
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Jorge Nuche | + 2021 word(s) | 8352 | 2020-08-04 04:34:45 | | | |
| 2 | Catherine Yang | -1 word(s) | 2020 | 2020-08-20 06:13:23 | | |
Video Upload Options
Review of the effect of COVID-19 on pulmonary circulation
1. Introduction
The coronavirus disease of 2019 (COVID-19) is an infectious disease that emerged in the Chinese city of Wuhan in December 2019 [1] and rapidly extended worldwide, leading to its declaration as a pandemic disease by the World Health Organization on 11 March 2020 [2]. By 30 June 2020, the rapid spread of the virus has caused more than 10,000,000 cases and more than 500,000 deaths all over the world (source: https://www.worldometers.info/coronavirus/).
COVID-19 is caused by the severe acute respiratory coronavirus 2 (SARS-CoV2), an RNA virus of the family Coronaviridae. Common symptoms include fever, cough, sore throat, and dyspnea [1][3], while the most severe cases develop pneumonia and acute respiratory distress syndrome (ARSD), requiring admission to the intensive care unit in up to 20% of hospitalized patients [4]. Reported data suggest an overall mortality rate ranging from 0.3 per 1000 cases in patients aged under 18 to 305 per 1000 cases in elderly people (>85-year-old) [4].
Precapillary pulmonary hypertension (PH) includes a variety of diseases leading to increased pulmonary artery pressure (≥25 mmHg) in the presence of normal pulmonary capillary wedge pressure (<15 mmHg) and a pulmonary vascular resistance >3 Wood units at rest [5]. Among them, pulmonary arterial hypertension (PAH) and chronic thromboembolic pulmonary hypertension (CTEPH) constitute a particular group due to its relation to poor quality of life and shortened survival [6]:
-
PAH is a rare, noncurable disease characterized by an aberrant pulmonary vascular remodeling that may be either idiopathic or related to different clinical conditions [6]. PAH-related histological changes include endothelial damage and dysfunction, vasoconstriction, vascular cell proliferation leading to vascular obliteration and wall thickening, and, eventually, to the formation of plexiform lesions [7]. Furthermore, there is a marked component of perivascular inflammation and microthrombosis [8].
-
CTEPH constitutes a different group of precapillary PH secondary to the obstruction of the pulmonary arteries by organized thrombus after a pulmonary embolism. These pulmonary vascular changes induce small-vessel vasculopathy consisting of altered vascular remodeling initiated or potentiated by a combination of defective angiogenesis, impaired fibrinolysis, and endothelial dysfunction [9].
The resultant increased pulmonary arterial pressures in PAH and CTEPH contribute to a further increase of the right ventricular afterload, thus leading to right ventricular dysfunction and reducing survival among affected patients, being heart failure the most common cause of death [10]. Hospitalizations for cardiovascular and non-cardiovascular diseases are common in these groups of patients and carry a high mortality risk, being especially high in patients admitted to the intensive care unit [11][12].
Patients with previous cardiovascular risk factors or a cardiovascular disease seem to be at a higher risk of developing severe forms of SARS-CoV-2, with higher rates of mortality described in this population [1][13][14]. Moreover, the shortage of equipment needed to care for critically ill patients in some areas has led to difficult decisions, and the patient’s short-term likelihood of surviving the acute medical episode has remained the rationale for rationing. As physicians, bearing this reality in mind, it seemed straightforward to assume that PAH and CTEPH patients were at a higher risk of developing severe forms of the disease and that their chances of recovering from such an insult were scarce.
Noteworthy, some case-series have repeatedly presented the unexpected favorable outcome of COVID-19 infection in this population, suggesting that these patients could be somehow protected against severe forms of the disease [15][16][17]. However, whether this protection is due to receiving therapy, to their special awareness about their particular risk factor or any other physiological condition related to the disease is still a remaining question.
With this comprehensive review, we aim to describe the effects of SARS-COV-2 in the pulmonary circulation and the possible differential features in patients exhibiting pulmonary microvasculopathy, such as PAH and CTEPH patients, since we believe it will help to further understand COVID-19 and its pulmonary consequences ((Figure 1).
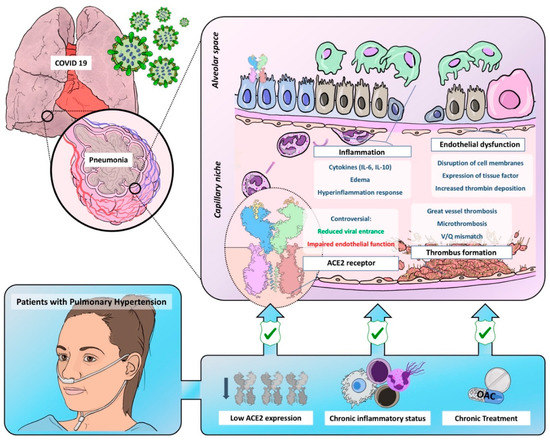
Figure 1. Graphic representation of common pathobiological characteristics between Coronavirus Disease of 2019 and pulmonary hypertension and the possible mechanisms leading to reduced virulence and severity in this population. ACE2 = angiotensin-converter enzyme 2; OAC = oral anticoagulant.
2. Endothelial dysfunction, inflammatory response, and thrombosis in COVID-19
Severe COVID-19 is characterized by a disproportional inflammatory response to an initial acute lung insult, which aggravates the lung injury and leads to a severe hypoxemic respiratory failure and, ultimately, to death [18][19][20][21][22][23]. Thus, in severe cases, the initial adaptive immune response necessary to eliminate the virus is followed by a severe and persistent inflammatory reaction, which appears to govern the clinical picture at this point, leading to further lung destruction and, therefore, ARDS. This exaggerated inflammation is mediated by several cytokines (IL-6, IL10, TNF-α, INF-γ), as well as mononuclear cells and neutrophils [18][19]. IL-6 is the central element of this cytokine storm by stimulating the expression of other cytokines and promoting edema [20]. The level of cytokines is higher in those patients who require admission to intensive care units in whom their lymphocyte count is lower. This finding suggests that the low absolute lymphocyte count observed in COVID-19 patients could be a consequence of the high serum cytokines concentrations that negatively regulate T-cell survival and proliferation [21]. Furthermore, T cells in COVID-19 express high levels of programmed cell death-1, an indicator of T-cell exhaustion [21]. Thus, it seems that the severe clinical picture described in COVID-19 patients is mediated by this inflammatory response responsible for both lung injury and impaired cellular immunity, which hampers the fight against the viral infection [21].
This immune response to the SARS-Cov-2 infection is more severe than that produced in other viral infections [22]. Older patients and those with previous comorbidities, which may present a chronic subclinical inflammatory status, could be at a higher risk of developing this cytokine storm. This could explain, at least in part, the worse prognosis described in these groups [14][22][23].
On histology, the lungs of COVID-19 patients present diffuse alveolar damage with “necrosis of alveolar lining cells, pneumocyte type 2 hyperplasia, and linear intra-alveolar fibrin deposition” [24]. The inflammatory infiltrates described in COVID-19 lungs differs from that described in H1N1 influenza lungs, with a predominant infiltration of CD4-positive T cells and a lower number of CD8-positive T cells and neutrophils in COVID-19 lungs [24]. Furthermore, there is also an extensive macrophagic infiltration contributing to the alveolar damage [25]
These inflammatory and the above-described procoagulant statuses are not isolated phenomena. The mechanisms leading to the activation of a coagulation cascade in COVID-19 are not well-defined but seem to be related to the inflammatory response also responsible for the endothelial damage and include the releasing of procoagulant factors [18][26]. COVID-19 pneumonia has some clinical, analytic, and histological similarities with macrophage activation syndrome (MAS), including the cytokine storm and high ferritin levels [25][27]. This is the rationale for the use of anti-cytokine therapies and corticosteroids in severe COVID-19 patients [27][28][29][30]. This MAS-like inflammatory response triggers the expression of the active tissue factor, which leads to a massive coagulation cascade activation, with the ulterior development of thrombotic events and widespread pulmonary microangiopathy [25]. These endothelial damages and coagulation activations are enhanced by local hypoxia constituting a deleterious vicious circle [25][31].
Furthermore, the detection of serum marker-specific neutrophil extracellular traps (NETs) in COVID-19 patients suggests a pivotal role of these structures in the marked inflammatory and prothrombotic responses. NETs are extracellular webs of chromatin, microbicidal proteins, and oxidative agents released by neutrophils to contain infections. The uncontrolled release of NETs leads to a procoagulant status and widespread inflammation [32]. Thus, an increased neutrophil-to-lymphocyte ratio is related to a severe COVID-19 course due to a disproportionate innate immune response leading to the release of cytokines and inflammatory infiltrates with tissue necrosis [33].
Thus, it seems that severe ARDS and widespread thrombosis are complementary phenomena leading to the severe clinical course described in COVID-19 patients [24].
3. Pulmonary Hypertension: A Paradigm of Endothelial Dysfunction
We have described how SARS-Cov-2 has an affinity for endothelial cells affecting not only pulmonary circulation but to other vascular territories (heart, gut, brain, and kidney). This becomes even more evident in patients with previous endothelial dysfunctions, such as hypertension or diabetes mellitus [18].
Endothelial dysfunction is one of the most relevant hallmarks and a critical contributor determining the onset and progression of PH. Pulmonary vascular endothelium controls the blood barrier integrity, the vascular tone by producing vasodilator and vasoconstrictor molecules and, also, optimizes the gas exchange in different conditions, being the main player in maintaining the vasomotor balance and vascular-tissue homeostasis. Also, the integrity of the endothelial barrier is essential for controlling an inflammatory and thrombosis-free surface. Therefore, an alteration of the endothelium leads to a pro-edematous, pro-vasoconstriction, prothrombotic and proinflammatory phenotype, which eventually alters the cell metabolism and oxidative stress, leading to a pro-proliferative and antiapoptotic phenotype all responsible in the last instance of the PH phenotype [24][34][35].
Pulmonary vascular remodeling in PAH and CTEPH is not only caused by the proliferation of different cells in the arterial wall but by the loss of precapillary arteries and by a severe chronic perivascular inflammatory infiltrate [8]. These conditions constitute a paradigm of endothelial dysfunction with a propensity to vasoconstriction; thrombosis; the production of reactive oxygen species; the expression of adhesion molecules (E-selectin, ICAM-1 and VCAM-1) and a massive release of cytokines and growth factors [8]. Thus, a devastating effect of COVID-19 would be expectable in these patients.
Few cases of COVID-19 in PAH and CTEPH patients have been reported. This observation may be explained by the low prevalence of these diseases and by the increased awareness of chronic patients with a special emphasis on social-distancing measure compliances [36][37]. However, we have recently published our surprisingly positive experience with COVID-19 in our PAH [15] and CPTEH [17] populations without any of them requiring intensive care and any deaths reported. Furthermore, a similar benign course has been observed in other centers [16][38], with no published reports suggesting particularly adverse outcomes [39] (Figure 2). Although the proportion of patients requiring intubation is similar to that described for the global COVID-19 population [4], it must be highlighted that these case-series represent a very biased population constituted only by those symptomatic patients who required hospitalization. We hypothesize that a high proportion of PAH and CTEPH patients infected by SARS-Cov-2 might have developed a mild or even an asymptomatic course and were not tested for SARS-Cov-2. Thus, the low prevalence of COVID-19 in PH patients might also be explained by this low proportion of patients with severe symptoms requiring medical attention (and, subsequently, being tested for SARS-Cov-2). The Spanish pulmonary hypertension registry is currently collecting information about all PAH and CTEPH patients diagnosed with COVID-19, aiming to improve our knowledge of the prognostic implications of the disease in this population.
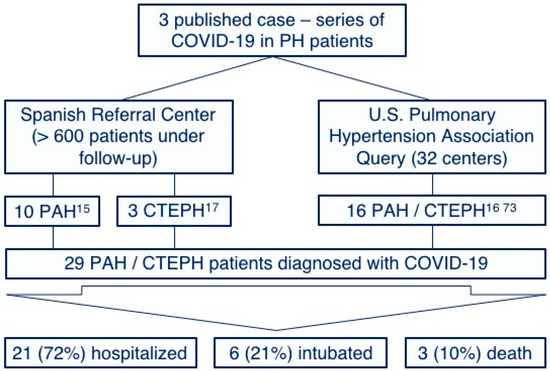
Figure 2. Reported cases and outcomes of the coronavirus disease of 2019 among pulmonary arterial hypertension and chronic thromboembolic pulmonary hypertension patients. PH: Pulmonary hypertension, PAH: Pulmonary arterial hypertension and CTEPH: Chronic thromboembolic pulmonary hypertension.
The chronic pathological findings in PAH and CTEPH somehow remind of those found in COVID-19 specimens. In fact, some experimental treatments tested for PAH such as recombinant ACE2, synthetic vasoactive intestinal peptide or IL-6 antagonists are already being tested in clinical trials for COVID-19 pneumonia, which support the hypothesis of pathophysiological similarities between both conditions [40]. This makes this population extremely vulnerable to acute conditions, especially to those causing severe respiratory failure [11]. In fact, infectious conditions are a common cause of decompensated right heart failure and death in these patients [39].
Although our observations of a benign course in PAH and CTEPH patients require caution, we described some pathophysiological characteristics that could explain this benign course [15][17].
4. Conclusions
Pulmonary damage associated to COVID-19 is a complex merge of different interrelated physiopathological processes in which endothelial dysfunction seems to play a pivotal role, leading to the development of thrombosis and vasomotor disturbances responsible for severe respiratory failure.
Although the scarcity of available data requires caution, the reported lethality of COVID-19 among PAH and CTEPH patients is low. The epidemiologic and prognostic impacts of COVID-19 in this population should be evaluated through international registries.
The role of chronic anticoagulation and specific PH therapies as protective measures against the described endothelial dysfunctions requires further investigation with placebo-controlled randomized trials, avoiding futile or even harmful interventions.
Last, due to severe endothelial dysfunctions, the higher incidences of thrombotic events and possible residual lung fibrosis, COVID-19 patients may require tight follow-ups to discard the possibility of PH as a chronic sequela of the disease.
References
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X.; et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020, 395, 497–506.
- World Health Organization. WHO Director-General’s Opening Remarks at the Media Briefing on COVID-19–11 March 2020. Available online: https://www.who.int/dg/speeches/detail/who-director-general-s-opening-remarks-at-the-media-briefing-on-covid-19---11-march-2020 (accessed on 12 March 2020).
- Zhai, P.; Ding, Y.; Wu, X.; Long, J.; Zhong, Y.; Li, Y. The epidemiology, diagnosis and treatment of COVID-19. Int. J. Antimicrob. Agents 2020, 55, 105955.
- Wiersinga, W.J.; Rhodes, A.; Cheng, A.C.; Peacock, S.J.; Prescott, H.C. Pathophysiology, Transmission, Diagnosis, and Treatment of Coronavirus Disease 2019 (COVID-19): A Review. JAMA 2020.
- Galiè, N.; Humbert, M.; Vachiery, J.L.; Gibbs, S.; Lang, I.; Torbicki, A.; Simonneau, G.; Peacock, A.; Vonk Noordegraaf, A.; Beghetti, M.; et al. 2015 ESC/ERS Guidelines for the diagnosis and treatment of pulmonary hypertension: The Joint Task Force for the Diagnosis and Treatment of Pulmonary Hypertension of the European Society of Cardiology (ESC) and the European Respiratory Society (ERS): Endorsed by: Association for European Paediatric and Congenital Cardiology (AEPC), International Society for Heart and Lung Transplantation (ISHLT). Eur. Heart J. 2016, 37, 67–119.
- Simonneau, G.; Montani, D.; Celermajer, D.S.; Denton, C.P.; Gatzoulis, M.A.; Krowka, M.; Williams, P.G.; Souza, R. Haemodynamic definitions and updated clinical classification of pulmonary hypertension. Eur. Respir. J. 2019, 53.
- Dai, Z.; Zhu, M.M.; Peng, Y.; Machireddy, N.; Evans, C.E.; Machado, R.; Zhang, X.; Zhao, Y.Y. Therapeutic Targeting of Vascular Remodeling and Right Heart Failure in Pulmonary Arterial Hypertension with a HIF-2α Inhibitor. Am. J. Respir. Crit. Care Med. 2018, 198, 1423–1434.
- Humbert, M.; Guignabert, C.; Bonnet, S.; Dorfmüller, P.; Klinger, J.R.; Nicolls, M.R.; Olschewski, A.J.; Pullamsetti, S.S.; Schermuly, R.T.; Stenmark, K.R.; et al. Pathology and pathobiology of pulmonary hypertension: State of the art and research perspectives. Eur. Respir. J. 2019, 53.
- Kim, N.H.; Delcroix, M.; Jais, X.; Madani, M.M.; Matsubara, H.; Mayer, E.; Ogo, T.; Tapson, V.F.; Ghofrani, H.A.; Jenkins, D.P. Chronic thromboembolic pulmonary hypertension. Eur. Respir. J. 2019, 53.
- Hoeper, M.M.; Kramer, T.; Pan, Z.; Eichstaedt, C.A.; Spiesshoefer, J.; Benjamin, N.; Olsson, K.M.; Meyer, K.; Vizza, C.D.; Vonk-Noordegraaf, A.; et al. Mortality in pulmonary arterial hypertension: Prediction by the 2015 European pulmonary hypertension guidelines risk stratification model. Eur. Respir. J. 2017, 50.
- Harder, E.M.; Small, A.M.; Fares, W.H. Primary cardiac hospitalizations in pulmonary arterial hypertension: Trends and outcomes from 2001 to 2014. Respir. Med. 2020, 161, 105850.
- Sztrymf, B.; Souza, R.; Bertoletti, L.; Jaïs, X.; Sitbon, O.; Price, L.C.; Simonneau, G.; Humbert, M. Prognostic factors of acute heart failure in patients with pulmonary arterial hypertension. Eur. Respir. J. 2010, 35, 1286–1293.
- Driggin, E.; Madhavan, M.V.; Bikdeli, B.; Chuich, T.; Laracy, J.; Bondi-Zoccai, G.; Brown, T.S.; Nigoghossian, C.; Zidar, D.A.; Haythe, J.; et al. Cardiovascular Considerations for Patients, Health Care Workers, and Health Systems During the Coronavirus Disease 2019 (COVID-19) Pandemic. J. Am. Coll. Cardiol. 2020, 75, 2352–2371.
- Guo, T.; Fan, Y.; Chen, M.; Wu, X.; Zhang, L.; He, T.; Wang, H.; Wan, J.; Wang, X.; Lu, Z. Cardiovascular Implications of Fatal Outcomes of Patients With Coronavirus Disease 2019 (COVID-19). JAMA Cardiol. 2020.
- Nuche, J.; Pérez-Olivares, C.; Segura de la Cal, T.; Jiménez López-Guarch, C.; Arribas Ynsaurriaga, F.; Escribano Subías, P. Clinical course of COVID-19 in pulmonary arterial hypertension patients. Rev. Esp. Cardiol. (Engl. Ed.) 2020.
- Horn, E.M.; Chakinala, M.; Oudiz, R.; Joseloff, E.; Rosenzweig, E.B. Could pulmonary arterial hypertension patients be at a lower risk from severe COVID-19? Pulm. Circ. 2020, 10.
- Cal, T.S.d.l.; Nuche, J.; López-Guarch, C.J.; Pérez-Olivares, C.; Velázquez, M.; Medrano, F.L.; Gude, M.J.L.; Charterina, S.A.; Ynsaurriaga, F.A.; Subías, P.E. Unexpected favourable course of Coronavirus Disease 2019 in chronic thromboembolic pulmonary hypertension patients. Arch. Bronconeumol. 2020.
- Connors, J.M.; Levy, J.H. Thromboinflammation and the hypercoagulability of COVID-19. J. Thromb. Haemost. 2020.
- Xu, Z.; Shi, L.; Wang, Y.; Zhang, J.; Huang, L.; Zhang, C.; Liu, S.; Zhao, P.; Liu, H.; Zhu, L.; et al. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir. Med. 2020, 8, 420–422.
- Geng, Y.J.; Wei, Z.Y.; Qian, H.Y.; Huang, J.; Lodato, R.; Castriotta, R.J. Pathophysiological characteristics and therapeutic approaches for pulmonary injury and cardiovascular complications of coronavirus disease 2019. Cardiovasc. Pathol. 2020, 47, 107228.
- Diao, B.; Wang, C.; Tan, Y.; Chen, X.; Liu, Y.; Ning, L.; Chen, L.; Li, M.; Wang, G.; Yuan, Z.; et al. Reduction and Functional Exhaustion of T Cells in Patients With Coronavirus Disease 2019 (COVID-19). Front. Immunol. 2020, 11, 827.
- Kronbichler, A.; Effenberger, M.; Eisenhut, M.; Lee, K.H.; Shin, J.I. Seven recommendations to rescue the patients and reduce the mortality from COVID-19 infection: An immunological point of view. Autoimmun. Rev. 2020.
- Li, P.; Chen, L.; Liu, Z.; Pan, J.; Zhou, D.; Wang, H.; Gong, H.; Fu, Z.; Song, Q.; Min, Q.; et al. Clinical Features and Short-term Outcomes of Elderly Patients With COVID-19. Int. J. Infect. Dis. 2020, 10, 245–250.
- Ackermann, M.; Verleden, S.E.; Kuehnel, M.; Haverich, A.; Welte, T.; Laenger, F.; Vanstapel, A.; Werlein, C.; Stark, H.; Tzankov, A.; et al. Pulmonary Vascular Endothelialitis, Thrombosis, and Angiogenesis in Covid-19. N. Engl. J. Med. 2020.
- Mc Gonagle, D.; O’Donnell, J.S.; Sharif, K.; Emery, P.; Bridgewood, C. Immune mechanisms of pulmonary intravascular coagulopathy in COVID-19 pneumonia. Lancet Rheumatol. 2020.
- Becker, R.C. COVID-19 update: Covid-19-associated coagulopathy. J. Thromb. Thrombolysis 2020.
- La Rosée, P.; Horne, A.; Hines, M.; von Bahr Greenwood, T.; Machowicz, R.; Berliner, N.; Birndt, S.; Gil-Herrera, J.; Girschikofsky, M.; Jordan, M.B.; et al. Recommendations for the management of hemophagocytic lymphohistiocytosis in adults. Blood 2019, 133, 2465–2477.
- Rios-Fernández, R.; Callejas-Rubio, J.L.; García-Rodríguez, S.; Sancho, J.; Zubiaur, M.; Ortego-Centeno, N. Tocilizumab as an Adjuvant Therapy for Hemophagocytic Lymphohistiocytosis Associated With Visceral Leishmaniasis. Am. J. Ther. 2016, 23, 1193–1196.
- Xu, X.; Han, M.; Li, T.; Sun, W.; Wang, D.; Fu, B.; Zhou, Y.; Zheng, X.; Yang, Y.; Li, X.; et al. Effective treatment of severe COVID-19 patients with tocilizumab. Proc. Natl. Acad. Sci. USA 2020, 117, 10970–10975.
- Ye, Z.; Wang, Y.; Colunga-Lozano, L.E.; Prasad, M.; Tangamornsuksan, W.; Rochwerg, B.; Yao, L.; Motaghi, S.; Couban, R.J.; Ghadimi, M.; et al. Efficacy and safety of corticosteroids in COVID-19 based on evidence for COVID-19, other coronavirus infections, influenza, community-acquired pneumonia and acute respiratory distress syndrome: A systematic review and meta-analysis. CMAJ 2020.
- Gupta, N.; Zhao, Y.Y.; Evans, C.E. The stimulation of thrombosis by hypoxia. Thromb. Res. 2019, 181, 77–83.
- Gupta, N.; Zhao, Y.Y.; Evans, C.E. The stimulation of thrombosis by hypoxia. Thromb. Res. 2019, 181, 77–83
- Tomar, B.; Anders, H.J.; Desai, J.; Mulay, S.R. Neutrophils and Neutrophil Extracellular Traps Drive Necroinflammation in COVID-19. Cells 2020, 9, 1383.
- Guignabert, C.; Tu, L.; Girerd, B.; Ricard, N.; Huertas, A.; Montani, D.; Humbert, M. New molecular targets of pulmonary vascular remodeling in pulmonary arterial hypertension: Importance of endothelial communication. Chest 2015, 147, 529–537.
- Guignabert, C. Dysfunction and Restoration of Endothelial Cell Communications in Pulmonary Arterial Hypertension: Therapeutic Implications; Springer: Singapore, 2020; pp. 147–155.
- Farha, S. COVID-19 and pulmonary hypertension. Cleve Clin. J. Med. 2020.
- Fernandes, T.M.; Papamatheakis, D.G.; Poch, D.S.; Kim, N.H. Letter to the Editor regarding “Could pulmonary arterial hypertension patients be at lower risk from severe COVID-19?”. Pulm. Circ. 2020, 10.
- Horn, E.; Chakinala, M.M.; Oudiz, R.; Joseloff, E.; Rosenzweig, E.B. EXPRESS: Author rebuttal to response regarding “Letter to the Editor regarding ‘Could pulmonary arterial hypertension patients be at lower risk from severe COVID-19?’. Pulm. Circ. 2020.
- Provencher, S.; Potus, F.; Bonnet, S. COVID-19 and the pulmonary vasculature. Pulm. Circ. 2020, 10.
- Magrone, T.; Magrone, M.; Jirillo, E. Focus on Receptors for Coronaviruses with Special Reference to Angiotensin-converting Enzyme 2 as a Potential Drug Target–A Perspective. Endocr. Metab. Immune Disord. Drug Targets 2020.





