
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Francisco R. Nieto | + 3311 word(s) | 3311 | 2021-07-21 08:08:14 | | | |
| 2 | Bruce Ren | -21 word(s) | 3290 | 2021-07-21 10:18:50 | | |
Video Upload Options
Tetrodotoxin (TTX) is a potent neurotoxin found mainly in puffer fish and other marine and terrestrial animals. TTX blocks voltage-gated sodium channels (VGSCs) which are typically classified as TTX-sensitive or TTX-resistant channels. VGSCs play a key role in pain signaling and some TTX-sensitive VGSCs are highly expressed by adult primary sensory neurons. During pathological pain conditions, such as neuropathic pain, upregulation of some TTX-sensitive VGSCs, including the massive re-expression of the embryonic VGSC subtype NaV1.3 in adult primary sensory neurons, contribute to painful hypersensitization. In addition, people with loss-of-function mutations in the VGSC subtype NaV1.7 present congenital insensitive to pain. TTX displays a prominent analgesic effect in several models of neuropathic pain in rodents. According to this promising preclinical evidence, TTX is currently under clinical development for chemo-therapy-induced neuropathic pain and cancer-related pain.
1. Introduction
2. Role of TTX-Sensitive Voltage-Gated Sodium Channels (VGSCs) in Neuropathic and Cancer Pain
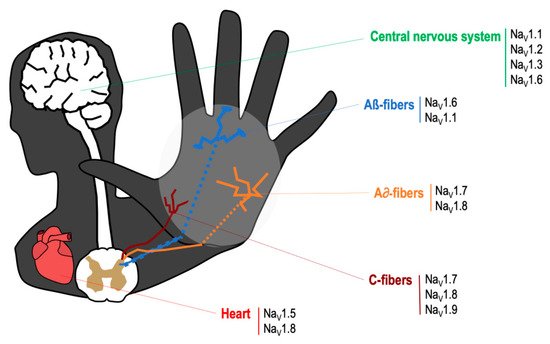
| Channel | Normal Localization [16] | Changes of Expression in Pain States |
|---|---|---|
| NaV1.1 | -CNS, PNS-Microglia | -Unclear after PNI in NP [16] |
| NaV1.2 | -CNS, very low expression in PNS-SC in lamina I/II | -Unclear after PNI in NP [16] |
| NaV1.3 | -Negligible in DRG (embryonic isoform)-SC in lamina I/II | Upregulated in DRG: CCI [21], PDN [22][23], SNI [24][25], SNL [26][27], traumatic nerve injury (human) [28] Upregulate in painful neuromas (human): [29] Trigeminal ganglion: trigeminal neuropathic pain [30][31][32] Upregulated in sciatic nerve: PDN [33] Upregulated in spinal cord: SCI [34] |
| NaV1.4 | -Skeletal muscle | |
| NaV1.6 | -Nodes of Ranvier-SC-PNS-Epidermal free nerve terminals-keratinocytes-Microglia | Upregulated in DRG: PDN [35], lumbar 5 ventral root transection [36], CINP (oxaliplatin) [37] Upregulated in sciatic nerve: CCI [38] Upregulated trigeminal ganglion: trigeminal neuropathic pain [30] |
| NaV1.7 | -PNS in all types of DRG neurons-SC-Epidermal free nerve terminals | Downregulated in DRG: SNL [39], SNI [24][25], traumatic nerve injury (human) [28] Upregulated in DRG: CCI [40], SNI [41], SNL [27], CINP (paclitaxel) [42], cancer-related pain (humans) [42], Herpesvirus quiescent infection [43], painful neuromas (human) [34] Upregulated in spinal cord: CINP (paclitaxel) [42] Upregulated trigeminal ganglion: trigeminal neuropathic pain [44] Upregulated in sciatic nerve: CCI [45] |
3. Effects of TTX in Preclinical Models of Neuropathic and Cancer Pain
3.1. Preclinical Studies on Neuropathic Pain
| Administration of TTX | TTX Doses | Effect (None, Moderate, Strong) | Pain Test | Pain Model | Reference |
|---|---|---|---|---|---|
| Sciatic nerve blockade | TTX osmotic pump | Strong | MA, TH | SNI and CCI | [61] |
| Topical DRG | 12.5–50 nM/5 µL | Strong (12.5–50 µg) | MA | SNL | [62] |
| Epidural | 25 nM/5 µL | Strong (25 µg) | MA | SNL | [62] |
| Topical median nerve | Gel pads with TTX | Strong | MA | CCI | [63] |
| Intraperitoneal | 25 nM/5 µL | None | MA | SNL | [62] |
| 8 µg | None | MA | CINP (vincristine) | [64] | |
| Subcutaneous | 1–6 µg | Strong | MA, TH, CA | CINP (paclitaxel) | [65] |
| 6 µg | Strong | MA | intraplantar capsaicin | [66] | |
| 0.3–6 µg | Strong (1–6 µg) | MA, TH | SNL | [57] | |
| Acute and subchronic TTX (1–6 µg) | Strong | MA, TH | CCI | [67] | |
| Acute and subchronic TTX (1–6 µg) | Moderate | MA, TH | CCI-intraorbital nerve | [67] | |
| 8 µg | Strong | MA, TH | burn-induced pain | [68] | |
| Intragastrical | 5–20 µg | Strong | MA, TH | Postherpetic Neuralgia (RTX) | [69] |
| Intramuscular | Acute and subchronic TTX (1–6 µg s.c.) | Strong | MA | Postherpetic Neuralgia (RTX) | [69] |
| 0.03–1 ug | Moderate | MH | CINP (oxaliplatin) | [70] | |
| Intrathecal | 10 µg | Strong | MA, TH | bone cancer pain | [71] |
3.2. Preclinical Studies on Cancer Pain
References
- Sá, K.N.; Moreira, L.; Baptista, A.F.; Yeng, L.T.; Teixeira, M.J.; Galhardoni, R.; de Andrade, D.C. Prevalence of chronic pain in developing countries: Systematic review and meta-analysis. Pain Rep. 2019, 4, e779.
- Jensen, T.S.; Baron, R.; Haanpää, M.; Kalso, E.; Loeser, J.D.; Rice, A.S.C.; Treede, R.D. A new definition of neuropathic pain. Pain 2011, 152, 2204–2205.
- Colloca, L.; Ludman, T.; Bouhassira, D.; Baron, R.; Dickenson, A.H.; Yarnitsky, D.; Freeman, R.; Truini, A.; Attal, N.; Finnerup, N.B.; et al. Neuropathic pain. Nat. Rev. Dis. Primers. 2017, 3, 17002.
- Watson, J.C.; Sandroni, P. Central Neuropathic Pain Syndromes. Mayo Clin. Proc. 2016, 91, 372–385.
- Costigan, M.; Scholz, J.; Woolf, C.J. Neuropathic pain: A maladaptive response of the nervous system to damage. Annu. Rev. Neurosci. 2009, 32, 1–32.
- Van Hecke, O.; Austin, S.K.; Khan, R.A.; Smith, B.H.; Torrance, N. Neuropathic pain in the general population: A systematic review of epidemiological studies. Pain 2014, 155, 654–662.
- Van den Beuken-van Everdingen, M.H.; Hochstenbach, L.M.; Joosten, E.A.; Tjan-Heijnen, V.C.; Janssen, D.J. Update on Prevalence of Pain in Patients with Cancer: Systematic Review and Meta-Analysis. J. Pain Symptom. Manag. 2016, 51, 1070–1090.
- Caraceni, A.; Shkodra, M. Cancer Pain Assessment and Classification. Cancers 2019, 11, 510.
- Finnerup, N.B.; Attal, N.; Haroutounian, S.; McNicol, E.; Baron, R.; Dworkin, R.H.; Gilron, I.; Haanpää, M.; Hansson, P.; Jensen, T.S.; et al. Pharmacotherapy for neuropathic pain in adults: A systematic review and meta-analysis. Lancet Neurol. 2015, 14, 162–173.
- Van den Beuken van Everdingen, M.H.J.; van Kuijk, S.M.J.; Janssen, D.J.A.; Joosten, E.A.J. Treatment of Pain in Cancer: Towards Personalised Medicine. Cancers 2018, 10, 502.
- Haumann, J.; Joosten, E.B.A.; Everdingen, M.H.J.V.D.B. Pain prevalence in cancer patients: Status quo or opportunities for improvement? Curr. Opin. Support Palliat. Care. 2017, 11, 99–104.
- Bates, D.; Schultheis, B.C.; Hanes, M.C.; Jolly, S.M.; Chakravarthy, K.V.; Deer, T.R.; Levy, R.M.; Hunter, C.W. A Comprehensive Algorithm for Management of Neuropathic Pain. Pain Med. 2019, 20 (Suppl. 1), S2–S12.
- Cooper, T.E.; Chen, J.; Wiffen, P.J.; Derry, S.; Carr, D.B.; Aldington, D.; Cole, P.; Moore, R.A. Morphine for chronic neuropathic pain in adults. Cochrane Database Syst. Rev. 2017, 5, CD011669.
- Van den Beuken-van Everdingen, M.H.; de Graeff, A.; Jongen, J.L.; Dijkstra, D.; Mostovaya, I.; Vissers, K.C.; National Guideline Working Group “Diagnosis Treatment of Cancer Pain”. Pharmacological Treatment of Pain in Cancer Patients: The Role of Adjuvant Analgesics, a Systematic Review. Pain Pract. 2017, 17, 409–419.
- Cardoso, F.C.; Lewis, R.J. Sodium channels and pain: From toxins to therapies. Br. J. Pharmacol. 2018, 175, 2138–2157.
- Nieto, F.R.; Cobos, E.J.; Tejada, M.Á.; Sánchez-Fernández, C.; González-Cano, R.; Cendán, C.M. Tetrodotoxin (TTX) as a therapeutic agent for pain. Mar. Drugs 2012, 10, 281–305.
- De Lera Ruiz, M.; Kraus, R.L. Voltage-Gated Sodium Channels: Structure, Function, Pharmacology, and Clinical Indications. J. Med. Chem. 2015, 58, 7093–7118.
- Fozzard, H.A.; Lipkind, G.M. The tetrodotoxin binding site is within the outer vestibule of the sodium channel. Mar. Drugs 2010, 8, 219–234.
- Latremoliere, A.; Woolf, C.J. Central sensitization: A generator of pain hypersensitivity by central neural plasticity. J. Pain 2009, 10, 895–926.
- Bennett, D.L.; Clark, A.J.; Huang, J.; Waxman, S.G.; Dib-Hajj, S.D. The Role of Voltage-Gated Sodium Channels in Pain Signaling. Physiol. Rev. 2019, 99, 1079–1151.
- Li, Y.; Zhang, X.; Fu, Z.; Zhou, Q. MicroRNA-212-3p Attenuates Neuropathic Pain via Targeting Sodium Voltage-gated Channel Alpha Subunit 3 (NaV 1.3). Curr. Neurovasc. Res. 2019, 16, 465–472.
- Cheng, K.I.; Wang, H.C.; Chuang, Y.T.; Chou, C.W.; Tu, H.P.; Yu, Y.C.; Chang, L.L.; Lai, C.S. Persistent mechanical allodynia positively correlates with an increase in activated microglia and increased P-p38 mitogen-activated protein kinase activation in streptozotocin-induced diabetic rats. Eur. J. Pain. 2014, 18, 162–173.
- Hong, S.; Morrow, T.J.; Paulson, P.E.; Isom, L.L.; Wiley, J.W. Early painful diabetic neuropathy is associated with differential changes in tetrodotoxin-sensitive and -resistant sodium channels in dorsal root ganglion neurons in the rat. J. Biol. Chem. 2004, 279, 29341–29350.
- Berta, T.; Poirot, O.; Pertin, M.; Ji, R.R.; Kellenberger, S.; Decosterd, I. Transcriptional and functional profiles of voltage-gated Na(+) channels in injured and non-injured DRG neurons in the SNI model of neuropathic pain. Mol. Cell Neurosci. 2008, 37, 196–208.
- Casals-Díaz, L.; Casas, C.; Navarro, X. Changes of voltage-gated sodium channels in sensory nerve regeneration and neuropathic pain models. Restor. Neurol. Neurosci. 2015, 33, 321–334.
- Black, J.A.; Cummins, T.R.; Plumpton, C.; Chen, Y.H.; Hormuzdiar, W.; Clare, J.J.; Waxman, S.G. Upregulation of a silent sodium channel after peripheral, but not central, nerve injury in DRG neurons. J. Neurophysiol. 1999, 82, 2776–2785.
- Lin, C.R.; Chen, K.H.; Yang, C.H.; Huang, H.W.; Sheen-Chen, S.M. Intrathecal miR-183 delivery suppresses mechanical allodynia in mononeuropathic rats. Eur. J. Neurosci. 2014, 39, 1682–1689.
- Coward, K.; Aitken, A.; Powell, A.; Plumpton, C.; Birch, R.; Tate, S.; Bountra, C.; Anand, P. Plasticity of TTX-sensitive sodium channels PN1 and brain III in injured human nerves. Neuroreport 2001, 12, 495–500.
- Black, J.A.; Nikolajsen, L.; Kroner, K.; Jensen, T.S.; Waxman, S.G. Multiple sodium channel isoforms and mitogen-activated protein kinases are present in painful human neuromas. Ann. Neurol. 2008, 64, 644–653.
- Liu, M.X.; Zhong, J.; Xia, L.; Dou, N.N.; Li, S.T. IL-6 contributes to Nav1.3 up-regulation in trigeminal nerve following chronic constriction injury. Neurol. Res. 2020, 42, 504–514.
- Liu, M.; Zhong, J.; Xia, L.; Dou, N.; Li, S. The expression of voltage-gated sodium channels in trigeminal nerve following chronic constriction injury in rats. Int. J. Neurosci. 2019, 129, 955–962.
- Yoon, J.H.; Son, J.Y.; Kim, M.J.; Kang, S.H.; Ju, J.S.; Bae, Y.C.; Ahn, D.K. Preemptive application of QX-314 attenuates trigeminal neuropathic mechanical allodynia in rats. Korean J. Physiol. Pharmacol. 2018, 22, 331–341.
- Aghdam, A.M.; Shahabi, P.; Karimi-Sales, E.; Ghiasi, R.; Sadigh-Eteghad, S.; Mahmoudi, J.; Alipour, M.R. Swimming Exercise Induced Reversed Expression of miR-96 and Its Target Gene NaV1.3 in Diabetic Peripheral Neuropathy in Rats. Chin. J. Physiol. 2018, 61, 124–129.
- Liu, J.; Wu, Y. Electro-acupuncture-modulated miR-214 prevents neuronal apoptosis by targeting Bax and inhibits sodium channel Nav1.3 expression in rats after spinal cord injury. Biomed. Pharmacother. 2017, 89, 1125–1135.
- Ren, Y.S.; Qian, N.S.; Tang, Y.; Liao, Y.H.; Yang, Y.L.; Dou, K.F.; Toi, M. Sodium channel Nav1.6 is up-regulated in the dorsal root ganglia in a mouse model of type 2 diabetes. Brain Res. Bull. 2012, 87, 244–249.
- Ding, H.H.; Zhang, S.B.; Lv, Y.Y.; Ma, C.; Liu, M.; Zhang, K.B.; Ruan, X.C.; Wei, J.Y.; Xin, W.J.; Wu, S.L. TNF-α/STAT3 pathway epigenetically upregulates Nav1.6 expression in DRG and contributes to neuropathic pain induced by L5-VRT. J. Neuroinflamm. 2019, 16, 29.
- Li, L.; Shao, J.; Wang, J.; Liu, Y.; Zhang, Y.; Zhang, M.; Zhang, J.; Ren, X.; Su, S.; Li, Y.; et al. MiR-30b-5p attenuates oxaliplatin-induced peripheral neuropathic pain through the voltage-gated sodium channel Nav1.6 in rats. Neuropharmacology 2019, 153, 111–120.
- Tseng, T.J.; Hsieh, Y.L.; Ko, M.H.; Hsieh, S.T. Redistribution of voltage-gated sodium channels after nerve decompression contributes to relieve neuropathic pain in chronic constriction injury. Brain Res. 2014, 1589, 15–25.
- Li, M.; Zhang, S.J.; Yang, L.; Fang, X.L.; Hu, H.F.; Zhao, M.Y.; Li, L.; Guo, Y.Y.; Shao, J.P. Voltage-gated sodium channel 1.7 expression decreases in dorsal root ganglia in a spinal nerve ligation neuropathic pain model. Kaohsiung J. Med. Sci. 2019, 35, 493–500.
- Jia, Q.; Dong, W.; Zhang, L.; Yang, X. Activating Sirt1 by resveratrol suppresses Nav1.7 expression in DRG through miR-182 and alleviates neuropathic pain in rats. Channels 2020, 14, 69–78.
- Tian, J.J.; Tan, C.Y.; Chen, Q.Y.; Zhou, Y.; Qu, Z.W.; Zhang, M.; Ma, K.T.; Shi, W.Y.; Li, L.; Si, J.Q. Upregulation of Nav1.7 by endogenous hydrogen sulfide contributes to maintenance of neuropathic pain. Int. J. Mol. Med. 2020, 46, 782–794.
- Li, Y.; North, R.Y.; Rhines, L.D.; Tatsui, C.E.; Rao, G.; Edwards, D.D.; Cassidy, R.M.; Harrison, D.S.; Johansson, C.A.; Zhang, H.; et al. DRG Voltage-Gated Sodium Channel 1.7 Is Upregulated in Paclitaxel-Induced Neuropathy in Rats and in Humans with Neuropathic Pain. J. Neurosci. 2018, 38, 1124–1136.
- Zhang, Q.; Martin-Caraballo, M.; Hsia, S.V. Modulation of Voltage-Gated Sodium Channel Activity in Human Dorsal Root Ganglion Neurons by Herpesvirus Quiescent Infection. J. Virol. 2020, 94, e01823-19.
- Yang, K.Y.; Kim, M.J.; Ju, J.S.; Park, S.K.; Lee, C.G.; Kim, S.T.; Bae, Y.C.; Ahn, D.K. Antinociceptive Effects of Botulinum Toxin Type A on Trigeminal Neuropathic Pain. J. Dent. Res. 2016, 95, 1183–1190.
- Stevens, A.M.; Liu, L.; Bertovich, D.; Janjic, J.M.; Pollock, J.A. Differential Expression of Neuroinflammatory mRNAs in the Rat Sciatic Nerve Following Chronic Constriction Injury and Pain-Relieving Nanoemulsion NSAID Delivery to Infiltrating Macrophages. Int. J. Mol. Sci. 2019, 20, 5269.
- Lindia, J.A.; Köhler, M.G.; Martin, W.J.; Abbadie, C. Relationship between sodium channel NaV1.3 expression and neuropathic pain behavior in rats. Pain 2005, 117, 145–153.
- Nassar, M.A.; Baker, M.D.; Levato, A.; Ingram, R.; Mallucci, G.; McMahon, S.B.; Wood, J.N. Nerve injury induces robust allodynia and ectopic discharges in Nav1.3 null mutant mice. Mol. Pain 2006, 2, 33.
- Nassar, M.A.; Levato, A.; Stirling, L.C.; Wood, J.N. Neuropathic pain develops normally in mice lacking both Na(v)1.7 and Na(v)1.8. Mol. Pain 2005, 1, 24.
- Chang, W.; Berta, T.; Kim, Y.H.; Lee, S.; Lee, S.Y.; Ji, R.R. Expression and Role of Voltage-Gated Sodium Channels in Human Dorsal Root Ganglion Neurons with Special Focus on Nav1.7, Species Differences, and Regulation by Paclitaxel. Neurosci. Bull. 2018, 34, 4–12.
- Waxman, S.G.; Merkies, I.S.J.; Gerrits, M.M.; Dib-Hajj, S.D.; Lauria, G.; Cox, J.J.; Wood, J.N.; Woods, C.G.; Drenth, J.P.H.; Faber, C.G. Sodium channel genes in pain-related disorders: Phenotype-genotype associations and recommendations for clinical use. Lancet Neurol. 2014, 13, 1152–1160.
- Xie, W.; Strong, J.A.; Zhang, J.M. Local knockdown of the NaV1.6 sodium channel reduces pain behaviors, sensory neuron excitability, and sympathetic sprouting in rat models of neuropathic pain. Neuroscience 2015, 291, 317–330.
- Chen, L.; Huang, J.; Zhao, P.; Persson, A.K.; Dib-Hajj, F.B.; Cheng, X.; Tan, A.; Waxman, S.G.; Dib-Hajj, S.D. Conditional knockout of NaV1.6 in adult mice ameliorates neuropathic pain. Sci. Rep. 2018, 8, 3845.
- Waxman, S.G.; Dib-Hajj, S.; Cummins, T.R.; Black, J.A. Sodium channels and pain. Proc. Natl. Acad. Sci. USA 1999, 96, 7635–7639.
- Dib-Hajj, S.D.; Fjell, J.; Cummins, T.R.; Zheng, Z.; Fried, K.; LaMotte, R.; Black, J.A.; Waxman, S.G. Plasticity of sodium channel expression in DRG neurons in the chronic constriction injury model of neuropathic pain. Pain 1999, 83, 591–600.
- Leo, S.; D’Hooge, R.; Meert, T. Exploring the role of nociceptor-specific sodium channels in pain transmission using Nav1.8 and Nav1.9 knockout mice. Behav. Brain Res. 2010, 208, 149–157.
- Zimmer, T.; Haufe, V.; Blechschmidt, S. Voltage-gated sodium channels in the mammalian heart. Glob. Cardiol. Sci. Pract. 2014, 2014, 449–463.
- Marcil, J.; Walczak, J.S.; Guindon, J.; Ngoc, A.H.; Lu, S.; Beaulieu, P. Antinociceptive effects of tetrodotoxin (TTX) in rodents. Br. J. Anaesth. 2006, 96, 761–768.
- Hong, B.; He, J.; Sun, J.; Le, Q.; Bai, K.; Mou, Y.; Zhang, Y.; Chen, W.; Huang, W. Analgesia Effect of Enteric Sustained-Release Tetrodotoxin Pellets in the Rat. Pharmaceutics 2020, 12, 32.
- Alguacil, L.F.; Pérez-García, C.; Salas, E.; González-Martín, C.; Castillo, C.; Polanco, M.J.; Herradón, G.; Morales, L. Subcutaneous tetrodotoxin and inflammatory pain. Br. J. Anaesth. 2008, 100, 729–730.
- González-Cano, R.; Tejada, M.Á.; Artacho-Cordón, A.; Nieto, F.R.; Entrena, J.M.; Wood, J.N.; Cendán, C.M. Effects of Tetrodotoxin in Mouse Models of Visceral Pain. Mar. Drugs 2017, 15, 188.
- Xie, W.; Strong, J.A.; Meij, J.T.A.; Zhang, J.M.; Yu, L. Neuropathic pain: Early spontaneous afferent activity is the trigger. Pain 2005, 116, 243–256.
- Lyu, Y.S.; Park, S.K.; Chung, K.; Chung, J.M. Low dose of tetrodotoxin reduces neuropathic pain behaviors in an animal model. Brain Res. 2000, 871, 98–103.
- Chen, J.J.; Lue, J.H.; Lin, L.H.; Huang, C.T.; Chiang, R.P.; Chen, C.L.; Tsai, Y.J. Effects of pre-emptive drug treatment on astrocyte activation in the cuneate nucleus following rat median nerve injury. Pain 2010, 148, 158–166.
- Nozaki-Taguchi, N.; Chaplan, S.R.; Higuera, E.S.; Ajakwe, R.C.; Yaksh, T.L. Vincristine-induced allodynia in the rat. Pain 2001, 93, 69–76.
- Nieto, F.R.; Entrena, J.M.; Cendán, C.M.; Del Pozo, E.; Vela, J.M.; Baeyens, J.M. Tetrodotoxin inhibits the development and expression of neuropathic pain induced by paclitaxel in mice. Pain 2008, 137, 520–531.
- Entrena, J.M.; Cobos, E.J.; Nieto, F.R.; Cendán, C.M.; Gris, G.; Del Pozo, E.; Zamanillo, D.; Baeyens, J.M. Sigma-1 receptors are essential for capsaicin-induced mechanical hypersensitivity: Studies with selective sigma-1 ligands and sigma-1 knockout mice. Pain 2009, 143, 252–261.
- Kayser, V.; Viguier, F.; Ioannidi, M.; Bernard, J.F.; Latrémolière, A.; Michot, B.; Vela, J.M.; Buschmann, H.; Hamon, M.; Bourgoin, S. Differential anti-neuropathic pain effects of tetrodotoxin in sciatic nerve-versus infraorbital nerve-ligated rats—Behavioral, pharmacological and immunohistochemical investigations. Neuropharmacology 2010, 58, 474–487.
- Salas, M.M.; McIntyre, M.K.; Petz, L.N.; Korz, W.; Wong, D.; Clifford, J.L. Tetrodotoxin suppresses thermal hyperalgesia and mechanical allodynia in a rat full thickness thermal injury pain model. Neurosci. Lett. 2015, 607, 108–113.
- Hong, B.; Sun, J.; Zheng, H.; Le, Q.; Wang, C.; Bai, K.; He, J.; He, H.; Dong, Y. Effect of Tetrodotoxin Pellets in a Rat Model of Postherpetic Neuralgia. Mar. Drugs 2018, 16, 195.
- Alvarez, P.; Levine, J.D. Antihyperalgesic effect of tetrodotoxin in rat models of persistent muscle pain. Neuroscience 2015, 311, 499–507.
- Zheng, Q.; Fang, D.; Cai, J.; Wan, Y.; Han, J.S.; Xing, G.G. Enhanced excitability of small dorsal root ganglion neurons in rats with bone cancer pain. Mol. Pain 2018, 8, 24.
- Colvin, L.A. Chemotherapy-induced peripheral neuropathy: Where are we now? Pain 2019, 160 (Suppl. 1), S1–S10.
- Klein, T.; Magerl, W.; Rolke, R.; Treede, R.D. Human surrogate models of neuropathic pain. Pain 2005, 115, 227–233.
- Bennett, M.I.; Kaasa, S.; Barke, A.; Korwisi, B.; Rief, W.; Treede, R.D.; IASP Taskforce for the Classification of Chronic Pain. The IASP classification of chronic pain for ICD-11: Chronic cancer-related pain. Pain 2019, 160, 38–44.




