Liver resection for malignant tumors should respect oncological margins while ensuring safety and improving the quality of life, therefore tumor staging, underlying liver disease and performance status should all be attentively assessed in the decision process. The concept of parenchyma-sparing liver surgery is nowadays used as an alternative to major hepatectomies to address deeply located lesions with intricate topography by means of complex multiplanar parenchyma-sparing liver resections, preferably under the guidance of intraoperative ultrasound. Regenerative liver surgery evolved as a liver growth induction method to increase resectability by stimulating the hypertrophy of the parenchyma intended to remain after resection (referred to as future liver remnant), achievable by portal vein embolization and liver venous deprivation as interventional approaches, and portal vein ligation and associating liver partition and portal vein ligation for staged hepatectomy as surgical techniques. Interestingly, although both strategies have the same conceptual origin, they eventually became caught in the never-ending parenchyma-sparing liver surgery vs. regenerative liver surgery debate. However, these strategies are both valid and must both be mastered and used to increase resectability.
- liver resection
- therapeutic options in liver surgery
- regenerative liver surgery
- parenchyma- sparing liver resection
1. Parenchyma-Sparing Liver Surgery
- -
-
systematic extended right posterior sectionectomy, as an alternative to right hemi-hepatectomy [23]—segment (S) 6–7 resection partially extended to S5 and/or S8 with right HV division; middle HV branches supply outflow of preserved S5 and/or S8;
- -
-
mini-upper transversal hepatectomy, as an alternative to right hemi-hepatectomy—S7–8 anatomic or limited resection with right HV division; inferior right HV [24], middle HV branches or distal CVs between right and middle HVs supply outflow of S5–6 [7];
- -
-
right upper transversal hepatectomy, as an alternative to right extended hemi-hepatectomy [25]—S7–S8–S4 superior anatomic or limited resection with right and middle HV division; inferior right HV and/or distal CVs between right, middle and left HVs supply outflow of S4 inferior–5–6;
- -
-
left upper transversal hepatectomy, as an alternative to left extended hemi-hepatectomy [7]—S2–S4 superior or S2–S4 superior–S8 anatomic or limited resection with left HV or left and middle HV division; distal CVs between left, middle and/or right HV supply outflow of S3–4 inferior–5;
- -
-
total upper transversal hepatectomy [7]—S2–S4 superior–S7–S8 anatomic or limited resection with right, middle and left HV division given the existence of an inferior right HV and CVs between hepatic HVs stumps, that provide outflow of S3–S4 inferior–S5–S6;
- -
-
mini-mesohepatectomy, as an alternative to central hepatectomy [26]—S4 superior–S8 anatomic or limited resection with middle HV division; distal CVs between middle HV and right and left HVs supply outflow of S5–S4 inferior;
- -
-
liver tunnel, as an alternative to central hepatectomy plus S1 segmentectomy [7][27]—S8 anatomic or limited resection with complete S1 removal;
- -
-
liver tunnel extended to segment 4 superior, as an alternative to central hepatectomy plus S1 segmentectomy [7][27]—S4 superior–S8 anatomic or limited resection with complete S1 removal and middle HV division; distal CVs between middle HV and right and left HVs supply outflow for S5–S4 inferior;
- -
-
systematic limited central, as an alternative to central hepatectomy—sparing the portal pedicle (P) for S8 dorsal and some of P4 and/or P5 pedicles (depending on tumor location). IOUS guides the right transection plane along the P8 dorsal, intersecting the P8 ventral and as few P5 pedicles as possible. The left transection plane is settled relative to tumor position between Cantlie’s line and the falciform ligament [28];
- -
-
left anterior sectorectomy, as an alternative to left hepatectomy for lesions invading the distal part of the umbilical portion of the left portal vein–resection of S3 and S4 inferior, while preserving the P2 and P4 superior.
2. Regenerative Liver Surgery
Regenerative surgery (RS) evolved as a method to increase resectability by stimulating the hypertrophy of the parenchyma intended to remain after resection, which is referred to as future liver remnant (FLR) [29]. This liver growth induction can be achieved by portal vein embolization (PVE) and liver venous deprivation (LVD) as interventional approaches, and portal vein ligation (PVL) and associating liver partition and portal vein ligation for staged hepatectomy (ALPPS) as surgical techniques.
PVE was conceived by Makuuchi et al. in 1990 as a tool to induce hypertrophy of the FLR and decrease the risk of liver failure after major hepatectomy, enabling major anatomical LR, which would otherwise not be feasible [30]. The mechanism behind this approach is based on the redirection of the portal flow, that stimulates the contralateral hypertrophy. PVE is associated with a low morbidity and mortality. However, the growth of FLR is limited to a volume by 40% at best, for most cases within a period of around 2 months [31]. This may lead to insufficient FLR and/or tumor progression while waiting for hypertrophy. PVL (open or minimally invasive surgery) is a feasible alternative to PVE. For patients undergoing PVE, major hepatectomy becomes feasible in 2/3 of cases with a similar overall survival to those without PVE [32]. Chemotherapy after PVE decreases the tumor progression rate and has not been shown to decrease liver hypertrophy. In about 1/3 of patients, PVE fails and leads to canceling of the planned LR (drop-out rate) [33].
Recently, liver venous deprivation (LVD), consisting of embolization of both the PV and one or two HVs of the hemi-liver, has been proposed as a promising way for improved regeneration (1–2 weeks) . Several studies comparing LVD to PVE reported improved FLR volume growth following LVD [34][35][36], as well as better FLR functional regeneration [37]. In particular, one study has shown a more than 75% increase in the kinetic growth rate of the FLR after LVD compared to PVE . Moreover, a 54% functional increase in the FLR 7 days after LVD has been reported [37]. However, literature data on LVD of a cirrhotic liver are lacking [38].
The two-stage hepatectomy (TSH) was introduced in 2000 as two successive surgical steps for removing multiple bilobar tumors that cannot be removed by a sole hepatectomy [39]. Usually, the response to neoadjuvant chemotherapy was used to select candi- dates with favorable tumor biology. TSH can be used by itself or combined with PVE or portal vein ligation (PVL) [40]. It usually has resection rates of up to 70–75%; the main reason for non-completion is disease progression between the two stages (around 90% of cases) [39][41]. The postoperative morbidity rate is around 20% after the 1st stage and 40% after the 2nd stage, with an overall mortality below 5% [42].
In 2012, Schnitzbauer et al. proposed combining PVL with in situ liver partition to obtain rapid FLR hypertrophy (in 7–10 days) as a new strategy to increase resectability [43], which was subsequently termed ALPPS [44]. One mechanism behind this technique is thought to trigger an inflammatory response that induces a growth rate of 22–35 mL daily, significantly superior to PVE (3–5 mL daily) [45]. However, this volume growth does not automatically equal an increase in liver function [46]. This strategy results in a FLR increase of up to 80% and above (compared to 40% in PVE/PVL), while shortening the interstage period to 1–2 weeks [47]. Moreover, ALPPS enables resection rates to increase to more than 90% [48][49][50], now being feasible even when using a minimally invasive approach [51].
Short-term results after ALPPS, that were initially a major concern, have been con- tinuously improved over time, now reaching 90-day mortality rates below 5% [61] and a relatively low major morbidity (21%) in high-volume centers.
To further increase resectability while reducing morbidity, scholars proposed a new techni- cal variant of ALPPS—parenchyma-sparing ALPPS (psALPPS)—that involves shifting the transection plane through segment 4 using IOUS guidance, preserving part of this segment along with the left lateral section [66]. Besides avoiding S4 necrosis, that is a source of complication when performing conventional ALPPS, a significant advantage of psALPPS lies in preventing major bile leaks at the transection surface by avoiding complete exclusion of S4 from the biliary system (as in conventional ALPPS). Parenchyma-sparing ALPPS offers the advantage of maximizing FLR while simultaneously reducing ischemic injury of S4 compared to conventional ALPPS (Figures 1 and 2). Moreover, when compared to stan- dard ALPPS, partitioning through segment 4, away from the umbilical portion of the left portal pedicle, protects against potential injuries of the vascular and biliary structures for segments 2 and 3. This new technical variant also embeds some of the main modifications already proposed, such as partial ALPPS, avoiding the transection beyond the middle HV, and delayed ALPSS [62]. It also adapts the concept of avoiding hilar dissection by adopt- ing a minimal hilar dissection (right side approach only) [62]. Using an extra-Glissonean approach to complete the hepatectomy during the second step further increases safety by avoiding re-dissection of the liver hilum.
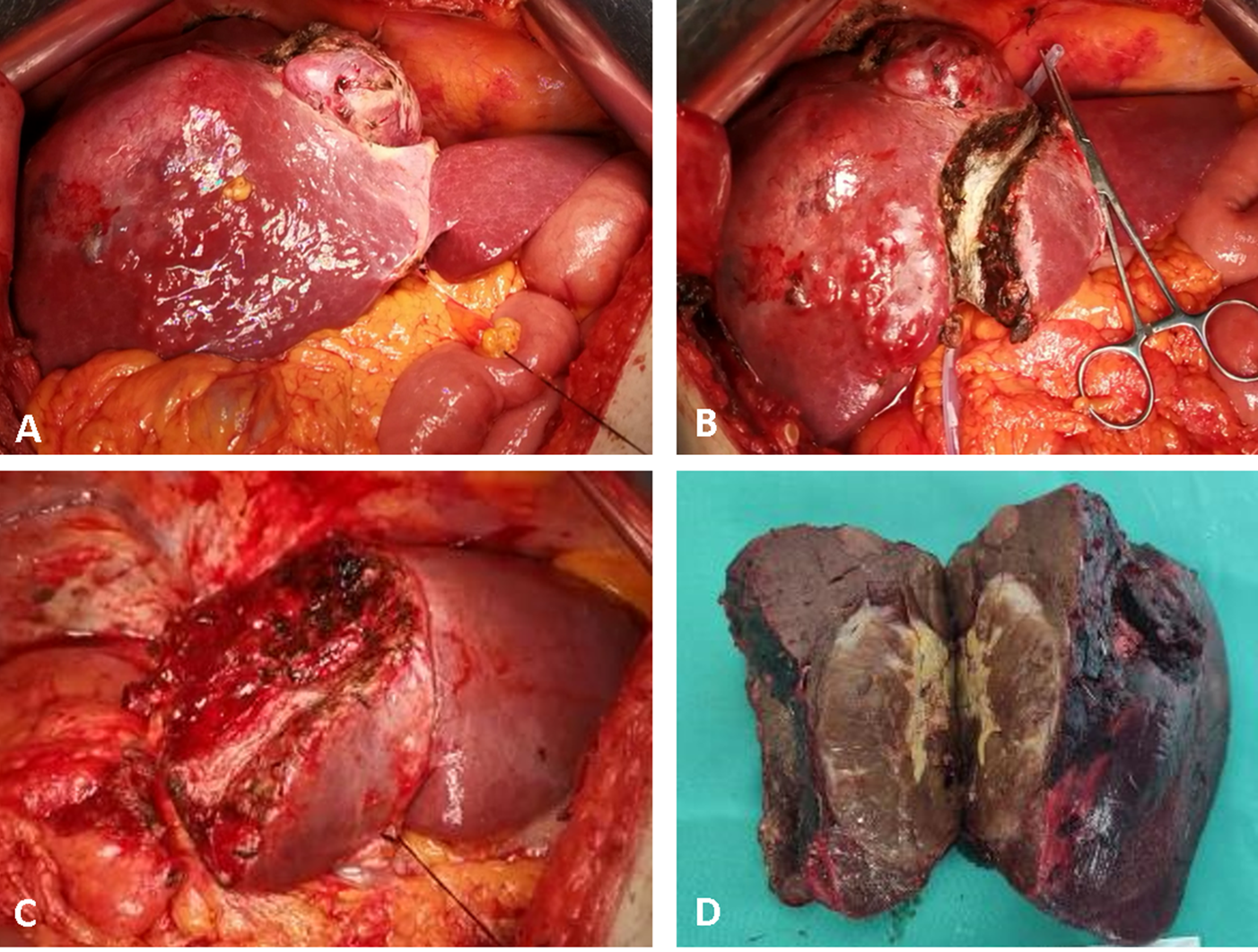
Figure 1. Intraoperative aspects of parenchymal sparing ALPPS in a 67-year-old male patient, for a large HCC located in segments 4, 5, 6, 7 and 8, with satellites in segment 4, on HBV chronic hepatitis. Stage 1: (A) intraoperative aspect at exploration; (B) ultrasound-guided partitioning of the liver through segment 4, adding the non-tumoral parenchyma of segment 4 to the FLR. Stage 2 after an interstage interval of 14 days; (C) remnant liver after completion of right hemi-hepatectomy non-anatomically extended to segment 4; (D) surgical specimen. No intraoperative adverse events were encountered during both operations, and only minor ascites after stage 2 were recorded as complications.
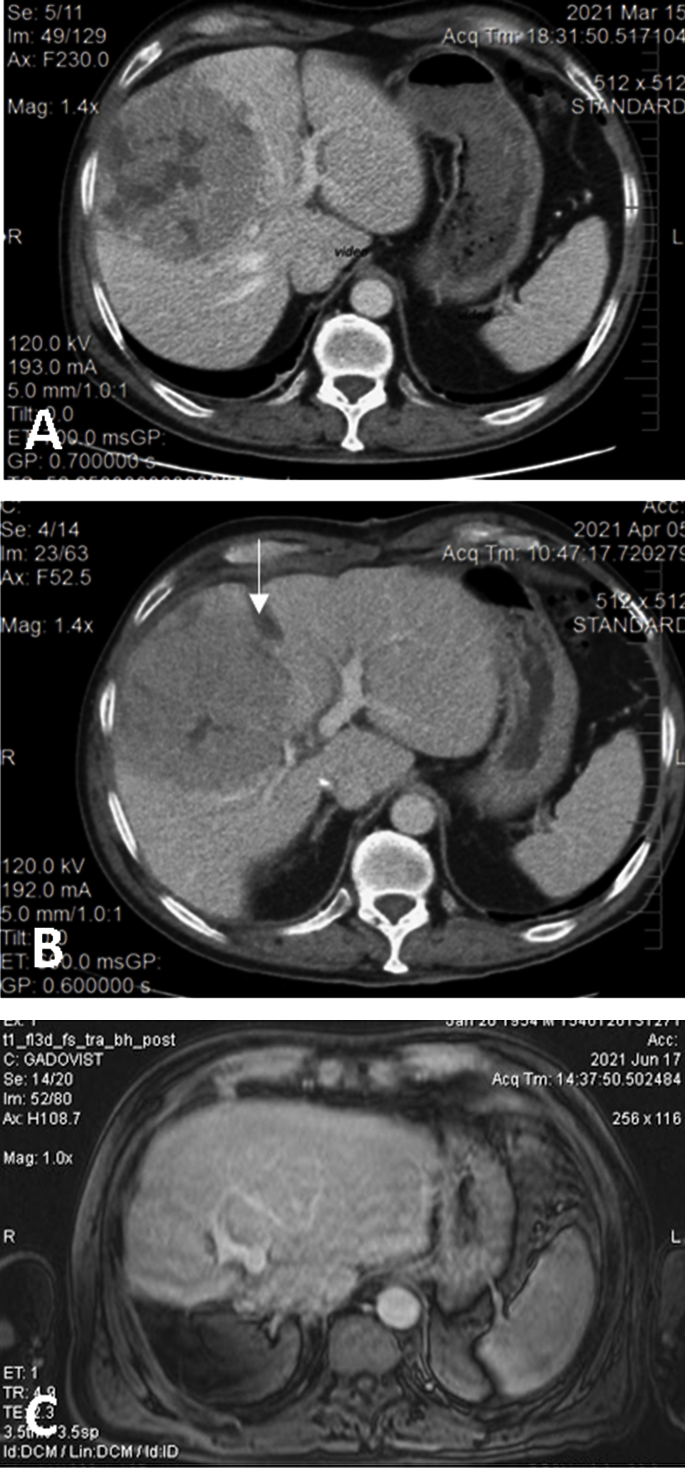
Figure 2. (A). Preoperative CT showing the large HCC located in segments 5 and 8 with extension to segments 6, 7, 4, compressing the middle hepatic vein; volumetry: volume of segments 2 and 3, 16.8% of total functional liver volume, volume of FLR 27.8%. (B) Interstage CT showing the liver partitioning, absence of contrast in the right portal vein (due to ligation), and sufficient growth of FLR (38.5% of total functional liver volume). (C) Postoperative CT with well-perfused, non-dilated bile ducts, and tumor-free remnant liver.
3. Parenchyma-Sparing vs. Regenerative Liver Surgery
3.1. Colorectal Liver Metastases
Downstaging of initially unresectable CRLM may be achieved using novel cytotoxic and biologic systemic therapy to achieve curative surgery with best results when carried out in tertiary referral centers with an expert multidisciplinary team [64][65]. Compared with non-PSS, perioperative outcomes are better in the case of PSS, with PSS also being associated with satisfactory oncological results. By sparing liver parenchyma, PSS allows repeat hepatectomy in the likely event of liver recurrence. PSS also showed beneficial OS and RFS rates [9]. Matsumura et al. showed that, in advanced CRLM, PS LR was not associated with more positive surgical margins or local recurrence when compared with major LR [66]. Mise et al. showed that PS LR impacts OS, RFS or liver-only RFS by allowing repeat LR, with similar perioperative morbimortality [67], while not increasing the local recurrence risk [16]. Neither survival, recurrence risk or site are influenced by the extent of a negative surgical margin [68]. In the setting of modern chemotherapy, Adam et al. demonstrated that R1 margins may yet be linked with similar OS [69]. The R1vasc approach is safe concerning oncological results in CRLM [70], yet hepatectomy en bloc with a vascular element is the preferable approach, given the confirmed true vascular invasion [71]. When compared to major LR, PS LR was linked to lower overall morbidity, fewer major complications and a shorter hospital stay while no significant differences were observed for postoperative liver insufficiency and positive resection margins [72]. The tumor burden (with a score ≥4.5) is related to a higher rate of positive resection margins both in major and PS LR [72]. Independently of tumor burden, the 5-year OS and RFS were similar for PS LR [72]. RS, such as PVE [73][74] and TSH [43][75], has made it possible for more patients with colorectal liver metastases (CRLMs) to benefit from curative LR [76][77]. Unresectable CRLMs are often related to a difficulty in completely removing all lesions and preserving as FLR at least two contiguous functional segments [78].3.2. Hepatocellular Carcinoma
There are several curative options for the treatment of hepatocellular carcinoma (HCC), e.g., usually intraoperative or percutaneous tumor ablation (for tumors <3 cm), hepatectomy and liver transplantation, particularly in the setting of advanced cirrhosis [79]. Targets of LR are improving recovery, reducing postoperative morbidity and ensuring a satisfactory function of a frequently cirrhotic liver [79]. R0 resection is no longer considered an absolute requirement, as R1 resection has become an accepted option for encapsulated HCC in contact with major vascular and biliary structures [80]. Hasegawa et al. proved that anatomical LR is oncologically superior to non-anatomical LR [81]. As anatomical LR included segmentectomies and sub-segmentectomies [81], it underlined that this strategy is also a parenchyma-sparing one. However, other studies showed no differences regarding oncological outcomes between anatomical and non-anatomical LR [82][83][84]. PS LRs are especially beneficial in case of cirrhosis. When compared to the right posterior sectionectomy, the right hepatectomy for HCC was more frequently associated with liver failure (9.4% vs. 2%), yet with similar 5-year OS (83% vs. 76%) and RFS (52% for both) [85]. Therefore, the right posterior sectionectomy is to be selected over the right hepatectomy in cases that allow complete tumor resection [79]. When the right posterior sectionectomy cannot ensure resectability, the systematic extended right posterior sectionectomy (SERPS) has been proved to be a feasible alternative to the right hepatectomy [23]. Liver failure risk and mortality are also increased due to an insufficient FLR in the setting of cirrhosis in patients with central tumors, for which extended right or left hepatectomy has been the standard recommended approach. An alternative is central or mesohepatectomy (S4–5–8 and middle HV resection), that preserves more parenchyma while ensuring complete tumor resection [79]. It has been shown that following a central hepatectomy, postoperative bilirubin levels >4 mg/dL are notably less common (2% vs. 39%) [86] and the liver failure risk is lower (1.7% vs. 10.6%) [87] than after an extended right/left hepatectomy; yet, the 5-year OS and RFS are similar [87]. As it is still a major hepatectomy, central hepatectomy is, however, to be avoided whenever possible. A feasible alternative is the systematic limited central hepatectomy [28]. The current practice involves performing parenchyma sparing, preferably anatomic LR, such as sub-segmentectomies, segmentectomies, bisegmentectomies, right posterior sectionectomy or central hepatectomy [79]. In case this is not feasible, non-anatomic LR such as SERPS [23] or systematic limited central hepatectomy [28] are options to be deployed whenever possible to avoid a major hepatectomy.3.3. Intrahepatic Cholangiocarcinoma
Anatomic and non-anatomic LRs are associated with comparable intraoperative bleeding and morbidity, but liver failure occurs more often following anatomic LR [88]. Non-anatomic LR has been linked with a higher rate of positive surgical margins, but this seemed to not impact the OS or the DFS [88]. However, it has also been shown that negative surgical margins are associated with a beneficial OS and PFS following resection for ICC [89]. Positive margins have been linked to inferior results in the long run and the OS and DFS proved to become gradually worse for a margin width >1 cm [90]. In this sense, R1 vascular resection is not recommended [91]. Non-anatomic LR has proved to be non-inferior to anatomic LR in terms of survival in the case of solitary ICC not invading contiguous organs or extrahepatic metastases, which shows that these patients, particularly in the context of cirrhosis, could benefit more from a non-anatomic hepatectomy, given the lower risk of liver failure [88].3.4. Hilar Cholangiocarcinoma
Resectability of hilar cholangiocarcinoma is mainly dictated by the intrahepatic tumor extension. Bismuth–Corlette type III tumors are usually resectable by performing a hemi-hepatectomy that can be extended, while Bismuth–Corlette type IV tumors are surgically manageable only in selected patients. Other aspects that determine resectability are the tumor invasion of the portal vein and/or hepatic artery, FLR in terms of both volume and functional status as well as its competent biliary drainage and PVE feasibility. Regardless of resection extent, en bloc S1 resection is advised [92]. Left liver resections are more beneficial as they allow sparing the right liver, hence left trisectionectomy is a feasible option for cases of Klatskin IV tumors that do not involve the right hepatic artery [92]. Mesohepatectomy should be taken into consideration when the bile ducts of S6–7 and S2–3 are not affected, and the portal and arterial branches of these sparable segments can be preserved [92]. Strategies to maximize liver functional status using PSS to reduce the extent of LR in Klatskin tumor have resulted in a lower overall mortality [92].3.5. Other Focal Liver Lesions
Indications for surgical resection of GIST include limited disease, progression refrac- tory to TKI and locally advanced or previously unresectable tumors that manifest favorable response to neoadjuvant therapy with TKI [93]. In the case of NELM, LR is the treatment of choice whenever feasible, since patient outcomes after resection have been reported to be favorable compared to those with unresectable tumors [120]. Repeat hepatectomy, if feasible, can be a good option for intra- hepatic recurrence and can provide long-term survival [94]. Regarding hemangiomas, as complications are rare, observation is justified in the absence of symptoms. LR is indicated in patients with abdominal (mechanical) complaints or complications or when diagnosis remains inconclusive. Enucleation is the preferred surgical method according to existing literature [95]. ALPPS may be also deployed in neuroendocrine liver metastases (NELMs) [96], and other rare indications, such as lymphoma [97]. In NELM, 2-year overall survival rates of 95.2% were reported [96].4. Conclusions
References
- Vauthey, J.N.; Baer, H.U.; Guastella, T.; Blumgart, L.H. Comparison of outcome between extended and nonextended liver resections for neoplasms. Surgery 1993, 114, 968–975.
- Gold, J.S.; Are, C.; Kornprat, P.; Jarnagin, W.R.; Gönen, M.; Fong, Y.; DeMatteo, R.P.; Blumgart, L.H.; D’Angelica, M. Increased use of parenchymal-sparing surgery for bilateral liver metastases from colorectal cancer is associated with improved mortality without change in oncologic outcome: Trends in treatment over time in 440 patients. Ann. Surg. 2008, 247, 109–117.
- Chouillard, E.; Cherqui, D.; Tayar, C.; Brunetti, F.; Fagniez, P.L. Anatomical bi- and trisegmentectomies as alternatives to extensive liver resections. Ann. Surg. 2003, 238, 29–34.
- Torzilli, G.; Montorsi, M.; Del Fabbro, D.; Palmisano, A.; Donadon, M.; Makuuchi, M. Ultrasonographically guided surgical approach to liver tumours involving the hepatic veins close to the caval confluence. Br. J. Surg. 2006, 93, 1238–1246.
- Torzilli, G.; Garancini, M.; Donadon, M.; Cimino, M.; Procopio, F.; Montorsi, M. Intraoperative ultrasonographic detection of communicating veins between adjacent hepatic veins during hepatectomy for tumours at the hepatocaval confluence. Br. J. Surg. 2010, 97, 1867–1873.
- Torzilli, G.; Procopio, F.; Costa, G. Adjuncts to hepatic resection—Ultrasound and emerging guidance systems. In Blumgart’s Surgery of the Liver, Pancreas, and Biliary Tract, 6th ed.; Jarnagin, W.R., Ed.; Elsevier Saunders: Philadelphia, PA, USA, 2016.
- Torzilli, G. Ultrasound-Guided Liver Surgery: An Atlas, 1st ed.; Springer: Milan, Italy, 2014.
- Torzilli, G.; McCormack, L.; Pawlik, T. Parenchyma-sparing liver resections. Int. J. Surg. 2020, 82, 192–197.
- Deng, G.; Li, H.; Jia, G.Q.; Fang, D.; Tang, Y.Y.; Xie, J.; Chen, K.F.; Chen, Z.Y. Parenchymal-sparing versus extended hepatectomy for colorectal liver metastases: A systematic review and meta-analysis. Cancer Med. 2019, 8, 6165–6175.
- Moris, D.; Ronnekleiv-Kelly, S.; Rahnemai-Azar, A.A.; Felekouras, E.; Dillhoff, M.; Schmidt, C.; Pawlik, T.M. Parenchymal-Sparing Versus Anatomic Liver Resection for Colorectal Liver Metastases: A Systematic Review. J. Gastrointest. Surg. 2017, 21, 1076–1085.
- Hosokawa, I.; Allard, M.A.; Mirza, D.F.; Kaiser, G.; Barroso, E.; Lapointe, R.; Laurent, C.; Ferrero, A.; Miyazaki, M.; Adam, R. Outcomes of parenchyma-preserving hepatectomy and right hepatectomy for solitary small colorectal liver metastasis: A LiverMetSurvey study. Surgery 2017, 162, 223–232.
- Burlaka, A.P.; Ganusevich, I.I.; Vovk, A.V.; Burlaka, A.A.; Gafurov, M.R.; Lukin, S.N. Colorectal Cancer and Mitochondrial Dysfunctions of the Adjunct Adipose Tissues: A Case Study. Biomed Res. Int. 2018, 2018, 2169036.
- Torzilli, G.; Viganò, L.; Cimino, M.; Imai, K.; Vibert, E.; Donadon, M.; Mansour, D.; Castaing, D.; Adam, R. Is Enhanced One-Stage Hepatectomy a Safe and Feasible Alternative to the Two-Stage Hepatectomy in the Setting of Multiple Bilobar Colorectal Liver Metastases? A Comparative Analysis between Two Pioneering Centers. Dig. Surg. 2018, 35, 323–332.
- Torzilli, G.; Serenari, M.; Viganò, L.; Cimino, M.; Benini, C.; Massani, M.; Ettorre, G.M.; Cescon, M.; Ferrero, A.; Cillo, U.; et al. Outcomes of enhanced one-stage ultrasound-guided hepatectomy for bilobar colorectal liver metastases compared to those of ALPPS: A multicenter case-match analysis. HPB 2019, 21, 1411–1418.
- Viganò, L.; Torzilli, G.; Cimino, M.; Imai, K.; Vibert, E.; Donadon, M.; Castaing, D.; Adam, R. Drop-out between the two liver resections of two-stage hepatectomy. Patient selection or loss of chance? Eur. J. Surg. Oncol. 2016, 42, 1385–1393.
- Mise, Y.; Aloia, T.A.; Brudvik, K.W.; Schwarz, L.; Vauthey, J.N.; Conrad, C. Parenchymal-sparing Hepatectomy in Colorectal Liver Metastasis Improves Salvageability and Survival. Ann. Surg. 2016, 263, 146–152.
- Burlaka, A.A.; Kolesnik, O.O. Parenchyma sparing multicomponent liver resection strategy for multiple bilobar synchronous colorectal cancer metastasis. Clin. Case Rep. 2020, 8, 661–666.
- Torzilli, G.; Viganò, L.; Gatti, A.; Costa, G.; Cimino, M.; Procopio, F.; Donadon, M.; Del Fabbro, D. Twelve-year experience of “radical but conservative” liver surgery for colorectal metastases: Impact on surgical practice and oncologic efficacy. HPB 2017, 19, 775–784.
- Alvarez, F.A.; Claria, R.S.; Oggero, S.; de Santibañes, E. Parenchymal-sparing liver surgery in patients with colorectal carcinoma liver metastases. World J. Gastrointest. Surg. 2016, 8, 407–423.
- Evrard, S.; Torzilli, G.; Caballero, C.; Bonhomme, B. Parenchymal sparing surgery brings treatment of colorectal liver metastases into the precision medicine era. Eur. J. Cancer. 2018, 104, 195–200.
- Torzilli, G.; Procopio, F.; Botea, F.; Marconi, M.; Del Fabbro, D.; Donadon, M.; Palmisano, A.; Spinelli, A.; Montorsi, M. One-stage ultrasonographically guided hepatectomy for multiple bilobar colorectal metastases: A feasible and effective alternative to the 2-stage approach. Surgery 2009, 146, 60–71.
- Donadon, M.; Torzilli, G. Intraoperative ultrasound in patients with hepatocellular carcinoma: From daily practice to future trends. Liver Cancer 2013, 2, 16–24.
- Torzilli, G.; Donadon, M.; Marconi, M.; Botea, F.; Palmisano, A.; Del Fabbro, D.; Procopio, F.; Montorsi, M. Systematic extended right posterior sectionectomy: A safe and effective alternative to right hepatectomy. Ann. Surg. 2008, 247, 603–611.
- Makuuchi, M.; Hasegawa, H.; Yamazaki, S.; Takayasu, K. Four new hepatectomy procedures for resection of the right hepatic vein and preservation of the inferior right hepatic vein. Surg. Gynecol. Obstet. 1987, 164, 68–72.
- Torzilli, G.; Procopio, F.; Donadon, M.; Del Fabbro, D.; Cimino, M.; Garcia-Etienne, C.A.; Montorsi, M. Upper transversal hepatectomy. Ann. Surg. Oncol. 2012, 19, 3566.
- Torzilli, G.; Palmisano, A.; Procopio, F.; Cimino, M.; Botea, F.; Donadon, M.; Del Fabbro, D.; Montorsi, M. A new systematic small for size resection for liver tumors invading the middle hepatic vein at its caval confluence: Mini-mesohepatectomy. Ann. Surg. 2010, 251, 33–39.
- Torzilli, G.; Procopio, F.; Viganò, L.; Costa, G.; Fontana, A.; Cimino, M.; Donadon, M.; Del Fabbro, D. The Liver Tunnel: Intention-to-treat Validation of a New Type of Hepatectomy. Ann. Surg. 2019, 269, 331–336.
- Botea, F.; Barcu, A.; Croitoru, A.; Tomescu, D.; Popescu, I. Limited Central Hepatectomy for Centrally Located Tumors: Is There a Place for Standardization? Surg. Gastroenterol. Oncol. 2019, 24, 170–183.
- Botea, F.; Barcu, A.; Verdea, C.; Kambakamba, P.; Popescu, I.; Linecker, M. Regenerative Liver Surgery—ALPPS and Associated Techniques. Chirurgia 2021, 116, 387–398.
- Makuuchi, M.; Thai, B.L.; Takayasu, K.; Takayama, T.; Kosuge, T.; Gunvén, P.; Yamazaki, S.; Hasegawa, H.; Ozaki, H. Preoperative portal embolization to increase safety of major hepatectomy for hilar bile duct carcinoma: A preliminary report. Surgery 1990, 107, 521–527.
- Pandanaboyana, S.; Bell, R.; Hidalgo, E.; Toogood, G.; Prasad, K.R.; Bartlett, A.; Lodge, J.P. A systematic review and meta-analysis of portal vein ligation versus portal vein embolization for elective liver resection. Surgery 2015, 157, 690–698.
- Shindoh, J.; Tzeng, C.W.; Aloia, T.A.; Curley, S.A.; Zimmitti, G.; Wei, S.H.; Huang, S.Y.; Gupta, S.; Wallace, M.J.; Vauthey, J.N. Portal vein embolization improves rate of resection of extensive colorectal liver metastases without worsening survival. Br. J. Surg. 2013, 100, 1777–1783.
- Eshmuminov, D.; Raptis, D.A.; Linecker, M.; Wirsching, A.; Lesurtel, M.; Clavien, P.A. Meta-analysis of associating liver partition with portal vein ligation and portal vein occlusion for two-stage hepatectomy. Br. J. Surg. 2016, 103, 1768–1782.
- Guiu, B.; Chevallier, P.; Denys, A.; Delhom, E.; Pierredon-Foulongne, M.A.; Rouanet, P.; Ramos, J. Simultaneous trans-hepatic portal and hepatic vein embolization before major hepatectomy: The liver venous deprivation technique. Eur. Radiol. 2016, 26, 4259–4267.
- Panaro, F.; Giannone, F.; Riviere, B.; Sgarbura, O.; Cusumano, C.; Deshayes, E.; Quenet, F. Perioperative impact of liver venous deprivation compared with portal venous embolization in patients undergoing right hepatectomy: Preliminary results from the pioneer center. Hepatobiliary Surg. Nutr. 2019, 8, 329–337.
- Laurent, C.; Fernandez, B.; Marichez, A.; Adam, J.P.; Papadopoulos, P.; Lapuyade, B.; Chiche, L. Radiological Simultaneous Portohepatic Vein Embolization (RASPE) Before Major Hepatectomy: A Better Way to Optimize Liver Hypertrophy Compared to Portal Vein Embolization. Ann. Surg. 2020, 272, 199–205.
- Le Roy, B.; Gallon, A.; Cauchy, F.; Pereira, B.; Gagnière, J.; Lambert, C.; Yoh, T.; Boyer, L.; Pezet, D.; Buc, E.; et al. Combined biembolization induces higher hypertrophy than portal vein embolization before major liver resection. HPB 2020, 22, 298–305.
- Guiu, B.; Herrero, A.; Panaro, F. Liver venous deprivation: A bright future for liver metastases-but what about hepatocellular carcinoma? Hepatobiliary Surg. Nutr. 2021, 10, 270–272.
- Adam, R.; Laurent, A.; Azoulay, D.; Castaing, D.; Bismuth, H. Two-stage hepatectomy: A planned strategy to treat irresectable liver tumors. Ann. Surg. 2000, 232, 777–785. [Google Scholar] [CrossRef]Adam, R.; Laurent, A.; Azoulay, D.; Castaing, D.; Bismuth, H. Two-stage hepatectomy: A planned strategy to treat irresectable liver tumors. Ann. Surg. 2000, 232, 777–785.
- Imai, K.; Adam, R.; Baba, H. How to increase the resectability of initially unresectable colorectal liver metastases: A surgical perspective. Ann. Gastroenterol Surg. 2019, 3, 476–486. [Google Scholar] [CrossRef] [PubMed]Imai, K.; Adam, R.; Baba, H. How to increase the resectability of initially unresectable colorectal liver metastases: A surgical perspective. Ann. Gastroenterol Surg. 2019, 3, 476–486.
- Jaeck, D.; Bachellier, P.; Nakano, H.; Oussoultzoglou, E.; Weber, J.C.; Wolf, P.; Greget, M. One or two-stage hepatectomy combined with portal vein embolization for initially nonresectable colorectal liver metastases. Am. J. Surg. 2003, 185, 221–229. [Google Scholar] [CrossRef]Jaeck, D.; Bachellier, P.; Nakano, H.; Oussoultzoglou, E.; Weber, J.C.; Wolf, P.; Greget, M. One or two-stage hepatectomy combined with portal vein embolization for initially nonresectable colorectal liver metastases. Am. J. Surg. 2003, 185, 221–229.
- Lam, V.W.; Laurence, J.M.; Johnston, E.; Hollands, M.J.; Pleass, H.C.; Richardson, A.J. A systematic review of two-stage hepatectomy in patients with initially unresectable colorectal liver metastases. HPB 2013, 15, 483–491. [Google Scholar] [CrossRef]Lam, V.W.; Laurence, J.M.; Johnston, E.; Hollands, M.J.; Pleass, H.C.; Richardson, A.J. A systematic review of two-stage hepatectomy in patients with initially unresectable colorectal liver metastases. HPB 2013, 15, 483–491.
- Kobayashi, K.; Yamaguchi, T.; Denys, A.; Perron, L.; Halkic, N.; Demartines, N.; Melloul, E. Liver venous deprivation compared to portal vein embolization to induce hypertrophy of the future liver remnant before major hepatectomy: A single center experience. Surgery 2020, 167, 917–923.
- Guiu, B.; Quenet, F.; Panaro, F.; Piron, L.; Cassinotto, C.; Herrerro, A.; Souche, F.R.; Hermida, M.; Pierredon-Foulongne, M.A.; Belgour, A.; et al. Liver venous deprivation versus portal vein embolization before major hepatectomy: Future liver remnant volumetric and functional changes. Hepatobiliary Surg. Nutr. 2020, 9, 564–576.
- Schnitzbauer, A.A.; Lang, S.A.; Goessmann, H.; Nadalin, S.; Baumgart, J.; Farkas, S.A.; Fichtner-Feigl, S.; Lorf, T.; Goralcyk, A.; Hörbelt, R.; et al. Right portal vein ligation combined with in situ splitting induces rapid left lateral liver lobe hypertrophy enabling 2-staged extended right hepatic resection in small-for-size settings. Ann. Surg. 2012, 255, 405–414.
- de Santibañes, E.; Clavien, P.A. Playing Play-Doh to prevent postoperative liver failure: The “ALPPS” approach. Ann. Surg. 2012, 255, 415–417.
- Moris, D.; Ronnekleiv-Kelly, S.; Kostakis, I.D.; Tsilimigras, D.I.; Beal, E.W.; Papalampros, A. Operative Results and Oncologic Outcomes of ALPPS versus TSH in Patients with Unresectable Colorectal Liver Metastases: A Systematic Review and Meta-Analysis. World J. Surg. 2018, 42, 806–815.
- Truant, S.; Baillet, C.; Deshorgue, A.C.; Leteurtre, E.; Hebbar, M.; Ernst, O.; Pruvot, F.R. Drop of total liver function in the inter-stages of the new associating liver partition and portal vein ligation for staged hepatectomy technique: Analysis of the “Auxiliary liver” by HIDA scintigraphy. Ann. Surg. 2016, 263, e33–e34.
- Enne, M.; Schadde, E.; Björnsson, B.; Alejandro, R.H.; Steinbruck, K.; Viana, E.; Campos, R.R.; Malago, M.; Clavien, P.A.; De Santibanes, E.; et al. ALPPS Registry Group. ALPPS as a salvage procedure after insufficient future liver remnant hypertrophy following portal vein occlusion. HPB 2017, 19, 1126–1129.
- Sandström, P.; Røsok, B.I.; Sparrelid, E.; Larsen, P.N.; Larsson, A.L.; Lindell, G.; Schultz, N.A.; Bjørnbeth, B.A.; Isaksson, B.; Rizell, M.; et al. ALPPS Improves Resectability Compared with Conventional Two-stage Hepatectomy in Patients with Advanced Colorectal Liver Metastasis: Results from a Scandinavian Multicenter Randomized Controlled Trial (LIGRO Trial). Ann. Surg. 2018, 267, 833–840.
- Linecker, M.; Stavrou, G.A.; Oldhafer, K.J.; Jenner, R.M.; Seifert, B.; Lurje, G.; Petrowsky, H. The ALPPS Risk Score: Avoiding Futile Use of ALPPS. Ann. Surg. 2016, 264, 763–771.
- Linecker, M.; Björnsson, B.; Stavrou, G.A.; Oldhafer, K.J.; Lurje, G.; Neumann, U.; Petrowsky, H. Risk Adjustment in ALPPS Is Associated with a Dramatic Decrease in Early Mortality and Morbidity. Ann. Surg. 2017, 266, 779–786.
- Machado, M.A.; Makdissi, F.F.; Surjan, R.C.; Basseres, T.; Schadde, E. Transition from open to laparoscopic ALPPS for patients with very small FLR: The initial experience. HPB 2017, 19, 59–66.
- Schadde, E.; Ardiles, V.; Slankamenac, K.; Tschuor, C.; Sergeant, G.; Amacker, N.; Baumgart, J.; Croome, K.; Hernandez-Alejandro, R.; Lang, H.; et al. ALPPS offers a better chance of complete resection in patients with primarily unresectable liver tumors compared with conventional-staged hepatectomies: Results of a multicenter analysis. World J. Surg. 2014, 38, 1510–1519.
- Olthof, P.B.; Tomassini, F.; Huespe, P.E.; Truant, S.; Pruvot, F.R.; Troisi, R.I.; Castro, C.; Schadde, E.; Axelsson, R.; Sparrelid, E.; et al. Hepatobiliary scintigraphy to evaluate liver function in associating liver partition and portal vein ligation for staged hepatectomy: Liver volume overestimates liver function. Surgery 2017, 162, 775–783.
- Lodge, J.P. ALPPS: The argument for. Eur. J. Surg. Oncol. 2017, 43, 246–248.
- Petrowsky, H.; Györi, G.; de Oliveira, M.; Lesurtel, M.; Clavien, P.A. Is partial-ALPPS safer than ALPPS? A single-center experience. Ann. Surg. 2015, 261, e90–e92.
- Alvarez, F.A.; Ardiles, V.; de Santibañes, M.; Pekolj, J.; de Santibañes, E. Associating liver partition and portal vein ligation for staged hepatectomy offers high oncological feasibility with adequate patient safety: A prospective study at a single center. Ann. Surg. 2015, 261, 723–732.
- Tanaka, K.; Kikuchi, Y.; Kawaguchi, D.; Murakami, T.; Hiroshima, Y.; Matsuo, K. Modified ALPPS Procedures Avoiding Division of Portal Pedicles. Ann. Surg. 2017, 265, e14–e20.
- Li, J.; Kantas, A.; Ittrich, H.; Koops, A.; Achilles, E.G.; Fischer, L.; Nashan, B. Avoid “All-Touch” by Hybrid ALPPS to Achieve Oncological Efficacy. Ann. Surg. 2016, 263, e6–e7.
- Wanis, K.N.; Linecker, M.; Madenci, A.L.; Müller, P.C.; Nüssler, N.; Brusadin, R.; Robles-Campos, R.; Hernandez-Alejandro, R. Variation in complications and mortality following ALPPS at early-adopting centers. HPB 2021, 23, 46–55.
- Botea, F.; Barcu, A.; Croitoru, A.; Tomescu, D.; Lupescu, I.; Dumitru, R.; Linecker, M. Parenchyma Sparing ALPPS—Ultrasound Guided Partition Through Segment 4 to Maximize Resectability (with video). Chirurgia 2022, 117, 81–93.
- de Santibañes, E.; Alvarez, F.A.; Ardiles, V.; Pekolj, J.; de Santibañes, M. Inverting the ALPPS paradigm by minimizing first stage impact: The Mini-ALPPS technique. Langenbecks Arch. Surg. 2016, 401, 557–563.
- Sakamoto, Y.; Inagaki, F.; Omichi, K.; Ohkura, N.; Hasegawa, K.; Kokudo, N. Associating Liver Partial Partition and Transileocecal Portal Vein Embolization for Staged Hepatectomy. Ann. Surg. 2016, 264, e21–e22.
- Makuuchi, M.; Thai, B.L.; Takayasu, K.; Takayama, T.; Kosuge, T.; Gunvén, P.; Yamazaki, S.; Hasegawa, H.; Ozaki, H. Preoperative portal embolization to increase safety of major hepatectomy for hilar bile duct carcinoma: A preliminary report. Surgery 1990, 107, 521–527.
- Gruenberger, T.; Bridgewater, J.; Chau, I.; García Alfonso, P.; Rivoire, M.; Mudan, S.; Lasserre, S.; Hermann, F.; Waterkamp, D.; Adam, R. Bevacizumab plus mFOLFOX-6 or FOLFOXIRI in patients with initially unresectable liver metastases from colorectal cancer: The OLIVIA multinational randomised phase II trial. Ann. Oncol. 2015, 26, 702–708.
- Guo, M.; Jin, N.; Pawlik, T.; Cloyd, J.M. Neoadjuvant chemotherapy for colorectal liver metastases: A contemporary review of the literature. World J. Gastrointest. Oncol. 2021, 13, 1043–1061.
- Pawlik, T.M.; Scoggins, C.R.; Zorzi, D.; Abdalla, E.K.; Andres, A.; Eng, C.; Curley, S.A.; Loyer, E.M.; Muratore, A.; Mentha, G.; et al. Effect of surgical gin status on survival and site of recurrence after hepatic resection for colorectal metastases. Ann. Surg. 2005, 241, 715–722.
- Adam, R.; Delvart, V.; Pascal, G.; Valeanu, A.; Castaing, D.; Azoulay, D.; Giacchetti, S.; Paule, B.; Kunstlinger, F.; Ghémard, O.; et al. Rescue surgery for unresectable colorectal liver metastases downstaged by chemotherapy: A model to predict long-term survival. Ann. Surg. 2004, 240, 644–665.
- Torzilli, G.; Adam, R.; Viganò, L.; Imai, K.; Goransky, J.; Fontana, A.; Toso, C.; Majno, P.; de Santibañes, E. Surgery of Colorectal Liver Metastases: Pushing the Limits. Liver Cancer 2016, 6, 80–89.
- Burlaka, A.A.; Kolesnik, O.O. Parenchyma sparing multicomponent liver resection strategy for multiple bilobar synchronous colorectal cancer metastasis. Clin. Case Rep. 2020, 8, 661–666.
- Matsumura, M.; Mise, Y.; Saiura, A.; Inoue, Y.; Ishizawa, T.; Ichida, H.; Matsuki, R.; Tanaka, M.; Takeda, Y.; Takahashi, Y. Parenchymal-Sparing Hepatectomy Does Not Increase Intrahepatic Recurrence in Patients with Advanced Colorectal Liver Metastases. Ann. Surg. Oncol. 2016, 23, 3718–3726.
- Oba, M.; Hasegawa, K.; Shindoh, J.; Yamashita, S.; Sakamoto, Y.; Makuuchi, M.; Kokudo, N. Survival benefit of repeat resection of successive recurrences after the initial hepatic resection for colorectal liver metastases. Surgery 2016, 159, 632–640.
- Andreou, A.; Gloor, S.; Inglin, J.; Di Pietro tinelli, C.; Banz, V.; Lachenmayer, A.; Kim-Fuchs, C.; Candinas, D.; Beldi, G. Parenchymal-sparing hepatectomy for colorectal liver metastases reduces postoperative morbidity while maintaining equivalent oncologic outcomes compared to non-parenchymal-sparing resection. Surg. Oncol. 2021, 38, 101631.
- van Lienden, K.P.; van den Esschert, J.W.; de Graaf, W.; Bipat, S.; Lameris, J.S.; van Gulik, T.M.; van Delden, O.M. Portal vein embolization before liver resection: A systematic review. Cardiovasc. Intervent. Radiol. 2013, 36, 25–34.
- Abdalla, E.K.; Hicks, M.E.; Vauthey, J.N. Portal vein embolization: Rationale, technique and future prospects. Br. J. Surg. 2001, 88, 165–175.
- Adam, R.; Miller, R.; Pitombo, M.; Wicherts, D.A.; de Haas, R.J.; Bitsakou, G.; Aloia, T. Two-stage hepatectomy approach for initially unresectable colorectal hepatic metastases. Surg. Oncol. Clin. N. Am. 2007, 16, 525–536.
- Rees, M.; Tekkis, P.P.; Welsh, F.K.; O’Rourke, T.; John, T.G. Evaluation of long-term survival after hepatic resection for metastatic colorectal cancer: A multifactorial model of 929 patients. Ann. Surg. 2008, 247, 125–135.
- Pulitanò, C.; Castillo, F.; Aldrighetti, L.; Bodingbauer, M.; Parks, R.W.; Ferla, G.; Garden, O.J. What defines ‘cure’ after liver resection for colorectal metastases? Results after 10 years of follow-up. HPB 2010, 12, 244–249.
- Charnsangavej, C.; Clary, B.; Fong, Y.; Grothey, A.; Pawlik, T.M.; Choti, M.A. Selection of patients for resection of hepatic colorectal metastases: Expert consensus statement. Ann. Surg. Oncol. 2006, 13, 1261–1268.
- Orcutt, S.T.; Anaya, D.A. Liver Resection and Surgical Strategies for Management of Primary Liver Cancer. Cancer Control 2018, 25, 1073274817744621.
- Donadon, M.; Terrone, A.; Procopio, F.; Cimino, M.; Palmisano, A.; Viganò, L.; Del Fabbro, D.; Di Tommaso, L.; Torzilli, G. Is R1 vascular hepatectomy for hepatocellular carcinoma oncologically adequate? Analysis of 327 consecutive patients. Surgery 2019, 165, 897–904.
- Hasegawa, K.; Kokudo, N.; Imamura, H.; Matsuyama, Y.; Aoki, T.; Minagawa, M.; Sano, K.; Sugawara, Y.; Takayama, T.; Makuuchi, M. Prognostic impact of anatomic resection for hepatocellular carcinoma. Ann. Surg. 2005, 242, 252–259.
- Marubashi, S.; Gotoh, K.; Akita, H.; Takahashi, H.; Ito, Y.; Yano, M.; Ishikawa, O.; Sakon, M. Anatomical versus non-anatomical resection for hepatocellular carcinoma. Br. J. Surg. 2015, 102, 776–784.
- Eltawil, K.M.; Kidd, M.; Giovinazzo, F.; Helmy, A.H.; Salem, R.R. Differentiating the impact of anatomic and non-anatomic liver resection on early recurrence in patients with Hepatocellular Carcinoma. World J. Surg. Oncol. 2010, 8, 43.
- Marubashi, S.; Gotoh, K.; Akita, H.; Takahashi, H.; Sugimura, K.; Miyoshi, N.; Sakon, M. Analysis of Recurrence Patterns After Anatomical or Non-anatomical Resection for Hepatocellular Carcinoma. Ann. Surg. Oncol. 2015, 22, 2243–2252.
- Yip, V.S.; Poon, R.T.; Chok, K.S.; Chan, A.C.; Dai, W.C.; Tsang, S.H.; Chan, S.C.; Lo, C.M.; Cheung, T.T. Comparison of Survival Outcomes Between Right Posterior Sectionectomy and Right Hepatectomy for Hepatocellular Carcinoma in Cirrhotic Liver: A Single-Centre Experience. World J. Surg. 2015, 39, 2764–2770.
- Lee, S.Y.; Sadot, E.; Chou, J.F.; Gönen, M.; Kingham, T.P.; Allen, P.J.; DeMatteo, R.P.; Jarnagin, W.R.; D’Angelica, M.I. Central hepatectomy versus extended hepatectomy for liver malignancy: A matched cohort comparison. HPB 2015, 17, 1025–1032.
- Chen, X.; Li, B.; He, W.; Wei, Y.G.; Du, Z.G.; Jiang, L. Mesohepatectomy versus extended hemihepatectomy for centrally located hepatocellular carcinoma. Hepatobiliary Pancreat. Dis. Int. 2014, 13, 264–270.
- Li, B.; Song, J.L.; Aierken, Y.; Chen, Y.; Zheng, J.L.; Yang, J.Y. Nonanatomic resection is not inferior to anatomic resection for primary intrahepatic cholangiocarcinoma: A propensity score analysis. Sci. Rep. 2018, 8, 17799.
- Li, M.X.; Bi, X.Y.; Li, Z.Y.; Huang, Z.; Han, Y.; Zhao, J.J.; Zhao, H.; Cai, J.Q. Impaction of surgical gin status on the survival outcome after surgical resection of intrahepatic cholangiocarcinoma: A systematic review and meta-analysis. J. Surg. Res. 2016, 203, 163–173.
- Spolverato, G.; Yakoob, M.Y.; Kim, Y.; Alexandrescu, S.; Ques, H.P.; Lamelas, J.; Aldrighetti, L.; Gamblin, T.C.; Maithel, S.K.; Pulitano, C.; et al. The Impact of Surgical gin Status on Long-Term Outcome after Resection for Intrahepatic Cholangiocarcinoma. Ann. Surg. Oncol. 2015, 22, 4020–4028.
- Machairas, N.; Prodromidou, A.; Molmenti, E.; Kostakis, I.D.; Sotiropoulos, G.C. Management of liver metastases from gastrointestinal stromal tumors: Where do we stand? J. Gastrointest. Oncol. 2017, 8, 1100–1108.
- Aoki, T.; Kubota, K.; Kiritani, S.; Arita, J.; Morizane, C.; Masui, T.; Kudo, A.; Komoto, I.; Hatano, E.; Ito, T.; et al. Survey of surgical resections for neuroendocrine liver metastases: A project study of the Japan Neuroendocrine Tumor Society (JNETS). J. Hepato Biliary Pancreat. Sci. 2021, 28, 489–497.
- Hoekstra, L.T.; Bieze, M.; Erdogan, D.; Roelofs, J.J.; Beuers, U.H.; van Gulik, T.M. Management of giant liver hemangiomas: An update. Expert Rev. Gastroenterol. Hepatol. 2013, 7, 263–268.
- Linecker, M.; Kambakamba, P.; Raptis, D.A.; Malagó, M.; Ratti, F.; Aldrighetti, L.; Frilling, A. ALPPS in neuroendocrine liver metastases not amenable for conventional resection—Lessons learned from an interim analysis of the International ALPPS Registry. HPB 2020, 22, 537–544.
- Alexandrescu, S.S.L.; Grigorie, R.; Tomescu, D.; Dobrea, C.; Popescu, I.; Hrehoret, D. Primary Hepatic Lymphoma Resected by ALPPS Procedure (Associating Liver Partition and Portal Vein Ligation for Staged Hepatectomy). JTMR 2016, 21, 153–158.
- Linecker, M.; Kambakamba, P.; Raptis, D.A.; Malagó, M.; Ratti, F.; Aldrighetti, L.; Frilling, A. ALPPS in neuroendocrine liver metastases not amenable for conventional resection—Lessons learned from an interim analysis of the International ALPPS Registry. HPB 2020, 22, 537–544.
- Alexandrescu, S.S.L.; Grigorie, R.; Tomescu, D.; Dobrea, C.; Popescu, I.; Hrehoret, D. Primary Hepatic Lymphoma Resected by ALPPS Procedure (Associating Liver Partition and Portal Vein Ligation for Staged Hepatectomy). JTMR 2016, 21, 153–158.
