
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Marco Valussi | + 8278 word(s) | 8278 | 2021-02-02 06:55:04 | | | |
| 2 | Nora Tang | -142 word(s) | 8136 | 2021-02-07 07:08:19 | | | | |
| 3 | Vicky Zhou | -5445 word(s) | 2691 | 2022-04-13 11:53:03 | | | | |
| 4 | Vicky Zhou | Meta information modification | 2691 | 2022-04-13 11:54:15 | | |
Video Upload Options
Bergamot essential oil (BEO) is the result of the mechanical manipulation (cold pressing) of the exocarp (flavedo) of the hesperidium of Citruslimon (L.) Osbeck Bergamot Group (synonym Citrus × bergamia Risso & Poit.), resulting in the bursting of the oil cavities embedded in the flavedo and the release of their contents. It is chemically dominated by monoterpene hydrocarbons (i.e., limonene), but with significant percentages of oxygenated monoterpenes (i.e., linalyl acetate) and of non-volatile oxygen heterocyclic compounds (i.e., bergapten).
The taxonomy, and consequently the nomenclature, of the genus Citrus, is particularly complicated and has been rapidly changing in recent years (see Table 1). For over 400 years, the centre of origin and biodiversity, along with the evolution and phylogeny of the Citrus species, have all been puzzling problems for botanists and the confusing and changing nomenclature of this taxon over the years can reflect intrinsic reproductive features of the species included in this genus, the cultural and geographical issues, and the rapidly evolving techniques used to clarify its phylogeny [1].
Because of the fragrance of the whole plant (flowers, leaves, fruits) and the delectability of the fruits, Citrus species have been exploited, domesticated, and selected by humankind for millennia. Classical Chinese literature mentions cultivated Mandarins and Pomelos as early as II millennia B.C.E., and citrons were depicted on the walls of the Egyptian Karnak Temple, dating 3000 years ago. Citrus fruits were known in ancient times even in India, Japan, Mesopotamia, Media, Persia, Palestine, and also in the Jewish culture, in Ancient Greece and Rome. In the Medieval period, several Citrus species were cherished and cultivated by the Arabs, who introduced many agronomical innovations and helped spread these plants across known countries [2].
The history of Citrus species distribution in Asia and Europe provides evidence of a complex domestication process, which has led to a high number of artificially obtained interspecific hybrids and cultivars, a fact that makes morphology-based classification very hard, given that fruits with similar morphology may belong to very distant species, and conversely, very similar or genetically almost indistinguishable varieties may give rise to very different morphologies [1][3].
Moreover, Citrus species show a very high frequency of spontaneous mutations and are a very sexually promiscuous taxon, with spontaneous hybridization being extremely common, another factor that makes it very hard to trace a solid and clear genealogy [1], and often these hybrids have been re-crossed with parent species, thus creating a variety of organisms that are sometimes genetically very close but morphologically very different, and vice versa, so much so that most of the Citrus fruits we normally consume are hybrids and not species (see below Table 2) [4]. Many citruses are capable of reproducing without fertilization (apomictic reproduction): specifically, they often present maternal nucellar embryogenesis, where nucellar tissue produces many clonal embryos, resulting in poly-embryonic seeds containing mainly or only nucellar embryos with the sole maternal genetic material. This is opposed to zygotic embryos, which contain genetic material of both parents (in mono-embryonic seeds). This process slows down or even stops the evolutionary process, thus making it again hard for taxonomists using morphological features to understand the true lineage [1][5][6].
Table 1. Taxonomy of the Citrus genus [7][8][9][10][11][12].
| Angiospermae |
| Eudicots |
| Superrosids |
| Rosids |
| Sapindales Juss. ex Bercht & J.Presl. |
| Rutaceae Juss. |
| ———Rutoideae (120 spp.) |
| ———Spathelipideae (5 spp.) |
| ———Aurantioideae (30 spp.) |
| ——————Clauseneae |
| ——————Citrae (Aurantieae) |
| —————————Triphasinae |
| —————————Balsamocitrinae |
| —————————Citrinae |
| ————————————Primitive Citrus fruits (Severinia, Pleiospermium, Burkillanthus, Limnocitrus, Hesperethusa) |
| ————————————Near Citrus fruits (Atalantia and Citropsis) |
| ————————————True Citrus fruits |
| ———————————————Clymenia |
| ———————————————Eremocitrus |
| ———————————————Microcitrus |
| ———————————————Poncirus |
| ———————————————Fortunella |
| ———————————————Citrus |
| ——————————————————Papeda |
| ——————————————————Eucitrus (Citrus) |
Table 2. Common Citrus Hybrids.
| These hybrids are often treated as separate species. Particularly important is the Mandarin-Pomelo hybridization axis, which leads to species with orange or red peeled fruits when ripe [1]: |
| Oranges. The Bitter Orange (Citrus × aurantium L. (sin Citrus × aurantium subsp. amara (Link) Engl)) is a pure Mandarin and Pomelo cross, the Sweet Orange (Citrus sinensis (L.) Osbeck) is a cross between a Mandarin (already crossed with a Pomelo) and a Pomelo. Both hybrids inherit the orange colour of the peel from the Mandarin. |
| Grapefruit (Citrus paradisi Macf.). These also derive from the gene pool of Mandarin and Pomelo, but with a higher contribution than the latter one, thanks to a cross between Sweet Orange and Pomelo. |
| Mandarins (Citrus reticulata Blanco). Mandarins and Clementine belong to the class of Mandarin hybrids, with genetic contributions from Sweet and Bitter Oranges and Grapefruit. |
| Lemons (Citrus limon (L.) Osbeck). “Real” Lemons have a light yellow peel and are derived from a single ancestral hybrid between male Citron and female Bitter Orange (i.e., 50% Citron, 25% Mandarin, 25% Pomelo). |
| Bergamot. This is a hybrid whose origin is still debated, but it seems to have been obtained by crossbreeding of some Citrus hybrids (Bitter Orange, Lemon, Lime) with their ancestors (Citron) or with other hybrids. |
Origins of the Genus Citrus
The first modern classification of the genus Citrus (in the Family Rutaceae) was formulated by Carl von Linné, who, in his “Species Plantarum”, clustered all Citrus fruits under two species: Citrus medica L. (Citron) along with its only variety Citrus medica var. limon (Lemon), and Citrus aurantium L. (Bitter Orange) along with its two varieties, Citrus aurantium var. grandis (Pomelo) and Citrus aurantium var. sinensis (Sweet Orange) [13]. All subsequent classifications in the following centuries were based on morphological and geographical data, and different authors recognized a diverse number of species.
A revolution in the taxonomy of Citrus has come with the development of laboratory techniques involving specific molecular markers, such as Random Amplification of Polymorphic DNA (RAPD), Sequence Characterized Amplified Region (SCAR), cpDNA Simple Sequence Repeats (SSR), Inter-Simple Sequence Repeat (ISSRs), Amplified Fragment Length Polymorphism (AFLP) and Restriction Fragment Length Polymorphisms (RFLPs) markers [3][14], and the triangulation with information from historical literature and contemporary data about the natural distribution of these species [5].
According to one study [15], there are at least 10 progenitor species. Of these, seven are original to Asia: Pomelo (Citrus maxima), the true Mandarin (C. reticulata), Citron (C. medica), Papeda (C. micrantha), Ichang’s Papeda (C. ichangensis), Mangshanyegan (C. mangshanensis), and Oval Kumquat (C. japonica). The remaining three species are Australian: Desert Lime (C. glauca), Round Lime (C. australis), and Lemon Caviar (C. australasica).
Recently, important genomic work [4][16] has provided the most up-to-date evolutionary tree for Citrus, thus challenging previous proposals. The paper by Wu and colleagues [4] that used single nucleotide polymorphism (SNP), proposes that at least three genera (Fortunella, Eremocitrus and Microcitrus) are nested in the Citrus clade, and that of the ten progenitor species, three are to be considered ancestral, namely Citrus maxima, Citrus reticulata and Citrus medica. Four main hybrids are derived from these ancestral species: Lemons, Limes, Oranges and Grapefruits [17][18].
Wu and colleagues [4] state that, according to their analysis of the accessions, “the centre of origin of Citrus species was the southeast foothills of the Himalayas, in a region that includes the eastern area of Assam, northern Myanmar and western Yunnan”, while inferences from modern distribution point towards the central-southern part of China as the primary biodiversity centre, with southeast Himalaya and southern China as two accessory centres [1]. Citrus probably moved from West to East Malaysia and Australia sometime in the Miocene/Pliocene [19].
Because of the intricacy of the taxonomy of the Citrus genus, from an agronomic perspective, when dealing with commercially important species, a pure Linnaean classification could be substituted by just using cultivar names grouped under general agronomic groups. At least eight groups have been recognized so far, the Sweet Orange Group (cultivar: navel, common, blood, acidless), the Mandarin Group (Satsuma, tangerine/clementine, Mediterranean Mandarin and Mandarin), the Lemon Group, the Grapefruit Group, the Lime Group, the Sour Orange Group (cultivar: Seville, Bouquet de fleur, Granita, Chinotto, Bergamot), the Pomelo Group, and the Citron group [13].
Taxonomy of Bergamot
The phylogeny and taxonomy of Bergamot (Figure 1) are particularly unclear, as it is its exact area of origin [20].
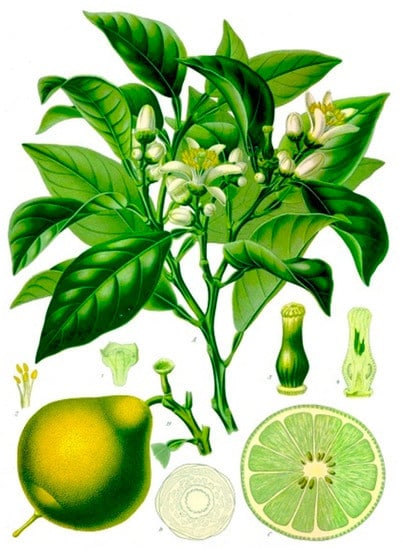
Figure 1. Bergamot plant and fruit, as illustrated in the traditional Köhler’s Medizinal-Pflanzen [21].
The plant is an interspecific hybrid, and this is testified by the high percentage of heterozygosity [22] and has been clustered with Bitter Orange [23]. The actual progenitor species of Bergamot have been claimed to be Citrus x aurantium (Bitter Orange) and C. x limon (Lemon), or C. x aurantium and C. x aurantifolia (Key Lime) [24], or C. x aurantium and C. medica [25][26].
Bergamot seems to have originated quite late in the phylogeny of the Citrus clade and was unknown to ancient authors. Some sources claim that the hybrid was originated directly in Calabria, or at least in the southern part of Italy [27], while others state that the hybrid first appearance was outside Italy (either in the Antilles or in Greece or the Canary Islands) and that it was brought to Calabria only later [22]. One source hypothesizes that the hybrid was brought to Italy by Columbus via the Spaniard city of Berga, which inspired the fruit name, Bergamot [22]. A more solid hypothesis for the name origin is that the term “Bergamot” may derive from the Turkish word “beg-a-mudi” which means “pears of the Prince”, due to its certain resemblance to Bergamot pears [27].
Some authors also identify four subgroups of Bergamot, namely the “Piccola” group, the “Torulosa” group, the “Melarosa” group, and the “Common” group. All Bergamot fruits that are interesting from an industrial point of view come from the “Common” group, which includes three cultivars:
“Castagnaro”, whose fruits give perhaps the least valuable essential oil (EO), but can be harvested for production for longer than any other cultivar.
“Femminiello”, less vigorous and strong than Castagnaro, with a low yield of a superior, excellent EO.
“Fantastico” (or “Inserto”), supposed to be a hybrid of the first two varieties, certainly introduced more recently (about 1940) but representing now a good part of all the trees in Calabria [22].
Concerning nomenclature, opinions of different authorities diverge, as explained in Table 3.
Table 3. Nomenclature of Bergamot according to different authorities and institutions.
| Authority | Binomial |
|---|---|
| Integrated Taxonomic Information System [28] | Citrus aurantium subsp. bergamia (Risso & Poit.) Wight & Arn. ex Engl |
| International Plant Names Index [29] | Citrus bergamia Risso |
| Tropicos [30] | Citrus × bergamia Risso & Poit. |
| World Checklist of Selected Plant Families [31] | Citrus limon (L.) Osbeck |
| U.S. Dep of Agriculture: Agricultural Research Service [32] | Citrus × limon (L.) Osbeck var. bergamia (Loisel.) ined. |
| Mabberley’s Plant-Book [12][33] | Citrus × limon, Bergamot group |
Definition of EO and Peculiarities of Citrus EOs
The International Organization for Standardization (ISO) definition (ISO 2020 document ISO 9235: 2013-2.11) states that an EO is a “product obtained from a natural raw material of plant origin, by steam distillation, by mechanical processes from the epicarp of Citrus fruits, or by dry distillation, after separation of the aqueous phase—if any—by physical processes”. Such includes three production methods, one of them exclusively reserved for Citrus EOs, which are therefore clearly separated from other EOs by production method and, therefore, by chemical composition. Distilled EOs contain only volatile molecules, while cold-pressed EOs may contain much heavier molecules and different chemical compounds from those normally found in distilled EOs. The second characteristic of Citrus fruit EOs is that, from an economic point of view, in many cases, the EO is a secondary product, while the fruit juice is the primary product, and other products can be obtained from the fruit (candied peels, “cells”, albedo, etc.). This is true for Lemons, Sweet Oranges, Grapefruits and Mandarins, but not for Bergamots and Bitter Oranges, whose fruit juice is generally produced in small quantities or even not at all. This fact partly explains the higher costs of both EOs [34].
Anatomy of Citrus fruits
The fruit of Citrus species is a modified berry called “hesperidium”, characterized by an approximately spherical shape, and by a very thick and robust outer rind or peel protecting the fluid-filled trichomes that make up the segments of the fruit juicy part (endocarp). The rind is divided in a superficial, thin, coloured exocarp called flavedo (from classical Latin flavus, or “blonde”) and a deeper and thicker spongy mesocarp called albedo (from the Latin albedo, derivative of albus, or “white”).
The flavedo is surrounded by a thick, waterproof cuticle and by various stomata in the superficial layer of the epidermis, it is made of small, dense collenchyma cells with chromoplasts, and contains many EO-bearing cavities called lumina or lacuna [22]. In the Bergamot fruit, these more or less spherical central cavities are surrounded by four to six layers of secretory cells and are formed in a schizolysigenous way [35], that is first by the separation of parenchymal cells (schizogenesis of the intercellular space), followed by further disintegration of cells in later stages of cavity development (lysogeny). These lacunae have no walls, are not connected, are found at different depths, and fill up with EO as the fruit ripens. They are of different sizes (the range is 0.4–0.6 mm in diameter) and they are conventionally divided in primary, secondary and tertiary cavities [34][36].
It is precisely this superficial distribution of the oil cavities that has made the method of cold-pressing or mechanical treatment the preferred one to obtain Citrus EOs [5][22].
Distribution and Cultivation of Bergamot
Around 90% of total BEO global production comes from Italy and specifically from a small area of fewer than 1400 hectares in Calabria [37]. Other regions of the world where Bergamot cultivation and BEO production have been tried are the African continent and the Americas, and lately China. Of all these producers, only the Ivory Coast has a commercially important share of the market, with 8–10% of global BEO production, although Brazil and China are emerging as global producers [22].
The introduction of Bergamot in Africa started only in the 1950s, and up until 1959, apart from a few trees in the Botanical Garden of Bingerville close to Abidjan, there was no record of the plants in this region. Other African countries such as Guinea, Liberia, Mali, Algeria, Morocco, and Tunisia only have small industrial productions with no indication of possible expansion [22].
One of the main problems for BEO production in Africa is that it appears to be up to 3–5 times lower per cultivated hectare than that of Calabria [38][39]. The reason for this has been sought in some differences in production technology, in the different climate, but above all in the characteristic composition of the soil. It can be added that a difference of three centuries of cultivation in Italy has allowed a better selection of plants [38][39].
The introduction of Bergamot in the Americas started in the US, specifically in Florida, Louisiana and California, where the tree was imported before 1815 but where it failed to thrive. The cultivations in South America were slightly more successful. Introduced since the 1960s there is only a modest industrial production of a good quality product in Argentina, Brazil and Uruguay [22].
The differences in the chemistry of BEO from various regions of the world are difficult to gauge because of their large variability, due to non-technological parameters according to Dugo and Bonaccorsi [22]. What can be stated is that most of the EOs produced in regions different from Calabria show values of β-pinene and γ-terpinene lower or close to the minima determined for Calabrian EOs and that in terms of enantiomeric composition it seems, from the scarce available chiral data, that EOs from the Ivory Coast and Uruguay agree with those of EOs from Calabria [22].
Distribution and Cultivation of Bergamot in Calabria
The Bergamot tree possibly originated as a seedling in southern Italy but, until the mid-sixteenth century, it was known only as an ornamental plant in Tuscany. It was first officially mentioned as an “aurantium stellatum et roseum” by Ferrari in 1646 in his “Hesperides sive de malorum aureorum cultura et usu”, and first described in 1708 by Volkamer as “gloria limonum fructus inter omnes nobilissimus” in his “Nurnbergische Hesperides”, where the importance of its essence was first reported [27].
It became famous in France by 1686, thanks to Francis Procopius, as the source of a precious perfume ingredient, named “Bergamot water”. It was also introduced in 1676 in Cologne by Paolo Feminis, who used it to create the famous Cologne water (eau de cologne), commercialised by his son-in-law, Gian Maria Farina [27].
This notoriety greatly stimulated the demand for the EO, and as a consequence in 1750 Nicola Parisi planted, in Reggio Calabria, the first Bergamot orchard (Rada Giunchi Estate). From there, then cultivation expanded around the city of Reggio Calabria and along the Straits of the Messina area [27]. The tree seems to be very sensitive to pedoclimatic conditions and hence cultivation was and is mainly limited to the region of Calabria [22]. After periods of expansion and reduction in the cultivated area, today Bergamot cultivation occurs mainly in a stretch of land of 100–140 km from Reggio di Calabria following the Ionian Coast, comprising 1000–1400 hectares between Villa San Giovanni and Monasterace, 800 of which are used by farmers who are part of the organization “Organizzazione Produttori Unionberg” [40].
References
- Zhong, G.; Nicolosi, E. Citrus Origin, Diffusion, and Economic Importance. In The Citrus Genome; Gentile, A., La Malfa, S., Deng, Z., Eds.; Springer International Publishing: Cham, Switzerland, 2020; pp. 5–21, ISBN 9783030153083.
- Calabrese, F. La Favolosa Storia Degli Agrumi; L’Epos: Palermo, Italy, 1998.
- Inglese, P.; Sortino, G. Citrus History, Taxonomy, Breeding, and Fruit Quality. In Oxford Research Encyclopedia of Environmental Science; Oxford University Press: Oxford, UK, 2019; ISBN 9780199389414.
- Wu, G.A.; Terol, J.; Ibanez, V.; López-García, A.; Pérez-Román, E.; Borredá, C.; Domingo, C.; Tadeo, F.R.; Carbonell-Caballero, J.; Alonso, R.; et al. Genomics of the origin and evolution of Citrus. Nature 2018, 554, 311–316, doi:10.1038/nature25447.
- Dugo, G.; Mondello, L. Citrus Oils: Composition, Advanced Analytical Techniques, Contaminants, and Biological Activity; CRC Press: Boca Raton, FL, USA, 2010; ISBN 9781439800294.
- Distefano, G.; Las Casas, G.; Deng, X.; Chai, L. Citrus Reproductive Biology from Flowering to Fruiting. In The Citrus Genome; Gentile, A., La Malfa, S., Deng, Z., Eds.; Springer International Publishing: Cham, Switzerland, 2020; pp. 167–176, ISBN 9783030153083.
- Chase, M.W.; Christenhusz, M.J.M.; Fay, M.F.; Byng, J.W.; Judd, W.S.; Soltis, D.E.; Mabberley, D.J.; Sennikov, A.N.; Soltis, P.S.; Stevens, P.F. An update of the Angiosperm Phylogeny Group classification for the orders and families of flowering plants: APG IV. Bot. J. Linn. Soc. 2016, 181, 1–20, doi:10.1111/boj.12385.
- Thorne, R.F. The classification and geography of the flowering plants: Dicotyledons of the class Angiospermae. Bot. Rev. 2000, 66, 441–647, doi:10.1007/BF02869011.
- Swingle, W.T. The botany of Citrus and its wild relations. Citrus Ind. 1967, 1, 190–422.
- Samuel, R.; Ehrendorfer, F.; Chase, M.W.; Greger, H. Phylogenetic analyses of Aurantioideae (Rutaceae) based on non-coding plastid DNA sequences and phytochemical features. Plant Biol. 2001, 3, 77–87.
- Dearaujo, E. What is Citrus? Taxonomic implications from a study of cp-DNA evolution in the tribe Citreae (Rutaceae subfamily Aurantioideae). Org. Divers. Evol. 2003, 3, 55–62.
- Bayer, R.J.; Mabberley, D.J.; Morton, C.; Miller, C.H.; Sharma, I.K.; Pfeil, B.E.; Rich, S.; Hitchcock, R.; Sykes, S. A molecular phylogeny of the orange subfamily (Rutaceae: Aurantioideae) using nine cpDNA sequences. Am. J. Bot. 2009, 96, 668–685, doi:10.3732/ajb.0800341.
- Dugo, G.; Di Giacomo, A. Citrus: The Genus Citrus; CRC Press: Boca Raton, FL, USA, 2002; ISBN 9780203216613.
- Goldschmidt, E.E. The Citrus Genome: Past, Present and Future. In The Citrus Genome; Gentile, A., La Malfa, S., Deng, Z., Eds.; Springer International Publishing: Cham, Switzerland, 2020; pp. 1–3, ISBN 9783030153083.
- Velasco, R.; Licciardello, C. A genealogy of the Citrus family. Nat. Biotechnol. 2014, 32, 640–642.
- Carbonell-Caballero, J.; Alonso, R.; Ibañez, V.; Terol, J.; Talon, M.; Dopazo, J. A Phylogenetic Analysis of 34 Chloroplast Genomes Elucidates the Relationships between Wild and Domestic Species within the Genus Citrus. Mol. Biol. Evol. 2015, 32, 2015–2035, doi:10.1093/molbev/msv082.
- Wu, G.A.; Prochnik, S.; Jenkins, J.; Salse, J.; Hellsten, U.; Murat, F.; Perrier, X.; Ruiz, M.; Scalabrin, S.; Terol, J.; et al. Sequencing of diverse mandarin, pummelo and orange genomes reveals complex history of admixture during Citrus domestication. Nat. Biotechnol. 2014, 32, 656–662.
- Curk, F.; Ollitrault, F.; Garcia-Lor, A.; Luro, F.; Navarro, L.; Ollitrault, P. Phylogenetic origin of limes and lemons revealed by cytoplasmic and nuclear markers. Ann. Bot. 2016, 117, 565–583, doi:10.1093/aob/mcw005.
- Schwartz, T.; Nylinder, S.; Ramadugu, C.; Antonelli, A.; Pfeil, B.E. The Origin of Oranges: A Multi-locus Phylogeny of Rutaceae Subfamily Aurantioideae. Syst. Bot. 2016, 40, 1053–1062, doi:10.1600/036364415X690067.
- Hodgson, R.W. Horticultural varieties of Citrus. In The Citrus Industry; Reuther, W., Webber, H.J., Batchelor, L.D., Eds.; Volume I.; University of California Press: Berkeley, CA, USA, 1967; pp. 431–592.
- Köhler, H.A. Köhler’s Medizinal-Pflanzen. Arch. Pharm. 1887, 225, 276–276.
- Dugo, G.; Bonaccorsi, I. Citrus Bergamia: Bergamot and its Derivatives; CRC Press: Boca Raton, FL, USA, 2013; ISBN 9781439862278.
- Uzun, A.; Yesiloglu, T.; Aka-Kacar, Y.; Tuzcu, O.; Gulsen, O. Genetic diversity and relationships within Citrus and related genera based on sequence related amplified polymorphism markers (SRAPs). Sci. Hortic. 2009, 121, 306–312.
- Federici, C.T.; Roose, M.L.; Scora, R.W. RFLP analysis of the origin of Citrus bergamia, Citrus jambhiri, and Citrus limonia. Proc. First Int. Citrus Biotechnol. Symp. 1998, 535, 55–64.
- Nicolosi, E.; Deng, Z.N.; Gentile, A.; La Malfa, S.; Continella, G.; Tribulato, E. Citrus phylogeny and genetic origin of important species as investigated by molecular markers. Theor. Appl. Genet. 2000, 100, 1155–1166, doi:10.1007/s001220051419.
- Li, X.; Xie, R.; Lu, Z.; Zhou, Z. The origin of cultivated Citrus as inferred from internal transcribed spacer and chloroplast DNA sequence and amplified fragment length polymorphism fingerprints. J. Am. Soc. Hortic. Sci. 2010, 135, 341–350.
- Maruca, G.; Laghetti, G.; Mafrica, R.; Turiano, D.; Hammer, K. The Fascinating History of Bergamot (Citrus Bergamia Risso & Poiteau), the Exclusive Essence of Calabria: A Review. JESE-A 2017, 6, doi:10.17265/2162-5298/2017.01.003.
- Guala, G. Integrated Taxonomic Information System (ITIS); National Museum of Natural History, Smithsonian Institution: Washington, DC, USA, 2019.
- Ipni. The International Plant Names Index; Royal Botanic Gardens, Kew: Richmond, UK, 2012.
- Tropicos. Available online: http://www.tropicos.org (accessed on 6 November 2020).
- Govaerts, R.H.A.; Faden, R.B. World Checklist of Selected Plant Families; Royal Botanic Gardens, Kew: Richmond, UK, 2016.
- Search Accessions GRIN-Global. Available online: https://npgsweb.ars-grin.gov/gringlobal/search (accessed on 6 November 2020).
- Mabberley, D.J. Mabberley’s Plant-Book, 3rd ed.; Cambridge University Press: Cambridge, UK, 2008.
- Fatta Del Bosco, S.; Abbate, L.; Mercati, F.; Napoli, E.; Ruberto, G. Essential Oils in Citrus. In The Citrus Genome; Gentile, A., La Malfa, S., Deng, Z., Eds.; Springer International Publishing: Cham, Switzerland, 2020; pp. 211–223, ISBN 9783030153083.
- Rapisarda, A.; Caruso, C.; Iauk, L.; Ragusa, S. Applicazione dell’analisi di immagine nello studio delle ghiandole oleifere dei frutti di alcune specie del genere Citrus. Essenze Deriv. Agrum. 1996, 66, 5–12.
- Svoboda, K.P.; Svoboda, T.G.; Syred, A.D. Secretory Structures of Aromatic and Medicinal Plants: A Review and Atlas of Micrographs; Microscopix Publications: Knighton, Wales, 2000.
- Amato, A.; Castellotti, T.; Gaudio, F.; Gaudio, G.; Lovecchio, R.; Pupo D’Andrea, M.R.; Peluso, R. L’agricoltura nella Calabria in cifre 2012; INEA–Istituto Nazionale di Economia Agraria: Roma, Italy, 2013. Available online: http://www.inea.it/pubbl/ (accessed on 6 November 2020).
- Giacomo, A.D. Bergamot in the calabrian economy and the role of consorzio del bergamotto. In Citrus Bergamia; CRC Press: Boca Raton, FL, USA, 2013; pp. 74–85, ISBN 9780429165887.
- Ciani, F.; Huggard, J.; Zervas, T. The Resilience of Bergamot Farmers to Economic Shocks in the Reggio Calabria Province. Reg. Mag. 2016, 302, 23–25, doi:10.1080/13673882.2016.11742705.
- Nesci, F.S.; Sapone, N. Bergamot–A Green and Multifunctional Asset Exclusive from the Province of Reggio Calabria. AEF 2014, 11, 376–379, doi:10.4028/www.scientific.net/AEF.11.376.

