from columnar titanium thin films sputtered on semiconductor surfaces. Electrochem. Commun. 2006, 8, 1840–1844.
- Gong, D.; Grimes, C.A.; Varghese, O.K.; Hu, W.; Singh, R.S.; Chen, Z.; Dickey, E. Titanium oxide nanotube arrays prepared by anodic oxidation. J. Mater. Res. 2001, 16, 3331–3334.
- Kasuga, T.; Hiramatsu, M.; Hoson, A.; Sekino, T.; Niihara, K. Formation of Titanium Oxide Nanotube. Langmuir 1998, 14, 3160–3163.
- Elumalai, V.; Sangeetha, D. Preparation of anion exchangeable titanate nanotubes and their effect on anion exchange membrane fuel cell. Mater. Des. 2018, 154, 63–72.
- Elumalai, V.; Sangeetha, D. Synergic effect of ionic liquid grafted titanate nanotubes on the performance of anion exchange membrane fuel cell. J. Power Sources 2018, 412, 586–596.
- Lu, Y.; Hu, Z.; Buregeya, I.; Pan, X.; Li, N.; Chen, S.; Liu, Y. Composite Anion Exchange Membranes from Quaternized Poly(arylene ether ketone) and Quaternized Titanate Nanotubes with Enhanced Ion Conductivity. Fuel Cells 2019, 19, 663–674.
- Shi, B.; Li, Y.; Zhang, H.; Wu, W.; Ding, R.; Dang, J.; Wang, J. Tuning the performance of anion exchange membranes by embedding multifunctional nanotubes into a polymer matrix. J. Membr. Sci. 2016, 498, 242–253.
- Di Vona, M.L.; Casciola, M.; Donnadio, A.; Nocchetti, M.; Pasquini, L.; Narducci, R.; Knauth, P. Anionic conducting composite membranes based on aromatic polymer and layered double hydroxides. Int. J. Hydrogen Energy 2017, 42, 3197–3205.
- Zeng, L.; Zhao, T.; Li, Y. Synthesis and characterization of crosslinked poly (vinyl alcohol)/layered double hydroxide composite polymer membranes for alkaline direct ethanol fuel cells. Int. J. Hydrogen Energy 2012, 37, 18425–18432.
- He, X.Y.; Cao, L.; He, G.W.; Zhao, A.Q.; Mao, X.L.; Huang, T.; Li, Y.; Wu, H.; Sun, J.; Jiang, Z.Y. A highly conductive and robust anion conductor obtained via synergistic manipulation in intra- and inter-laminate of layered double hydroxide nanosheets. J. Mater. Chem. A 2018, 6, 10277–10285.
- Hu, Y.; Tsen, W.-C.; Chuang, F.-S.; Jang, S.-C.; Zhang, B.; Zheng, G.; Wen, S.; Liu, H.; Qin, C.; Gong, C. Glycine betaine intercalated layered double hydroxide modified quaternized chitosan/polyvinyl alcohol composite membranes for alkaline direct methanol fuel cells. Carbohydr. Polym. 2018, 213, 320–328.
- Zhao, S.; Tsen, W.-C.; Hu, F.; Zhong, F.; Liu, H.; Wen, S.; Zheng, G.; Qin, C.; Gong, C. Layered double hydroxide-coated silica nanospheres with 3D architecture-modified composite anion exchange membranes for fuel cell applications. J. Mater. Sci. 2019, 55, 2967–2983.
- Gong, C.; Zhao, S.; Tsen, W.-C.; Hu, F.; Zhong, F.; Zhang, B.; Liu, H.; Zheng, G.; Qin, C.; Wen, S. Hierarchical layered double hydroxide coated carbon nanotube modified quaternized chitosan/polyvinyl alcohol for alkaline direct methanol fuel cells. J. Power Sources 2019, 441, 227176.
- Sinha, A.; Dhanjai; Zhao, H.; Huang, Y.; Lu, X.; Chen, J.; Jain, R. MXene: An emerging material for sensing and biosensing. TrAC Trends Anal. Chem. 2018, 105, 424–435.
- Li, C.; Sun, G.; Ren, S.; Liu, J.; Wang, Q.; Wu, Z.; Sun, H.; Jin, W. Casting Nafion–sulfonated organosilica nano-composite membranes used in direct methanol fuel cells. J. Membr. Sci. 2006, 272, 50–57.
- Adjemian, K.T.; Lee, S.J.; Srinivasan, S.; Benziger, J.; Bocarsly, A.B. Silicon oxide Nafion composite membranes for proton-exchange membrane fuel cell operation at 80–140 °C. J. Electrochem. Soc. 2002, 149, A256–A261.
- Sgreccia, E.; Narducci, R.; Knauth, P.; Di Vona, M. Silica Containing Composite Anion Exchange Membranes by Sol–Gel Synthesis: A Short Review. Polymers 2021, 13, 1874.
- Zuo, X.; Yu, S.; Xu, X.; Xu, J.; Bao, R.; Yan, X. New PVDF organic–inorganic membranes: The effect of SiO2 nanoparticles content on the transport performance of anion-exchange membranes. J. Membr. Sci. 2009, 340, 206–213.
- Liu, G.; Tsen, W.-C.; Jang, S.-C.; Hu, F.; Zhong, F.; Zhang, B.; Wang, J.; Liu, H.; Wang, G.; Wen, S.; et al. Composite membranes from quaternized chitosan reinforced with surface-functionalized PVDF electrospun nanofibers for alkaline direct methanol fuel cells. J. Membr. Sci. 2020, 611, 118242.
- Wang, E.D.; Zhao, T.S.; Yang, W.W. Poly (vinyl alcohol)/3-(trimethylammonium) propyl-functionalized silica hybrid membranes for alkaline direct ethanol fuel cells. Int. J. Hydrogen Energy 2010, 35, 2183–2189.
- Yang, C.-C.; Chiu, S.-S.; Kuo, S.-C.; Liou, T.-H. Fabrication of anion-exchange composite membranes for alkaline direct methanol fuel cells. J. Power Sources 2012, 199, 37–45.
- Kumar, S.R.; Juan, C.-H.; Liao, G.-M.; Lin, J.-S.; Yang, C.-C.; Ma, W.-T.; You, J.-H.; Lue, S.J. Fumed Silica Nanoparticles Incorporated in Quaternized Poly(Vinyl Alcohol) Nanocomposite Membrane for Enhanced Power Densities in Direct Alcohol Alkaline Fuel Cells. Energies 2015, 9, 15.
- Lu, Y.; Armentrout, A.A.; Li, J.; Tekinalp, H.L.; Nanda, J.; Ozcan, S. A cellulose nanocrystal-based composite electrolyte with superior dimensional stability for alkaline fuel cell membranes. J. Mater. Chem. A 2015, 3, 13350–13356.
- Pan, J.F.; He, Y.B.; Wu, L.; Jiang, C.X.; Wu, B.; Mondal, A.N.; Cheng, C.L.; Xu, T.W. Anion exchange membranes from hot-pressed electrospun QPPO-SiO2 hybrid nanofibers for acid recovery. J. Membr. Sci. 2015, 480, 115–121.
- Yang, C.C.; Chiu, S.J.; Chien, W.C.; Chiu, S.S. Quaternized poly(vinyl alcohol)/alumina composite polymer membranes for alkaline direct methanol fuel cells. J. Power Sources 2010, 195, 2212–2219.
- Vinodh, R.; Sangeetha, D. Comparative study of composite membranes from nano-metal-oxide-incorporated polymer electrolytes for direct methanol alkaline membrane fuel cells. J. Appl. Polym. Sci. 2012, 128, 1930–1938.
- Nonjola, P.T.; Mathe, M.K.; Modibedi, R.M. Chemical modification of polysulfone: Composite anionic exchange membrane with TiO2 nano-particles. Int. J. Hydrogen Energy 2013, 38, 5115–5121.
- Xie, F.; Gao, X.; Hao, J.; Yu, H.; Shao, Z.; Yi, B. Preparation and properties of amorphous TiO2 modified anion exchange membrane by impregnation-hydrolysis method. React. Funct. Polym. 2019, 144, 104348.
- Msomi, P.; Nonjola, P.; Ndungu, P.; Ramontja, J. Poly (2, 6-dimethyl-1, 4-phenylene)/polysulfone anion exchange membrane blended with TiO2 with improved water uptake for alkaline fuel cell application. Int. J. Hydrogen Energy 2020, 45, 29465–29476.
- Derbali, Z.; Fahs, A.; Chailan, J.-F.; Ferrari, I.; Di Vona, M.; Knauth, P. Composite anion exchange membranes with functionalized hydrophilic or hydrophobic titanium dioxide. Int. J. Hydrogen Energy 2017, 42, 19178–19189.
- Chu, Y.; Chen, Y.; Chen, N.; Wang, F.; Zhu, H. A new method for improving the ion conductivity of anion exchange membranes by using TiO2 nanoparticles coated with ionic liquid. RSC Adv. 2016, 6, 96768–96777.
- Chen, Y.; Li, Z.; Chen, N.; Li, R.; Zhang, Y.; Li, K.; Wang, F.; Zhu, H. A new method for improving the conductivity of alkaline membrane by incorporating TiO2-ionic liquid composite particles. Electrochim. Acta 2017, 255, 335–346.
More
Information
Subjects:
Polymer Science
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.9K
Entry Collection:
Environmental Sciences
Revisions:
2 times
(View History)
Update Date:
29 Nov 2021
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





