
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Tae-Hyung Kim | + 1752 word(s) | 1752 | 2021-11-15 09:01:45 | | | |
| 2 | Nora Tang | Meta information modification | 1752 | 2021-11-17 10:28:06 | | |
Video Upload Options
Unlike the 2D cultures, which grow by attaching to the bottom as a monolayer, 3D cell culture refers to cells aggregated and expressed as a single tissue or form. Moreover, the 3D-cultured cells are attached to an artificially created ECM environment to interact with or grow with the surrounding environment. Therefore, unlike 2D cell cultures, cell growth in a 3D environment allows cells to grow in multiple directions rather than in a single direction in vitro, which is similar to in vivo conditions. Upon comparison, the 3D cell culture exhibits several advantages: (1) A similar biomimetic model, which is more physiologically relevant. (2) A 3D culture exhibits a high level of structural complexity and maintains homeostasis for longer. (3) 3D models can indicate how different types of cells interact. (4) 3D cultures can reduce the use of animal models. (5) They are a good simulator for the treatment of disease groups including cancer tumors.
1. Anchorage-Independent Approaches
1.1. Hanging-Drop Method

1.2. Rotary Cell Culture System
2. Anchorage-Independent Approaches
2.1. Porous Scaffold
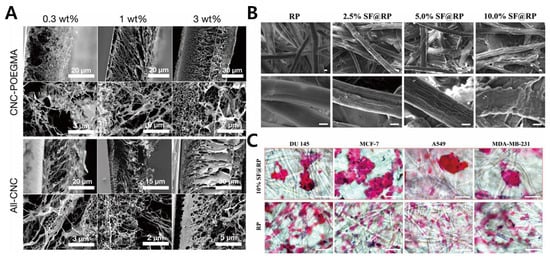
2.2. Fibrous Scaffold
2.3. Gel-Based Scaffolds
References
- Ham, S.L.; Joshi, R.; Thakuri, P.S.; Tavana, H. Liquid-based three-dimensional tumor models for cancer research and drug discovery. Exp. Biol. Med. 2016, 241, 939–954.
- Kelm, J.M.; Timmins, N.E.; Brown, C.J.; Fussenegger, M.; Nielsen, L.K. Method for generation of homogeneous multicellular tumor spheroids applicable to a wide variety of cell types. Biotechnol. Bioeng. 2003, 83, 173–180.
- Mitteregger, R.; Vogt, G.; Rossmanith, E.; Falkenhagen, D. Rotary cell culture system (RCCS): A new method for cultivating hepatocytes on microcarriers. Int. J. Artif. Organs 1999, 22, 816–822.
- Foty, R. A simple hanging drop cell culture protocol for generation of 3D spheroids. J. Vis. Exp. 2011, 2720.
- Tung, Y.-C.; Hsiao, A.Y.; Allen, S.G.; Torisawa, Y.-S.; Ho, M.; Takayama, S. High-throughput 3D spheroid culture and drug testing using a 384 hanging drop array. Analyst 2011, 136, 473–478.
- Ivascu, A.; Kubbies, M. Rapid generation of single-tumor spheroids for high-throughput cell function and toxicity analysis. J. Biomol. Screen. 2006, 11, 922–932.
- Hsiao, A.Y.; Tung, Y.-C.; Kuo, C.-H.; Mosadegh, B.; Bedenis, R.; Pienta, K.J.; Takayama, S. Micro-ring structures stabilize microdroplets to enable long term spheroid culture in 384 hanging drop array plates. Biomed. Microdevices 2012, 14, 313–323.
- Lin, R.Z.; Chang, H.Y. Recent advances in three-dimensional multicellular spheroid culture for biomedical research. Biotechnol. J. 2008, 3, 1172–1184.
- Zhao, L.; Xiu, J.; Liu, Y.; Zhang, T.; Pan, W.; Zheng, X.; Zhang, X. A 3D printed hanging drop dripper for tumor spheroids analysis without recovery. Sci. Rep. 2019, 9, 1–14.
- Park, J.; Kim, H.; Park, J.-K. Microfluidic channel-integrated hanging drop array chip operated by pushbuttons for spheroid culture and analysis. Analyst 2020, 145, 6974–6980.
- RA, V.; Kumari, S.; Poddar, P.; Dhara, D.; Maiti, S. Poly (N-isopropylacrylamide)-Based Polymers as Additive for Rapid Generation of Spheroid via Hanging Drop Method. Macromol. Biosci. 2020, 20, 2000180.
- Lv, D.; Hu, Z.; Lu, L.; Lu, H.; Xu, X. Three-dimensional cell culture: A powerful tool in tumor research and drug discovery. Oncol. Lett. 2017, 14, 6999–7010.
- Jiang, N.; Chen, Z.; Li, B.; Guo, S.; Li, A.; Zhang, T.; Fu, X.; Si, S.; Cui, Y. Effects of rotary cell culture system-simulated microgravity on the ultrastructure and biological behavior of human MDA-MB-231 breast cancer cells. Precis. Radiat. Oncol. 2019, 3, 87–93.
- Chen, Z.Y.; Jiang, N.; Guo, S.; Li, B.B.; Yang, J.Q.; Chai, S.B.; Yan, H.F.; Sun, P.M.; Zhang, T.; Sun, H.W. Effect of simulated microgravity on metabolism of HGC-27 gastric cancer cells. Oncol. Lett. 2020, 19, 3439–3450.
- Nyberg, S.L.; Hardin, J.; Amiot, B.; Argikar, U.A.; Remmel, R.P.; Rinaldo, P. Rapid, large-scale formation of porcine hepatocyte spheroids in a novel spheroid reservoir bioartificial liver. Liver Transplant. 2005, 11, 901–910.
- Fischbach, C.; Chen, R.; Matsumoto, T.; Schmelzle, T.; Brugge, J.S.; Polverini, P.J.; Mooney, D.J. Engineering tumors with 3D scaffolds. Nat. Methods 2007, 4, 855–860.
- Dhiman, H.K.; Ray, A.R.; Panda, A.K. Three-dimensional chitosan scaffold-based MCF-7 cell culture for the determination of the cytotoxicity of tamoxifen. Biomaterials 2005, 26, 979–986.
- Weaver, V.M.; Lelièvre, S.; Lakins, J.N.; Chrenek, M.A.; Jones, J.C.; Giancotti, F.; Werb, Z.; Bissell, M.J. β4 integrin-dependent formation of polarized three-dimensional architecture confers resistance to apoptosis in normal and malignant mammary epithelium. Cancer Cell 2002, 2, 205–216.
- Zhang, X.; Wang, W.; Yu, W.; Xie, Y.; Zhang, X.; Zhang, Y.; Ma, X. Development of an in vitro multicellular tumor spheroid model using microencapsulation and its application in anticancer drug screening and testing. Biotechnol. Prog. 2005, 21, 1289–1296.
- Ohtaka, K.; Watanabe, S.; Iwazaki, R.; Hirose, M.; Sato, N. Role of extracellular matrix on colonic cancer cell migration and proliferation. Biochem. Biophys. Res. Commun. 1996, 220, 346–352.
- Hollister, S.J. Porous scaffold design for tissue engineering. Nat. Mater. 2005, 4, 518–524.
- Zhang, M.; Boughton, P.; Rose, B.; Lee, C.S.; Hong, A.M. The use of porous scaffold as a tumor model. Int. J. Biomater. 2013, 2013, 396056.
- Nemati, S.; Kim, S.-J.; Shin, Y.M.; Shin, H. Current progress in application of polymeric nanofibers to tissue engineering. Nano Converg. 2019, 6, 36.
- Park, M.; Lee, G.; Ryu, K.; Lim, W. Improvement of Bone Formation in Rats with Calvarial Defects by Modulating the Pore Size of Tricalcium Phosphate Scaffolds. Biotechnol. Bioprocess Eng. 2019, 24, 885–892.
- Sun, M.T.; O’Connor, A.J.; Milne, I.; Biswas, D.; Casson, R.; Wood, J.; Selva, D. Development of Macroporous Chitosan Scaffolds for Eyelid Tarsus Tissue Engineering. J. Tissue Eng. Regen. Med. 2019, 16, 595–604.
- Zhang, H.-W.; Hu, X.-B.; Qin, Y.; Jin, Z.-H.; Zhang, X.-W.; Liu, Y.-L.; Huang, W.-H. Conductive polymer coated scaffold to integrate 3D cell culture with electrochemical sensing. Anal. Chem. 2019, 91, 4838–4844.
- Or, T.; Saem, S.; Esteve, A.; Osorio, D.A.; De France, K.J.; Vapaavuori, J.; Hoare, T.; Cerf, A.; Cranston, E.D.; Moran-Mirabal, J.M. Patterned cellulose nanocrystal aerogel films with tunable dimensions and morphologies as ultra-porous scaffolds for cell culture. ACS Appl. Nano Mater. 2019, 2, 4169–4179.
- Fu, J.; Li, X.B.; Wang, L.X.; Lv, X.H.; Lu, Z.; Wang, F.; Xia, Q.; Yu, L.; Li, C.M. One-Step Dip-Coating-Fabricated Core–Shell Silk Fibroin Rice Paper Fibrous Scaffolds for 3D Tumor Spheroid Formation. ACS Appl. Bio Mater. 2020, 3, 7462–7471.
- Girard, Y.K.; Wang, C.; Ravi, S.; Howell, M.C.; Mallela, J.; Alibrahim, M.; Green, R.; Hellermann, G.; Mohapatra, S.S.; Mohapatra, S. A 3D fibrous scaffold inducing tumoroids: A platform for anticancer drug development. PLoS ONE 2013, 8, e75345.
- Bae, C.-S.; Lee, C.-M.; Ahn, T. Encapsulation of Apoptotic Proteins in Lipid Nanoparticles to Induce Death of Cancer Cells. Biotechnol. Bioprocess Eng. 2020, 25, 264–271.
- Feng, S.; Duan, X.; Lo, P.-K.; Liu, S.; Liu, X.; Chen, H.; Wang, Q. Expansion of breast cancer stem cells with fibrous scaffolds. Integr. Biol. 2013, 5, 768–777.
- Shang, M.; Soon, R.H.; Lim, C.T.; Khoo, B.L.; Han, J. Microfluidic modelling of the tumor microenvironment for anti-cancer drug development. Lab Chip 2019, 19, 369–386.
- Caliari, S.R.; Burdick, J.A. A practical guide to hydrogels for cell culture. Nat. Methods 2016, 13, 405–414.
- Ashworth, J.C.; Morgan, R.L.; Lis-Slimak, K.; Meade, K.A.; Jones, S.; Spence, K.; Slater, C.E.; Thompson, J.L.; Grabowska, A.M.; Clarke, R.B.; et al. Preparation of a User-Defined Peptide Gel for Controlled 3D Culture Models of Cancer and Disease. J. Vis. Exp. 2020, e61710.
- Hainline, K.M.; Gu, F.; Handley, J.F.; Tian, Y.F.; Wu, Y.; de Wet, L.; Vander Griend, D.J.; Collier, J.H. Self-Assembling Peptide Gels for 3D Prostate Cancer Spheroid Culture. Macromol. Biosci. 2019, 19, 1800249.
- Jung, J.P.; Moyano, J.V.; Collier, J.H. Multifactorial optimization of endothelial cell growth using modular synthetic extracellular matrices. Integr. Biol. 2011, 3, 185–196.





