
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Chrysoula (Chrysa) Voidarou | + 1139 word(s) | 1139 | 2021-09-22 08:23:15 | | | |
| 2 | Theodoros Varzakas | Meta information modification | 1139 | 2021-09-24 08:02:32 | | | | |
| 3 | Aura Daraba | + 5 word(s) | 1144 | 2021-09-25 11:04:12 | | | | |
| 4 | Aura Daraba | + 5 word(s) | 1144 | 2021-09-25 11:31:31 | | | | |
| 5 | Aura Daraba | + 5 word(s) | 1144 | 2021-09-25 11:32:22 | | | | |
| 6 | Aura Daraba | + 5 word(s) | 1144 | 2021-09-25 11:32:58 | | | | |
| 7 | JOCELYN C LEE | + 837 word(s) | 1976 | 2021-09-25 16:28:14 | | | | |
| 8 | JOCELYN C LEE | -98 word(s) | 1878 | 2021-09-27 00:12:50 | | | | |
| 9 | JOCELYN C LEE | -98 word(s) | 1878 | 2021-09-27 00:14:47 | | | | |
| 10 | Nora Tang | -51 word(s) | 1925 | 2021-09-27 03:26:02 | | | | |
| 11 | Nora Tang | -51 word(s) | 1925 | 2021-09-27 03:27:06 | | |
Video Upload Options
The food industry’s failure in planning and designing of and in implementing a Food Safety Management System and its foundation elements leads, in most instances, to compromised food safety and subsequent foodborne illness outbreaks. This phenomenon was noticed, worldwide, for all food processors, but with a much higher incidence in the medium- and small-sized food processing plants. Our study focuses on the importance of Food Safety Management System (FSMS), Critical Control Points Hazard Analysis (HACCP) and the Prerequisite Programs (PRPs) as the foundation of HACCP, in preventing foodborne outbreaks.
1. Introduction
The General Principles of Food Hygiene issued by Codex Alimentarius define Food Hygiene as: “All conditions and measures necessary to ensure the safety and suitability of food at all stages of the food chain; Properly applied prerequisite programs, including Good Hygiene Practices (GHPs), Good Agricultural Practices (GAPs) and Good Manufacturing Practices (GMPs), along with training and traceability, should provide the foundations for an effective HACCP system" [1].
In September 2005, ISO incorporated Hazard Analysis and Critical Control Point (HACCP) principles for food safety into the ISO 22000 quality management system. In this way, there has been an integration of HACCP program and principles to quality management systems and prerequisite programs (PRPs) for the improvement of the quality and safety of the food chain in the food industry.
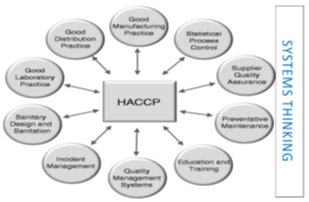
Figure 1. ‘Prerequisite programs (PRPs) adapted from What is a Management System? Available online: https://images.app.goo.gl/Aqez5sSfpbvjQqmK7 (accessed 20 June 2021).
It is well known that food chain includes organizations that are either directly involved with food or not directly involved with food processing and come in contact with either food or food ingredients [2].
ISO 22000:2018 standard allows international trading by assuring reliability, food quality, and food safety.
The aim of this review paper is to show the paramount importance of food safety management systems and their reliance on prerequisite programs for the implementation of effective preventive controls. Even though this may seem to be a very basic principle to practice, there have been incidents that this has been neglected which consequently led to cross-contamination and foodborne illness. The case study in this document is one such incident. Moreover, the implementation of management tools such as HAZOP, FMEA, Ishikawa and Pareto will be described. Finally, correlation with microbiological criteria is discussed in the context of EU regulation 2073/2005.
2. Identified Barriers to Implementing Food Safety Management Systems FSMS
The potential for food and beverages cross-contamination may occur at any and all food processing steps including transportation from the farm fields to processing facilities, processing steps in the food manufacturing facilities (due to deficient SSOPs, lack of personnel training, deficient GHPs, deficient good process practices (GPP), food packaging and storage). The most frequently identified barriers to implementing Food Safety Systems are: a) Lack of prerequisite programs (ranking #1), and b) Lack of knowledge about HACCP (ranking #2).[3].
Prerequisite GHPs must be in place first, before an effective FSMS, hazard analysis, and critical control and operational procedures. PRPs are encompassing the entire FSMS now, not only operational GHPs as in the past. Deficient point HACCP can be implemented [4]. Nowadays, PRPs include environmental criteria. PRPs are recognized to be an established foundation for the implementation of HACCP.
Meanwhile, HACCP and PRPs have gone unacknowledged as interdependent. For example, they are often taught separately, with more emphasis on HACCP training and certification than on PRPs. Case in hand, the ISO 22000:2005 on FSMSs contained minuscule guidance on PRP requirements that a supplemental specification for the food industry [5] on PRPs was developed for it to be benchmarked and approved by the Global Food Safety Initiative (GFSI) [6]. Thus, the symbiotic significance of PRPs to the manufacture of safe food is predominantly apparent. Recalls associated with pathogens such as Listeria or Salmonella are more often caused by PRP failure (post-process cross-contamination and | or unsanitary production environments) rather than the failure or mismanagement of critical control points (CCPs) in a HACCP plan [7].
GMP regulations guidance are designed to control the risk of food contamination with filth, dirt, scum, allergens, biofilm, chemicals, microbial particulates, and other mediums during food processing.
The most frequent cause of contamination is Cross-contamination of food by food handlers. Good food worker hygiene (GHP) is necessary because the cleanliness and behavior of workers determine the risk level (low risk or high risk) of cross-contamination from worker to food products and food contact surfaces. Trained, clean, sanitary, good hygiene-minded workers are of the utmost importance to produce safe food.
SSOPs are written in order to ensure sanitary conditions in food facilities. The SSOP procedures are specific to a plant but also may be similar to plants of the same or similar food sector categories. All SSOP procedures must be adequately documented and validated. When the facility’s SSOPs are in a developmental stage cGMPs could assist as a guide to the food facility.
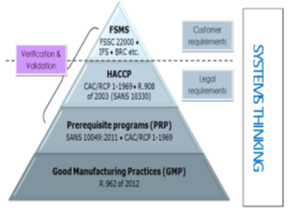
Figure 2. Adapted from Prerequisite Programs Ensure Food Safety Available online: https://images.app.goo.gl/jFnRtoUP3ByyLyn77 (accessed on 20 June 2021).
PRPs provide the hygienic foundations for any food operations. The terms prerequisite programs PRPs, Good Manufacturing Practices (GMPs), Good Hygienic Practices (GHPs) and sanitary standard operating practices (SSOPs) are interchangeable in diverse parts of the globe but carry the same applicable definition (Figure 1). “Prerequisite programs” (the term) is now frequently used for systems in support of HACCP principles, all-in written and spoken languages and practices. In other words, “The concept of supporting good practices is widely accepted” [8].
Many challenges faced today are extremely complex, and cannot be looked at from one perspective only, but they can and should be approached by looking at the system within. Looking at the system within, and having the courage to do so, restructure of the system can take place. By using systems thinking, complex problems can be solved [9].
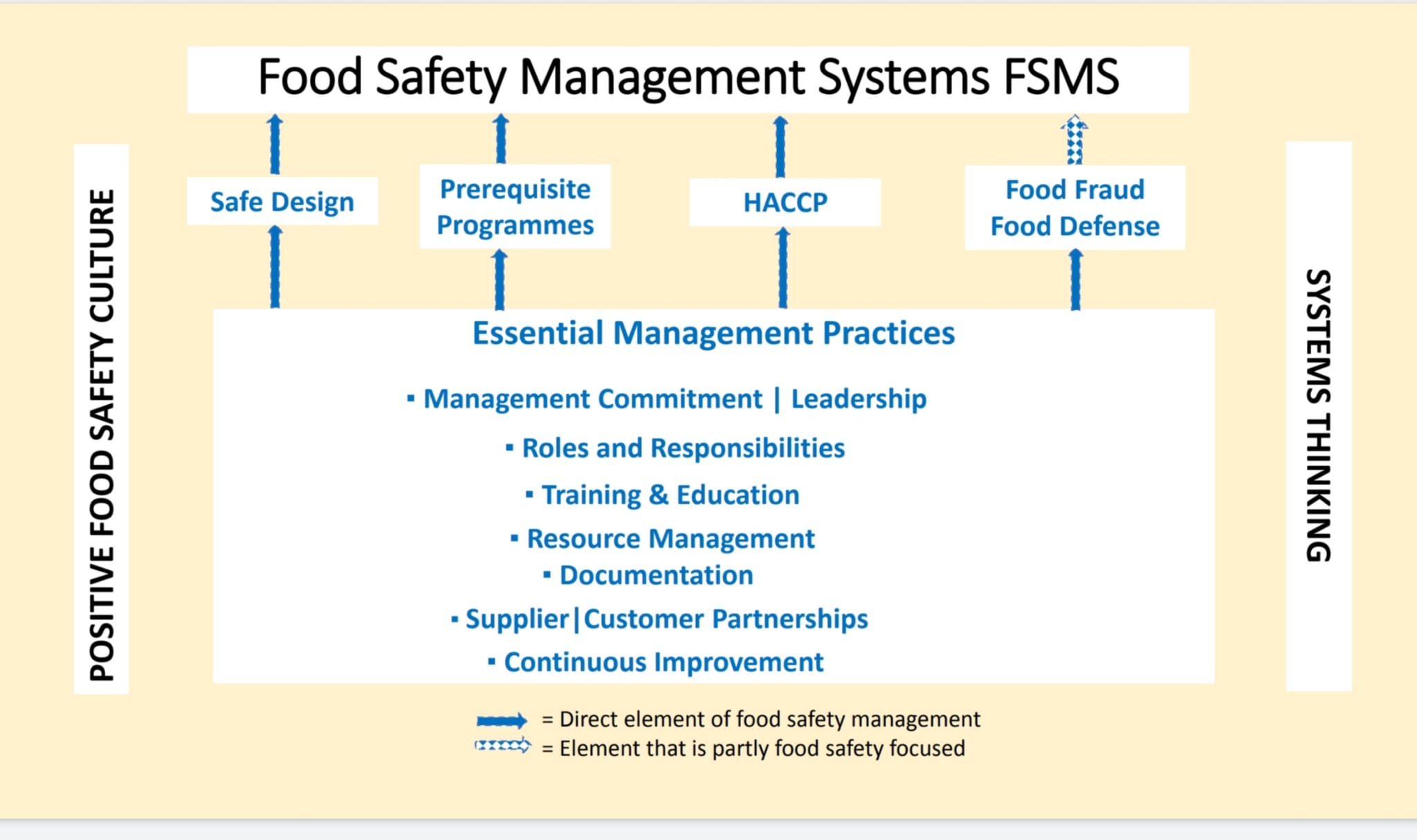
Figure 3. The interaction between FSMS, necessary good management practices, positive food safety
culture and systems thinking Source: (Adapted from “Food Safety for the 21st Century”, Figure 9.2,
page 164) [8].
3. Other Management Tools in Conjunction with ISO 22000 (Fmea Analysis-Pareto and Ishikawa Diagrams)
RPN assessment is carried out and corrective actions are proposed per identified hazard. Following calculation of RPN’ (after undertaking corrective actions), a new classification of Hazardous Elements is shown (Table 1, [10]).
| Defective Products | Estimated Corrective Actions Result | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Production Step | Hazards | Causes | S | O | D | RPN | Corrective Actions |
S’ | O’ | D’ | RPN’ |
| Receiving of raw materials Tr ≤ 5 °C Refrigeration Tr ≤ −18 °C freezer Tr = 18–22 °C Environment |
Physical, Chemical or Microbiological | Wrong handling | 8 | 5 | 4 | 160 | Not required | 7 | 2 | 2 | 28 |
Quantification of risk assessment was carried out by determining RPN per identified processing hazard. The highest RPNs (225, 225, 180 and 144, respectively) were correlated with receiving, storage and distribution, packaging and cooling and corrective actions were undertaken. Following the application of corrective actions, RPN values were below the upper acceptable limit of 130 [10].
Another case study involved FMEA model applied in conjunction with cause-and-effect analysis for the risk assessment of octopus processing ( Octopus vulgaris ). The highest RPNs (378, 294, 280, 252, 245 and 144, respectively) were correlated with chemically contaminated product, decomposed raw materials, scombrotoxin presence in the final product, incorrectly labelled product, storage in cans (foreign matter) and defective products, and corrective actions were undertaken. Following the application of corrective actions, new RPN values led to considerably lower values (below the upper acceptable limit of 130) [11][12].
Failure Mode and Effect Analysis (FMEA) has been applied for the risk assessment of snails manufacturing. Quantification of risk assessment was carried out by determination of the RPN per identified processing hazard. The highest RPNs (280, 240, 147, 144, respectively) corresponded to sterilization of tins, bioaccumulation of heavy metals, packaging of shells and poisonous mushrooms, and corrective actions were undertaken. Following the application of corrective actions, new RPNs led to lower values (below the upper acceptable limit of 130). The application of Ishikawa diagram led to converging results thus corroborating the validity of conclusions derived from risk assessment and FMEA [13].
4. ‘Process-Based’ Microbiological Criteria
However, the question as to what exactly is a quality food cannot be answered as easily as one might think, and the vagueness in the definitions of what constitutes quality has been noted by researchers such as Deming: «The difficulty in defining quality is in translating the future needs of the user into quantifiable characteristics, in a way which enables a product to be designed so as to provide satisfaction to the consumer at the price they will pay» [14].
Generally, there is an agreement among academics and researchers that there are two main aspects to food quality: objective and subjective. The former regards the physical features of a product and is related to quality control and food technology. The latter reflects the evaluation and judgments of consumers regarding perceived product quality characteristics. In any case, while food quality is an inherently complex and multifaceted concept it definitely has food safety at its core. Being the most essential variable for food quality, food safety is regulated by state legislation, in order to ensure that consumers purchase food products that fulfill their expectations regarding safety. In order to implement food quality and safety controls it is necessary to have real-time monitoring at critical points in the process. Rapid and precise methods of analysis are vital to guarantee product quality and safety, as well as compliance with labeling. The fast detection of spoilage agents such as bacteria, pathogens and other microbial contaminants in food production and processing is necessary, to reduce spoilage and secure a safe supply of food [15][16].
When testing results are not satisfactory for whichever type of criteria, food producers must apply the measures determined by the regulation. If there is a tendency towards repeated unsatisfactory results, food production companies must immediately take steps to prevent the occurrence of microbial hazards.
Depending on the particular characteristics of each food, food producers must guarantee compliance of a given product to relevant criteria which must have been set by authorities, without fail. These criteria regard the following products (Table 7 captures the main micro-organisms, their toxins, metabolites found in selected food categories).
5. Discussion
The case study highlights the critical importance of the successful implementation of a food safety management system and its reliance on prerequisite programs such as hazard analysis, SSOPs, microbiological criteria testing, management commitment, and leadership, etc.
Leadership is essential for positive food safety culture. Leaders must demonstrate ideal model behavior. The top damaging characteristic in food safety management is a food safety culture that nurtures fear. Amidst this “fear culture”, employees are disparaged from reporting potential incidents; they may cover up errors mistakes oversights, thereby increasing opportunities for incidents. We may presume this case study is one of toxic-negative-fear culture wherein there were no indications of any form of food safety management nor the important prerequisite programs [17].
Foodborne illness outbreak incidents, such as this case study, in the last decades, have deteriorated consumers' trust and have understandably generated a growing negative misperception on the enormity of food safety management systems [18].
We strongly believe that the implementation of food safety management systems along with other management tools such as Ishikawa, FMEA, Pareto, HAZOP could help the food industry to avoid the lack of good hygiene practices issues and to effectively and proactively monitor the critical control points (CCPs) and operational prerequisites (oPRPs). Training of all employees will play a catalytic factor in this direction and the adaptation of a positive food safety culture will minimize any likely risks, hazards, obstacles, and constraints. These steps will be beneficial not only for the company but also for the consumers who will enjoy safe, healthy, delicious, and nutritious food.
References
- Codex Committee on Food Hygiene. Guidelines for the Application of the Hazard Analysis Critical Control Point (HACCP) System, in Training Considerations for the Application of the HACCP System to Food Processing and Manufacturing; WHO/FNU/FOS/93.3 II, 1993; WHO: Geneva, Germany, 1995.
- British Standards Institute. PAS 220: 2008, Prerequisite Programmes on Food Safety for Food Manufacturing; BSI: London, UK, 2008.
- International Organization for Standardization. Food Safety Management Systems–Requirements for Any Organization in the Food Chain; BS EN ISO 22000; International Organization for Standardization: Geneva, Germany, 2005.
- Mortimore, S.E.; Warren, B.R. Prerequisite programs: Current perspectives in food manufacturing. Perspect. Public Health 2014, 134, 191.
- Wallace, C.W.; Sperber, W.H.; Mortimore, S.E. Food Safety for the 21st Century/Managing HACCP and Food Safety throughout the Global Supply Chain, 2nd ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2018; pp. 8–9.
- Makwela, M. Centre of Excellence in Food Security COE Article: Systems Thinking Can Be Used to Solve Complex Food Contamination Problems; Centre of Excellence in Food Security: Bellville, South Africa, 2016.
- Regulation (EC) No 178/2002. Available online: https://eur-lex.europa.eu/legal-content/EN/ALL/?uri=celex%3A32002R0178 (accessed on 20 April 2021).
- Motarjemi, Y.; Lelieveld, H. (Eds.) Food Safety Management a Practical Guide for the Food Industry. In Changes in Organizational Culture Leading to Incidents; Academic Press: London, UK, 2014.
- Srey, S.; Jahid, I.K.; Ha, S.D. Biofilm formation in food industries: A food safety concern. Food Control 2013, 31, 572–585.
- Ferone, M.; Gowen, A.; Fanning, S.; Scannell, A.G.M. Microbial detection and identification methods: Bench top assays to omics approaches. Compr Rev Food Sci Food Saf. 2020, 19, 3106–3129.





