Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Pasquale Mone | + 1187 word(s) | 1187 | 2021-11-11 04:16:56 | | | |
| 2 | Camila Xu | Meta information modification | 1187 | 2021-11-12 01:46:48 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Mone, P. L-Arginine. Encyclopedia. Available online: https://encyclopedia.pub/entry/15914 (accessed on 10 May 2026).
Mone P. L-Arginine. Encyclopedia. Available at: https://encyclopedia.pub/entry/15914. Accessed May 10, 2026.
Mone, Pasquale. "L-Arginine" Encyclopedia, https://encyclopedia.pub/entry/15914 (accessed May 10, 2026).
Mone, P. (2021, November 11). L-Arginine. In Encyclopedia. https://encyclopedia.pub/entry/15914
Mone, Pasquale. "L-Arginine." Encyclopedia. Web. 11 November, 2021.
Copy Citation
l-Arginine is a semi-essential amino acid involved in numerous biological processes. It is a substrate for different enzymatic reactions and is metabolized using three major known pathways in the body: (1) Arginase metabolizes l-Arginine to l-ornithine, (2) l-Arginine decarboxylase metabolizes l-Arginine to agmatine, and (3) nitric oxide (NO) synthase (NOS) uses l-Arginine to form NO and citrulline.
l-Arginine
acute respiratory syndrome
COVID-19
nitric oxide
1. Functional Role of l-Arginine in NO Formation
l-Arginine is the substrate used for NO production by NOS [1]; due to its ability to cause NO generation, which has been shown to be a major endothelial relaxation factor (able to increase vasodilation and reduce arterial blood pressure [2][3][4][5]), l-Arginine has considerable potential in becoming a tool to tackle cardiovascular issues [6]. For instance, in patients with known endothelial dysfunction, l-Arginine supplementation (6–8 g per day) has been shown to improve endothelial function and ultimately lower blood pressure [6].
Three isoforms of NOS have been identified; two of them (endothelial NOS [7][8] and neuronal NOS [9][10]) are expressed constitutively, while the last one is inducible and is mainly involved in the inflammatory/immune response [11][12][13][14].
In the reaction carried out by NOS, electrons are transferred to heme in the N-terminal domain [15][16]. Electrons are taken from nicotinamide adenine dinucleotide phosphate (NADP) using flavin adenine dinucleotide in the C-terminal reductase domain [17]. Once electrons are transferred to the N terminal oxygenase domain, NO and citrulline are formed via l-Arginine oxidation [1][18][19]. For NOS to function properly, there needs to be an ample amount of l-Arginine available for this reaction [20]. In addition, NADP, glutathione, tetrahydrobiopterin, and oxygen are needed for proper functioning [2][21].
A substrate competition occurs between NOS and arginase [22][23]. Although the affinity for l-Arginine in NOS is much higher than arginase, the speed of the reaction allows for substrate concentration. The speed of arginase rection is a thousand times faster than NOS [24]. Since these two enzymes compete for a common substrate, arginase will reduce the amount of L-Arginine available for NOS to use [25][26], ultimately decreasing the amount of NO produced.
2. Effects of l-Arginine on the Immune System
A large part of a normal immune system depends on the amount of l-Arginine available in the body. Arginase is known to represent an integral part of certain granulocyte subsets, which can be released locally or systematically once there is an immune response. In addition, there is an accumulation of immature myeloid cells that express arginase, which is released when fighting off specific illnesses. These myeloid cells that express arginase can decrease the amount of l-Arginine [27][28][29].
T cell function has been shown to depend on l-Arginine levels [30][31]. A decreased ability of lymphocytes to proliferate has been reported in critically ill septic patients and correlated to reduced availability of l-Arginine [32]. Moreover, l-Arginine administration has been found to be beneficial to maintain immune homeostasis (Figure 1), especially in terms of T cell and macrophage function [33]. In fact, l-Arginine is essential in the macrophage M1-to-M2 switch [34].
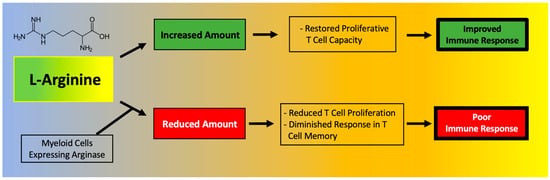
Figure 1. Main effects of l-Arginine on the immune system.
A deficiency in l-Arginine has been shown to lead to a reduction in T cell proliferation and to cause a diminished response in T cell-mediated memory [35]. In vitro assays have validated that L-Arginine can restore the function of T cells [36]. Mechanistically, the immunosuppressive effects of myeloid-derived suppressor cells (MDSCs) due to l-Arginine depletion and lymphocyte mitochondrial dysfunction have been demonstrated in models of cancer [30].
The expansion of MDSCs observed in COVID-19 has been directly correlated to enhanced arginase activity and lymphopenia [37]. Monocytic MDSCs were significantly expanded in the blood of COVID-19 patients and were strongly associated with disease severity; MDSCs were shown to suppress T cell proliferation and IFNγ production, at least in part through an arginase-dependent mechanism, strongly indicating a role for these cells in the dysregulated COVID-19 immune response [38]. Indeed, MDSCs express high levels of arginase, which metabolizes l-Arginine to ornithine and urea, effectively depleting this amino acid from the microenvironment [39]. l-Arginine depletion is known to inhibit T cell receptor signaling, eventually resulting in T cell dysfunction [40] and to increase the generation of reactive oxygen species (ROS), thereby exacerbating inflammation [39][41].
In a recent study focused on COVID-19, Dr. Claudia Morris and colleagues were able to determine the bioavailability of l-Arginine in three cohorts: asymptomatic healthy adults, adults hospitalized with COVID-19, and children hospitalized with COVID-19; they found that both adults and children affected by COVID-19 display significantly lower levels of plasma l-Arginine (as well as l-Arginine bioavailability) compared to controls [42]. Additionally, a low l-Arginine-to-ornithine ratio observed in COVID-19 patients [42] indicates an elevation of arginase activity in these patients. In another study, plasmatic L-Arginine levels were shown to inversely correlate with the severity of COVID-19 [43]. This study also revealed that the expression of the activated GPIIb/IIIa complex (PAC-1), known to be involved in platelet activation and thromboembolic events [44], is higher on platelets of patients with severe COVID-19 compared to healthy controls and inversely correlated with the plasmatic concentration of l-Arginine [43].
These pieces of evidence seem to go against the recently proposed strategy of l-Arginine depletion in COVID-19, based on the assumption that some steps in the viral lifecycle of SARS-CoV-2 could depend on l-Arginine residues (for instance, the nucleocapsid protein has a 6.9% l-Arginine content) [45].
In fact, a decrease in the bioavailability of l-Arginine has been shown to cause a diminished T cell response and function, eventually leading to an increased susceptibility to infections [46][47]. Twelve weeks of continuous l-Arginine supplementation significantly decreased the level of IL-21 [48], while NO has been shown to suppress the proliferation and function of human Th17 cells [49], which have been implied in the pathogenesis of the cytokine storm and of hyperinflammatory phenomena observed in COVID-19 patients [50][51][52][53]. Higher l-Arginine levels are associated with lower levels of CCL-20, a ligand for CCR6, a part of the chemotaxis system that is induced in response to coronavirus infections [51].
In vitro assays have demonstrated that the proliferative capacity of T cells is significantly reduced in COVID-19 patients and can be restored through l-Arginine supplementation [37]. Corroborating these findings, recent metabolomics data indicates that l-Arginine pathways are altered in COVID-19 patients [54] and an increased mRNA expression of arginase has also been found in the peripheral blood mononuclear cells (PBMCs) of COVID-19 patients [55].
Of note, circulating levels of metabolites of the l-Arginine pathway can be affected by arginase activity in red blood cells [56], which is known to be affected by oxidative stress and can contribute to endothelial dysfunction observed in COVID-19 [57]; furthermore, l-Arginine metabolism is known to be altered in hemolysis [58]. The exquisite balance between arginase and NOS activity has also been shown to influence the inflammatory responses of gut resident macrophages [34].
To actually test l-Arginine in COVID-19 patients, based on the rationale described above, we designed a randomized clinical trial to study the effects of adding l-Arginine orally (Bioarginina®, 1.66 g twice per day) to standard therapy in patients hospitalized for COVID-19. The interim results, recently published [59], revealed that patients who received l-Arginine had a significantly reduced duration of the in-hospital stay, and a diminished respiratory support, compared to patients in the placebo arm.
References
- Andrew, P.J.; Mayer, B. Enzymatic function of nitric oxide synthases. Cardiovasc. Res. 1999, 43, 521–531.
- Gambardella, J.; Khondkar, W.; Morelli, M.B.; Wang, X.; Santulli, G.; Trimarco, V. Arginine and Endothelial Function. Biomedicines 2020, 8, 277.
- Jorens, P.G.; Vermeire, P.A.; Herman, A.G. L-Arginine-dependent nitric oxide synthase: A new metabolic pathway in the lung and airways. Eur. Respir. J. 1993, 6, 258–266.
- Tousoulis, D.; Kampoli, A.M.; Tentolouris, C.; Papageorgiou, N.; Stefanadis, C. The role of nitric oxide on endothelial function. Curr. Vasc. Pharmacol. 2012, 10, 4–18.
- Dormanns, K.; Brown, R.G.; David, T. The role of nitric oxide in neurovascular coupling. J. Theor. Biol. 2016, 394, 1–17.
- Ströhle, A.; von Bibra, H.; Hahn, A. l-Arginine and vascular health. Med. Monatsschr. Pharm. 2016, 39, 515–520.
- Albrecht, E.W.; Stegeman, C.A.; Heeringa, P.; Henning, R.H.; van Goor, H. Protective role of endothelial nitric oxide synthase. J. Pathol. 2003, 199, 8–17.
- Chatterjee, A.; Black, S.M.; Catravas, J.D. Endothelial nitric oxide (NO) and its pathophysiologic regulation. Vascul. Pharmacol. 2008, 49, 134–140.
- Sezen, S.F.; Lagoda, G.; Burnett, A.L. Neuronal nitric oxide signaling regulates erection recovery after cavernous nerve injury. J. Urol. 2012, 187, 757–763.
- Bredt, D.S.; Snyder, S.H. Nitric oxide, a novel neuronal messenger. Neuron 1992, 8, 3–11.
- Chakravortty, D.; Hensel, M. Inducible nitric oxide synthase and control of intracellular bacterial pathogens. Microbes Infect. 2003, 5, 621–627.
- Lee, M.; Rey, K.; Besler, K.; Wang, C.; Choy, J. Immunobiology of Nitric Oxide and Regulation of Inducible Nitric Oxide Synthase. Results Probl. Cell Differ. 2017, 62, 181–207.
- Sharma, J.N.; Al-Omran, A.; Parvathy, S.S. Role of nitric oxide in inflammatory diseases. Inflammopharmacology 2007, 15, 252–259.
- Schini, V.B.; Busse, R.; Vanhoutte, P.M. Inducible nitric oxide synthase in vascular smooth muscle. Arzneimittelforschung 1994, 44, 432–435.
- Stuehr, D.J. Enzymes of the l-Arginine to nitric oxide pathway. J. Nutr. 2004, 134, 2748S–2751S.
- Forstermann, U.; Sessa, W.C. Nitric oxide synthases: Regulation and function. Eur. Heart J. 2012, 33, 829–837.
- Mori, M. Regulation of nitric oxide synthesis and apoptosis by arginase and arginine recycling. J. Nutr. 2007, 137, 1616S–1620S.
- Agapie, T.; Suseno, S.; Woodward, J.J.; Stoll, S.; Britt, R.D.; Marletta, M.A. NO formation by a catalytically self-sufficient bacterial nitric oxide synthase from Sorangium cellulosum. Proc. Natl. Acad. Sci. USA 2009, 106, 16221–16226.
- Jáchymová, M.; Martásek, P.; Panda, S.; Roman, L.J.; Panda, M.; Shea, T.M.; Ishimura, Y.; Kim, J.J.; Masters, B.S. Recruitment of governing elements for electron transfer in the nitric oxide synthase family. Proc. Natl. Acad. Sci. USA 2005, 102, 15833–15838.
- Rajapakse, N.W.; Mattson, D.L. Role of L-Arginine in nitric oxide production in health and hypertension. Clin. Exp. Pharmacol. Physiol. 2009, 36, 249–255.
- Stuehr, D.J.; Kwon, N.S.; Nathan, C.F. FAD and GSH participate in macrophage synthesis of nitric oxide. Biochem. Biophys. Res. Commun. 1990, 168, 558–565.
- Durante, W.; Johnson, F.K.; Johnson, R.A. Arginase: A critical regulator of nitric oxide synthesis and vascular function. Clin. Exp. Pharmacol. Physiol. 2007, 34, 906–911.
- Morris, C.R. Mechanisms of vasculopathy in sickle cell disease and thalassemia. Hematology Am. Soc. Hematol. Educ. Program. 2008, 2008, 177–185.
- Wu, G.; Morris, S.M., Jr. Arginine metabolism: Nitric oxide and beyond. Biochem. J. 1998, 336, 1–17.
- S Clemente, G.; van Waarde, A.; F Antunes, I.; Dömling, A.; H Elsinga, P. Arginase as a Potential Biomarker of Disease Progression: A Molecular Imaging Perspective. Int. J. Mol. Sci. 2020, 21, 5291.
- Benson, R.C.; Hardy, K.A.; Morris, C.R. Arginase and arginine dysregulation in asthma. J. Allergy 2011, 2011, 736319.
- Popovic, P.J.; Zeh, H.J., 3rd; Ochoa, J.B. Arginine and immunity. J. Nutr. 2007, 137, 1681s–1686s.
- Szefel, J.; Danielak, A.; Kruszewski, W.J. Metabolic pathways of l-Arginine and therapeutic consequences in tumors. Adv. Med. Sci. 2019, 64, 104–110.
- Steggerda, S.M.; Bennett, M.K.; Chen, J.; Emberley, E.; Huang, T.; Janes, J.R.; Li, W.; MacKinnon, A.L.; Makkouk, A.; Marguier, G.; et al. Inhibition of arginase by CB-1158 blocks myeloid cell-mediated immune suppression in the tumor microenvironment. J. Immunother. Cancer 2017, 5, 101.
- Geiger, R.; Rieckmann, J.C.; Wolf, T.; Basso, C.; Feng, Y.; Fuhrer, T.; Kogadeeva, M.; Picotti, P.; Meissner, F.; Mann, M.; et al. l-Arginine Modulates T Cell Metabolism and Enhances Survival and Anti-tumor Activity. Cell 2016, 167, 829–842.e813.
- Li, P.; Yin, Y.L.; Li, D.; Kim, S.W.; Wu, G. Amino acids and immune function. Br. J. Nutr. 2007, 98, 237–252.
- Uhel, F.; Azzaoui, I.; Gregoire, M.; Pangault, C.; Dulong, J.; Tadie, J.M.; Gacouin, A.; Camus, C.; Cynober, L.; Fest, T.; et al. Early Expansion of Circulating Granulocytic Myeloid-derived Suppressor Cells Predicts Development of Nosocomial Infections in Patients with Sepsis. Am. J. Respir. Crit. Care Med. 2017, 196, 315–327.
- Tepaske, R.; Velthuis, H.; Oudemans-van Straaten, H.M.; Heisterkamp, S.H.; van Deventer, S.J.; Ince, C.; Eysman, L.; Kesecioglu, J. Effect of preoperative oral immune-enhancing nutritional supplement on patients at high risk of infection after cardiac surgery: A randomised placebo-controlled trial. Lancet 2001, 358, 696–701.
- Rath, M.; Muller, I.; Kropf, P.; Closs, E.I.; Munder, M. Metabolism via Arginase or Nitric Oxide Synthase: Two Competing Arginine Pathways in Macrophages. Front. Immunol. 2014, 5, 532.
- Zhu, X.; Pribis, J.P.; Rodriguez, P.C.; Morris, S.M., Jr.; Vodovotz, Y.; Billiar, T.R.; Ochoa, J.B. The central role of arginine catabolism in T-cell dysfunction and increased susceptibility to infection after physical injury. Ann. Surg. 2014, 259, 171–178.
- Bronte, V.; Zanovello, P. Regulation of immune responses by l-Arginine metabolism. Nat. Rev. Immunol. 2005, 5, 641–654.
- Reizine, F.; Lesouhaitier, M.; Gregoire, M.; Pinceaux, K.; Gacouin, A.; Maamar, A.; Painvin, B.; Camus, C.; Le Tulzo, Y.; Tattevin, P.; et al. SARS-CoV-2-Induced ARDS Associates with MDSC Expansion, Lymphocyte Dysfunction, and Arginine Shortage. J. Clin. Immunol. 2021, 41, 515–525.
- Falck-Jones, S.; Vangeti, S.; Yu, M.; Falck-Jones, R.; Cagigi, A.; Badolati, I.; Osterberg, B.; Lautenbach, M.J.; Ahlberg, E.; Lin, A.; et al. Functional monocytic myeloid-derived suppressor cells increase in blood but not airways and predict COVID-19 severity. J. Clin. Investig. 2021, 131, e144734.
- Dean, M.J.; Ochoa, J.B.; Sanchez-Pino, M.D.; Zabaleta, J.; Garai, J.; Del Valle, L.; Wyczechowska, D.; Baiamonte, L.B.; Philbrook, P.; Majumder, R.; et al. Severe COVID-19 Is Characterized by an Impaired Type I Interferon Response and Elevated Levels of Arginase Producing Granulocytic Myeloid Derived Suppressor Cells. Front. Immunol. 2021, 12, 695972.
- Rodriguez, P.C.; Ochoa, A.C. Arginine regulation by myeloid derived suppressor cells and tolerance in cancer: Mechanisms and therapeutic perspectives. Immunol. Rev. 2008, 222, 180–191.
- Lucas, R.; Czikora, I.; Sridhar, S.; Zemskov, E.A.; Oseghale, A.; Circo, S.; Cederbaum, S.D.; Chakraborty, T.; Fulton, D.J.; Caldwell, R.W.; et al. Arginase 1: An unexpected mediator of pulmonary capillary barrier dysfunction in models of acute lung injury. Front. Immunol. 2013, 4, 228.
- Rees, C.A.; Rostad, C.A.; Mantus, G.; Anderson, E.J.; Chahroudi, A.; Jaggi, P.; Wrammert, J.; Ochoa, J.B.; Ochoa, A.; Basu, R.K.; et al. Altered amino acid profile in patients with SARS-CoV-2 infection. Proc. Natl. Acad. Sci. USA 2021, 118.
- Sacchi, A.; Grassi, G.; Notari, S.; Gili, S.; Bordoni, V.; Tartaglia, E.; Casetti, R.; Cimini, E.; Mariotti, D.; Garotto, G.; et al. Expansion of Myeloid Derived Suppressor Cells Contributes to Platelet Activation by L-Arginine Deprivation during SARS-CoV-2 Infection. Cells 2021, 10, 2111.
- Chen, Z.; Mondal, N.K.; Ding, J.; Koenig, S.C.; Slaughter, M.S.; Griffith, B.P.; Wu, Z.J. Activation and shedding of platelet glycoprotein IIb/IIIa under non-physiological shear stress. Mol. Cell Biochem. 2015, 409, 93–101.
- Grimes, J.M.; Khan, S.; Badeaux, M.; Rao, R.M.; Rowlinson, S.W.; Carvajal, R.D. Arginine depletion as a therapeutic approach for patients with COVID-19. Int. J. Infect. Dis. 2021, 102, 566–570.
- Ochoa, J.B.; Bernard, A.C.; O’Brien, W.E.; Griffen, M.M.; Maley, M.E.; Rockich, A.K.; Tsuei, B.J.; Boulanger, B.R.; Kearney, P.A.; Morris, S.M., Jr. Arginase I expression and activity in human mononuclear cells after injury. Ann. Surg. 2001, 233, 393–399.
- Ochoa, J.B.; Bernard, A.C.; Mistry, S.K.; Morris, S.M., Jr.; Figert, P.L.; Maley, M.E.; Tsuei, B.J.; Boulanger, B.R.; Kearney, P.A. Trauma increases extrahepatic arginase activity. Surgery 2000, 127, 419–426.
- Liao, S.Y.; Linderholm, A.; Showalter, M.R.; Chen, C.H.; Fiehn, O.; Kenyon, N.J. L-Arginine as a potential GLP-1-mediated immunomodulator of Th17-related cytokines in people with obesity and asthma. Obes. Sci. Pract. 2021, 7, 339–345.
- Niedbala, W.; Alves-Filho, J.C.; Fukada, S.Y.; Vieira, S.M.; Mitani, A.; Sonego, F.; Mirchandani, A.; Nascimento, D.C.; Cunha, F.Q.; Liew, F.Y. Regulation of type 17 helper T-cell function by nitric oxide during inflammation. Proc. Natl. Acad. Sci. USA 2011, 108, 9220–9225.
- Xu, Z.; Shi, L.; Wang, Y.; Zhang, J.; Huang, L.; Zhang, C.; Liu, S.; Zhao, P.; Liu, H.; Zhu, L.; et al. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir. Med. 2020, 8, 420–422.
- Thomassen, M.J.; Buhrow, L.T.; Connors, M.J.; Kaneko, F.T.; Erzurum, S.C.; Kavuru, M.S. Nitric oxide inhibits inflammatory cytokine production by human alveolar macrophages. Am. J. Respir. Cell Mol. Biol. 1997, 17, 279–283.
- Hotez, P.J.; Bottazzi, M.E.; Corry, D.B. The potential role of Th17 immune responses in coronavirus immunopathology and vaccine-induced immune enhancement. Microbes. Infect. 2020, 22, 165–167.
- Wu, D.; Yang, X.O. TH17 responses in cytokine storm of COVID-19: An emerging target of JAK2 inhibitor Fedratinib. J. Microbiol. Immunol. Infect. 2020, 53, 368–370.
- D’Alessandro, A.; Akpan, I.; Thomas, T.; Reisz, J.; Cendali, F.; Gamboni, F.; Nemkov, T.; Thangaraju, K.; Katneni, U.; Tanaka, K.; et al. Biological and Clinical Factors contributing to the Metabolic Heterogeneity of Hospitalized Patients with and without COVID-19. Cells 2021, 10, 2293.
- Derakhshani, A.; Hemmat, N.; Asadzadeh, Z.; Ghaseminia, M.; Shadbad, M.A.; Jadideslam, G.; Silvestris, N.; Racanelli, V.; Baradaran, B. Arginase 1 (Arg1) as an Up-Regulated Gene in COVID-19 Patients: A Promising Marker in COVID-19 Immunopathy. J. Clin. Med. 2021, 10, 1051.
- Renoux, C.; Fort, R.; Nader, E.; Boisson, C.; Joly, P.; Stauffer, E.; Robert, M.; Girard, S.; Cibiel, A.; Gauthier, A.; et al. Impact of COVID-19 on red blood cell rheology. Br. J. Haematol. 2021, 192, e108–e111.
- Hoffmann, M.; Kleine-Weber, H.; Schroeder, S.; Kruger, N.; Herrler, T.; Erichsen, S.; Schiergens, T.S.; Herrler, G.; Wu, N.H.; Nitsche, A.; et al. SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell 2020, 181, 271–280.e278.
- Morris, C.R.; Kato, G.J.; Poljakovic, M.; Wang, X.; Blackwelder, W.C.; Sachdev, V.; Hazen, S.L.; Vichinsky, E.P.; Morris, S.M., Jr.; Gladwin, M.T. Dysregulated arginine metabolism, hemolysis-associated pulmonary hypertension, and mortality in sickle cell disease. JAMA 2005, 294, 81–90.
- Fiorentino, G.; Coppola, A.; Izzo, R.; Annunziata, A.; Bernardo, M.; Lombardi, A.; Trimarco, V.; Santulli, G.; Trimarco, B. Effects of adding L-Arginine orally to standard therapy in patients with COVID-19: A randomized, double-blind, placebo-controlled, parallel-group trial. Results of the first interim analysis. EClinicalMedicine 2021, 40, 101125.
- Yan, Z.; Yang, M.; Lai, C.L. Long COVID-19 Syndrome: A Comprehensive Review of Its Effect on Various Organ Systems and Recommendation on Rehabilitation Plans. Biomedicines 2021, 9, 966.
- Paul, B.D.; Lemle, M.D.; Komaroff, A.L.; Snyder, S.H. Redox imbalance links COVID-19 and myalgic encephalomyelitis/chronic fatigue syndrome. Proc. Natl. Acad. Sci. USA 2021, 118, e2024358118.
- Paneroni, M.; Pasini, E.; Vitacca, M.; Scalvini, S.; Comini, L.; Pedrinolla, A.; Venturelli, M. Altered Vascular Endothelium-Dependent Responsiveness in Frail Elderly Patients Recovering from COVID-19 Pneumonia: Preliminary Evidence. J. Clin. Med. 2021, 10, 2558.
- Roe, K. A role for T-cell exhaustion in Long COVID-19 and severe outcomes for several categories of COVID-19 patients. J Neurosci. Res. 2021, in press.
More
Information
Subjects:
Allergy
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.2K
Revisions:
2 times
(View History)
Update Date:
12 Nov 2021
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





