Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Atsushi Sakamoto | + 2236 word(s) | 2236 | 2021-10-29 04:49:41 | | | |
| 2 | Catherine Yang | -3 word(s) | 2233 | 2021-11-11 07:27:19 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Sakamoto, A. Marine-Derived Omega-3 Polyunsaturated Fatty Acids and Heart Failure. Encyclopedia. Available online: https://encyclopedia.pub/entry/15898 (accessed on 14 May 2026).
Sakamoto A. Marine-Derived Omega-3 Polyunsaturated Fatty Acids and Heart Failure. Encyclopedia. Available at: https://encyclopedia.pub/entry/15898. Accessed May 14, 2026.
Sakamoto, Atsushi. "Marine-Derived Omega-3 Polyunsaturated Fatty Acids and Heart Failure" Encyclopedia, https://encyclopedia.pub/entry/15898 (accessed May 14, 2026).
Sakamoto, A. (2021, November 11). Marine-Derived Omega-3 Polyunsaturated Fatty Acids and Heart Failure. In Encyclopedia. https://encyclopedia.pub/entry/15898
Sakamoto, Atsushi. "Marine-Derived Omega-3 Polyunsaturated Fatty Acids and Heart Failure." Encyclopedia. Web. 11 November, 2021.
Copy Citation
Heart failure (HF) is a rapidly growing global public health problem. Since HF results in high mortality and re-hospitalization, new effective treatments are desired. Although it remains controversial, omega 3 polyunsaturated fatty acids (n-3 PUFAs), such as the eicosapentaenoic acid and docosahexaenoic acid, have been widely recognized to have benefits for HF.
omega 3 polyunsaturated fatty acid
Heart failure
eicosapentaenoic acid
docosahexaenoic acid
cardiovascular disease
1. Introduction
Heart failure (HF) is a rapidly growing global public health problem both in developed and developing countries, with an estimated prevalence of over 37.7 million patients worldwide [1]. Despite recent developments of HF treatments, including pharmacologic and device therapy, HF results in high mortality and re-hospitalization. Therefore, innovations regarding HF treatments are desired.
2. n-3 PUFA on Heart Failure
HF is a condition in which the heart is unable to pump enough blood to meet demand. Since HF is a multifactorial syndrome, its mortalities and progressions differ from their underling etiologies, such as IHD, valvular heart disease, hypertension, arrhythmia, cardiac myopathies, congenital heart disease, endocrine and metabolic diseases, infection, and certain drugs. Several prior prospective observational studies and randomized control trials proved that consumption of fish or fish oil containing n-3 PUFA decreased IHD mortality (e.g., myocardial infarction (MI) and sudden cardiac death) in patients with or without pre-diagnosed CVD [2][3][4]. In addition, recent studies have shown that fish and/or n-3 PUFA intake also prevented the new onset of HF and rehospitalization [5][6][7][8][9][10]. However, conflicting results were reported from other groups [11][12][13], meaning that the n-3 PUFA-mediated effects on HF are once again viewed as uncertain. In this section, we summarize existing clinical evidence regarding primary and secondary prevention of HF, and discuss the considerable points which may cause such heterogenous results.
3. n-3 PUFA-Mediated Cardiac Protection from Basic and Translational Research
Despite controversy regarding the benefits of n-3 PUFA for HF prevention in clinical trials, a significant amount of supportive evidence from basic and clinical translational research has been reported. Although such follow-up research tends to contain an inherent bias, by which positive data are likely to be reported, each study has well-explained pathophysiological mechanisms of preventive effects of PUFA against HF (Figure 1). The evidence of n-3 PUFA-mediated cardiac protection and HF prevention both from basic and translational research was summarized.
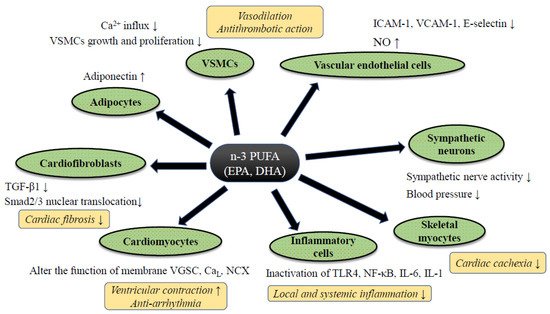
Figure 1. The putative mechanism of n-3 PUFA-mediated cardiac protection against heart failure. Abbreviations: CaL; L type calcium channel, IL-6/-1; ICAM-1; intercellular adhesion molecule-1, Interleukin-6/-1, NCX: sodium calcium exchanger, NF-κB; nuclear factor-kappa B, NO; nitric oxide, TGF-β1; transforming growth factor-beta 1, TLR4; Toll-like receptor 4, VGSC; voltage-gated sodium channel, VCAM-1; vascular cell adhesion molecule-1, VSMCs; vascular smooth muscle cells. Abbreviations: ADMA: Asymmetric Dimethylarginine, DB: Double blind trial, ESLVD: End-systolic left ventricle diameters, Etv/Atv: Early rapid right ventricular filling/late right ventricular filling, FMD: Flow-mediated dilatation, LAEF: Left atrial ejection fraction, LVESVI: Left ventricular end-systolic volume indexed to body surface area, MC: Multi-center trial, MCP-1: Monocyte chemoattractant protein 1, NYHA: New York Heart Association class, OL: Open label trial, PC: Placebo-control trial, PRSP: Prospective trial, RDM: Randomized trial, SC: Single-center trial, ST2: Suppression of tumorigenicity 2, TDI: Tissue Doppler imaging. Downward allows represent the decrease or suppression, and upward allows the increase or enhancement.
3.1. Anti-Inflammatory Effect of n-3 PUFA
It is well known that the inflammatory cytokines (e.g., interleukin-1β and TNF-α), which can reduce both systolic and diastolic cardiac function to advance cardiac remodeling through abnormal calcium handling in cardiomyocytes and enhancement of cardiac fibrosis via an activation of fibroblasts, are elevated both in the blood and local heart tissue of HF patients [14]. In addition, systemic inflammatory cytokines are considered one of the major causes of cachexia during HF [15][16]. Thus, the activation of local and/or systemic inflammatory reaction plays a pivotal role for the pathogenesis and progression of HF.
Classically, eicosanoids originated from n-3 PUFA are known to enhance the production of anti-inflammatory cytokines at the site of inflammation, whereas essential n-6 PUFAs, such as AA, are considered as substrates of pro-inflammatory eicosanoids, which promote vascular permeability, leucocyte infiltration and activation, and pro-inflammatory cytokine release [17]. The conventional interpretation is that the n-3 PUFAs antagonize the production and action of the inflammatory eicosanoid derived from AA metabolites. Indeed, in a pressure-overloaded HF rodent model (induced by aortic contraction surgery), the n-3 PUFA supplementation reduced serum TNF-α as well as pro-inflammatory eidosanoid (i.e., thromboxane B2), and prevented abnormal LV remodeling [18]. In addition, a number of human clinical investigations revealed that n-3 PUFA supplementation reduced the pro-inflammatory cytokines in terms of lower circulating levels of cytokines, e.g., TNF-α, IL-1, and IL-6 [10][19][20][21]. Basic studies also support the idea that n-3 PUFA would downregulate the pro-inflammatory pathways, such as NF-κB [22] and NLRP3 inflammasome [23], or upregulate anti-inflammatory intra-cellular signaling pathways, including peroxisome proliferator-activated receptor (PPAR) α/γ transcriptional activation [24]. Meanwhile, another study suggested the effect of low dose n-3 PUFA on NF-κB pathway activation in a cultured macrophage, potentially enhancing pro-inflammatory cytokines production [25]. The immunomodulatory activity of n-3 PUFAs has not been clearly explained yet and should be investigated further in detail.
Adiponectin, a peptide hormone released from adipose tissue, is known to show a cardio-protective effect through anti-inflammatory reaction. PPARγ-dependent adiponectin secretion is thought to correlate with n-3 PUFA-mediated cardiac protection, and the administration of n-3 PUFA increased the circulation of adiponectin in a dose-dependent manner both in animal [18][26] and human [27] studies.
The free FA specific membrane receptor family, a family of orphan G-protein coupled receptors (GPR), has been detected in the last 10 years. GPR120, also known as a free fatty acid receptor (FFR) 4, was detected as a receptor for n-3 PUFA, regulating downstream of intra-cellular signaling [28]. Eclov et al. revealed that the expression of FFR4 was markedly higher than other subtypes of FFRs in cardiomyocytes as well as fibroblasts isolated from mice [29]. In a rat heart study, a high-fat diet increased the expression level of FFR4 [30]. The involvement of FFR4 in the n-3 PUFA-mediated cardiac protection has been actively investigated in cardiac fibroblasts rather than cardiac myocytes. In a rodent pressure-overloaded HF model, EPA inhibited the transforming growth factor-beta 1 (TGF-β1) pro-fibrotic pathway via FFR signaling in cardiac fibroblasts and suppressed the entire cardiac fibrosis without requirement of EPA localization into the cellular membrane [29]. In addition, the activation of FFR4 by n-3 PUFA further stimulated endothelial nitric oxide synthase (eNOS) and promoted intracellular nitric oxide (NO), which leads to the suppression of TGF-β1 induced smad2/3 nuclear translocation and inhibition of pro-fibrotic gene transcription. It remains to be elucidated whether FFR4 is also expressed in human cardiomyocytes or cardiac fibroblasts.
In order to obtain enhanced benefits of n-3 PUFA-mediated cardiac protection, it would be better to identify the effective n-3 PUFA metabolites rather than to take a high dose of n-3 PUFA. Recently, the essential PUFA derived large class of cell signaling lipid mediators, named specialized pro-resolving mediators (SPMs; resolvins, protectins, lipoxins, and maresins) were discovered and indicated to exert the resolution of inflammatory reactions at nanomolar levels of concentration [31][32]. Halade et al., in a mouse coronary ligation model, reported that leukocytes, which immigrate from the splenic reservoir to the acutely infarcted myocardium, express lipoxygenases and abundant SPMs, predominantly derived from DHA to resolve the local acute inflammatory reaction [33]. Other rodent studies investigating the post-MI rodent heart revealed that the exogenous delivery of resolvins reduced infarction size [34] and excessive inflammation and fibrosis [35], leading to improved cardiac function [35]. Precursor of resolvins (i.e., 18-hydroxyeicosapentaenoic acid) administration in a pressure overload HF model mouse was also reported to ameliorate cardiac inflammation and fibrosis and preserve systolic function [36]. A recent clinical study revealed that plasma resolvin D1 levels were markedly decreased in patients with chronic HF compared with healthy subjects, suggesting a defect of resolvin biosynthesis in HF conditions [37]. To date, medicine purified or produced from SPMs has not been available or reached the stage of clinical application.
3.2. Effects on Myocardial Energy Metabolism and Mitochondrial Function of n-3 PUFA
Alteration of energetic substrate utilization in myocardium and modification of its intra-cellular signaling pathway also play important roles for the pathogenesis and progression of HF [38]. Previous animal studies revealed that the dietary n-3 PUFA intake altered the mitochondrial membrane phospholipid composition in cardiomyocytes, which led to a decrease in myocardial oxygen consumption without loss of ventricular power generation [39][40]. Furthermore, mitochondrial permeability transition pore (mPTP) opening, by which mitochondrial swelling and apoptotic cell death are activated, is suppressed by n3-PUFA (especially by DHA) supplementation [41][42][43]. Thus, it is considered that n-3 PUFA could protect the heart through improvement in the cardiac mitochondrial function as well as the efficiency of the ATP production [39][40][41][42][43].
In HF, lipotoxicity by excessive serum free FAs, which is caused by chronic adrenergic stimulation, is one of the critical pathophysiological mechanisms to exacerbate HF. Excessive free FA exposure to cardiomyocytes causes uncoupled mitochondrial respiration and reactive oxygen species (ROS) production [44], which thereby causes energy depletion and further impairs contraction in the failing heart [45]. Saturated fatty acids (SFA) (e.g., palmitate and stearate) are reported as the main components of serum free FAs [46]. Since unsaturated FAs, especially n-3 PUFA, are reported to act counter to the behavior of SFA, it is widely accepted that n-3 PUFA may mitigate SFA-induced lipotoxicity.
Mitochondria are dynamic organelles, which can continuously alter their morphology to maintain a number of cellular processes, such as cell cycle, immunity, apoptosis, and mitochondrial quality control. Mitochondrial dynamics play key roles for the pathophysiology of HF [47][48]. Specific fusion-related (e.g., mitofusin 1, mitofusin 2, and optic atrophy 1) and fission-related (e.g., dynamin related protein 1 (Drp1), mitochondrial fission 1 protein, and mitochondrial fission factor) proteins are involved in the regulation of mitochondrial dynamics [49]. We, in our previous investigation, reported that EPA activated phosphorylation of AMP-activated protein kinase (AMPK; a key enzyme for cellular energy homeostasis), altered mitochondrial morphology (relatively elongated mitochondria by suppression in Drp1) in myocardium, and thereby protected myocytes from SFA-induced cardiac lipotoxicity [50]. Despite the attractive pathophysiological findings of n-3 PUFA for cardiac protection against lipotoxicity, further in vivo animal and/or human investigations are warranted.
3.3. Anti-Arrhythmic Property by n-3 PUFA
Patients with HF are highly comorbid with arrhythmia, and the incidence of arrhythmia certainly worsens the rate of mortality of HF [51]. n-3 PUFAs are reported to reduce both atrial and ventricular arrhythmia, which may cause an improvement in the mortality of patients with HF. In the GISSI-HF trial, the reason for CVD risk reduction was mainly presumed to be due to anti-arrhythmic effects [6]. A number of basic experimental studies have revealed direct and/or indirect alteration in the electrophysiological behavior of plasma membrane ion channels of cardiomyocytes, such as the sodium, potassium and calcium channels, as well as the sodium-calcium exchanger [52][53]. In isolated mammalian cardiomyocytes (e.g., neonatal and adult rat ventricular myocytes), n-3 PUFA exhibited the inhibition of sodium current in a dose-dependent manner [54][55]. In addition, n-3 PUFA suppressed an intracellular Ca2+ wave, which was propagated from sarcoplasmic reticulum (SR) Ca2+ release by isoproterenol stimulation [56], suggesting a contribution of n-3 PUFA to suppression of arrhythmogenicity (i.e., myocardial triggered activity and abnormal automaticity) during HF.
Although the detailed mechanism remains unknown, n-3 PUFA may be able to improve the autonomic nervous system. An impaired autonomic tone is one risk-factor of a fatal arrhythmic event and sudden cardiac death in patients with dilated cardiomyopathy [57] and ischemic cardiomyopathy [58]. Some clinical studies revealed that n-3 PUFA supplementation improved heart rate variability (an index of autonomic tone) in patients with ischemic and non-ischemic cardiomyopathy [59][60][61][62]. Thus, n-3 PUFA may improve the mortality of HF patients through the autonomic nervous system and thereby suppression of anti-arrhythmogenicity.
3.4. Anti-Hypertensive Effect, Improvement of Vascular Endothelial Function, and Modulation of Autonomic Nervous System Activity by n-3 PUFA
It is well known that hypertension is a critical factor in developing HF. Previous investigations revealed the beneficial effects of fish oil on hypertension. Morris et. al. reported that the intake of fish oils reduced blood pressure (BP) by 3.0 mmHg in systole and by 1.5 mmHg in diastole (95% CI: systolic BP (4.5–1.5), diastolic BP (2.2–0.8)) [63]. Geleijnse et. al. also revealed that fish oils (mean intake 3.7 g/day) reduced BP by 2.1 mmHg in systole and 1.6 mmHg in diastole (95% CI: systolic BP (1.0–3.2), p < 0.01, diastolic BP (1.0–2.2), p < 0.01), and this anti-hypertensive effect of fish oil was obvious especially in the elderly and patients with hypertension [64]. The n-3 PUFA has been reported to release NO from vascular endothelial cells both in vivo [65] and in vitro [66]. In addition, DHA is reported to activate NO synthase and concentration of tetrahydrobiopterin in the central nervous system, which may increase local NO availability and exert tonic inhibition of central sympathetic outflow [67]. Thus, n-3 PUFA may suppress the progression of HF, not only through BP lowering, but also the correction of autonomic imbalance.
3.5. Anti-Thrombotic and Anti-Atherosclerotic Effects and Prevention of HF by n-3 PUFA
The anti-thrombotic and anti-atherosclerotic effects by n-3 PUFA can contribute to HF prevention through the risk reduction of ischemic heart disease [68][69]. The n-3 PUFA is reported to suppress the synthesis of platelet-derived thromboxane A2 (TXA2), which causes platelet aggregation and vasoconstriction [70], and to increase the plasminogen activator inhibitor-1 with reduction of fibrinogen [71]. Atherosclerotic plaque stabilization by n-3 PUFA has been reported. Matsumoto et al. reported that EPA significantly suppressed the development of atherosclerotic lesions in atherosclerosis-prone mice with reduced production of matrix metalloproteinases released by macrophages in a PPARα-dependent fashion [72]. RCTs of patients awaiting carotid endarterectomy also revealed that the supplementation of n-3 PUFA substantially increased tissue concentration of EPA and DHA, and decreased macrophage infiltration and thickened the fibrous cap in the human carotid artery [73].
References
- Ziaeian, B.; Fonarow, G.C. Epidemiology and aetiology of heart failure. Nat. Rev. Cardiol. 2016, 13, 368–378.
- Marik, P.E.; Varon, J. Omega-3 dietary supplements and the risk of cardiovascular events: A systematic review. Clin. Cardiol. 2009, 32, 365–372.
- Mozaffarian, D.; Rimm, E.B. Fish intake, contaminants, and human health: Evaluating the risks and the benefits. JAMA 2006, 296, 1885–1899.
- Leon, H.; Shibata, M.C.; Sivakumaran, S.; Dorgan, M.; Chatterley, T.; Tsuyuki, R.T. Effect of fish oil on arrhythmias and mortality: Systematic review. BMJ 2008, 337, a2931.
- Belin, R.J.; Greenland, P.; Martin, L.; Oberman, A.; Tinker, L.; Robinson, J.; Larson, J.; Van Horn, L.; Lloyd-Jones, D. Fish intake and the risk of incident heart failure: The Women’s Health Initiative. Circ. Heart Fail 2011, 4, 404–413.
- Tavazzi, L.; Maggioni, A.P.; Marchioli, R.; Barlera, S.; Franzosi, M.G.; Latini, R.; Lucci, D.; Nicolosi, G.L.; Porcu, M.; Tognoni, G. Effect of n-3 polyunsaturated fatty acids in patients with chronic heart failure (the GISSI-HF trial): A randomised, double-blind, placebo-controlled trial. Lancet 2008, 372, 1223–1230.
- Mozaffarian, D.; Lemaitre, R.N.; King, I.B.; Song, X.; Spiegelman, D.; Sacks, F.M.; Rimm, E.B.; Siscovick, D.S. Circulating long-chain omega-3 fatty acids and incidence of congestive heart failure in older adults: The cardiovascular health study: A cohort study. Ann. Intern Med. 2011, 155, 160–170.
- Wilk, J.B.; Tsai, M.Y.; Hanson, N.Q.; Gaziano, J.M.; Djousse, L. Plasma and dietary omega-3 fatty acids, fish intake, and heart failure risk in the Physicians’ Health Study. Am. J. Clin. Nutr. 2012, 96, 882–888.
- Yamagishi, K.; Iso, H.; Date, C.; Fukui, M.; Wakai, K.; Kikuchi, S.; Inaba, Y.; Tanabe, N.; Tamakoshi, A. Fish, omega-3 polyunsaturated fatty acids, and mortality from cardiovascular diseases in a nationwide community-based cohort of Japanese men and women the JACC (Japan Collaborative Cohort Study for Evaluation of Cancer Risk) Study. J. Am. Coll. Cardiol. 2008, 52, 988–996.
- Nodari, S.; Triggiani, M.; Campia, U.; Manerba, A.; Milesi, G.; Cesana, B.M.; Gheorghiade, M.; Dei Cas, L. Effects of n-3 polyunsaturated fatty acids on left ventricular function and functional capacity in patients with dilated cardiomyopathy. J. Am. Coll. Cardiol. 2011, 57, 870–879.
- Dijkstra, S.C.; Brouwer, I.A.; van Rooij, F.J.; Hofman, A.; Witteman, J.C.; Geleijnse, J.M. Intake of very long chain n-3 fatty acids from fish and the incidence of heart failure: The Rotterdam Study. Eur. J. Heart Fail 2009, 11, 922–928.
- Levitan, E.B.; Wolk, A.; Mittleman, M.A. Fish consumption, marine omega-3 fatty acids, and incidence of heart failure: A population-based prospective study of middle-aged and elderly men. Eur. Heart J. 2009, 30, 1495–1500.
- Levitan, E.B.; Wolk, A.; Mittleman, M.A. Fatty fish, marine omega-3 fatty acids and incidence of heart failure. Eur. J. Clin. Nutr. 2010, 64, 587–594.
- Van Linthout, S.; Tschope, C. Inflammation - Cause or Consequence of Heart Failure or Both? Curr. Heart Fail Rep. 2017, 14, 251–265.
- Lavine, K.J.; Sierra, O.L. Skeletal muscle inflammation and atrophy in heart failure. Heart Fail Rev. 2017, 22, 179–189.
- von Haehling, S.; Ebner, N.; Dos Santos, M.R.; Springer, J.; Anker, S.D. Muscle wasting and cachexia in heart failure: Mechanisms and therapies. Nat. Rev. Cardiol. 2017, 14, 323–341.
- Calder, P.C. Marine omega-3 fatty acids and inflammatory processes: Effects, mechanisms and clinical relevance. Biochim. Biophys. Acta 2015, 1851, 469–484.
- Duda, M.K.; O’Shea, K.M.; Tintinu, A.; Xu, W.; Khairallah, R.J.; Barrows, B.R.; Chess, D.J.; Azimzadeh, A.M.; Harris, W.S.; Sharov, V.G.; et al. Fish oil, but not flaxseed oil, decreases inflammation and prevents pressure overload-induced cardiac dysfunction. Cardiovasc. Res. 2009, 81, 319–327.
- Mehra, M.R.; Lavie, C.J.; Ventura, H.O.; Milani, R.V. Fish oils produce anti-inflammatory effects and improve body weight in severe heart failure. J. Heart Lung Transplant 2006, 25, 834–838.
- Mantzioris, E.; Cleland, L.G.; Gibson, R.A.; Neumann, M.A.; Demasi, M.; James, M.J. Biochemical effects of a diet containing foods enriched with n-3 fatty acids. Am. J. Clin. Nutr. 2000, 72, 42–48.
- Lennie, T.A.; Chung, M.L.; Habash, D.L.; Moser, D.K. Dietary fat intake and proinflammatory cytokine levels in patients with heart failure. J. Card Fail 2005, 11, 613–618.
- Zhao, Y.; Joshi-Barve, S.; Barve, S.; Chen, L.H. Eicosapentaenoic acid prevents LPS-induced TNF-alpha expression by preventing NF-kappaB activation. J. Am. Coll. Nutr. 2004, 23, 71–78.
- Yan, Y.; Jiang, W.; Spinetti, T.; Tardivel, A.; Castillo, R.; Bourquin, C.; Guarda, G.; Tian, Z.; Tschopp, J.; Zhou, R. Omega-3 fatty acids prevent inflammation and metabolic disorder through inhibition of NLRP3 inflammasome activation. Immunity 2013, 38, 1154–1163.
- Gani, O.A.; Sylte, I. Molecular recognition of docosahexaenoic acid by peroxisome proliferator-activated receptors and retinoid-X receptor alpha. J. Mol. Graph Model 2008, 27, 217–224.
- Wang, C.; Xiong, B.; Huang, J. The Role of Omega-3 Polyunsaturated Fatty Acids in Heart Failure: A Meta-Analysis of Randomised Controlled Trials. Nutrients 2016, 9, 18.
- Duda, M.K.; O’Shea, K.M.; Lei, B.; Barrows, B.R.; Azimzadeh, A.M.; McElfresh, T.E.; Hoit, B.D.; Kop, W.J.; Stanley, W.C. Dietary supplementation with omega-3 PUFA increases adiponectin and attenuates ventricular remodeling and dysfunction with pressure overload. Cardiovasc. Res. 2007, 76, 303–310.
- Itoh, M.; Suganami, T.; Satoh, N.; Tanimoto-Koyama, K.; Yuan, X.; Tanaka, M.; Kawano, H.; Yano, T.; Aoe, S.; Takeya, M.; et al. Increased adiponectin secretion by highly purified eicosapentaenoic acid in rodent models of obesity and human obese subjects. Arterioscler. Thromb Vasc. Biol. 2007, 27, 1918–1925.
- Hirasawa, A.; Tsumaya, K.; Awaji, T.; Katsuma, S.; Adachi, T.; Yamada, M.; Sugimoto, Y.; Miyazaki, S.; Tsujimoto, G. Free fatty acids regulate gut incretin glucagon-like peptide-1 secretion through GPR120. Nat. Med. 2005, 11, 90–94.
- Eclov, J.A.; Qian, Q.; Redetzke, R.; Chen, Q.; Wu, S.C.; Healy, C.L.; Ortmeier, S.B.; Harmon, E.; Shearer, G.C.; O’Connell, T.D. EPA, not DHA, prevents fibrosis in pressure overload-induced heart failure: Potential role of free fatty acid receptor 4. J. Lipid Res. 2015, 56, 2297–2308.
- Cornall, L.M.; Mathai, M.L.; Hryciw, D.H.; McAinch, A.J. Diet-induced obesity up-regulates the abundance of GPR43 and GPR120 in a tissue specific manner. Cell Physiol. Biochem. 2011, 28, 949–958.
- Schwab, J.M.; Chiang, N.; Arita, M.; Serhan, C.N. Resolvin E1 and protectin D1 activate inflammation-resolution programmes. Nature 2007, 447, 869–874.
- Serhan, C.N. Discovery of specialized pro-resolving mediators marks the dawn of resolution physiology and pharmacology. Mol. Aspects Med. 2017, 58, 1–11.
- Halade, G.V.; Norris, P.C.; Kain, V.; Serhan, C.N.; Ingle, K.A. Splenic leukocytes define the resolution of inflammation in heart failure. Sci. Signal. 2018, 11.
- Keyes, K.T.; Ye, Y.; Lin, Y.; Zhang, C.; Perez-Polo, J.R.; Gjorstrup, P.; Birnbaum, Y. Resolvin E1 protects the rat heart against reperfusion injury. Am. J. Physiol. Heart Circ. Physiol. 2010, 299, H153–H164.
- Kain, V.; Ingle, K.A.; Colas, R.A.; Dalli, J.; Prabhu, S.D.; Serhan, C.N.; Joshi, M.; Halade, G.V. Resolvin D1 activates the inflammation resolving response at splenic and ventricular site following myocardial infarction leading to improved ventricular function. J. Mol. Cell Cardiol. 2015, 84, 24–35.
- Endo, J.; Sano, M.; Isobe, Y.; Fukuda, K.; Kang, J.X.; Arai, H.; Arita, M. 18-HEPE, an n-3 fatty acid metabolite released by macrophages, prevents pressure overload-induced maladaptive cardiac remodeling. J. Exp. Med. 2014, 211, 1673–1687.
- Chiurchiu, V.; Leuti, A.; Saracini, S.; Fontana, D.; Finamore, P.; Giua, R.; Padovini, L.; Incalzi, R.A.; Maccarrone, M. Resolution of inflammation is altered in chronic heart failure and entails a dysfunctional responsiveness of T lymphocytes. FASEB J. 2019, 33, 909–916.
- Saotome, M.; Ikoma, T.; Hasan, P.; Maekawa, Y. Cardiac Insulin Resistance in Heart Failure: The Role of Mitochondrial Dynamics. Int. J. Mol. Sci. 2019, 20, 3552.
- Pepe, S.; McLennan, P.L. Cardiac membrane fatty acid composition modulates myocardial oxygen consumption and postischemic recovery of contractile function. Circulation 2002, 105, 2303–2308.
- Pepe, S.; McLennan, P.L. (n-3) Long chain PUFA dose-dependently increase oxygen utilization efficiency and inhibit arrhythmias after saturated fat feeding in rats. J. Nutr. 2007, 137, 2377–2383.
- Khairallah, R.J.; Sparagna, G.C.; Khanna, N.; O’Shea, K.M.; Hecker, P.A.; Kristian, T.; Fiskum, G.; Des Rosiers, C.; Polster, B.M.; Stanley, W.C. Dietary supplementation with docosahexaenoic acid, but not eicosapentaenoic acid, dramatically alters cardiac mitochondrial phospholipid fatty acid composition and prevents permeability transition. Biochim. Biophys. Acta 2010, 1797, 1555–1562.
- Khairallah, R.J.; O’Shea, K.M.; Brown, B.H.; Khanna, N.; Des Rosiers, C.; Stanley, W.C. Treatment with docosahexaenoic acid, but not eicosapentaenoic acid, delays Ca2+-induced mitochondria permeability transition in normal and hypertrophied myocardium. J. Pharmacol. Exp. Ther. 2010, 335, 155–162.
- Galvao, T.F.; Khairallah, R.J.; Dabkowski, E.R.; Brown, B.H.; Hecker, P.A.; O’Connell, K.A.; O’Shea, K.M.; Sabbah, H.N.; Rastogi, S.; Daneault, C.; et al. Marine n3 polyunsaturated fatty acids enhance resistance to mitochondrial permeability transition in heart failure but do not improve survival. Am. J. Physiol. Heart Circ. Physiol. 2013, 304, H12–H21.
- Tominaga, H.; Katoh, H.; Odagiri, K.; Takeuchi, Y.; Kawashima, H.; Saotome, M.; Urushida, T.; Satoh, H.; Hayashi, H. Different effects of palmitoyl-L-carnitine and palmitoyl-CoA on mitochondrial function in rat ventricular myocytes. Am. J. Physiol. Heart Circ. Physiol. 2008, 295, H105–H112.
- Opie, L.H.; Knuuti, J. The adrenergic-fatty acid load in heart failure. J. Am. Coll. Cardiol. 2009, 54, 1637–1646.
- Yli-Jama, P.; Meyer, H.E.; Ringstad, J.; Pedersen, J.I. Serum free fatty acid pattern and risk of myocardial infarction: A case-control study. J. Intern. Med. 2002, 251, 19–28.
- Chen, L.; Gong, Q.; Stice, J.P.; Knowlton, A.A. Mitochondrial OPA1, apoptosis, and heart failure. Cardiovasc. Res. 2009, 84, 91–99.
- Montaigne, D.; Marechal, X.; Coisne, A.; Debry, N.; Modine, T.; Fayad, G.; Potelle, C.; El Arid, J.M.; Mouton, S.; Sebti, Y.; et al. Myocardial contractile dysfunction is associated with impaired mitochondrial function and dynamics in type 2 diabetic but not in obese patients. Circulation 2014, 130, 554–564.
- Benard, G.; Karbowski, M. Mitochondrial fusion and division: Regulation and role in cell viability. Semin. Cell Dev. Biol. 2009, 20, 365–374.
- Sakamoto, A.; Saotome, M.; Hasan, P.; Satoh, T.; Ohtani, H.; Urushida, T.; Katoh, H.; Satoh, H.; Hayashi, H. Eicosapentaenoic acid ameliorates palmitate-induced lipotoxicity via the AMP kinase/dynamin-related protein-1 signaling pathway in differentiated H9c2 myocytes. Exp. Cell Res. 2017, 351, 109–120.
- Masarone, D.; Limongelli, G.; Rubino, M.; Valente, F.; Vastarella, R.; Ammendola, E.; Gravino, R.; Verrengia, M.; Salerno, G.; Pacileo, G. Management of Arrhythmias in Heart Failure. J. Cardiovasc. Dev. Dis. 2017, 4, 3.
- London, B.; Albert, C.; Anderson, M.E.; Giles, W.R.; Van Wagoner, D.R.; Balk, E.; Billman, G.E.; Chung, M.; Lands, W.; Leaf, A.; et al. Omega-3 fatty acids and cardiac arrhythmias: Prior studies and recommendations for future research: A report from the National Heart, Lung, and Blood Institute and Office Of Dietary Supplements Omega-3 Fatty Acids and their Role in Cardiac Arrhythmogenesis Workshop. Circulation 2007, 116, e320–e335.
- Richardson, E.S.; Iaizzo, P.A.; Xiao, Y.F. Electrophysiological mechanisms of the anti-arrhythmic effects of omega-3 fatty acids. J. Cardiovasc. Transl. Res. 2011, 4, 42–52.
- Xiao, Y.F.; Kang, J.X.; Morgan, J.P.; Leaf, A. Blocking effects of polyunsaturated fatty acids on Na+ channels of neonatal rat ventricular myocytes. Proc. Natl. Acad. Sci. USA 1995, 92, 11000–11004.
- Leifert, W.R.; McMurchie, E.J.; Saint, D.A. Inhibition of cardiac sodium currents in adult rat myocytes by n-3 polyunsaturated fatty acids. J. Physiol. 1999, 520 Pt 3, 671–679.
- Leifert, W.R.; Dorian, C.L.; Jahangiri, A.; McMurchie, E.J. Dietary fish oil prevents asynchronous contractility and alters Ca(2+) handling in adult rat cardiomyocytes. J. Nutr. Biochem. 2001, 12, 365–376.
- Fauchier, L.; Babuty, D.; Cosnay, P.; Fauchier, J.P. Prognostic value of heart rate variability for sudden death and major arrhythmic events in patients with idiopathic dilated cardiomyopathy. J. Am. Coll. Cardiol. 1999, 33, 1203–1207.
- La Rovere, M.T.; Bigger, J.T., Jr.; Marcus, F.I.; Mortara, A.; Schwartz, P.J. Baroreflex sensitivity and heart-rate variability in prediction of total cardiac mortality after myocardial infarction. ATRAMI (Autonomic Tone and Reflexes After Myocardial Infarction) Investigators. Lancet 1998, 351, 478–484.
- Nodari, S.; Metra, M.; Milesi, G.; Manerba, A.; Cesana, B.M.; Gheorghiade, M.; Dei Cas, L. The role of n-3 PUFAs in preventing the arrhythmic risk in patients with idiopathic dilated cardiomyopathy. Cardiovasc. Drugs Ther. 2009, 23, 5–15.
- Radaelli, A.; Cazzaniga, M.; Viola, A.; Balestri, G.; Janetti, M.B.; Signorini, M.G.; Castiglioni, P.; Azzellino, A.; Mancia, G.; Ferrari, A.U. Enhanced baroreceptor control of the cardiovascular system by polyunsaturated Fatty acids in heart failure patients. J. Am. Coll. Cardiol. 2006, 48, 1600–1606.
- Christensen, J.H.; Gustenhoff, P.; Korup, E.; Aaroe, J.; Toft, E.; Moller, J.; Rasmussen, K.; Dyerberg, J.; Schmidt, E.B. Effect of fish oil on heart rate variability in survivors of myocardial infarction: A double blind randomised controlled trial. BMJ 1996, 312, 677–678.
- La Rovere, M.T.; Staszewsky, L.; Barlera, S.; Maestri, R.; Mezzani, A.; Midi, P.; Marchioli, R.; Maggioni, A.P.; Tognoni, G.; Tavazzi, L.; et al. n-3PUFA and Holter-derived autonomic variables in patients with heart failure: Data from the Gruppo Italiano per lo Studio della Sopravvivenza nell’Insufficienza Cardiaca (GISSI-HF) Holter substudy. Heart Rhythm 2013, 10, 226–232.
- Morris, M.C.; Sacks, F.; Rosner, B. Does fish oil lower blood pressure? A meta-analysis of controlled trials. Circulation 1993, 88, 523–533.
- Geleijnse, J.M.; Giltay, E.J.; Grobbee, D.E.; Donders, A.R.; Kok, F.J. Blood pressure response to fish oil supplementation: Metaregression analysis of randomized trials. J. Hypertens. 2002, 20, 1493–1499.
- Wu, Y.; Zhang, C.; Dong, Y.; Wang, S.; Song, P.; Viollet, B.; Zou, M.H. Activation of the AMP-activated protein kinase by eicosapentaenoic acid (EPA, 20:5 n-3) improves endothelial function in vivo. PLoS ONE 2012, 7, e35508.
- Okuda, Y.; Kawashima, K.; Sawada, T.; Tsurumaru, K.; Asano, M.; Suzuki, S.; Soma, M.; Nakajima, T.; Yamashita, K. Eicosapentaenoic acid enhances nitric oxide production by cultured human endothelial cells. Biochem. Biophys. Res. Commun. 1997, 232, 487–491.
- Engstrom, K.; Saldeen, A.S.; Yang, B.; Mehta, J.L.; Saldeen, T. Effect of fish oils containing different amounts of EPA, DHA, and antioxidants on plasma and brain fatty acids and brain nitric oxide synthase activity in rats. Ups J. Med. Sci. 2009, 114, 206–213.
- Yokoyama, M.; Origasa, H.; Matsuzaki, M.; Matsuzawa, Y.; Saito, Y.; Ishikawa, Y.; Oikawa, S.; Sasaki, J.; Hishida, H.; Itakura, H.; et al. Effects of eicosapentaenoic acid on major coronary events in hypercholesterolaemic patients (JELIS): A randomised open-label, blinded endpoint analysis. Lancet 2007, 369, 1090–1098.
- Bhatt, D.L.; Steg, P.G.; Miller, M.; Brinton, E.A.; Jacobson, T.A.; Ketchum, S.B.; Doyle, R.T., Jr.; Juliano, R.A.; Jiao, L.; Granowitz, C.; et al. Cardiovascular Risk Reduction with Icosapent Ethyl for Hypertriglyceridemia. N. Engl. J. Med. 2019, 380, 11–22.
- Swann, P.G.; Venton, D.L.; Le Breton, G.C. Eicosapentaenoic acid and docosahexaenoic acid are antagonists at the thromboxane A2/prostaglandin H2 receptor in human platelets. FEBS Lett. 1989, 243, 244–246.
- Haglund, O.; Mehta, J.L.; Saldeen, T. Effects of fish oil on some parameters of fibrinolysis and lipoprotein(a) in healthy subjects. Am. J. Cardiol. 1994, 74, 189–192.
- Matsumoto, M.; Sata, M.; Fukuda, D.; Tanaka, K.; Soma, M.; Hirata, Y.; Nagai, R. Orally administered eicosapentaenoic acid reduces and stabilizes atherosclerotic lesions in ApoE-deficient mice. Atherosclerosis 2008, 197, 524–533.
- Thies, F.; Garry, J.M.; Yaqoob, P.; Rerkasem, K.; Williams, J.; Shearman, C.P.; Gallagher, P.J.; Calder, P.C.; Grimble, R.F. Association of n-3 polyunsaturated fatty acids with stability of atherosclerotic plaques: A randomised controlled trial. Lancet 2003, 361, 477–485.
More
Information
Subjects:
Cardiac & Cardiovascular Systems
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.0K
Entry Collection:
Hypertension and Cardiovascular Diseases
Revisions:
2 times
(View History)
Update Date:
11 Nov 2021
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





