
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Alexandra Rippa | + 3537 word(s) | 3537 | 2021-11-17 05:00:40 | | | |
| 2 | Jason Zhu | Meta information modification | 3537 | 2021-11-23 02:51:24 | | |
Video Upload Options
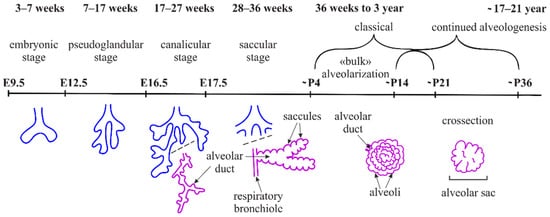
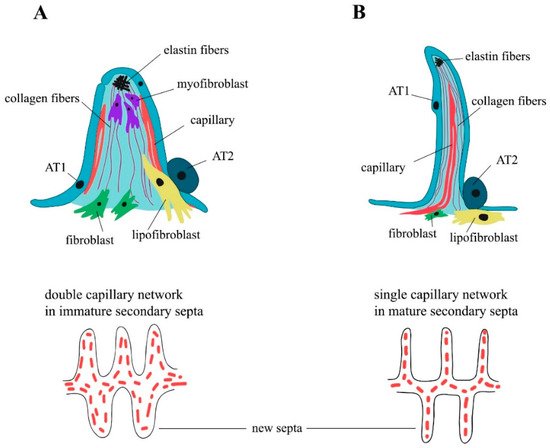
Alveologenesis is the final stage of lung maturation, when an alveolar region is divided into smaller units called alveoli via the process known as secondary septation. Each of the formed septa serves as a new gas exchange surface, and all together, they dramatically increase the respiratory surface area. Alveologenesis is divided into 2 phases: classical and continued. During the classical alveologenesis, the secondary septa are formed and the number of alveoli increases. During the continued alveologenesis, the maturation and thinning of the septa occur and the size of alveoli increases. The disruption of alveologenesis leads to the simplification of the alveoli, as seen in preterm infants diagnosed with bronchopulmonary dysplasia (BPD), a widespread pulmonary disease that is often connected with lifelong respiratory failure.
1. Introduction
2. Briefly about Embryonic Lung Development
3. Elastogenesis and Collagens in Alveologenesis
4. Postnatal Alveologenesis
References
- Pieretti, A.C.; Ahmed, A.M.; Roberts, J.D., Jr.; Kelleher, C.M. A Novel in vitro Model to Study Alveologenesis. Am. J. Respir. Cell Mol. Biol. 2013, 50, 459–469.
- Akram, K.M.; Yates, L.L.; Mongey, R.; Rothery, S.; Gaboriau, D.C.A.; Sanderson, J.; Hind, M.; Griffiths, M.; Dean, C.H. Live Imaging of Alveologenesis in Precision-Cut Lung Slices Reveals Dynamic Epithelial Cell Behaviour. Nat. Commun. 2019, 10, 1178.
- Gkatzis, K.; Taghizadeh, S.; Huh, D.; Stainier, D.Y.R.; Bellusci, S. Use of three-dimensional organoids and lung-on-a-chip methods to study lung development, regeneration and disease. Eur. Respir. J. 2018, 52, 1800876.
- Li, Y.; Wu, Q.; Sun, X.; Shen, J.; Chen, H. Organoids as a Powerful Model for Respiratory Diseases. Stem Cells Int. 2020, 2020, 5847876.
- Zepp, J.A.; Morley, M.P.; Loebel, C.; Kremp, M.M.; Chaudhry, F.N.; Basil, M.C.; Leach, J.P.; Liberti, D.C.; Niethamer, T.K.; Ying, Y.; et al. Genomic, epigenomic, and biophysical cues controlling the emergence of the lung alveolus. Science 2021, 371, eabc3172.
- Vila Ellis, L.; Cain, M.P.; Hutchison, V.; Flodby, P.; Crandall, E.D.; Borok, Z.; Zhou, B.; Ostrin, E.J.; Wythe, J.D.; Chen, J. Epithelial Vegfa Specifies a Distinct Endothelial Population in the Mouse Lung. Dev. Cell 2020, 52, 617–630.e6.
- Shannon, J.M. Induction of Alveolar Type II Cell Differentiation in Fetal Tracheal Epithelium by Grafted Distal Lung Mesenchyme. Dev. Biol. 1994, 166, 600–614.
- Shannon, J.M.; Nielsen, L.D.; Gebb, S.A.; Randell, S.H. Mesenchyme specifies epithelial differentiation in reciprocal recombinants of embryonic lung and trachea. Dev. Dyn. 1998, 212, 482–494.
- Zepp, J.A.; Zacharias, W.J.; Frank, D.B.; Cavanaugh, C.A.; Zhou, S.; Morley, M.P.; Morrisey, E.E. Distinct Mesenchymal Lineages and Niches Promote Epithelial Self-Renewal and Myofibrogenesis in the Lung. Cell 2017, 170, 1134–1148.e10.
- Riccetti, M.; Gokey, J.J.; Aronow, B.; Perl, A.-K.T. The elephant in the lung: Integrating lineage-tracing, molecular markers, and single cell sequencing data to identify distinct fibroblast populations during lung development and regeneration. Matrix Biol. 2020, 91–92, 51–74.
- Burgstaller, G.; Oehrle, B.; Gerckens, M.; White, E.S.; Schiller, H.B.; Eickelberg, O. The instructive extracellular matrix of the lung: Basic composition and alterations in chronic lung disease. Eur. Respir. J. 2017, 50, 1601805.
- Zhou, Y.; Horowitz, J.C.; Naba, A.; Ambalavanan, N.; Atabai, K.; Balestrini, J.; Bitterman, P.B.; Corley, R.A.; Ding, B.-S.; Engler, A.J.; et al. Extracellular matrix in lung development, homeostasis and disease. Matrix Biol. 2018, 73, 77–104.
- Mereness, J.A.; Mariani, T.J. The critical role of collagen VI in lung development and chronic lung disease. Matrix Biol. Plus 2021, 10, 100058.
- Loscertales, M.; Nicolaou, F.; Jeanne, M.; Longoni, M.; Gould, D.B.; Sun, Y.; Maalouf, F.I.; Nagy, N.; Donahoe, P.K. Type IV collagen drives alveolar epithelial-endothelial association and the morphogenetic movements of septation. BMC Biol. 2016, 14, 59.
- Mereness, J.A.; Bhattacharya, S.; Wang, Q.; Ren, Y.; Pryhuber, G.S.; Mariani, T.J. Type VI collagen promotes lung epithelial cell spreading and wound-closure. PLoS ONE 2018, 13, e0209095.
- Suki, B.; Ito, S.; Stamenovic, D.; Lutchen, K.R.; Ingenito, E.P. Biomechanics of the lung parenchyma: Critical roles of collagen and mechanical forces. J. Appl. Physiol. 2005, 98, 1892–1899.
- Hautamaki, R.D.; Kobayashi, D.K.; Senior, R.M.; Shapiro, S.D. Requirement for macrophage elastase for cigarette smoke-induced emphysema in mice. Science 1997, 277, 2002–2004.
- Ozsvar, J.; Yang, C.; Cain, S.A.; Baldock, C.; Tarakanova, A.; Weiss, A.S. Tropoelastin and Elastin Assembly. Front. Bioeng. Biotechnol. 2021, 9, 643110.
- Papke, C.L.; Yanagisawa, H. Fibulin-4 and fibulin-5 in elastogenesis and beyond: Insights from mouse and human studies. Matrix Biol. 2014, 37, 142–149.
- Shin, S.J.; Yanagisawa, H. Recent updates on the molecular network of elastic fiber formation. Essays Biochem. 2019, 63, 365–376.
- Yamauchi, Y.; Tsuruga, E.; Nakashima, K.; Sawa, Y.; Ishikawa, H. Fibulin-4 and -5, but not Fibulin-2, are Associated with Tropoelastin Deposition in Elastin-Producing Cell Culture. ACTA Histochem. Cytochem. 2010, 43, 131–138.
- Noda, K.; Dabovic, B.; Takagi, K.; Inoue, T.; Horiguchi, M.; Hirai, M.; Fujikawa, Y.; Akama, T.O.; Kusumoto, K.; Zilberberg, L.; et al. Latent TGF-β binding protein 4 promotes elastic fiber assembly by interacting with fibulin-5. Proc. Natl. Acad. Sci. USA 2013, 110, 2852–2857.
- Mecham, R.P. Elastin in lung development and disease pathogenesis. Matrix Biol. 2018, 73, 6–20.
- Lindahl, P.; Karlsson, L.; Hellström, M.; Gebre-Medhin, S.; Willetts, K.; Heath, J.K.; Betsholtz, C. Alveogenesis failure in PDGF-A-deficient mice is coupled to lack of distal spreading of alveolar smooth muscle cell progenitors during lung development. Dev. Camb. Engl. 1997, 124, 3943–3953.
- Boström, H.; Willetts, K.; Pekny, M.; Levéen, P.; Lindahl, P.; Hedstrand, H.; Pekna, M.; Hellström, M.; Gebre-Medhin, S.; Schalling, M.; et al. PDGF-A Signaling Is a Critical Event in Lung Alveolar Myofibroblast Development and Alveogenesis. Cell 1996, 85, 863–873.
- Mandeville, I.; Aubin, J.; LeBlanc, M.; Lalancette-Hébert, M.; Janelle, M.-F.; Tremblay, G.M.; Jeannotte, L. Impact of the Loss of Hoxa5 Function on Lung Alveogenesis. Am. J. Pathol. 2006, 169, 1312–1327.
- Hrycaj, S.M.; Marty-Santos, L.; Cebrian, C.; Rasky, A.J.; Ptaschinski, C.; Lukacs, N.W.; Wellik, D.M. Hox5 genes direct elastin network formation during alveologenesis by regulating myofibroblast adhesion. Proc. Natl. Acad. Sci. USA 2018, 115, E10605–E10614.
- Parks, W.C.; Shapiro, S.D. Matrix metalloproteinases in lung biology. Respir. Res. 2001, 2, 10–19.
- Li, C.; Lee, M.K.; Gao, F.; Webster, S.; Di, H.; Duan, J.; Yang, C.-Y.; Bhopal, N.; Peinado, N.; Pryhubar, G.; et al. The secondary crest myofibroblast PDGFRα controls elastogenesis pathway via a secondary tier of signaling networks during alveologenesis. Development 2019, 146, dev.176354.
- Chailley-Heu, B.; Boucherat, O.; Barlier-Mur, A.-M.; Bourbon, J.R. FGF-18 is upregulated in the postnatal rat lung and enhances elastogenesis in myofibroblasts. Am. J. Physiol.-Lung Cell. Mol. Physiol. 2005, 288, L43–L51.
- Bruce, M.C.; Pawlowski, R.; Tomashefski, J.F. Changes in Lung Elastic Fiber Structure and Concentration Associated with Hyperoxic Exposure in the Developing Rat Lung. Am. Rev. Respir. Dis. 1989, 140, 1067–1074.
- Bruce, M.C.; Bruce, E.N.; Janiga, K.; Chetty, A. Hyperoxic exposure of developing rat lung decreases tropoelastin mRNA levels that rebound postexposure. Am. J. Physiol.-Lung Cell. Mol. Physiol. 1993, 265, L293–L300.
- Blanco, L.N.; Frank, L. The Formation of Alveoli in Rat Lung during the Third and Fourth Postnatal Weeks: Effect of Hyperoxia, Dexamethasone, and Deferoxamine. Pediatr. Res. 1993, 34, 334–340.
- Dylag, A.M.; Haak, J.; Yee, M.; O’Reilly, M.A. Pulmonary mechanics and structural lung development after neonatal hyperoxia in mice. Pediatr. Res. 2020, 87, 1201–1210.
- O’Dell, B.L.; Kilburn, K.H.; McKenzie, W.N.; Thurston, R.J. The lung of the copper-deficient rat. A model for developmental pulmonary emphysema. Am. J. Pathol. 1978, 91, 413–432.
- Kida, K.; Thurlbeck, W.M. The effects of beta-aminopropionitrile on the growing rat lung. Am. J. Pathol. 1980, 101, 693–710.
- Wendel, D.P.; Taylor, D.G.; Albertine, K.H.; Keating, M.T.; Li, D.Y. Impaired Distal Airway Development in Mice Lacking Elastin. Am. J. Respir. Cell Mol. Biol. 2000, 23, 320–326.
- Kim, H.Y.; Pang, M.-F.; Varner, V.D.; Kojima, L.; Miller, E.; Radisky, D.C.; Nelson, C.M. Localized Smooth Muscle Differentiation Is Essential for Epithelial Bifurcation during Branching Morphogenesis of the Mammalian Lung. Dev. Cell 2015, 34, 719–726.
- Goodwin, K.; Mao, S.; Guyomar, T.; Miller, E.; Radisky, D.C.; Košmrlj, A.; Nelson, C.M. Smooth muscle differentiation shapes domain branches during mouse lung development. Development 2019, 146, dev.181172.
- Peng, T.; Tian, Y.; Boogerd, C.J.; Lu, M.M.; Kadzik, R.S.; Stewart, K.M.; Evans, S.M.; Morrisey, E.E. Coordination of heart and lung co-development by a multipotent cardiopulmonary progenitor. Nature 2013, 500, 589–592.
- Zhang, W.; Menke, D.B.; Jiang, M.; Chen, H.; Warburton, D.; Turcatel, G.; Lu, C.-H.; Xu, W.; Luo, Y.; Shi, W. Spatial-temporal targeting of lung-specific mesenchyme by a Tbx4 enhancer. BMC Biol. 2013, 11, 111.
- Kumar, M.E.; Bogard, P.E.; Espinoza, F.H.; Menke, D.B.; Kingsley, D.M.; Krasnow, M.A. Defining a mesenchymal progenitor niche at single-cell resolution. Science 2014, 346, 1258810.
- Bellusci, S.; Grindley, J.; Emoto, H.; Itoh, N.; Hogan, B.L. Fibroblast growth factor 10 (FGF10) and branching morphogenesis in the embryonic mouse lung. Development 1997, 124, 4867–4878.
- Weaver, M.; Dunn, N.R.; Hogan, B.L. Bmp4 and Fgf10 play opposing roles during lung bud morphogenesis. Development 2000, 127, 2695–2704.
- Abler, L.L.; Mansour, S.L.; Sun, X. Conditional gene inactivation reveals roles for Fgf10 and Fgfr2 in establishing a normal pattern of epithelial branching in the mouse lung. Dev. Dyn. 2009, 238, 1999–2013.
- Clément, R.; Blanc, P.; Mauroy, B.; Sapin, V.; Douady, S. Shape Self-Regulation in Early Lung Morphogenesis. PLoS ONE 2012, 7, e36925.
- El Agha, E.; Herold, S.; Alam, D.A.; Quantius, J.; MacKenzie, B.; Carraro, G.; Moiseenko, A.; Chao, C.-M.; Minoo, P.; Seeger, W.; et al. Fgf10 -positive cells represent a progenitor cell population during lung development and postnatally. Development 2014, 141, 296–306.
- Zepp, J.A.; Morrisey, E.E. Cellular crosstalk in the development and regeneration of the respiratory system. Nat. Rev. Mol. Cell Biol. 2019, 20, 551–566.
- Bellusci, S.; Furuta, Y.; Rush, M.G.; Henderson, R.; Winnier, G.; Hogan, B.L. Involvement of Sonic hedgehog (Shh) in mouse embryonic lung growth and morphogenesis. Development 1997, 124, 53–63.
- Menshykau, D.; Blanc, P.; Unal, E.; Sapin, V.; Iber, D. An interplay of geometry and signaling enables robust lung branching morphogenesis. Development 2014, 141, 4526–4536.
- Frank, D.B.; Penkala, I.J.; Zepp, J.A.; Sivakumar, A.; Linares-Saldana, R.; Zacharias, W.J.; Stolz, K.G.; Pankin, J.; Lu, M.; Wang, Q.; et al. Early lineage specification defines alveolar epithelial ontogeny in the murine lung. Proc. Natl. Acad. Sci. USA 2019, 116, 4362–4371.
- Schittny, J.C. Development of the lung. Cell Tissue Res. 2017, 367, 427–444.
- Mund, S.I.; Stampanoni, M.; Schittny, J.C. Developmental alveolarization of the mouse lung. Dev. Dyn. 2008, 237, 2108–2116.
- Li, R.; Li, X.; Hagood, J.; Zhu, M.-S.; Sun, X. Myofibroblast contraction is essential for generating and regenerating the gas-exchange surface. J. Clin. Investig. 2020, 130, 2859–2871.
- Ushakumary, M.G.; Riccetti, M.; Perl, A.T. Resident interstitial lung fibroblasts and their role in alveolar stem cell niche development, homeostasis, injury, and regeneration. Stem Cells Transl. Med. 2021, 10, 1021–1032.
- Orr-Urtreger, A.; Lonai, P. Platelet-derived growth factor-A and its receptor are expressed in separate, but adjacent cell layers of the mouse embryo. Development 1992, 115, 1045–1058.
- Gouveia, L.; Betsholtz, C.; Andrae, J. Expression analysis of platelet-derived growth factor receptor alpha and its ligands in the developing mouse lung. Physiol. Rep. 2017, 5, e13092.
- Boström, H.; Gritli-Linde, A.; Betsholtz, C. PDGF-a/PDGF alpha-receptor signaling is required for lung growth and the formation of alveoli but not for early lung branching morphogenesis: Lung Development in PDGF Receptor Alpha-Deficient Mice. Dev. Dyn. 2002, 223, 155–162.
- Prodhan, P.; Kinane, T.B. Developmental paradigms in terminal lung development. BioEssays 2002, 24, 1052–1059.
- Burri, P.H. Structural Aspects of Postnatal Lung Development–Alveolar Formation and Growth. Neonatology 2006, 89, 313–322.
- Tsao, P.-N.; Matsuoka, C.; Wei, S.-C.; Sato, A.; Sato, S.; Hasegawa, K.; Chen, H.; Ling, T.-Y.; Mori, M.; Cardoso, W.V.; et al. Epithelial Notch signaling regulates lung alveolar morphogenesis and airway epithelial integrity. Proc. Natl. Acad. Sci. USA 2016, 113, 8242–8247.
- Schittny, J.C.; Djonov, V.; Fine, A.; Burri, P.H. Programmed Cell Death Contributes to Postnatal Lung Development. Am. J. Respir. Cell Mol. Biol. 1998, 18, 786–793.
- Bruce, M.C.; Honaker, C.E.; Cross, R.J. Lung Fibroblasts Undergo Apoptosis Following Alveolarization. Am. J. Respir. Cell Mol. Biol. 1999, 20, 228–236.
- McGowan, S.E.; McCoy, D.M. Fibroblasts Expressing PDGF-Receptor-Alpha Diminish During Alveolar Septal Thinning in Mice. Pediatr. Res. 2011, 70, 44–49.
- Hagan, A.S.; Zhang, B.; Ornitz, D.M. Identification of an FGF18-expressing alveolar myofibroblast that is developmentally cleared during alveologenesis. Development 2020, 147, dev.181032.
- Srinivasan, S.; Strange, J.; Awonusonu, F.; Bruce, M.C. Insulin-like growth factor I receptor is downregulated after alveolarization in an apoptotic fibroblast subset. Am. J. Physiol.-Lung Cell. Mol. Physiol. 2002, 282, L457–L467.
- Branchfield, K.; Li, R.; Lungova, V.; Verheyden, J.M.; McCulley, D.; Sun, X. A three-dimensional study of alveologenesis in mouse lung. Dev. Biol. 2016, 409, 429–441.
- Kugler, M.C.; Loomis, C.A.; Zhao, Z.; Cushman, J.C.; Liu, L.; Munger, J.S. Sonic Hedgehog Signaling Regulates Myofibroblast Function during Alveolar Septum Formation in Murine Postnatal Lung. Am. J. Respir. Cell Mol. Biol. 2017, 57, 280–293.
- Pozarska, A.; Rodríguez-Castillo, J.A.; Surate Solaligue, D.E.; Ntokou, A.; Rath, P.; Mižíková, I.; Madurga, A.; Mayer, K.; Vadász, I.; Herold, S.; et al. Stereological monitoring of mouse lung alveolarization from the early postnatal period to adulthood. Am. J. Physiol.-Lung Cell. Mol. Physiol. 2017, 312, L882–L895.
- Schittny, J.C.; Mund, S.I.; Stampanoni, M. Evidence and structural mechanism for late lung alveolarization. Am. J. Physiol. Lung Cell. Mol. Physiol. 2008, 294, L246–L254.
- Tschanz, S.A.; Salm, L.A.; Roth-Kleiner, M.; Barré, S.F.; Burri, P.H.; Schittny, J.C. Rat lungs show a biphasic formation of new alveoli during postnatal development. J. Appl. Physiol. (1985) 2014, 117, 89–95.
- Weibel, E.R. Morphological basis of alveolar-capillary gas exchange. Physiol. Rev. 1973, 53, 419–495.
- Knudsen, L.; Ochs, M. The micromechanics of lung alveoli: Structure and function of surfactant and tissue components. Histochem. Cell Biol. 2018, 150, 661–676.





